Abstract
The essential oil from the aerial part of Satureja horvatii spp. macrophylla, an endemic plant species of northwest Greece has been analyzed using GC-MS. Thirty-nine compounds were identified, among which carvacrol was the major constituent (46.8%). The antifungal effect of the essential oil was investigated in vitro against 3 phytopathogenic fungi of tomato, using fumigant and contact assays. Fumigant assay was more effective than contact assay against all phytopathogens. The essential oil inhibited completely the mycelial growth of Fusarium oxysporum f. sp. lycopersici (123 µL L-1 air) and Alternaria alternatα (100 µL L-1 air), while it significantly reduced the growth of Botrytis cinerea (91%).
Tomato (Lycopersicon esculentum Mill.), one of the major vegetables after potato worldwide, is known to possess high nutritional and economic value. 1 Tomato plants can be infected, among others, with Fusarium oxysporum f. sp. lycopersici, a soil born phytopathogenic fungus, which causes tomato wilt, one of the most serious tomato diseases. The loss in yield of tomatoes may reach 80%. 2 This phytopathogen is very difficult to manage because it persists for a very long time with chlamidospores in infected soils, without the presence of the host. The usual inefficiency of chemical treatments and soil solarization to control Fusarium wilt 3 and the risk of varieties losing resistance due to emergence of new races, increase the interest of finding safe and environmentally friendly alternatives. 4 The 2 phytopathogenic fungi Alternaria alternata and Botrytis cinerea affect tomato plants causing also postharvest diseases of tomato fruits with a maximum 30% loss during storage, transport, and marketing. 5 Investigation of alternatives to the conventional fungicides is of particular interest, firstly because of the short period between harvest and consumption, and secondly because of the consumers’ demand for safe food.
Among natural compounds, many plant extracts, and essential oils of aromatic plants have presented strong antifungal properties 6 against plant pathogens. 7,8 The volatile compounds of the essential oils are of great interest for the protection of fruits and vegetables from post-harvest diseases, because of their easy handling. 9 In particular, the essential oils that are used for flavoring foods have a low toxicity towards mammals and are “Generally recognized as safe” (GRAS). Different compounds of essential oils may express antifungal properties through different mode of actions, resulting, in comparison to fungicides, a reduced risk of pathogens developing resistance. 10,11
Satureja horvatii ssp. macrophylla (family Lamiaceae) is an endemic, aromatic, perennial shrub which grows wild in mountainous areas of northwestern Greece. It is used as a honey plant for apiculture and as flavoring for cooking. It is also known for its healing properties. 12 Plant extracts or essential oils have also been found to possess antimicrobial 13 -19 and antifungal properties. 20 -24 In particular, the essential oil of S. horvatii exerts antimicrobial action against some medically important bacteria and yeasts. 15,25
Essential oils, which are mixtures of low molecular weight volatile terpenes, give to the plant a characteristic odor and taste that depend not only on the genotype, but are also influenced by environmental factors. Four different chemotypes of S. horvatii ssp. macrophylla were found to be associated with bioclimatic factors. 26
The in vitro antifungal activity of S. horvatii ssp. macrophylla against phytopathogenic fungi was investigated for the first time in this study. The aims of these experiments were firstly to evaluate the antifungal properties of this aromatic plant against 3 tomato pathogens, B. cinerea, A. alternata and F. oxysporum f.sp. lycopersici, and secondly to identify the chemical composition of the essential oil of the plant.
Results and Discussion
The essential oil was analyzed by gas chromatography–mass spectrometry (GC-MS). Thirty-nine (39) compounds, representing 96.6% of the total components, appeared in the chromatogram (Table 1). Oxygenated monoterpenes dominated (64.7%), with carvacrol (46.8% of total) being the major component, and thymol found in low percentage (3.0%). Μonoterpene hydrocarbons formed 24.6% of the total, with p-cymene (11.1%) and γ-terpinene (7.0%), the 2 biosynthetic precursors of carvacrol and thymol, dominating. Sesquiterpenes (9.9%) comprised a small part of the essential oil, in which caryophyllene (4.8%) was the major constituent.
