Abstract
To investigate the hepatoprotective activities of a polysaccharide extracted from the fruit of Ribes odoratum Wendl. (ROWFP) in a mouse model of high-fat-sucrose diet (HFD)-induced nonalcoholic fatty liver disease (NAFLD). The NAFLD model was induced in C57BL/6 mice by feeding them an HFD for 12 weeks. The mice were randomly divided into the following 5 groups: control group, HFD group, 10-mg/kg ROWFP group, 100-mg/kg ROWFP group, and 200-mg/kg ROWFP group. The levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), total triglycerides (TG), total cholesterol (TC), and high-density lipoprotein (HDL) in the serum were analyzed by enzyme-linked immunosorbent assay. The liver ultrastructure was observed via optical microscopy. The oil red O-stained lipid droplets of the fresh liver samples were analyzed, and the lipid content was semiquantified. CD68 expression in the liver tissue and serum levels of the inflammatory factors (interleukin [IL]-1β, IL-6, and tumor necrosis factor-alpha [TNF-α]) were measured to reflect the inflammation status. The degree of liver fibrosis was determined by sirius red staining. When compared with the control group, the levels of AST, ALT, TG, TC, IL-1β, IL-6, TNF-α, and CD68 in the HFD group were increased, while the HDL level was decreased. Severe liver damage, lipid accumulation, and liver fibrosis were also observed in the HFD model group. When compared with the model group, ROWFP treatment (100 mg/kg or 200 mg/kg) significantly attenuated the HFD-induced hepatic damage. This study supports the hepatoprotective effect of ROWFP against HFD-induced NAFLD.
The liver is one of the most significant organs involved with clearing metabolites and exogenous compounds from the body. Therefore, liver disease seriously affects the quality of life for patients. Nonalcoholic fatty liver disease (NAFLD) has emerged as a common risk factor for chronic liver disease. 1,2 It is rapidly becoming a major health care problem worldwide and affects 15%-30% of the population in Asia. 3 Unfortunately, no effective treatment is available for NAFLD. 4 Thus, the development of methods for NAFLD prevention and/or treatment is an urgent need.
Although the underlying mechanism of NAFLD is not completely understood, an imbalance in lipid metabolism that causes hepatic lipid accumulation is considered to be a key factor in its initiation and development. 5,6 Studies have found that high-level dietary fat can lead to lipid accumulation in the liver, which is one of the main causes of NAFLD. 7,8 A high-fat diet (HFD) can induce obesity, diabetes, hyperlipidemia, and other metabolic syndromes that are usually associated with a fatty liver. 9 Studies also suggest that lipid accumulation induces oxidative stress and inflammation, which causes secondary damage to the liver and aggravates the nonalcoholic fatty liver. 10,11 Therefore, the development of drugs with hypolipidemic and anti-inflammatory properties might be an effective strategy for the treatment of NAFLD.
Ribes odoratum Wendl. is a deciduous shrub that is mainly used for decoration, 12 with no relevant pharmacological activity having yet been reported. However, in Xining, China, people have found that consuming the fruits of R. odoratum Wendl. reduces the levels of fat in circulation. The aim of this study is to evaluate the hepatoprotective activities of a polysaccharide extracted from the fruit of R. odoratum Wendl. (ROWFP) by utilizing a mouse model of HFD-induced NAFLD.
Experiment
Test Compounds, Chemicals, and Reagents
The fruit of R. odoratum Wendl. was provided by Qinghai Academy of Agriculture and Forestry (number: 20180716) and was identified by Professor Mingcheng Ma. The alanine aminotransferase (ALT) enzyme-linked immunosorbent assay (ELISA) kit, aspartate aminotransferase (AST) ELISA kit, total triglycerides (TG) ELISA kit, total cholesterol (TC) ELISA kit, high-density lipoprotein (HDL) ELISA kit, tumor necrosis factor-alpha (TNF–α) ELISA kit, interleukin (IL)-1β ELISA kit, and IL-6 ELISA kit were purchased from Tsz Biosciences (Greater Boston, USA). The oil Red O (Sudan III) was purchased from Sigma-Aldrich (St. Louis, MO, USA). Analytical grade chemicals and reagents were used.
ROWFP Preparation
The powder, produced after drying and grinding the pulp, was sieved in order to remove any seeds and then double-extracted with petroleum ether (boiling point, 60-90 °C) at room temperature for 24 hours upon continuous stirring. The defatted sample was then extracted with 80% ethanol at 60 °C for 2 hours to remove the monosaccharides, oligosaccharides, and other small molecules. The residue was extracted with distilled water (80 °C, 1:20 w/v) 3 times (3 hours for each). The filtrate was subsequently combined, concentrated, and then mixed with ethanol up to 80% at 4 °C overnight. The solution was centrifuged at 4000 rmp/min for 15 minutes, collected the precipitate, and dissolved in water (5% w/v). Next, deproteinization (Sevag method) and depigmentation (diethylaminoethyl cellulose chromatography) were performed, and the polysaccharide was obtained after freeze drying.
