Abstract
Background
High-fat diet (HFD)-induced nonalcoholic fatty liver disease (NAFLD) is a key link between several medical conditions and is becoming a major health concern affecting thousands of people globally. Andrographolide, an active ingredient extracted from Andrographis paniculate, has shown anti-inflammatory and anti-fibrosis effects in recent studies.
Objective
This study aims to explore whether andrographolide can mitigate HFD-induced NAFLD and the underlying mechanism.
Methods
We treated C57B/6 mice consuming a high-fat diet (HFD) with varying concentrations of andrographolide. Enzyme-linked immunosorbent assay (ELISA) was employed to evaluate liver function indicators in circulating blood. Haematoxylin and Eosin and Oil Red O staining were used to assess inflammation infiltration and lipid deposits in liver tissues. Reverse transcription-quantitative polymerase chain reaction (RT-qPCR) and immunoblotting were used to analyze the expression levels of genes and proteins.
Results
ELISA revealed that andrographolide dose-dependently mitigated the HFD-induced elevations of glucose, alanine aminotransferase, and aspartate aminotransferase in circulating blood. Histological analysis indicated that andrographolide dose-dependently reduced HFD-induced inflammation infiltration and lipid deposition. RT-qPCR and immunoblotting analyses showed that andrographolide dose-dependently inhibited the HFD-induced overproduction of tumor necrosis factor (TNF)-α, interleukin (IL)-1β, IL-6, and the up-regulation of p-p65 and NLRP3 proteins.
Conclusion
Together, these results suggest that andrographolide treatment can dose-dependently mitigate HFD-induced liver function impairments, lipid accumulation, pro-inflammatory cytokine overproduction, and inflammatory responses in the liver of mice by regulating the NF-κB signaling pathway. However, it is important to note that our results are observational and do not conclusively demonstrate the necessity of the NF-κB signaling pathway's involvement in the NAFLD-alleviating effects of andrographolide. This highlights the need for further validation at both the gene and protein levels through additional in vitro and in vivo experiments.
Keywords
Introduction
The liver, a complex immune organ, functions as a physiological bridge connecting foreign molecules derived from the gut and the peripheral circulatory system. Consequently, the liver must maintain constant immunosurveillance for infectious pathogens and liver malignancies while tolerating normal foreign molecules with immunogenic or inflammatory potential.1,2 These complex functions of the liver also make the liver vulnerable to various diseases. One such disease is nonalcoholic fatty liver disease (NAFLD), the most prevalent chronic liver disease, affecting approximately 25% of the global population and is projected to become the leading cause of liver transplantation by 2030.3,4 Although the etiology of NAFLD is complex and multifactorial, it is currently considered a hepatic manifestation of metabolic abnormalities associated with insulin resistance, obesity, and cholesterol dysregulation.3,5 Hence, a high-fat diet (HFD), which predisposes individuals to fat metabolism disorders, is significantly corrected with the prevalence of NAFLD.6,7 For instance, it has been reported that patients with type 2 diabetes exhibit a NAFLD prevalence of up to 75%, and severely obese patients show prevalence rates as high as 90%.8,9
An HFD contributes to the development and progression of NAFLD through multiple mechanisms, including but not limited to the induction of an inflammatory response in hepatocytes.6,10 Briefly, when HDF is consumed over a long period, excess free fatty acids (FFAs) absorbed by small intestinal epithelial cells are translocated to the liver. These FFAs exceed the processing capacity of hepatocytes, leading to lipotoxicity-induced cell damage and the overproduction of various pro-inflammatory cytokines, such as tumor necrosis factor (TNF)-α, interleukin (IL)-1β, IL-6, and IL-10. High levels of these pro-inflammatory cytokines attract immune cells and cause alterations in the components of the extra-cellular matrix, ultimately resulting in liver fibrosis.1,6,11 Therefore, alleviating the inflammatory response in the liver can help mitigate the development and progression of HFD-induced NAFLD. 12 For example, Zhu and colleagues revealed that exercise-induced irisin decreased inflammation and improved HFD-induced NAFLD in mice by regulating the MD2-TLR4 pathway.
