Abstract
Nonalcoholic fatty liver disease (NAFLD) is paralleling the insulin resistance and obesity epidemic and is regarded as liver metabolic syndrome, and its prevalence rate is increasing rapidly. The best explanation for the occurrence and development of NAFLD is the “multiple hit” hypothesis instead of the “two-hit” hypothesis. At present, NAFLD therapies are limited. The nuclear factor erythroid 2-related factor 2 (Nrf2) signaling pathway is a key pathway in oxidative stress. Its downstream proteins/enzymes are regulated. Metabolic enzymes and antioxidant proteins/enzymes play a vital role in cell defense protection and have attracted attention in the field of antioxidant research in recent years. This paper summarizes the regulatory mechanism of the Nrf2 signaling pathway and the research progress of Nrf2 activators in NAFLD to provide guidance for NAFLD therapy in the future.
Nonalcoholic fatty liver disease (NAFLD) is a complicated chronic liver disease worldwide that is characterized by excessive fat accumulation in the liver. 1 The well-known explanation for the occurrence and development of NAFLD is the “multiple hit” hypothesis. Such hits include hormones excreted from adipose tissue, insulin resistance, nutritional factors, gut microbiota, and genetic and epigenetic factors. In the general population, there has been a growing amount of evidence indicating the high prevalence of NAFLD in the past 20 years. A recent meta-analysis predicted that the global prevalence of NAFLD had reached 25.2%, and in the Middle East and South America, it was 31.8% and 30.4%, respectively. In addition, the prevalence of NAFLD in Europe and Asia was verified to be 23.7% and 27.4%, respectively. 2 Therefore, the health problem of NAFLD has aroused wide public concern. Unfortunately, there are no effective treatments in the management of NAFLD despite the significant advances that have been made in illuminating the pathophysiology of NAFLD. Currently, lifestyle changes such as diet and exercise remain the primary treatment of NAFLD, which are mainly dependent on patient compliance. 3 Because of the increasing NAFLD prevalence and the lack of effective treatment, it is urgently necessary to explore effective and safe treatments for patients with NAFLD.
Nuclear factor erythroid-derived 2-like 2 (Nrf2) is a transcription factor that contains a highly conserved basic region-leucine zipper (bZIP) structure. Nrf2 is a member of the Cap “n” Collar (CNC) family and plays a vital cytoprotective role in the activation of cellular antioxidant enzymes. 4,5 Furthermore, Nrf2 has also been shown to play a role in the resistance to oxidation and the regulation of lipid metabolism in models of NAFLD and remains a potential strategy in the prevention and therapy of NAFLD. 6 Therefore, researchers have paid much attention to identifying Nrf2 activators, which may be used as protective agents for NAFLD.
Relationship Between NAFLD and the Nrf2 Pathway
NAFLD is a multifactorial chronic disease with a complex pathogenesis in which oxidative stress plays a key role. 6 The pathologic spectrum of NAFLD ranges from steatosis to steatohepatitis, liver fibrosis, liver cirrhosis, and even hepatocellular carcinoma, and the common characteristic of the disease is the accumulation of lipid droplets in liver cells. 7 Lipid storage in the liver is limited, but in patients with NAFLD, the storage capacity is surpassed, leading to hepatocyte dysfunction and death. Excessive oxidation of fatty acids promotes reactive oxygen species (ROS) generation in hepatic mitochondria, which become overburdened, leading to oxidative stress in the liver. 8 Oxidative stress damages lipids, proteins, and DNA by activating proinflammatory cells such as Kupffer cells and stimulating the release of inflammatory cytokines. Furthermore, overloaded ROS may directly deplete the antioxidant capacity in the liver by reducing glutathione (GSH) and restraining the expression and activity of downstream antioxidant enzymes. 9,10 Finally, chronic inflammation and oxidative stress promote liver diseases, such as liver fibrosis, cirrhosis, and even hepatoma. In conclusion, reducing oxidative stress is expected to improve the progression of NAFLD.