General Methods
Total carbohydrate content was determined by the phenol-sulfuric acid method using rhamnose, galacturonic acid, glucose, galactose, xylose, and arabinose as standard (according to the determination of monosaccharide composition in ROWFP). 13 Uronic acid content was determined by the m-hydroxydiphenyl colorimetric method using galacturonic acid as a standard. 14 Protein content was determined by the Coomassie brilliant blue method using bovine serum albumin as a standard. 15
The molecular weight (MW) distributions of the polysaccharide samples were determined using high-performance gel permeation chromatography (HPGPC) on a TSK-GEL G3000 PWXL column (7.8 mm × 300 mm, 10 µm), and the monosaccharide compositions of the polysaccharide samples were analyzed by high-performance liquid chromatography (HPLC) using a Bonshell ASB C18 column (100 × 4.6 mm i.d., 2.7 µm, 90 Å) with 1-phenyl-3-methyl-5-pyrazolone (PMP) precolumn derivatization. 16 The HPGPC and HPLC were carried out by using an LC-10ADvp HPLC pump (Shimadzu, Tokyo, Japan) equipped with a refractive index RID-10A detector and an SPD-10Avp ultraviolet-visible detector (Shimadzu, Tokyo, Japan).
Fourier-transform-infrared (FT-IR) spectra were obtained on a Thermo IS50 FT-IR spectrometer with a DTGS detector in the range of 400-4000 cm−1. The sample was measured as a film on a potassium bromide disc. Morphological analyses of the polysaccharide samples were conducted using a field emission scanning electron microscope (SU-8010, Hitachi, Japan).
Animals
Male C57BL/6 mice (6-8 weeks) were obtained from the Changzhou CAVENS Experimental Animal Co. Ltd. and housed individually in a ventilated unit under a 12‐hour light/dark cycle, with a temperature of 22 ± 2 °C and humidity level of 40%-60%. The animals were given free access to water and standard pelleted rodent food ad libitum. All of the animal studies were approved by the Institutional Animal Care and Use Committee of the Chinese Academy of Sciences, China, under the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All efforts were made to minimize the suffering of the animals.
NAFLD Model
The animals were allowed to adjust to the new environment for 1 week after their arrival. The NAFLD model was induced by feeding with the HFD. The high-fat formulation HFD is based on the literature, 17 the composition of the HFD was: 506.8 kcal/100 g; lard, 10 g/100 g; cholesterol, 2 g/100 g; egg yolk powder, 5 g/100 g; sucrose, 10 g/100 g; propylthiouracil, 2 g/100 g; basal diet, 72.8 g/100 g.
Experimental Groups
The animals were randomly divided into the following 5 groups (n = 8 mice/group): control group, HFD group, 10 mg/kg ROWFP group, 100 mg/kg ROWFP group, and 200 mg/kg ROWFP group. In the control group, the mice were fed a normal diet for 12 weeks, and 1 mL of distilled water was administered daily starting in the fifth week. In the HFD group, the mice were fed an HFD for 12 weeks, and 1 mL of distilled water was administered daily starting in the fifth week. In the ROWFP groups, the mice were fed an HFD for 12 weeks, and the administration of the ROWFP solution (10 mg/kg/day, 100 mg/kg/day, or 200 mg/kg/day with 1 mL of distilled water as solvent) was started at the fifth week. All of the drugs were administered via oral gavage.
Detection of Biochemical Markers
The animals were anesthetized using ether, and 1 mL of blood was collected through a cardiac puncture. The blood was allowed to clot and then centrifuged at 4000 g for 10 minutes. The serum was collected for the following assays. The serum levels of ALT, AST, TG, TC, HDL, TNF-α, IL-1β, and IL-6 were analyzed by ELISA (Tsz Biosciences, Greater Boston, USA).
Oil Red O Staining
The fresh liver tissue was cut into 8 µm thick sections with Cryostat (OCT, SAKURA, Japan). The sections were fixed in 4% paraformaldehyde for 30 minutes. Then, the slices were stained with 0.3% oil red O (ORO, Sigma Aldrich, USA) for 30 minutes at room temperature and washed with phosphate-buffered saline (PBS) 3 times. Finally, the liver sections were mounted onto slides and sealed with glycerin. The hepatic lipid accumulation was assessed using an Olympus-BX 53 microscope (Tokyo, Japan). Histological analysis of the droplets was semiquantified using the ImageJ software. Three regions were analyzed for each section.
