Abstract
Detailed chemical composition of the essential oil of
The yield of essential oil of
Chemical Composition of the Essential Oil of Aerial Parts of
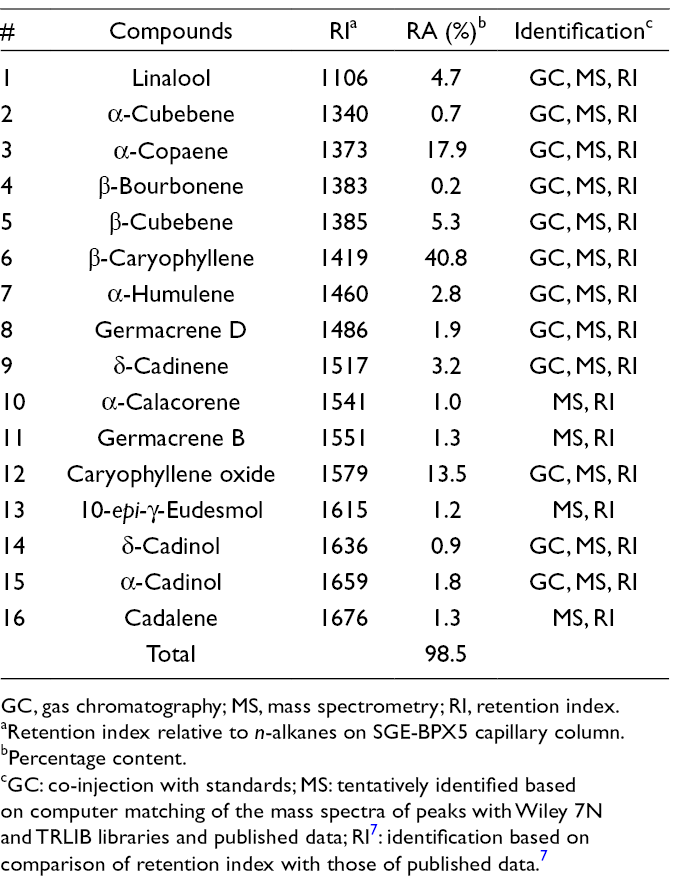
GC, gas chromatography; MS, mass spectrometry; RI, retention index.
a
bPercentage content.
cGC: co-injection with standards; MS: tentatively identified based on computer matching of the mass spectra of peaks with Wiley 7N and TRLIB libraries and published data; RI 7 : identification based on comparison of retention index with those of published data. 7
The chemical composition of the essential oil of
Generally, essential oil composition within the
The antioxidant activity of the essential oil of
Antioxidant Activity of the Essential Oil From

BHA, butylated hydroxyanisole; EDTA, disodium edetate; CUPRAC, cupric reducing antioxidant capacity; FRAP, ferric reducing antioxidant power; DPPH, α,α-diphenyl-β-picrylhydrazyl; ABTS, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid).
Different superscript letters in the same row indicate significant difference (
Furthermore, the oil was able to scavenge stable radicals, which was tested via α,α-diphenyl-β-picrylhydrazyl (DPPH) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) spectrophotometric assays, reaching IC50 values of 25.36 ± 1.61 and 54.06 ± 2.4 mg/mL, respectively. Moreover, Formisano et al.
3
subjected the essential oil of
Enzyme Inhibitory Activities (IC50; mg/mL) of the Essential Oil From

xDifferent superscripts in the same row indicate significant differences (
Experimental
Plant Material
The aerial parts (leaves, buds, and flowers) of
Isolation and Analysis of the Essential Oils
The air-dried and grounded plant material (2.5 kg) was submitted for 5 hours to hydrodistillation by using a British-type Clevenger apparatus. The constituents of the oil were analyzed by means of gas chromatography-flame ionization detector (GC-FID) and GC-mass spectrometry (MS). Briefly, the GC-FID analysis of the essential oil was performed using a Thermo-Finnigan Trace GC/A1300 (E.I.) equipped with an SGE/BPX5 MS capillary column (30 m × 0.25 mm i.d., 0.25 µm). Helium was the carrier gas, at a flow rate of 1 mL/min. Injector temperature was set at 220°C. The program used was 50°C to 150°C at a rate of 3°C/min, held isothermal for 10 min, and finally raised to 250°C at 10°C/min. Diluted sample (1/100, v/v, in methylene chloride), 1.0 µL, was injected manually and in the splitless mode. Quantitative data of the oil was obtained from FID area percentage data. The GC-MS analysis of the oil was performed with a Thermo-Finnigan Trace GC/Trace DSQ/A1300 (E.I. Quadrapole) with the same column and an electron ionization system with I.E. of 70 eV. Carrier gas was He at a flow rate of 1 mL/min. Injector and MS transfer line temperatures were set at 220°C and 290°C, respectively. The temperature program was same as described above. The identification of compounds was based on comparison of their relative retention times with those of authentic samples on the SGE/BPX5 capillary column and by matching their mass spectra with those obtained from authentic samples and/or the Wiley 7N and TRLIB libraries spectra and published data. 7
Biological Activity
Antioxidant activities of the samples were investigated by using β-carotene-linoleic acid, phosphomolybdenum, free radical scavenging DPPH radical and ABTS radical cation, ferrous ion chelating, and reducing power assays (potassium ferricyanide, cupric ion reducing, and ferric reducing antioxidant power). For enzyme inhibitory activities, the essential oils were analyzed toward α-amylase and tyrosinase. All assays are already described in the literature. 10 Results of biological activities are presented as IC50 values, which represent the concentration of the oil (mg/mL) required to inhibit 50% of the initial concentration of oxidant/enzyme.
Statistical Analysis
All the assays were carried out in triplicate. The results were expressed as mean and standard deviation values. Statistical differences between the samples were analyzed using Student’s
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by project No. RO0418 (sustainable systems and technologies, improving crop production for higher quality of production of food, feed, and raw materials, under conditions of changing climate) funded by the Ministry of Agriculture, Czech Republic.