Currently, although numerous investigations into the pathophysiology of NAFLD have been carried out, no pharmacological agent has been officially approved for the treatment of NAFLD. Herbal medicine, defined as whole medicinal plants and their unpurified extracts with medical properties, has been applied globally to improve liver diseases, including NAFLD.13,14 For example, mixture 919 syrup, a Chinese herbal medicine, was found to alleviate HFD-induced NAFLD in a mouse model by inhibiting the NF-κB signaling pathway-mediated inflammatory response. 15 Andrographis paniculate, also known as the “King of Bitters”, is a medicinal plant with anti-inflammatory and antibacterial properties. 16 It was found to reduce steatohepatitis and liver injury in mice fed a high-fat and high-cholesterol diet. 17 Andrographolide, a natural compound extracted from Andrographis paniculate, was found to inhibit AIM2 inflammasome-mediated pyroptosis and contribute to the amelioration of radiation-induced lung inflammation and fibrosis. 18 Additionally, andrographolide has been widely reported to inhibit the activation of the NF-κB signaling pathway, which is persistently activated and considered to play a crucial role in the progression of NAFLD.19,20 This inhibition has been observed in multiple inflammation-related cell models.21–23 These findings suggest that andrographolide might play a role in the mitigative effects of Andrographis paniculate on steatohepatitis and liver injury by affecting the inflammation in hepatocytes. However, this hypothesis needs to be validated by further experiments.
In this study, we investigated whether andrographolide could mitigate lipid deposition, the inflammatory response in hepatocytes, and liver injury in an HFD-induced NAFLD mouse model. We also examined whether the NF-κB signaling pathway was involved in the mitigative effects of andrographolide. Our findings will clarify the role of andrographolide in alleviating HFD-induced inflammatory response in hepatocytes, providing a potential alternative treatment for NAFLD.
Materials and Methods
All procedures related to animals in this study were conducted in strict accordance with the Guidelines on the Care and Use of Laboratory Animals issued by the Chinese Council on Animal Research and the Guidelines of Animal Care and received approval from the Ethical and Welfare Committee for Animal Experiments of Yunnan University of Traditional Chinese Medicine.
Animal Management and Experimental Design
A total of 60 male C57BL/6 mice, aged 6 weeks, were purchased from Chengdu Dossy Experimental Animals Co., Ltd (Chengdu, China). The mice were housed in standard cages maintained at a temperature of 22 ± 2 °C, with a 12-h light/dark cycle and a humidity level of 40–60%. Bedding was refreshed every 2 days. After a 1-week acclimation period, the mice were randomly allocated into 4 groups: Chow (n = 15), HFD (n = 15), HFD + LA (n = 15), and HFD + HA (n = 15). During the 12-week feeding period, the Chow group received a standard laboratory diet containing 10% kcal from fat, 20% kcal from protein, and 70% kcal from carbohydrates (Yes Service Biotech, Shanghai, China; Cat No: D12450B). The HFD, HFD + LA, and HFD + HA groups were fed an HFD consisting of 60% kcal from fat, 20% kcal from protein, and 20% kcal from carbohydrates (Yes Service Biotech, Shanghai, China; Cat No: D12492). The detailed ingredient lists of the chow diet and HFD are shown in Supplementary Table 1 and Supplementary Table 2, respectively. Beginning in the fifth week and continuing through the 12th week, each mouse in the Chow and HFD groups was gavaged daily with 1 ml of distilled water at 10 am. Mice in the HFD + LA and HFD + HA groups received a daily gavage of 1 ml of distilled water containing either a low dose (50 mg/kg) or a high dose (200 mg/kg) of andrographolide (Cat No: HY-N0191; MCE, NJ, USA) at the same time each day. Before the administration, mice in the HFD + LA and HFD + HA groups were weighed daily at 9:00 am, and the doses were prepared accordingly based on their body weight (50 mg/kg and 200 mg/kg, respectively), then dissolved in 1 ml of distilled water. This treatment was administered for 8 consecutive weeks. The doses of andrographolide were chosen according to previous studies.24,25 All mice had free access to water and food, which were replenished daily in each cage. After 12 weeks of feeding, the mice were fasted for 12 h before being sacrificed. Liver tissue and serum samples were collected for further experiments. Figure 1 A shows the sketch of the experimental design.

Sketch map of the experimental design (A) and molecular formula of andrographolide (B). Chow: fed with standard laboratory diet; HFD: fed with high-fat diet; HFD + LA: fed with high-fat diet and treated with 50 mg/kg andrographolide; and HFD + HA: fed with high-fat diet and treated with 200 mg/kg andrographolide.
Enzyme-Linked Immunosorbent Assay (ELISA)
The level of glucose (Cat No: A154-1-1) and the activities of alanine aminotransferase (ALT; Cat No: C009-2-1) and aspartate aminotransferase (AST; Cat No: C010-2-1) in the collected serum were measured using commercial ELISA kits from Nanjing Jiancheng (Nanjing, China), following the manufacturer's protocols. In brief, 100 μl of serum was added to each well of the reaction plate, which was then sealed with a membrane and incubated at room temperature for 2 h. Afterward, biotinylated antibody, horseradish peroxidase-labeled streptavidin, and tetramethylbenzidine solution were sequentially added to each well. The plate was left to incubate overnight in the dark at room temperature. Finally, 50 μl of stop solution was added to halt the reaction, and the optical density at 450 nm (OD450) was measured using a microplate reader (ThermoFisher, Shanghai, China).