General Histological Survey of the Livers
The C57BL/6 mice were killed by cervical dislocation, and their livers were excised, washed in a phosphate buffer, and absorbed with tissue paper. The hepatic tissue was fixed in 10% formalin and embedded in paraffin. The paraffin-embedded tissue was cut into 5 µm sections, placed on slides, deparaffinized in xylene, hydrated in decreasing concentrations of ethanol, and washed in water. Then, conventional hematoxylin and eosin (HE) staining was performed. After the HE staining, the sections were observed under a light microscope.
Sirius Red Staining
Liver fibrosis was evaluated by sirius red staining, as previously described (Sigma, 365548-5G). 18 The areas of portal tracts, centrilobular veins, and the surrounding regions were examined with the collagen contents. The histological analysis was assessed using an Olympus-BX53 light microscope (Tokyo, Japan). Three different regions per slice were captured and semiquantified (200×). The degree of liver fibrosis was determined by the following formula: (area of fibrosis/entire area) × 100%.
Liver Histopathological Examination
Pathological analysis and semiquantitative evaluation of the liver were performed. The liver tissue was briefly fixed in 4% paraformaldehyde for 4 hours, dehydrated in a series of ethanol-water solutions, and embedded in paraffin wax. Anti-CD68 (1:800, ab31630, Abcam, Cambridge, UK) was used for the detection of a CD68 protein. The histological analysis was performed using an Olympus-BX53 light microscope.
Statistical Analysis
The data are based on at least 6 independent experiments and presented as the mean ± SD. The analyses were performed with the statistical program for social sciences software (IBM SPSS, International Business Machines Corporation, Armonk City, NY, USA). The difference was determined using a one-way analysis of variance followed by a Bonferroni correction. In all of the cases, the differences were considered significant if P < 0.05.
Results
Analysis of ROWFP
The contents of total carbohydrate and uronic acid of ROWFP were determined to be 81.35% and 39.67%, and no protein was determined in it. HPGPC analysis showed that the ROWFP exhibited 2 main elution peaks, of which molecular weights were estimated to be 8059 Da and 4066 Da. The monosaccharide composition analysis (Figure 1) indicated that the ROWFP was mainly composed of rhamnose, galacturonic acid, glucose, galactose, xylose, and arabinose, with a molar ratio of 1.00:18.29:4.86:3.31:2.53:9.22.

High-performance liquid chromatography chromatograms of 1-phenyl-3-methyl-5-pyrazolone derivatization for (A) 9 monosaccharide standards (mannose, glucuronic acid, rhamnose, galacturonic acid, glucose, galactose, xylose, arabinose, and fucose) and (B) polysaccharide extracted from the fruit of Ribes odoratum Wendl.
FT-IR spectra of ROWFP (Figure 2) showed the absorptions at 3335 cm−1 caused by the O-H stretching vibration, 2924 cm−1 caused by the C-H stretching vibration, and 1738 cm−1 attributed to C=O stretching vibration of carboxyl groups. The absorptions at 1438 cm−1, 1328 cm−1 are corresponding to the various vibration from C-H, and those at 1074 cm−1, 1051 cm−1, and 1017 cm−1 are assigned to C-O stretching vibration, respectively. The spectrum revealed absorptions at 916 cm−1 and 830 cm−1, which indicated the presence of both β-glycosidic and α-glycosidic linkages. 16,19,20

Fourier transform-infrared spectra of the polysaccharide extracted from the fruit of Ribes odoratum Wendl.
The scanning electron micrographs of ROWFP were shown in Figure 3 at magnifications of 500, 2000, and 5000. The result indicates that ROWFP exhibits continuous lamellar and fibrous structure. The lamellar structures have some microspores (70-260 µm in diameter), and the diameter of fibrous structures is 3-10 μm. The surface of ROWFP adheres to some round and subround particles.
