Introduction
The Zingiberaceae family, the largest family in the order Zingiberales, is a monocotyledon-bearing aromatic and medicinal family with roughly 52 genera and 1300 species that may be found in the tropics from Malaysia to India.1,2 The native Zingiberaceae species were important traditional medicinal plants as well as providing sources of spices and food for ethnic groups in many countries around the world, such as China, India, Thailand, Laos, and Vietnam.2,3 Several species of the Zingiberaceae family (eg, Alpinia galanga, Curcuma longa, Zingiber officinale, Hedychium coronarium, Kaempferia galanga, etc) were well-known for their effects in treating various diseases, such as digestive disorders, hypertension, diabetes mellitus (DM), infection, inflammation, antioxidant, anti-microbial, anti-cancer, antiparasitic, analgesic, hepatoprotective activities, etc, as reported in previous studies.3,4 In terms of chemical composition profiles, some species (eg, A. galanga, C. longa, Curcuma thorelii, Curcuma rhabdota, Curcuma petiolata, Distichochlamys citrea, Z. officinale, H. coronarium, K. galanga, etc) were previously reported to be rich in essential oils. Moreover, various chemical components, such as flavonoids, tannins, resins, phenols, volatile fatty acids, etc, were isolated from their different parts.4–7 Essential oils (EOs) are volatile substances with low molecular weight and various biological activities that have been known.
8
As a source of natural chemicals for treating illnesses including diabetes, anti-cancer, hepatoprotective, antioxidant, and antimicrobial activities,
4
EOs are currently gaining popularity as a natural substitute in the health industry. EOs are therefore extremely important in the pharmaceutical, food, agricultural, cosmetic, and health industries.9,10
In the process of finding new medicinal sources from the Zingiberaceae family, two aromatic species named Wurfbainia schmidtii (K.Schum.) Škorničk. & A.D.Poulsen and Zingiber atroporphyreus Škorničk. & Q.B. Nguyên were studied. The genus Wurfbainia Giseke is widely distributed from the Himalayas to South China and Western and Central Malesia,
11
especially among them, W. schmidtii is the native range of Indo-China, that is, Cambodia, Laos, Thailand, and Vietnam. It is a rhizomatous geophyte and grows primarily in the wet tropical biome.
12
Meanwhile, the genus Zingiber Mill is native to tropical and subtropical Asia and includes 208 accepted species.
12
In 2015, Z. atroporphyreus, a new Zingiber species from Vietnam, was reported by Leong-Skornickova and colleagues.
13
In the literature, all species in these two genera are utilized extensively for medicine, food, and ornamental purposes. Specifically, W. schmidtii was used for tonic, carminative, and stomachic properties and for treating the symptoms of dizziness and digestive disorders. Moreover, it is also used for herbal soap and spas due to its very attractive aromatic smell. Additionally, the young leaves were used for food, as previously documented.3,11 However, until now, there have been no studies that have noted the chemical composition or pharmacological effects of Z. atroporphyreus.
Currently, DM, a chronic disease condition characterized by unusually high blood glucose levels, is brought on by an endocrine metabolic imbalance in the body.
14
It is expected that by 2045, the International Diabetes Federation (IDF) will have estimated globally about 12.2% (783.2 million) of the 20-79 age group with diabetes, and especially, global diabetes-related health spending is estimated at $1.054 billion. Additionally, IDF estimates that diabetes prevalence is expected to increase in middle-income, high-income, and low-income countries by 21.1%, 12.2%, and 11.9%, respectively.
15
In traditional medical systems, numerous varieties of herbs have been used to treat diabetes for a very long period. These herbs are thought to have a high economic benefit and low adverse effects when used to treat diabetes. Therefore, natural medicinal sources that support and treat diabetes are increasingly receiving attention from scientists. Among herbal sources, the members of Zingiberaceae family are also used to support and treat diabetes, such as Aframomum melegueta, C. longa, D. citrea (Black Ginger), and Z. officinale.4,14 Therefore, natural medicinal sources that support and treat diabetes are increasingly receiving attention from scientists. The objective of the present study, EOs from two plant species (W. schmidtii and Z. atroporphyreus) were evaluated for their chemical profile and inhibitory activities against carbohydrate-hydrolyzing enzymes, namely α-glucosidase and α-amylase. The findings of this study will provide information about the antidiabetic potential of two plant species collected in Vietnam.