The Kelch-like-ECH-associated protein 1 (Keap1)-Nrf2-antioxidative response element (ARE) signaling pathway is regarded as a vital antioxidative mechanism because of its ability to improve the oxidative stress response induced by internal or external oxidation and chemicals. 11 The Nrf2 gene contains 6 highly conserved epichlorohydrin (EHC) domains (Nrf2-EHC homology, Neh), named Neh1-6 (Figure 1). 12 The C-terminus of Neh1 contains the bZIP DNA region, which binds to the musculoaponeurotic fibrosarcoma protein (Maf) to form a heterodimer that binds to DNA and recognizes ARE. Neh2 contains 2 important conserved regions, DLG and ETGE, which bind to KEAP1 and then anchor Nrf2 to the cytoplasm. 13 Neh3, which is located at the C-terminus and binds with a chromo-ATPase/helicase DNA-binding protein (CHD6), is closely related to ARE transcriptional activity. 14 Neh4 and Neh5 act as transcriptional activators interacting with the cyclic adenosine monophosphate response element (CREB)-binding protein (CBP) and initiate transcription. 15 The Neh6 region is rich in serine and is a regulatory region for Nrf2 degradation independent of KEAP1. 16
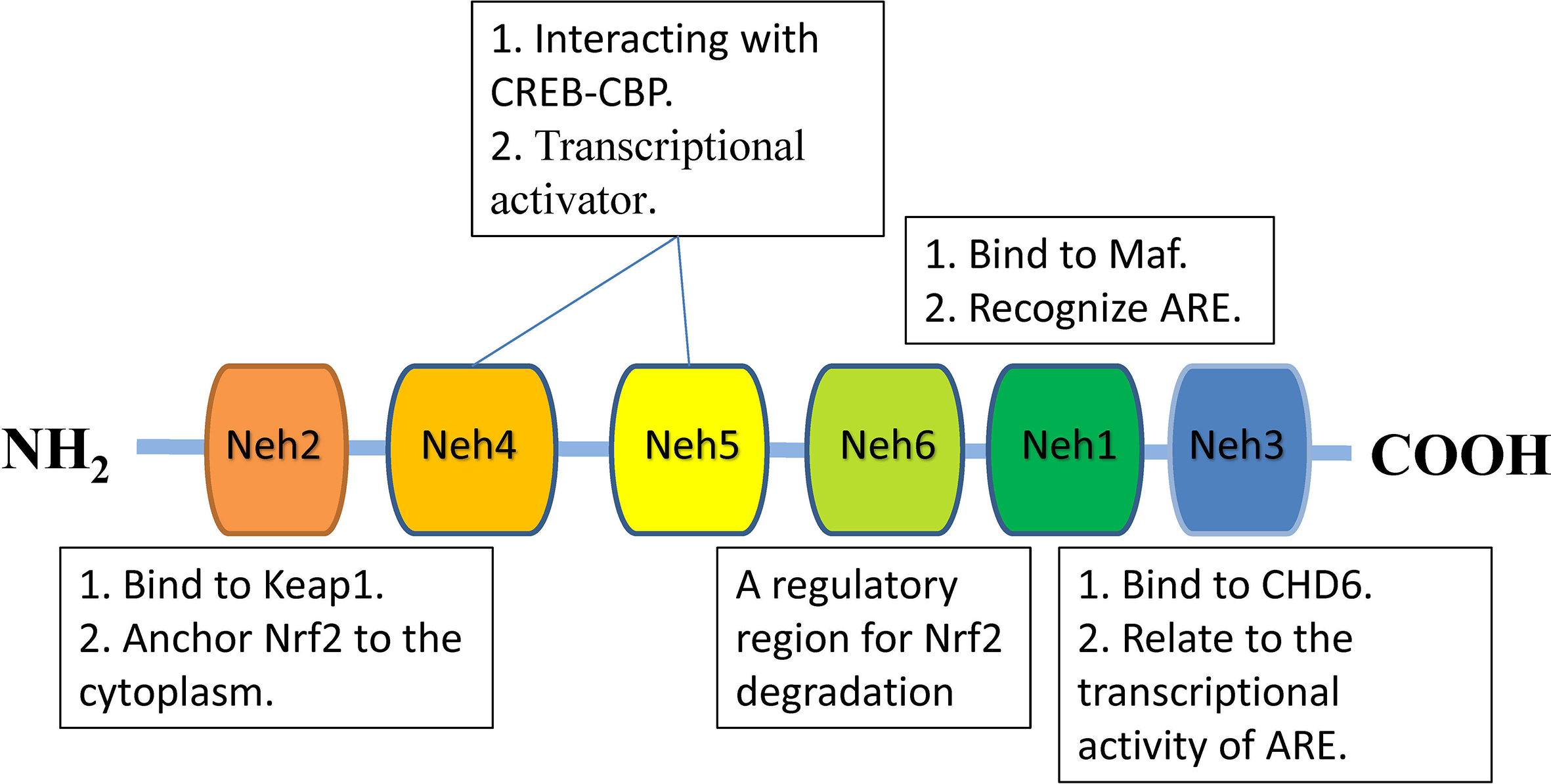
The structure of Nrf2. ARE, antioxidative response element; CBP, CREB-binding protein; CREB, cyclicadenosine monophosphate response element; Keap1, Kelch-like-ECH-associated protein 1; Maf, musculoaponeurotic fibrosarcoma; Nrf2, nuclearfactor erythroid-derived 2-like 2.