Histological Analysis
The freshly collected liver tissues were fixed in 4% paraformaldehyde for 48 h. After fixation, the tissues were rinsed multiple times with tap water, dehydrated using a gradient alcohol series, soaked in paraffin, and then embedded. For Haematoxylin and Eosin (HE) staining analysis, the paraffin sections (5 μm thick) were first incubated at 80°C for 20 min. They were then de-paraffinized and soaked in haematoxylin solution (Solarbio, Beijing, China) for 15 min. Following this, the sections were rinsed with tap water for re-bluing, stained in eosin, hydrated through a gradient alcohol series, cleared with xylene, and mounted with neutral resin before being air-dried, observed, and photographed under a microscope. For Oil Red O staining analysis, tissue sections were fixed with formaldehyde calcium for 10 min, washed with distilled water, and then rinsed with 60% isopropanol. The sections were stained with Oil Red O solution (Solarbio, Beijing, China) for 10 min, followed by color separation using 60% isopropanol and washing with distilled water. Subsequently, the tissue sections were counterstained with Mayer Haematoxylin, washed with tap water for 2 min, rinsed with distilled water, and mounted using glycerine gelatin. Finally, the sections were observed and captured under a microscope.
Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR) Analysis
The collected liver tissues were homogenized, and total RNA was extracted using Trizol Reagent (TransGen, Beijing, China) according to the manufacturer's protocol. The RNA concentration and purity were assessed using a spectrophotometer (ThermoFisher, NC, USA) and confirmed by 1% agarose gel electrophoresis. The total RNA was then reverse transcribed into complementary DNA (cDNA) using the RevertAid First Stand cDNA Synthesis Kit (ThermoFisher, Shanghai, China) following the manufacturer's instructions. Next, the relative expression levels of Tnf (encoding TNF-α), Il6 (encoding IL-6), Il1b (encoding IL-1β), Rela (encoding nuclear factor NF-κB p65 subunit, p65), and Nlrp3 (encoding NLR family pyrin domain containing 3, NLRP3) were measured relative to the Gapdh (encoding GAPDH) gene. The resulting Ct values were analyzed using the 2−ΔΔC method in Excel software. The sequences of the gene primers are presented in Table 1.
Sequences of Primers Used in the RT-qPCR Experiment.

Immunoblotting Analysis
Total proteins were extracted from the collected liver tissues using RIPA lysis buffer (Solarbio, Beijing). The concentrations of the extracted proteins were measured using a BCA kit (Beyond, Shanghai, China). The proteins were then denatured at 100°C for 10 min. Equivalent amounts of protein from each group were loaded onto a 12% sodium dodecyl sulfate-polyacrylamide gel and subjected to electrophoresis. Following electrophoresis, the proteins were transferred onto a polyvinylidene fluoride membrane. The membrane was then blocked with 5% skim milk in TBST blocking for 2 h. After blocking, the membranes were incubated with primary antibodies overnight at 4°C. The next day, the membranes were incubated with a secondary antibody (donkey anti-rabbit IgG antibody, Cat No: ab6701, Abcam, Shanghai, China) and treated with enhanced chemiluminescent reagents to visualize the protein bands. The primary antibodies used in this experiment included GAPDH (Cat No: 200306; ZEN-BIOSCIENCE, Chengdu, China) and Ki67 (Cat No: 16667; Abcam, Shanghai, China). The intensity of the protein bands was measured using ImageJ software.
Statistical Analysis
Data are expressed as the mean ± standard error. Statistical analyses were conducted using SPSS 26 (IBM Corporation, NY, USA). Specifically, continuous variables were first tested for normal distribution and variance homogeneity. For data meeting these assumptions, an independent samples analysis of variance (ANOVA) was conducted for preliminary hypothesis testing, followed by post-hoc analysis using the least significant difference (LSD) method. For data not meeting these criteria, the Kruskal-Wallis test was employed, with the Tukey method used for post-hoc comparisons. A p-value of less than 0.05 was considered statistically significant for all hypothesis tests. Graphs were generated using Origin 2024 Pro (Originlab, MA, USA), Adobe Photoshop 2022, and Adobe Illustrator 2022 (Adobe Systems Incorporated, CA, USA).