Results and Discussion
Chemical Components of Essential Oils
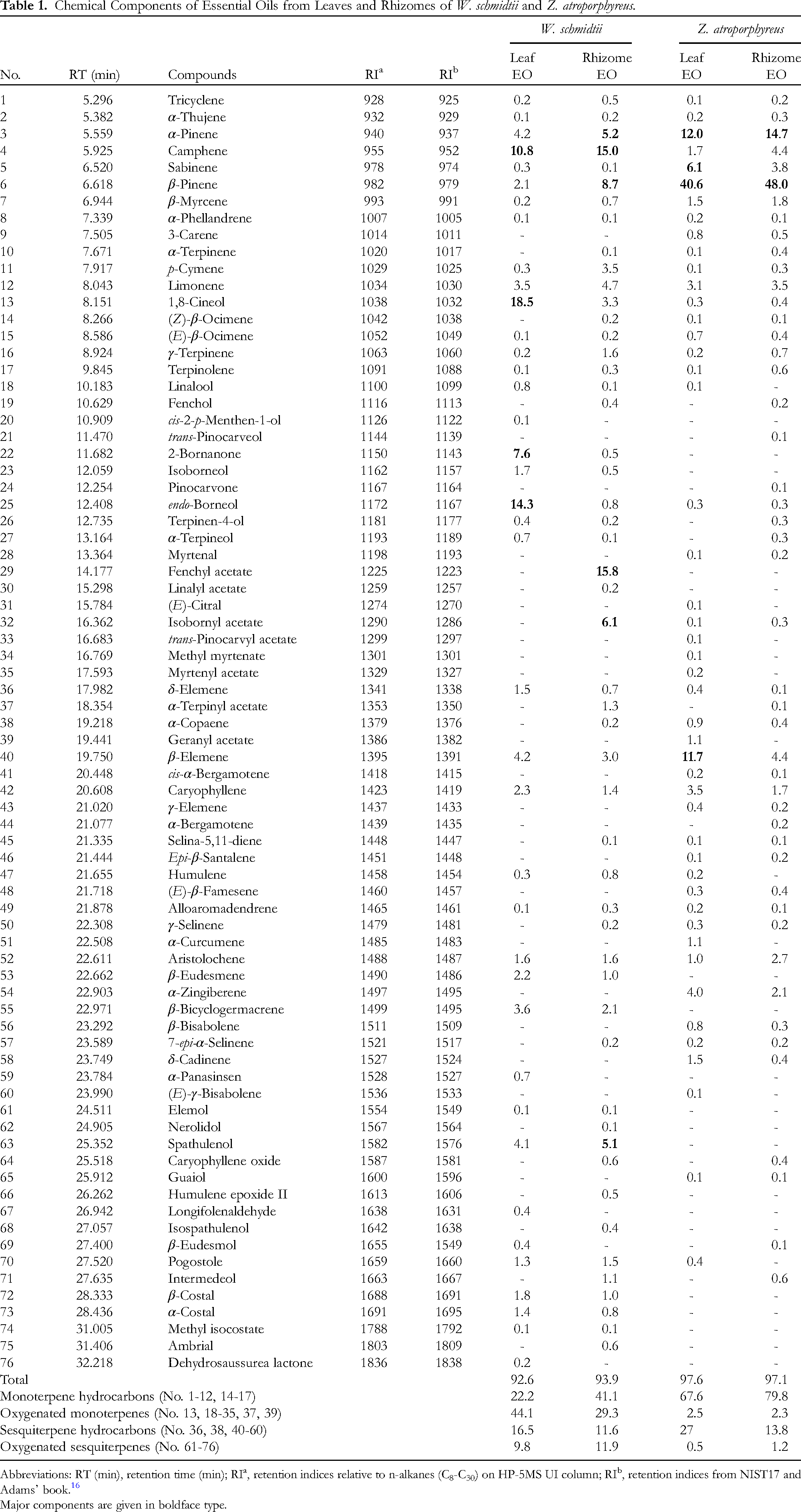
The GC/MS analysis (Supplemental Figures 1-4) provided information on the chemical components of the EOs from the leaves and rhizomes of W. schmidtii and Z. atroporphyreus, the results of which are presented in Table 1.