The mechanism of action for Nrf2 has been reviewed in detail at present. 17 Briefly, during homeostatic conditions, Nrf2 is also expressed but is located in the cytoplasm in combination with Keap1, which mediates the ubiquitinylation and degradation of Nrf2 (Figure 2). Upon exposure to oxidative or electrophilic stress, cysteine residues in Keap1 are modified 18 and stimulate the release of Nrf2. The Nrf2 protein, which dissociates from Keap1, relocates to the nucleus and dimerizes with bZIP proteins such as Maf 19 to bind with AREs and then promotes the expression of ARE-mediated downstream target genes, including hundreds of antioxidant and cytoprotective enzymes. NAD(P)H quinone oxidoreductase 1 (NQO1), heme oxygenase 1 (HO-1), and GSH are the most important antioxidant proteins among many downstream target proteins of Nrf2.

The regulation of the Nrf2/ARE signaling pathway. (a) Under homeostatic conditions, the Keap1 dimer binds with Nrf2 in the cytoplasm, leading to ubiquitination and then degradation of Nrf2. (b) Under oxidative or electrophilic stress, Keap1 is modified, and the capacity to bind Nrf2 is weaker. Nrf2 dissociates and transfers to the nucleus and binds with Maf to recognize AREs and stimulates the transcription of a series of class II metabolic enzymes, antioxidant proteins/enzymes, proteasome/molecular chaperones, anti-inflammatory factors, and phase III metabolic enzymes (drug transporters). ARE, antioxidative response element; Keap1, Kelch-like-ECH-associated protein 1; Maf, musculoaponeurotic fibrosarcoma; Nrf2, nuclearfactor erythroid-derived 2-like 2.