Results
Andrographolide Improved HFD-induced Elevations in Serum Concentrations of Glucose, ALT, and AST
Compared to mice in the Chow group, mice that consumed an HFD for 12 weeks showed significantly higher serum concentrations of glucose (0.3863 ± 0.006 and 0.1626 ± 0.002 μmol/mL; p < 0.001; Figure 2 A), ALT (103.50 ± 0.164 and 46.27 ± 0.767 pg/mL; p < 0.001; Figure 2 B), and AST (221.14 ± 0.252 and 89.04 ± 0.119 ng/L; p < 0.001; Figure 2 C). Treatment with 50 mg/kg and 200 mg/kg andrographolide significantly reduced the serum concentrations of glucose (0.3484 ± 0.005 and 0.2373 ± 0.007 μmol/mL; p < 0.001), ALT (94.45 ± 0.103 and 66.07 ± 0.080 pg/mL; p < 0.001), and AST (198.60 ± 0.305 and 160.57 ± 0.142 ng/L; p < 0.001) in HFD-fed mice (Figure 2). The effects of andrographolide on these indicators were dose-dependent (Figure 2).
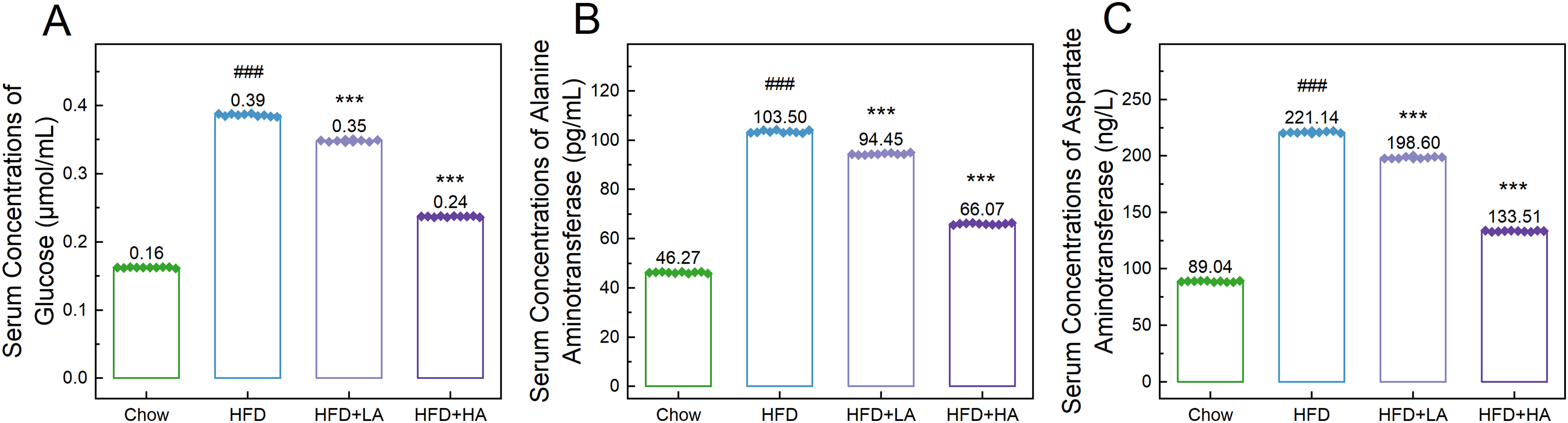
Andrographolide decreases the concentrations of detected serum makers. A-C: Bar charts with dots showing the serum concentrations of glucose (A), alanine aminotransferase (B), and aspartate aminotransferase (C) in the mice from each group (n = 10). Data are expressed as mean ± standard error, and one-way analysis of variance was employed for statistical analyses: ###, p < 0.001 compared to the Chow group; ***, p < 0.001 compared to the HFD group.
Andrographolide Mitigated HFD-induced Lipid Deposition and Inflammation Injury in Liver
The Red Oil O staining results showed minimal lipid accumulation with very few and small red-stained lipid droplets in the liver of mice from the Chow group (Figure 3 A). In contrast, severe lipid accumulation with numerous large red-stained lipid droplets was observed in mice from the HFD group (Figure 3 B). After andrographolide treatment, mice in the HFD + LA group exhibited moderate to severe lipid accumulation with many large red-stained lipid droplets in their livers (Figure 3 C), while those in the HFD + HA group showed moderate accumulation with several large red-stained lipid droplets in their livers (Figure 3 D).