As for W. schmidtii EOs, the most dominant components of the leaf EO were oxygenated monoterpenes (44.1%), followed by monoterpene hydrocarbon (22.2%), while the rhizome EO contained mostly monoterpene hydrocarbons (41.1%) and oxygenated monoterpenes (29.3%). Specifically, the major components of W. schmidtii leaf EO were 1,8-cineol (18.5%), endo-borneol (14.3%), camphene (10.8%), and 2-bornanone (7.6%), while the rhizome EO contained mostly fenchyl acetate (15.8%) camphene (15.0%), β-pinene (8.7%), isobornyl acetate (6.1%), α-pinene (5.2%), and spathulenol (5.1%). Noticeably, camphene, α-pinene, and spathulenol can be found in both leaf and rhizome EOs of W. schmidtii with significant contents (5%-10%). The GC/MS results also showed the resemblance between Z. atroporphyreus leaf and rhizome EOs in terms of chemical composition. To be more specific, the major chemical components of the leaf and rhizome EOs were β-pinene (40.6% and 48.0%), α-pinene (12.0% and 14.7%), β-elemene (11.7% and 4.4%), and sabinene (6.1% and 3.8%), respectively.
The main chemical compositions of EOs of Zingiber species were α-zingiberene, ar-curcumene, geranial, sabinene, and camphene. Interestingly, the EOs of Z. atroporphyreus expressed the distinction, in comparison to those of other Zingiber species, with β-pinene, α-pinene, β-elemene, and sabinene as the most dominant components.
17
As for the Wurfbainia genus, there has been some information on the chemical composition of the EOs of these species. According to a chemical review of the essential oils prepared from several species of the Amomum genus, which are synonyms with several species of the Wurfbainia genus, there were several differences in the chemical compositions.
18
Specifically, α-pinene (10.4%-31.3%) and β-pinene (29.9%-58.5%) were the major compounds of Wurfbainia uliginosa (syn: Amomum uliginosum) rhizomes and Wurfbainia villosa (syn: Amomum villosum) leaves while β-caryophyllene (26.6%-37.4%) and α-humulene (12.5%-16.5%) were the most dominant in the EOs of Wurfbainia longiligularis (syn: Amomum longiligulare) leaves, stems, and roots. The fruit EO of Wurfbainia villosa was found to contain camphor (22.3%-37.9%), borneol acetate (15.5%-30.5%), and camphene (6.7%-7.6%). Some studies on Wurfbainia vera (syn: Amomum kravanh) fruit revealed that 1,8-cineole was present in the EO with the highest content (34.0%-78.0%). The present study revealed that the chemical compositions of W. schmidtii EOs were similar to that of W. vera, W. aromatica, Amomum subulatum, and Amomum tsao-ko with 1,8-cineole as the most major compound.