Nrf2 is pivotal for maintaining cellular redox homeostasis, providing protection against oxidative or electrophilic stresses, and suppressing NAFLD promotion and progression. In Nrf2+/+ and Nrf2−/− mice fed a high-fat diet (HFD), livers from Nrf2−/− mice had a conspicuous increase in steatosis and suffered more oxidative stress than Nrf2+/+ mice as assessed by a decrease in GSH and growing fatty acid accumulation compared with Nrf2+/+ mice. Insulin resistance could be reversed in Nrf2+/+ group mice treated with TBE-31, which is a new drug for NAFLD treatment, but not in Nrf2−/− mice. 20 -22 Additionally, spontaneous dysfunction of the intestinal barrier and promotion of the inflammatory response to lipopolysaccharide (LPS) stimulated the development of nonalcoholic steatohepatitis (NASH) in Nrf2−/− mice. 23 Bach1, a repressor of Nrf2, was upregulated because of hepatocyte-specific Sirt6 and was found to correlate with an increased risk of NAFLD. 24 Thus, Nrf2 has attracted attention as an effective target for NAFLD. Many Nrf2 activators have also been found and are used to stimulate Nrf2 expression to improve the cellular antioxidant ability against diseases. 25
Chemical Activators of Nrf2 in NAFLD
Flavonoids
Flavonoids are a class of bioactive antioxidants found in various fruits, vegetables, and plants. 26 Studies have shown their anti-inflammatory, antioxidant, and antitumor properties. Furthermore, flavonoids have also exhibited therapeutic properties in NAFLD by enhancing cellular antioxidant protection. 27,28 Meanwhile, many flavonoids have been proven to be Nrf2-ARE activators (Table 1).
Chemical Activators of the Nrf2 Signaling Pathway in NAFLD.

Abbreviations: ↓:decrease; ↑:increase;AKT, protein kinase B; ALT, alanine transaminase; AMPK, adenosine monophosphate-activated protein kinase; AST, aspartate transaminase; CAT, catalase; CPT: triterpenic acids-enriched fractions from Cyclocarya paliurus;GPx, glutathione peroxidase; GSH, glutathione; GSSG, glutathione disulphide; HO-1, heme oxygenase 1; JNK, c-Jun N-terminal kinase; Keap1, Kelch-like-ECH-associated protein 1 ; LFP: polyphenol-rich loquat fruit extract;lncRNA, long noncoding RNA; MDA, malondialdehyde; mRNA, messenger RNA; NAFLD, nonalcoholic fatty liver disease; NF-κB, nuclear factor kappa B; NQO1, NAD(P)H quinone oxidoreductase 1; Nrf2, nuclear factor erythroid-derived 2-like 2; PE: punicalagin enriched pomegranate extract; PI3K, phosphatidylinositol 3‑kinase; PPAR, peroxisome proliferator-activated receptor; SOD, superoxide dismutase; TC, total cholestrol; TG, triglycerides.
7-Mono-O-(β-hydroxyethyl)-rutoside (MonoHER) has been reported to activate the antioxidant defense system of Nrf2 by improving the ratio of GSH/glutathione disulfide (GSSG) and increasing the expression levels of the HO-1 and glutathione peroxidase 3 (GPx3) genes in Ldlr−/− mice. 27 Scutellarin (4′,5,6-trihydroxy flavonoid-7-glucuronide) increased the level of Nrf2 protein in C57BL/6J mice fed an HFD. Total cholesterol (TC), triglyceride (TG), and low-density lipoprotein cholesterol in serum were reduced in groups that were fed scutellarin. The authors proved that scutellarin could activate the peroxisome proliferator-activated receptor-gamma (PPARγ)-PGC-1a-Nrf2 pathway and accelerate the expression of HO-1, glutathione S transferase (GST), and NQO1, while it significantly inhibited NF-κB and Keap1 at the mRNA and protein levels. 45 Similarly, Fan et al reported that scutellarin prevented NAFLD based on activation with Nrf2, which increased the expression of HO-1 and NQO1, and the enhancing effect is possibly related to the phosphatidylinositol 3‑kinase/protein kinase B pathway. 46 Apigenin (4′,5,7-trihydroxyflavone), a flavonoid present in many fruits and leafy vegetables, can protect against HFD-induced hepatic steatosis in rats 49 and inhibit lipid peroxidation, thereby exerting protective effects against hepatic steatosis. 50 In a study, Feng et al found that apigenin promoted Nrf2 translocation into nuclei; increased the activities of antioxidative enzymes, such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px) in vivo; attenuated the HFD-induced increase in TG; and modified hepatic pathological changes (steatosis, lipid droplets) in rats. Further mechanistic research verified that the role of apigenin in activating PPARγ was inhibited by binding with Nrf2. 30 Kumar et al found that 7,8-dihydroxyflavone had activity against alcohol-induced and HFD-induced liver toxicity via upregulation of Nrf2. 51 According to the above results, flavonoids are effective in the treatment of NAFLD in animal models. However, the application of flavonoids in the clinical therapy of NAFLD needs more research. In particular, the potential involvement of general cytotoxicity toward normal cells should be fully excluded.