Andrographolide alleviates high-fat diet-induced liver fat accumulation in mice. A-D: Microscopy images showing representative Oil Red O-stained sections of the liver from mice in the Chow (A), HFD (B), HFD + LA (C), and HFD + HA (D) groups.
According to the HE staining results, the livers of mice in the Chow group appeared normal with well-organized hepatocytes and no signs of steatosis, inflammation, or inflammatory cell infiltration (Figure 4 A). In contrast, significant lipid accumulation was observed within hepatocytes of the HFD group, evident as large white vacuoles displacing the cytoplasm and nuclei to the periphery, but there was no significant presence of inflammatory cells (Figure 4 B). After andrographolide treatment, mice in the HFD + LA group exhibited moderate to severe steatosis, with many hepatocytes containing large lipid vacuoles, and scattered inflammatory cells were present in their livers (Figure 4 C). Mice in the HFD + HA group showed moderate steatosis with numerous large and small lipid vacuoles within hepatocytes, along with a noticeable presence of inflammatory cells in their livers (Figure 4 D).

Andrographolide attenuates high-fat diet-induced liver inflammatory injuries in mice. A-D: Microscopy images showing representative Hematoxylin & Eosin-stained sections of the liver from mice in the Chow (A), HFD (B), HFD + LA (C), and HFD + HA (D) groups.
Andrographolide Mitigated HFD-induced Inflammation in Liver by Inhibiting NF-κB Signaling Pathway
RT-qPCR results showed that the relative expression levels of Il1β, Il6, and Tnf genes in the HFD group (6.26 ± 0.19, 5.13 ± 0.22, and 7.05 ± 0.18, respectively) were significantly higher than those in the Chow group (1.05 ± 0.04, 1.03 ± 0.06, and 0.98 ± 0.03, respectively; p < 0.001; Figure 5 A). After andrographolide treatment, mice in the HFD + LA and HFD + HA groups showed significantly lower relative expression levels of Il1β (3.87 ± 0.26 and 1.91 ± 0.05; p < 0.001), Il6 (2.96 ± 0.16 and 1.74 ± 0.05; p < 0.001), and Tnf (2.32 ± 0.19 and 1.35 ± 0.08; p < 0.001) genes compared to those in the HFD group (Figure 5 A).
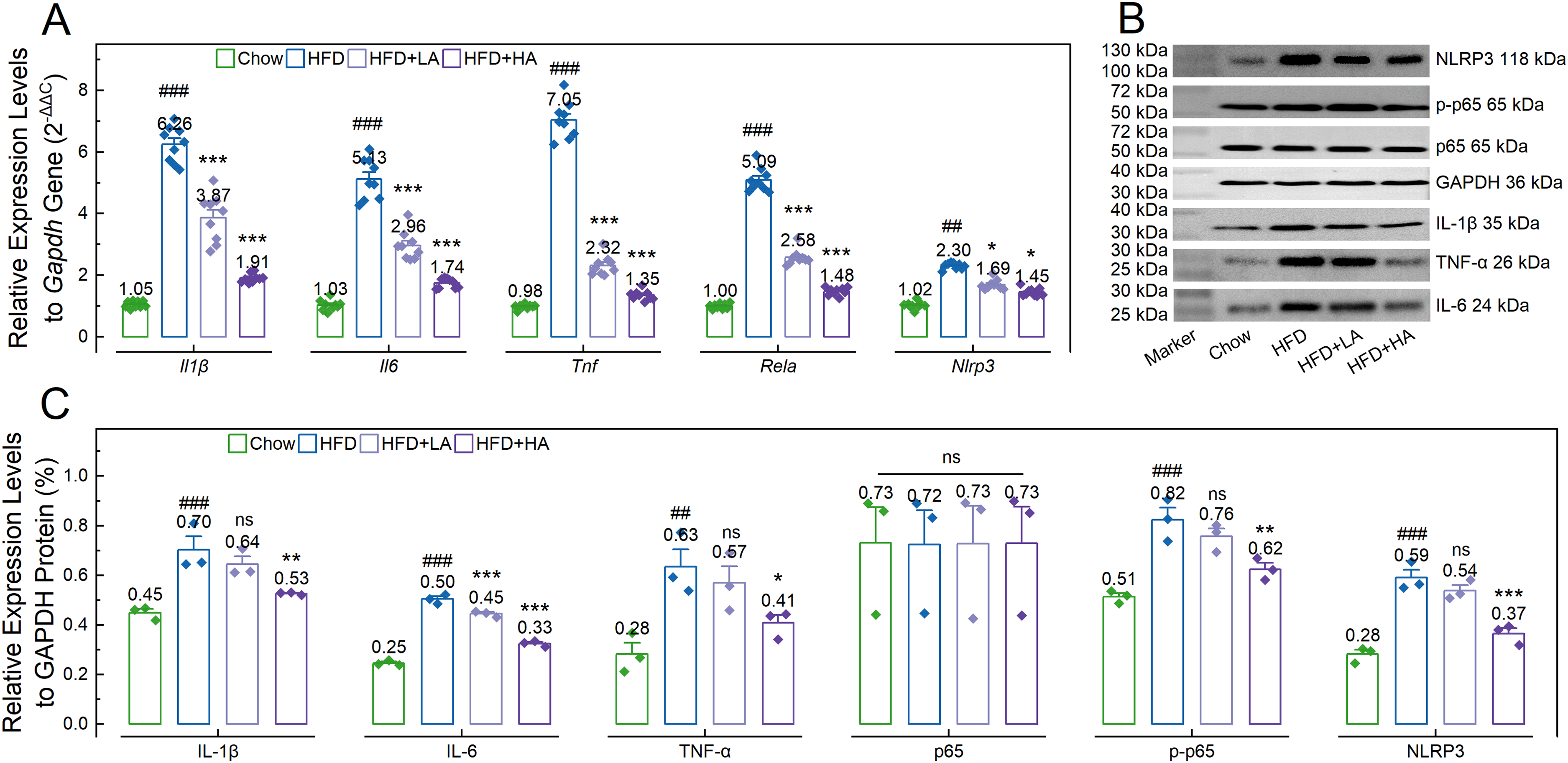