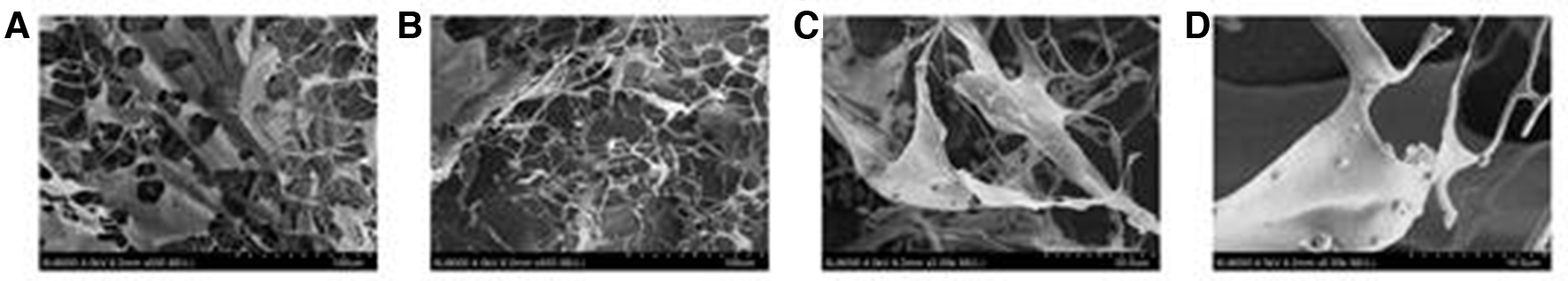
Scanning electron micrographs of the ROWFP. (A) Morphology of ROWFP at 500× (scalebar 100 µm). (B) Morphology of ROWFP at 500× (scalebar 100 µm). (C) Morphology of ROWFP at 2000× (scalebar 20 µm). (D) Morphology of ROWFP at 5000× (scalebar 10 µm). ROWFP, polysaccharide extracted from the fruit of Ribes odoratum Wendl.
ROWFP Reduced HFD-Induced Liver Damage in the C57BL/6 Mice
As shown in Figure 4(A) and (B) when compared with the control group, the serum levels of AST and ALT, which are markers of liver injury, were significantly increased in the HFD group (P < 0.01). When compared with the HFD group, 100 mg/kg and 200 mg/kg ROWFP treatment significantly reduced the AST and ALT levels. In contrast, the 10 mg/kg ROWFP treatment did not reduce the levels of AST and ALT. The HE staining results agreed with this finding and suggested reduced HFD-induced liver damage after the ROWFP treatment. As shown in Figure 4(C), the liver sections from the control group showed normal cell morphology, with well-preserved cytoplasm, a prominent nucleus, a central vein (CV), and a compact arrangement of hepatocytes. In contrast, the liver cells of the HFD group showed significant anomalies, with hydropic changes in centrilobular hepatocytes, and necrosis of the neutrophils being observed. Interestingly, the mice treated with the ROWFP (100 mg/kg and 200 mg/kg) exhibited attenuated HFD-induced liver damage, as evidenced by the presence of hepatic cords and the absence of inflammatory cells and necrosis. The 10 mg/kg ROWFP-treated group showed no protective effect on HFD-induced liver injury.

ROWFP reduces HFD-induced liver damage in C57BL/6 mice. ROWFP reduced the levels of ALT (A) and AST (B) in the C57BL/6 mice serum. (C) Different concentrations of ROWFP protected against HFD-induced histopathological damage (n = 6). ## P < 0.01 when compared with the control group, *P < 0.05, **P < 0.01 when compared with the HFD group. ALT, alanine aminotransferase; AST, aspartate aminotransferase; HFD, high-fat diet; ROWFP, polysaccharide extracted from the fruit of Ribes odoratum Wendl.
ROWFP Improved HFD-Induced Dyslipidemia in the C57BL/6 Mice
As shown in Figure 5(A)-(D), when compared with the control group, the TG and TC contents in the serum of the HFD group were increased, while the HDL level was decreased (P < 0.01). When compared with the HFD group, the 100 mg/kg and 200 mg/kg ROWFP treatment reduced TG and TC levels and increased HDL levels (P < 0.01). The results of ORO staining also showed that HFD induced severe lipid accumulation in the liver, which was attenuated by 100 mg/kg or 200 mg/kg ROWFP treatment. The 10 mg/kg ROWFP treatment did not protect against HFD-induced dyslipidemia.

ROWFP improves HFD-induced dyslipidemia in C57BL/6 mice. ROWFP reduced the levels of TG (A) and TC (B), and increased HDL levels (C) in the C57BL/6 mice serum. (D) The results of oil red O staining showed that the ROWFP treatment significantly improved HFD-induced lipid accumulation (n = 6). # P < 0.05, ## P < 0.01 when compared with the control group, *P < 0.05, **P < 0.01 when compared with the HFD group. HFD, high-fat diet; TC, total cholesterol; TG, triglycerides; ROWFP, polysaccharide extracted from the fruit of Ribes odoratum Wendl.