Curcumin
Curcumin (1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione), extracted from the dry rhizome of Curcuma longa, exerts hepatoprotective effects by suppressing lipid synthesis. 34 Many studies have been reported that clarify the roles of Nrf2 activation and liver protection from curcumin against NAFLD. 34,35 Yan et al verified that curcumin could inhibit SREBP-1c and induce the expression of CYP3A and CYP7A1 and simultaneously stimulate the expression of Nrf2. The study revealed that curcumin modulated the Nrf2/FXR pathway and improved NAFLD. 34 In 1 study, the authors observed that curcumin could inhibit HFD-induced elevation of serum aminotransferases and lipids, decrease tumor necrosis factor-alpha, interleukin-6, and malondialdehyde (MDA) levels in serum and liver, and increase GSH, HO-1, and SOD levels in rats. The in vivo study also showed that curcumin treatment increased Nrf2 protein in liver cell nuclei in mouse livers. Hence, curcumin appeared to exert its effects, at least in part, by activating the Nrf2 signaling pathway. 35 Overall, curcumin is effective in animals with NAFLD. However, its specificity for Nrf2 activity needs to be further verified.
Triterpenic Acid-Enriched Fractions From Cyclocarya paliurus
Cyclocarya paliurus (CP) is a Chinese herb whose leaves have antioxidant, hypoglycemic, and hypolipidemic activities. 52 -55 Previous research has revealed that CP can improve NAFLD by increasing the impact of SOD and GSH-Px and decreasing MDA levels in rats. 56 Furthermore, the triterpenic acid-enriched fraction from CP (CPT) could also reduce the content of hepatic lipid peroxide in NAFLD rats. 33,57 In 1 study, the authors designed a study to illuminate the function and mechanism of CPT against NAFLD in vivo and in vitro. The study found that CPT could inhibit HFD-induced activities of alanine transaminase (ALT), aspartate transaminase (AST), and alkaline phosphatase (ALP) in serum; decrease TG, TC, and free fatty acid levels; increase SOD expression and the GSH/GSSG ratio; and reduce MDA levels in vivo. An in vitro study also showed that CPT increased the protein and mRNA expression of Nrf2, NQO1, and HO-1 in HepG2 cells, which could be blocked by the Nrf2 inhibitor brusatol. 33 Clearly, the mechanistic details of CPT actions in the Nrf2 pathway deserve further research.
Gastrodin
Gastrodin (GSTD), a water-soluble extract of the root of Gastrodia elata BI., is regarded as a treatment for nervous system diseases such as vertebral basilar artery insufficiency, vertigo, and epilepsy in the clinic in China. 58,59 The increase in SOD activity and decrease in ROS production and the levels of proinflammatory cytokines by using GSTD in neurons and brain tissues under pathological conditions proved the antioxidative activity of GSTD, and the activity was connected with the phenomenon of the Nrf2 pathway. 60 -64 Qu et al reported that GSTD had antioxidative activity and the ability to improve lipid metabolism in both oleic acid-induced HL-7702 cells and HFD-induced NAFLD mice by activating adenosine monophosphate-activated protein kinase and promoting the nuclear distribution of Nrf2. 36 The toxicity of GSTD deserves exploration in the future.
NK-252
NK-252 (12.5-100 μmol/L) is a well-known Nrf2 activator.