Andrographolide inhibits high-fat diet-induced up-regulation of the NF-κB signaling pathway in the livers of mice from each group. A: Bar charts with dots showing the relative expression levels of Il1β, Il6, Tnf, Rela, and Nlrp3 genes to the Gapdh gene (n = 9); B and C: Representative gel images (B) and bar charts with dots (C) showing the relative expression levels of tumor necrosis factor (TNF)-α, interleukin (IL)-1β, IL-6, nuclear factor NF-κB p65 subunit (p65), phosphorylated p65 (p-p65), and NLR family pyrin domain containing 3 (NLRP3) proteins to the housekeeping protein GADPH (n = 3). In A and C, data are expressed as mean ± standard error, and one-way analysis of variance was employed for statistical analyses: ##, p < 0.01; ###, p < 0.001 compared to the Chow group; ns, p > 0.05; *, p < 0.05; **, p < 0.01; ***, p < 0.001 compared to the HFD group.
Similarly, the relative expression levels of the IL-1β, IL-6, and TNF-α proteins in the HFD group (0.70 ± 0.05, 0.50 ± 0.01, and 0.63 ± 0.07, respectively) were also significantly higher than those in the Chow group (0.45 ± 0.02, 0.25 ± 0.01, and 0.28 ± 0.05, respectively; p ≤ 0.002; Figure 5 B and C). After andrographolide treatment, mice in the HFD + LA group showed similar relative expression levels of IL-1β (0.64 ± 0.03; p = 0.240) and TNF-α (0.57 ± 0.07; p = 0.439) proteins compared to those in the HFD group, while showing a significantly lower level of (0.45 ± 0.01; p < 0.001) IL-6 protein compared to those in the HFD group (Figure 5 B and C). Mice in the HFD + HA group showed significantly lower levels of these proteins (0.53 ± 0.01, 0.33 ± 0.01, and 0.41 ± 0.05, respectively; p ≤ 0.021) compared to those in the HFD group (Figure 5 B and C).
Additionally, RT-qPCR results showed that the relative expression levels of Rela and Nlrp3 genes in the Chow group (1.00 ± 0.03 and 0.98 ± 0.03) were significantly lower compared to those in the HFD group (5.09 ± 0.13 and 7.05 ± 0.20; p ≤ 0.007). The relative expression levels of the Rela and Nlrp3 genes in the HFD group were significantly higher than those in the HFD + LA (2.58 ± 0.08 and 2.32 ± 0.11; p ≤ 0.014) and HFD + HA (1.48 ± 0.04 and 1.35 ± 0.05; p ≤ 0.037) groups (Figure 5 A). At the protein level, although the relative expression level of p65 was similar among groups (p = 0.974), mice in the Chow group showed significantly lower relative expression levels of p-p65 and NLRP3 proteins (0.51 ± 0.01 and 0.28 ± 0.02) compared to those in the HFD groups (0.82 ± 0.05 and 0.59 ± 0.03; p < 0.001). These levels were similar to those in the HFD + LA group (0.76 ± 0.03 and 0.54 ± 0.02; p ≥ 0.183) but significantly higher than those in the HFD + HA group (0.62 ± 0.03 and 0.37 ± 0.03; p ≤ 0.003; Figure 5 B and C).