Chemical Composition of S. horvatii ssp. macrophylla Essential Oil.

Abbreviations: RI, Retention Index relative to n-alkanes on BPX-5 capillary column; RT, Retention Time; Tr., Trace < 0.1%.
To assess the antifungal activity of S. horvatii ssp. macrophylla, we tested an emulsion of the essential oil and an aqueous solution prepared from the dry plant tissue (details in the experimental section). For this screening we used the agar wells diffusion method described by Souza et al., 27 with some modifications. In this, Potato Dextrose Agar (PDA) was mixed with conidia suspension of the test fungus and wells prepared in the medium were filled with the extract. For both extracts, we prepared solutions at different concentrations. The aqueous solution was selected for the assay because it was found bioactive on A549 cancer cells. 28 According to the results, the essential oil emulsion showed significant antifungal activity, inhibiting the mycelium growth of all 3 fungi at a concentration of 15 µL mL-1 (Table 2), in contrast to the aqueous solution, which was ineffective. The aqueous solution had decreased concentrations of essential oil compounds due to their low solubility in water. This might contribute to the ineffectiveness of the aqueous solution against the tested phytopathogens.
Effect of S. horvatii spp. macrophylla Essential Oil Emulsion Against the 3 Tested Phytopathogenic Fungi as Determined by the Agar Wells Diffusion Method. Values Are the Mean of Inhibitory Zone ±Standard Error (Mm). Means With the Same Letter Present Non-Significant Difference (LSD P = 0.05).
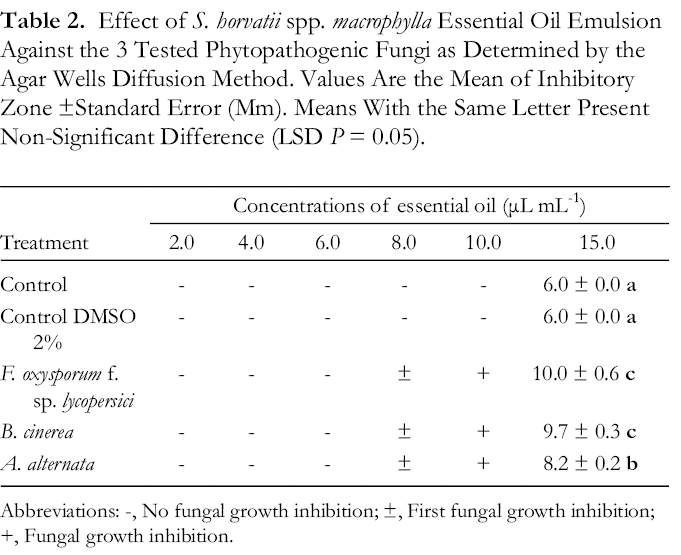
Abbreviations: -, No fungal growth inhibition; ±, First fungal growth inhibition; +, Fungal growth inhibition.
Because of the important antifungal activity of the essential oil emulsion, we further studied the activity of S. horvatii ssp. macrophylla essential oil against the 3 phytopathogens by fumigant and contact assays. In contact assay the essential oil was incorporated into the PDA used for fungal growth, while in the fumigant assay the inoculum and the essential oil were not in contact. To compare the 2 assays, we used the same amount of essential oil per plate for each treatment. The results shown in Figure 1(A–C) and Figure 2(A–C) respectively, represent the % mycelium growth in relation to the control treatment. The value 100 was attributed to the maximum growth of the control.

Fumigant effect of S. horvatii ssp. macrophylla essential oil on radical growth (% of control) of A. alternata, and F. oxysporum f. sp. lycopersici after 7 days of exposure and of B. cinerea after 5 days. Means with the same letter present non-significant differences (LSD p<0.05). C: untreated control, C2: 2μL per plate, C3: 3μL per plate, C4: 4μL per plate, C5: 5μL per plate, C6: 6μL per plate.