41
It attenuated choline-deficient
Clusterin
Clusterin, a multifunctional glycoprotein isolated from ram rete testis fluid, is upregulated and plays vital role in liver diseases. 65,66 A study by Park et al suggested that clusterin could improve liver steatosis and hepatitis induced by a methionine and choline-deficient diet by increasing Nrf2 protein and HO-1 mRNA in mice. 32 Meanwhile, the immune reactivity for NF-κB and F4/80 was lower in the livers of hepatocyte-specific clusterin-over expressing mice than in those of wild-type mice.
Osteocalcin
Previous research has proven that there is a negative correlation between serum osteocalcin levels and the concentrations of ALT and AST. 67 Osteocalcin treatment improved mouse hepatic TG accumulation and liver injury. The levels of MDA and the ratio of oxidized glutathione in the liver induced by HFD were significantly protected due to the treatment of osteocalcin. Research on the mechanism demonstrated that osteocalcin intervention not only promoted Nrf2 nuclear translocation but also inhibited phosphorylation of the c-Jun N-terminal kinase pathway in vivo. 42
Other Activators
A large number of other compounds/extracts, including black sesame seed extract, 68 green tea extract, 37 lemon balm extract ALS-L1023, 39 punicalagin-enriched pomegranate extract, 43 silybin, 47 polyphenol-rich loquat fruit extract, 40 berberine 31 , and chitosan oligosaccharide, 69 have been demonstrated to attenuate inflammation, steatosis, and liver fibrosis in various NAFLD models, which might be related to activating the Nrf2 antioxidant system (Table 1; Figure 3).
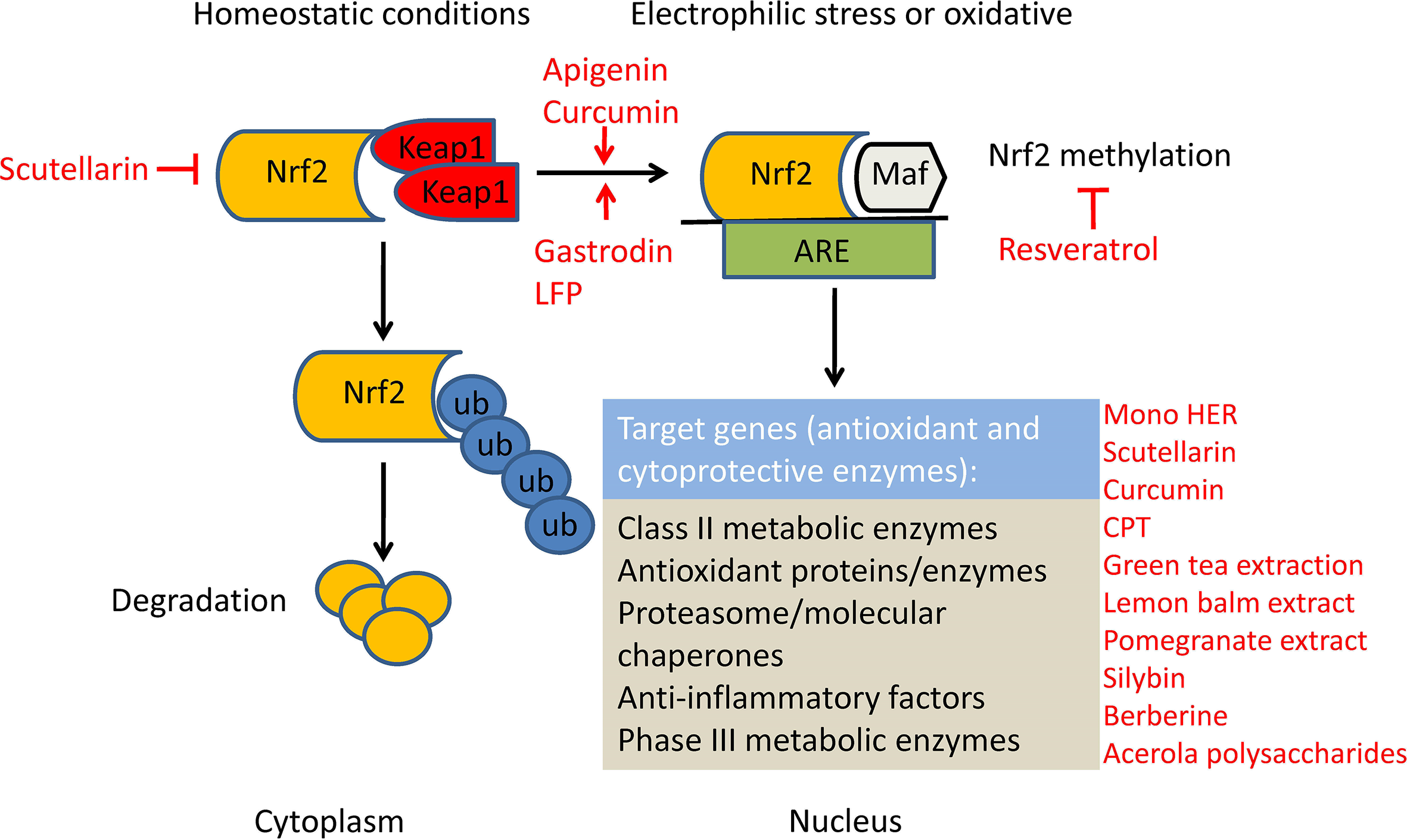
Natural compounds activating Nrf2 in NAFLD. ARE, antioxidativeresponse element; CPT, triterpenic acids-enriched fractions from Cyclocarya paliurus; Keap1, Kelchlike-ECH-associated protein 1; LFP, polyphenol-rich loquat fruit extract; Maf,musculoaponeurotic fibrosarcoma; MonoHER, 7-mono-O-(β-hydroxyethyl)-rutoside; NAFLD, nonalcoholicfatty liver disease; Nrf2, nuclearfactor erythroid-derived 2-like 2.
Conclusions