Discussion
NAFLD is a key link between several medical conditions, including obesity, diabetes, cardiovascular, and kidney disease, 26 and is becoming a major health concern affecting thousands of people globally. 27 Although there is no complete cure for NAFLD, recent studies have shown that many herbs or herbal ingredients have the potential to ease the progression of NAFLD.14,28 Andrographolide, an active ingredient extracted from Andrographis paniculate, has shown anti-inflammation and anti-fibrosis effects on the lungs 18 and exerts potential mitigative effects on NAFLD. Here, we explored whether andrographolide treated can mitigate HFD-induced NAFLD in a mice model.
HFD-induced NAFLD mice are a commonly used in vivo model for NAFLD-related studies,29,30 and alterations in hepatic metabolism have been observed in mouse models of obesity, diabetes, and fatty liver diseases. 31 Hence, in this study, to explore the hepatic protective effects of andrographolide, we developed a NAFLD in vivo model by feeding C57BL/6 mice an HFD consisting of 60% kcal from fat, 20% kcal from protein, and 20% kcal from carbohydrates for 12 weeks (Figure 1 A). After 12 weeks of HFD consumption, mice in the HFD group showed significantly higher levels of liver function indicators, including glucose, ALT, and AST, in the circulating system (Figure 2), indicating that the hepatocytes in their liver were damaged. 32 Additionally, severe lipid accumulation with numerous large red-stained lipid droplets was also observed in the liver of mice in the HFD group (Figure 3 and Figure 4), indicating HFD-induced lipotoxicity and cell damage. 33 Meanwhile, RT-qPCR and immunoblotting analyses showed significantly elevated expression of pro-inflammatory cytokines, including TNF-α, IL6, and IL-1β (Figure 5), indicating HFD-induced overproduction of pro-inflammatory cytokines and an inflammation response in the liver. 34 Collectively, these results suggest that we successfully developed NAFLD in mice.
To explore the potential effects of andrographolide on NAFLD, we treated the HFD-fed mice with low and high doses of andrographolide for 8 weeks (Figure 1 A). Mice in the HFD + LA and HFD + HA groups showed significantly lower levels of these 3 liver function indicators 32 in the circulating system compared to those in the HFD group (Figure 2). Although the concentrations of these indicators in the HFD + LA and HFD + HA groups were still significantly higher than those in the Chow group, it can be assumed that the HFD-induce liver damage was significantly mitigated by andrographolide treatment, and this effect was dose-dependent. Mice in the HFD + LA and HFD + HA groups also showed dose-dependent decreases in lipid accumulation and Oil Red O-stained lipid droplets in the liver sections (Figure 3 and Figure 4), indicating that HFD-induced lipotoxicity and cell damage were dose-dependently attenuated by andrographolide treatment. Similarly, Ran et al found that andrographolide was capable of ameliorating hepatic steatosis by suppressing FATP2-mediated fatty acid uptake in mice with NAFLD, explaining the reasons for decreased lipid accumulation and Oil Red O-stained lipid droplets in our study. 35 Meanwhile, 200 mg/kg of andrographolide treatment significantly down-regulated the expression levels of these 3 considered pro-inflammatory cytokines (Figure 5). Although the down-regulation of TNF-α and IL-1β were not significant in the mice from the HFD + LA group, it can be suggested that a high dose of andrographolide treatment could effectively mitigate HFD-induced overproduction of pro-inflammatory cytokines and inflammation responses in the liver. These results support the findings of Cabrera et al, who observed that andrographolide ameliorated inflammation and fibrogenesis and attenuated inflammasome activation in experimental non-alcoholic steatohepatitis. 36
Moreover, considering that andrographolide is a novel NF-κB inhibitor 37 and exerts pharmacological effects by regulating the NF-κB signaling pathway,37,38 we also evaluated the expression levels of p65, p-p65, and NLRP3 proteins, which are key checkpoints of the NF-κB signaling pathway. It was observed that, although the expression level of p65 was similar among groups, mice in the HFD group showed significantly elevated expression levels of p-p65 and NLRP3 (Figure 5), which was consistent with the findings of Huang et al., 39 indicating that the HFD-induced inflammatory responses were likely mediated by the activation of the NF-κB signaling pathway. Although the expression levels of p-p65 and NLRP3 in the HFD + LA group were not decreased, those in the HFD + HA group were significantly lower than those in the HFD group and similar to those in the Chow group (Figure 5), indicating that high levels of andrographolide treatment could effectively down-regulate the NF-κB signaling pathway. This might be one of the underlying mechanisms of andrographolide's NAFLD mitigative effects. Andrographolide has been found to enhance NF-κB subunit p65 Ser536 dephosphorylation through activation of protein phosphatase 2A in vascular smooth muscle cells. 21 However, whether this mechanism applies to hepatocytes needs to be further investigated. Besides the NF-κB signaling pathway, andrographolide has also been reported to modulate other inflammation-related signaling pathways, including PI3 K/AKT-eNOS, 40 JAK-STAT, 41 and HIF1α/VEGF 42 signaling pathways. Whether these signaling pathways are involved in the mitigative effects of andrographolide also warrants further investigation. Additionally, the expression conditions of other lipid-lowering drug target genes associated with NAFLD43–46 should also be examined in further studies.