Contact action of S. horvatii ssp. macrophylla essential oil on radical growth (% of control T) of A. alternata and F. oxysporum f. sp. lycopersici after 7 days of incubation and of B. cinerea after 5 days. Means with the same letter do not present significant difference (LSD P<0.05). C: control, T: 2% Tween 80, C2: 2 µL per plate, C3: 3 µL per plate, C4: 4 µL per plate, C5: 5 µL per plate, C6: 6 µL per plate.
In both assays the essential oil was found to inhibit the growth of all tested fungi in a dose – dependent manner. The fumigant action of the essential oil was found to be very effective against A. alternata (Figure 1(A)) and F. oxysporum f. sp. lycopersici (Figure 1(C)). Total inhibition of fungal growth was observed at 6 µL per plate for A. alternata (MIC 100 µL L-1 air) and 7 µL per plate for F. oxysporum f. sp. lycopersici (MIC 123 µL L-1 air). For these fungi, the effect of the essential oil was found to be fungistatic and not fungitoxic. However, the fumigant assay has not been proven to be equally effective on B. cinerea (Figure 1(B)). Although the essential oil significantly reduced the mycelial growth of B. cinerea, it did not inhibit it completely, even at the highest concentration used (6 µL per plate). It is noted that in the untreated control of B. cinerea, the colony covered almost the entire plate after 5 days of incubation.
On the other hand, the contact effect on the mycelial growth was weaker (Figure 2(A-C)) than the fumigant effect for all fungi tested. According to probit analysis, the dose required to achieve 50% colony growth inhibition (ED50) by the latter assay was calculated as 4.4 µL of essential oil per plate (219 µL L-1 medium) for F. oxysporum f. sp. lycopersici and 18.2 µL per plate (911 µl L-1 medium) for Α. alternata. In addition, the concentration causing 50% inhibition of Botrytis cinerea mycelial growth (ED50 value) was determined as 286 µL L-1 medium (PDA) for the contact assay, while only 27 µL L-1 air produced the same effect in the fumigant assay. However, as shown in Figure 1(B) 1 and Figure 2(B) and 2, the essential oil significantly reduced the growth of B. cinerea in both fumigant (91% of control) and contact assays (53% of control) (Figure 2(B)) after 5 days of exposure.
According to the results, the essential oil inhibited more effectively the mycelial growth of the tested fungi using the fumigant assay compared to the contact one. These findings are consistent with the results of other researchers 9,29 who suggest that this could be due to the lipophilic nature of the essential oil compounds, which have more affinity with the fungal mycelium than with the growth medium. It should be noted that to conduct a contact assay, the essential oil is incorporated into the molten growth medium (PDA) at 45°C. At this temperature, some of the most volatile essential oil compounds, such as the monoterpenic hydrocarbons, can be lost due to their high volatility. 28 It is noted that the preparation of the growth medium was accompanied by a characteristic odor. So, although we applied the same amount of essential oil per plate to conduct fumigant and contact assays, the actual concentration of the essential oil might not be exactly the same. This could be another reason for the different results of the 2 bioassays.