The contemporary pathophysiological model of NAFLD consists of parallel pathways with dynamic crosstalk that culminate in steatosis and inflammation and ultimately fibrosis, cirrhosis, and hepatocellular carcinoma. The accumulation of hepatocellular lipids is increased in obesity. Increased metabolism of hepatocellular lipid content in obesity induces the development of NAFLD. The detrimental effect of excess accumulation of lipids and their derivatives is related to NAFLD. The Nrf2-related antioxidant system has been verified to play key roles in promoting oxidative stress in NAFLD. Nrf2 activation could effectively ameliorate NAFLD-induced inflammation, serum and intrahepatic lipid levels, and pathological changes in the liver in vitro and in vivo, which indicates that Nrf2 is a promising target for NAFLD therapy. However, there are some difficulties that should be solved in future research.
First, current studies use animal or cell models of NAFLD to research the antioxidant stress role of Nrf2 activators. These data indicate that Nrf2 activators could improve NAFLD. However, it remains to be explored whether Nrf2 activators can prevent advanced stages of NAFLD, such as liver fibrosis and liver cirrhosis. It is also of great significance to use primate models to explore the roles of Nrf2 activators. Second, the authors did not exclude mechanisms other than the Nrf2 pathway. Third, the side effects of Nrf2 activators are not well researched. The poor bioavailability of some Nrf2 activators also obstructs their application in clinical NAFLD therapy. The most important point is that Nrf2 activators can promote the development of tumors and chemotherapy resistance. This relationship with cancer and the difficulties with Nrf2 activator use need to be explained by further research.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Zhejiang Provincial Natural Science Foundation-Zhejiang Pharmaceutical Society Joint Fund (No. LYY18H280004, LY17H290007) and the Special Research Project on Hospital Pharmacy of the Zhejiang Pharmaceutical Association (2018ZYY24). This project was supported by the National Natural Science Foundation of China (Grant No. 82074186). ”Ten thousand plan”- high level talents special support plan of Zhejiang province, China (Grant No. ZJWR0108035).