18
In Vitro α-Glucosidase and α-Amylase Inhibitory Effects
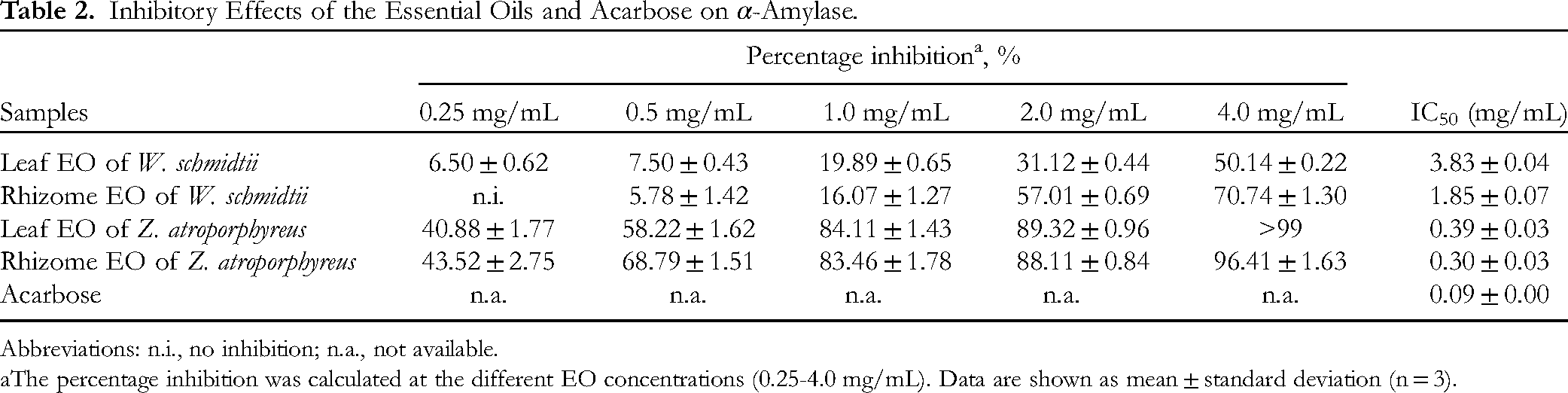
The EOs from W. schmidtii and Z. atroporphyreus leaves and rhizomes were tested for their ability to inhibit α-amylase, and the results are shown in Table 2 and Supplemental Table 1. At concentrations ranging between 0.25 and 1.0 mg/mL, the EO from W. schmidtii leaves appeared to be slightly better at inhibiting the enzyme compared to the EO from its rhizomes. However, the effect is not concentration-dependent manner with the leaf EO no longer exerting stronger inhibitory activity than the rhizome EO. In detail, the former inhibited α-amylase by 31.12% on average while the latter inhibited the enzyme by 57.01% at the concentration of 2.0 mg/mL. Similarly, the inhibitory effect of the rhizome EO was estimated to be about 40% higher than that of its leaf counterpart at a concentration of 4 mg/mL. As a result, the IC50 value of rhizome EO (1.85 ± 0.07 mg/mL) was significantly lower than that of the leaf EO (3.83 ± 0.04 mg/mL), signifying its stronger inhibitory activity against α-amylase. The results also demonstrated comparable inhibition percentages between the two EOs from Z. atroporphyreus across the monitored concentration range. As presented in Table 2, no significant difference in the IC50 values of the two EOs was noted. In comparison with the W. schmidtii EOs, the Z. atroporphyreus EOs had much lower IC50 values and accordingly exhibited higher inhibitory effects against the enzyme. In general, all the examined EO samples may possess a weaker anti-α-amylase activity compared to acarbose (IC50 = 0.09 ± 0.00 mg/mL).
Previous studies have documented the inhibitory potential of Zingiber species against α-amylase, but there is currently no existing literature that provides data on this specific activity for EOs from the Wurfbainia genus. For example, the EO from Z. officinale was shown to inhibit α-amylase.
19
Several plant species within the Zingiberaceae family have been investigated for their potential in this bioactivity. For example, the EO obtained from both fresh and dried rhizomes of Curcuma longa demonstrated a significantly lower IC50 value compared to acarbose, a well-known α-amylase inhibitor.
20
Additionally, a recent study demonstrates EO of Curcuma caesia rhizomes exhibits profound α-amylase inhibitory activity.
21
Reports also indicate that the EOs extracted from the seeds of A. melegueta and A. danielli exerted α-amylase inhibition, although their efficacy was not as potent as acarbose.
20
The inhibitory effect of the EOs from W. schmidtii may be due to the presence of monoterpenes in their chemical composition. In our study, the major constituents identified in the W. schmidtii EOs were pinenes and camphene. Previous research has suggested that these compounds could potentially contribute to inhibitory effects against α-amylase.20,22 The anti-α-amylase activity observed in this species could also be due to synergistic or additive effects of the various constituents present in the EOs.
As for α-glucosidase inhibitory activity of the EOs, the Z. atroporphyreus EOs showed no inhibition in the tested concentrations while the W. schmidtii EOs exhibited very weak inhibition against α-glucosidase (Table 3). According to the findings of our study, if the EOs had an inhibitory effect on α-amylase but not α-glucosidase, they would have impacts on the early stages of carbohydrate digestion, specifically the breakdown of starch. However, it would have limited effects on the later stages, where glucose is released from disaccharides and oligosaccharides. This selective inhibition could potentially lead to a slower and more controlled release of glucose from foods rich in starch. Such an effect could be valuable in the development of natural-based medications for managing type 2 diabetes. In a previous study, the flower EO of Hornstedtia scyphifera showed potential α-glucosidase inhibitory activity with the IC50 value of 21.44 μg/mL.