The GC-MS analysis showed that the essential oil of S. horvatii ssp. macrophylla used in the experiment belongs to the cavacrol chemotype. According to the literature, essential oils rich in the monoterpenic phenols carvacrol and thymol are produced by aromatic plants belonging to the genera Origanum, Satureja and Thymus and possess significant antifungal effects against soil-borne pathogens such as F. oxysporum 30,31 and post-harvest pathogens like A. alternata and B. cinerea. 9,20 -22,31,32 These reports are in accordance with our findings. According to the results of the chemical analysis, the essential oil of S. horvatii spp. macrophylla contains, in addition to the above mentioned monoterpenic phenols, other compounds such as 1,8-cineole, limonene, 33 linalool, p-cymene, α-pinene, α-terpinene, terpinen-4-ol, and thymoquinone, 6 which have been reported to exhibit antifungal activity. These compounds affect the cell growth and morphology of fungi and can cause cell death, mainly by inhibition of the cell wall formation and disruption of the cell membrane. 6 Essential oil compounds could also penetrate the fungal cells, due to their lipophilic nature and act on different intracellular targets. 27
Our findings showed that the essential oil of S. horvatii ssp. macrophylla, which belongs to the carvacrol chemotype, possesses a strong antifungal activity against the 3 important phytopathogenic fungi tested (A. alternatα, B. cinerea, F. oxysporum f. sp. lycopersici). The volatile phase effect of the essential oil was found to be more effective than the contact phase effect. The antifungal action increased in a dose dependent manner. The use of essential oils as alternatives to conventional fungicides is of interest because they are biodegradable and ecofriendly. Nevertheless, further work is needed to investigate the in vivo effect of the S. horvatii ssp. macrophylla volatile essential oil compounds on the organoleptic characteristics of tomato fruits and the application on soil against F. oxysporum f. sp. lycopersici.
Experimental
Plant Material
Wild grown aerial plant parts of S. horvatii ssp. macrophylla were collected from populations at the flowering stage in September, at a mountainous area (800 m altitude) of municipality Ziros, Epirus, northwest Greece. The plant material was freeze dried, as previously described. 28
Isolation of the Essential Oil
The essential oil was obtained from part of the freeze-dried plant material by hydrodistillation (20 g L-1) for 2 hours, using a Clevenger type apparatus, dried over anhydrous sodium sulfate and stored in hermetically sealed vials under N2 at 4°C in the dark until use and analysis.
Preparation of Essential Oil Emulsion
To test the antifungal activity of the essential oil, an emulsion (20 µL mL-1) using DMSO was prepared. Subsequently, a series of essential oil dilutions was performed (2-4-6-8-10-15 μL mL-1). The final concentration of all emulsions in DMSO was 2%.
Preparation of Aqueous-Soluble Solution of the Dried Methanol Extract
Equipment suitable for mixing and pulverizing plant tissues (Polymix, Kinematica) was used for the homogenization of part of the freeze-dried plant material. Particles less than 0.2 mm were extracted by sonication with methanol (10% w/v) using an ultrasound bath. After centrifugation (2.250 x g, 10 minutes) and filtration the solvent (MeOH) was removed from the extract under a stream of N2, at 40°C and the remaining dry extract was re-dissolved in sterile dd H2O. The final concentrations prepared were 0, 2, 4, 6, 8, 10 and 15 mg dw mL-1.
Test Pathogens
The phytopathogenic fungus Fusarium oxysporum f. sp. lycopersici (ΒPIC-2550) was provided by Benaki Phytopathological Institute (Kifissia, Athens, Greece). Alternaria alternata and Botrytis cinerea were isolated from infected tomato fruits and stock cultures were obtained from single spore colonies. Cultures of each of the tested fungi were maintained on Potato Dextrose Agar (PDA) and stored at 4°C.
Agar Wells Diffusion Method
The antifungal activity of the extracts was determined by the agar wells diffusion method. Briefly, conidia from the test fungi were scraped from the agar surface and suspended in sterile distilled water. The suspension was diluted with sterile water to adjust the conidial concentration to 106 CFU mL-1 using a hematocytometer slide. Potato Dextrose Agar (PDA) (18 ml) was inoculated at 40 to 45°C with the conidial suspension (2 ml), mixed and transferred immediately to a 90 mm Petri dish. Wells were made on the solid media plates using sterile glass tubes (6 mm diameter) and subsequently filled with 50 µL either of the test aqueous solution or the essential oil emulsion at the appropriate concentration. A negative control for the aqueous solution was prepared with the same procedure using methanol, while 2% DMSO was used as a negative control for the essential oil treatment. For each fungus, 3 replicates were performed. The Petri dishes were incubated at 25°C in the dark. After 6 days, the diameter of inhibition zones was measured in mm.