However, there are some limitations in this study. Firstly, our results are observational and only demonstrated that the NF-κB signaling pathway is involved in the mitigative effects of andrographolide. Additionally, we used only a single mouse model. Therefore, further studies are needed, including in vitro research, experiments using other in vivo models, and eventually, human studies, to verify these mitigative effects and their mechanisms at both the gene and protein levels. Future research should aim to further elucidate the specific mechanisms by which andrographolide inhibits the NF-κB signaling pathway or other pathways. It should also explore the potential effects of combining andrographolide with other drugs. This research could provide theoretical support and new insights for developing novel drugs to treat NAFLD.
Conclusion
In summary, our findings collectively indicate that andrographolide treatment can dose-dependently alleviate liver function impairments, lipid accumulation, excessive production of pro-inflammatory cytokines, and inflammatory responses in the livers of mice induced by an HFD. The mitigation of these NAFLD effects by andrographolide may be linked to the down-regulation of the NF-κB signaling pathway. Our study elucidates the role of andrographolide in reducing HFD-induced inflammatory responses in hepatocytes, offering a potential alternative treatment for NAFLD. Further in vitro and in vivo studies are warranted to further explore their underlying mechanisms and potential applications in NAFLD treatment.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241302020 - Supplemental material for Andrographolide Inhibits the NF-κB Signaling Pathway and Alleviates High-Fat Diet-Induced Hepatic Inflammation in a Mouse Model of Nonalcoholic Fatty Liver Disease
Supplemental material, sj-docx-1-npx-10.1177_1934578X241302020 for Andrographolide Inhibits the NF-κB Signaling Pathway and Alleviates High-Fat Diet-Induced Hepatic Inflammation in a Mouse Model of Nonalcoholic Fatty Liver Disease by Weiwen Hu, Yilin Chen, Wenhong Tan, Yirong Wang, Die Huang and Hao Yuan in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X241302020 - Supplemental material for Andrographolide Inhibits the NF-κB Signaling Pathway and Alleviates High-Fat Diet-Induced Hepatic Inflammation in a Mouse Model of Nonalcoholic Fatty Liver Disease
Supplemental material, sj-docx-2-npx-10.1177_1934578X241302020 for Andrographolide Inhibits the NF-κB Signaling Pathway and Alleviates High-Fat Diet-Induced Hepatic Inflammation in a Mouse Model of Nonalcoholic Fatty Liver Disease by Weiwen Hu, Yilin Chen, Wenhong Tan, Yirong Wang, Die Huang and Hao Yuan in Natural Product Communications
Footnotes
Author's Contributions
Weiwen Hu and Yilin Chen designed and conducted the experiment; Weiwen Hu, Yilin Chen, Wenhong Tan, Yirong Wang, and Hao Yuan collected and analyzed the data; Weiwen Hu prepared all tables and figures and wrote the main text of the manuscript. All authors reviewed and approved the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Yunnan Provincial Science and Technology Department 2023 Science and Technology Plan Project in the Field of Biomedicine: Research and Development of Traditional Medicine Inheritance and Prescription Experiences of Six Ethnic Minorities Including the Lahu Ethnic Group in Yunnan, Xingdian Talent Support Program-Youth Talent Special Project, (grant number 202302AA310006, 166).
Ethical Approval
This study was approved by the Ethical and Welfare Committee for Animal Experiments of Yunnan University of Traditional Chinese Medicine, Kunming City, Yunnan Province, China (Approval No: DW-2024-002; Approval Date: 2024.1.3).
Statement of Human and Animal Rights
All procedures related to animals in this study were conducted in strict accordance with the Guidelines on the Care and Use of Laboratory Animals issued by the Chinese Council on Animal Research and the Guidelines of Animal Care, Yunnan Province, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