23
The leaf EO of Alpinia vietnamica was reported to the α-glucosidase inhibitory assay with the IC50 value of 115.18 ± 4.87 µg/mL.
24
Materials and Methods
Plant Materials
The fresh leaves and rhizomes of W. schmidtii were collected from Dinh Mountain, Tan Thanh District, Ba Ria-Vung Tau Province, Vietnam in July-2022, while the fresh leaves and rhizomes of Z. atroporphyreus were collected from Xuan Son National Park, Tan Son District, Phu Tho Province, Vietnam in September-2022. The plants were identified by Assoc. Prof. Dr Nguyen Hoang Tuan. Voucher specimens HCWS-028 and HCZA-038, respectively, were deposited at the Laboratory of the Department of Chemistry, Vinh University, Nghe An Province, Vietnam.
Extraction of the Essential Oils
The EOs were obtained by hydrodistillation which was carried out in a Clevenger-type distillation as previously described with slight modifications.7,25 The experiment was repeated three times and carried out until no further EO could be distilled (approximately 3.5 h each). The leaf and rhizome EOs were treated with anhydrous sodium sulfate to remove the traces of water and stored in sealed vials in the dark at 4°C for further analysis.
GC/MS Analysis
GC/MS analysis was performed using an Agilent Technologies 7890B GC system equipped with an HP-5MS UI column (30 m × 0.25 mm, 0.25 μm film thickness) and coupled to an Agilent 5977B MSD. Helium was used as the carrier gas (1.0 mL/min). The EO was dissolved in dichloromethane (1:100 v/v) then the sample (1.0 μL) was injected, a split ratio of 25:1. The GC oven temperature was kept at 60°C for 1 min and programmed to 240°C at a rate of 4°C/min, and maintained at 240°C for 4 min. Mass spectra were taken at 70 eV. The injector, MS Quad, MS source, and transfer line temperatures were set at 300°C, 150°C, 230°C, and 300°C, respectively. The mass range was from m/z 50 to 550 (2.0 scan/s).
Constituent identification of the EO was based on a comparison of their mass spectra and retention indices (RI) with those reported by the NIST17 database and Adams’ book. The relative percentages of compounds were calculated by the relative area of the total ion chromatogram (TIC) peaks of volatile components.
In Vitro α-Amylase Inhibitory Effect
A diluted EO (0.25-4 mg/mL) in 5% DMSO was combined with 10 μL of α-amylase solution in a phosphate buffer with pH 6.9 and then subjected to incubation at 37°C for 15 min. To initiate the reaction, 15 μL of a 0.25% starch solution was introduced, followed by another 15-min incubation at 37°C. A blank sample was prepared similarly, except that the α-amylase solution was omitted. To terminate the reaction, 50 μL of 1 M HCl was introduced. Subsequently, 100 μL of KI3 solution was added. The spectrophotometer was used to analyze the absorbance at 595 nm.
26
The percentage inhibition of enzymatic activity was computed as follows:

where A and B represent the absorbance of the test sample and the blank, respectively. IC50 values (expressed in mg/mL) were employed to assess the activity of the EOs. Acarbose served as a reference standard.
In Vitro α-Glucosidase Inhibitory Effect
The assay was carried out by mixing 50 μL of an EO diluted in 5% DMSO with 40 μL of α-glucosidase (0.05 U) in phosphate buffer (pH 6.8), followed by a 20-min incubation at 37°C. After this step, 40 μL of 5 mM 4-nitrophenyl-β-D-glucopyranoside (p-NPG) was added and another incubation (20 min, 37°C) was performed. To stop the reaction, 130 μL of 0.2 M sodium carbonate solution was added, and the changes in absorbance were measured at 405 nm.
27
The concentration range was 0.25-4 mg/mL. Acarbose served as a reference standard.