Fumigant and Contact Assays
Petri dishes (90 mm) with 20 ml PDA medium culture were prepared to conduct the fumigant and contact antifungal activities of the essential oil. For assessing the fumigant effect towards mycelial growth, the essential oil was applied on a sterile filter paper (6 mm), which was placed at the center of a Petri lid. 9 The contact assay was performed after mixing of the essential oil with molten PDA medium (45°C), as described by Abdolahi et al. 34 For both methods, the same essential oil quantities (0-2-3-4-5-6 μL) per Petri plate were tested, while mycelial disks of 6 mm diameter were cut out from the periphery of 7 days’ old cultures of the test pathogens and inoculated upside down on the PDA. The Petri dishes were sealed with parafilm to prevent loss of essential oil vapors. Three replicates were performed for each treatment and the Petri dishes were incubated at 25°C in the dark. The diameter of fungal growth was measured in mm every 24 hours, for five (5) to seven (7) consecutive days, depending upon the growth rate (control) of the phytopathogenic fungi tested. The relative mycelial growth of treatment compared to control was calculated as a percentage (%). The minimum concentration of essential oil showing complete inhibition of fungal growth (MIC) was determined by visual inspection, while the concentration causing 50% inhibition of mycelial growth on control media (ED50 value) was calculated by probit analysis. The inocula that were completely inhibited after exposure to the essential oil were transferred to a new PDA Petri dish to investigate the mode of essential oil effect (fungistatic or fungitoxic). The viability was evaluated after 3 to 4 days.
GC/MS Analysis
The essential oil was analyzed by a gas chromatograph (Master GC, Dani) coupled with a Time-of-flight (TOF) mass spectrometer (MS, Dani) equipped with an autosampler (Master AS, Dani). A capillary column BPX-5 (30 m x 0.25 mm x 0.25 μm) was used for the analysis. The chromatographic conditions used for the analysis were previously described by Yfanti et al. 28 Briefly, helium (He) was used as carrier gas at a flow rate of 0.7 mL min-1 and a split ratio of 1:30. The oven temperature program raised from 60°C to 110°C with a rate of 3°C min-1, held isothermal for 10 minutes, raised to 150°C with a rate of 3°C min-1 and to 280°C with a rate of 30°C min-1 and then held isothermal for 5 minutes. The mass spectrometer was operated in the electron ionization mode (70 eV), the acquisition rate was 5 spectra s-1 and the spectra ranged from 50 to 550 m/z. For the analysis, the essential oil was diluted in n-hexane (1:400), while 1 µL of the tested sample was injected in splitless mode. The compounds were identified by comparison of their mass spectra with the NIST library data. Their retention indices relative to n-alkanes on a BPX-5 capillary column were compared with the literature.
Statistical Analysis
Analysis was performed with SPSS software. Data from these studies were analyzed by one way analysis of variance (Anova). Mean comparisons were performed by Fisher’s least significant differences (LSD) to examine if the difference among treatments was significant at P < 0.05.
Footnotes
Acknowledgments
The authors would like to thank the research infrastructure network of “OPENSCREEN-GR,” MIS 5002691, implemented under the Action ‘‘Reinforcement of the Research and Innovation Infrastructure’’, funded by the Operational Programme ‘‘Competitiveness, Entrepreneurship and Innovation’’ (NSRF 2014–2020) and cofinanced by Greece and the European Union (European Regional Development Fund), for providing access to the facilities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The manuscript was co-financed by Greece / European Regional Development Fund (ERDF), OP EPIRUS 2014-2020 and the European Union / European Structural and Investment Funds (ESIF). Title: “Treatment of Epirus Meat with Medicinal Plants and Herbs to Produce Bio-Functions Meat”, MIS: 5033186.