ROWFP Reduced HFD-Induced Inflammation
As shown in Figure 6(A)-(C), when compared with the control group, the HFD group showed increased serum levels of TNF-α, IL-1β, and IL-6 (P < 0.01). Treatment with 100 mg/kg or 200 mg/kg ROWFP reduced the levels of inflammatory cytokines (P < 0.01). The positive Kupffer cells were labeled by CD68 staining to determine the inflammation status. As shown in Figure 6(D), the liver of the HFD-fed mice showed an increase in the number of CD68 positive Kupffer cells while treatment with 100 mg/kg or 200 mg/kg ROWFP effectively inhibited this phenotype. The 10 mg/kg ROWFP treatment did not change HFD-induced inflammation.

ROWFP reduces HFD-induced inflammation. ROWFP reduced the levels of IL-1β (A), IL-6 (B), and TNF-α (C) in the C57BL/6 mice serum. (D) The results of CD68 staining showed that the ROWFP treatment significantly improved the HFD-induced increase in the number of CD68 positive marking Kupffer cells (n = 6). # P < 0.05, ## P < 0.01 when compared with the control group, *P < 0.05, **P < 0.01 when compared with the HFD group. HFD, high-fat diet; IL, interleukin; TNF-α, tumor necrosis factor-alpha; ROWFP, polysaccharide extracted from the fruit of Ribes odoratum Wendl.
ROWFP Reduced HFD-Induced Liver Fibrosis
As shown in Figure 7, there was no liver fibrosis in the control mice. The livers in the HFD group showed severe fibrosis. Treatment with 100 mg/kg or 200 mg/kg ROWFP significantly attenuated HFD-induced liver fibrosis, although the 10 mg/kg ROWFP treatment did not show a protective effect in HFD-induced liver fibrosis.

The results of sirius red staining showed that ROWFP reduces HFD-induced liver fibrosis. HFD, high-fat diet; ROWFP, polysaccharide extracted from the fruit of Ribes odoratum Wendl.
Discussion
In this study, for the first time, the protective effect of ROWFP against HFD-induced NAFLD was demonstrated. The severity of liver steatosis, inflammation, and fibrosis was attenuated by the ROWFP treatment.
NAFLD is a serious health problem that is related to hepatic lipid metabolic disorders. 21 Hyperlipidemia is considered the main cause of NAFLD, and excessive HFD is one of the main causes of NAFLD. 22,23 An excessive HFD increases blood lipid levels (such as TG and TC), and excess lipids can accumulate in the liver and cause liver damage. When the lipids in the liver are not relieved for a long period of time, liver damage will gradually increase and eventually develop into liver fibrosis. 24,25 In the present study, we found that an HFD caused a lipid metabolism disorder in mice, significantly increased the serum TG and TG levels, and significantly reduced HDL levels. At the same time, we also found that HFD induced liver damage in mice and significantly increased the serum levels of AST and ALT and that liver fibrosis appeared in the mice after 12 weeks of eating an HFD. Interestingly, the administration of ROWFP reduced HFD-induced lipid disorders, lipid accumulation, liver injury, and liver fibrosis. All of these results suggest the protective effects of ROWFP in mice livers against HFD-induced NAFLD.
Inflammation, which plays an important role in many pathological conditions, is associated with enhanced expression of adhesive molecules in the vasculature, which results in infiltration of large populations of neutrophils and monocytes/macrophages. 26,27 Studies have found that under NAFLD conditions, the liver develops inflammation and releases a large number of inflammatory cytokines (eg, TNF-α, IL-1β, and IL-6), further aggravating the liver damage. 28,29 In the present study, we did find a high level of inflammatory cytokines (TNF-α, IL-1β, and IL-6) in the serum of the HFD group, which was accompanied by increased CD68 staining in the liver tissue. All of these results support the idea that inflammation occurs in NAFLD. Remarkably, the administration of ROWFP significantly reduced the expression of inflammatory factors and CD68. Taken together, our results indicate that ROWFP protects against HFD-induced liver damage, and the mechanism might be via its anti-inflammatory properties.
Conclusion
In conclusion, ROWFP exerts a hepatoprotective effect in HFD-induced NAFLD in the C57BL/6 mice, and the protective effect might be through its lipid-lowering and anti-inflammatory properties.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (31870333), Qinghai Province High-end Innovative Thousand Talents Program, Youth Innovation Promotion Association, CAS, Qinghai Provincial Science Foundation (2019-ZJ-7023, 2017-SF-A8), Innovation Platform for the Development and Construction of Special Project of Key Laboratory of Tibetan Medicine Research of Qinghai Province (2017-ZJ-Y11), and Qinghai Province International Cooperation Project (2018-HZ-812).
