Abstract
Previous studies have revealed that green tea polyphenol (GTP) could protect against liver injury due to oxidative stress. However, the mechanism underlying the bioactive actions of GTP in the liver has not been systematically evaluated. This study aimed to investigate the effect of GTP on the activation of the nuclear factor erythroid-2-related factor 2 (Nrf2)-Kelch-like ECH-associated protein 1 (keap1) pathway, using in silico and in vivo methods. Furthermore, the regulation of Nrf2 downstream target antioxidant response element (ARE) was also evaluated. The high-performance liquid chromatography analysis indicated that GTP includes 9 major compounds, and molecule docking analysis demonstrated that most of these polyphenols have a strong binding affinity with the keap1 Kelch domain, where keap1 binds to the Neh2 domain of Nrf2. Remarkably, the predominant compound of GTP, that is, epigallocatechin gallate, displayed the best binding affinity score, which can fully occupy all 3 polar subpockets of the keap1 Kelch domain. The Nrf2, keap1, and Nrf2 downstream target gene expression levels were changed in the livers compared to the control group. It showed that the Nrf2 expression level was significantly upregulated in GTP-induced mice liver across most treatments, while the keap1 expression level remained unchanged. Subsequently, we observed a significant increasing trend in the expression of the downstream ARE, including antioxidative enzymes, liver phase II enzymes, and liver efflux transporters in mice livers. The present study demonstrated that GTP could activate the Nrf2 signaling pathway by interrupting the Nrf2-keap1 protein–protein interaction
Introduction
The liver is the largest organ and digestive gland in the digestive system of the human body. It plays a vital role in the metabolism and detoxification of multiple endogenous and exogenous harmful substances, such as food, drugs, and environmental pollutants.1,2 Antitubercular, antiretroviral, immunosuppressive drugs, and xenobiotics have been shown to induce free radical generation during their biotransformation in the liver.3,4 Liver has a remarkable ability to scavenge free radicals through its intracellular antioxidant system. Oxidative stress is related to the pathogenesis of several chronic liver diseases manifested as necrosis, cholestasis, fibrosis, and cirrhosis. 5 In the human body, oxidative stress also plays a key role in several diseases, including cancers, cardiovascular diseases, and neurodegenerative diseases. 6
The nuclear factor erythroid-2-related factor 2 (Nrf2) pathway is the major pathway that responds to reactive species and redox potentials, and its activation is a key defense mechanism against oxidative stress. 7 Nrf2 is an important transcription factor responsible for the transcription of several antioxidant and cytoprotective genes. 8 Under physiological conditions, Nrf2 is located in the cytoplasm bound to its repressor Kelch-like ECH-associated protein 1 (keap1). Under oxidative stress conditions, Nrf2 is released from keap1, translocated to the nucleus, and subsequently triggers the transcriptional activation of ARE-dependent genes. These include phase II drug-metabolizing enzymes such as NADPH quinone oxidoreductase 1, UDP-glucuronyl transferase (ugt), glutamate-cysteine ligase catalytic subunit (gclc), hemeoxygenase-1; antioxidants, such as catalase (CAT), glutathione (GSH), glutathione peroxidase (GSH-Px); drug transporters, such as p-glycoprotein (p-gp) and multidrug-resistant proteins (mrps).9–11 GSH redox system can maintain cell redox balance and prevent oxidative damage. At the same time, it also plays an important role in free radical scavenging. The relationship between the GSH redox system and Nrf2 antioxidation is that Nrf2 regulates GSH biosynthesis, and is mainly the rate-limiting enzyme regulating GSH biosynthesis, γ-glutamylcysteine synthetase. Nrf2 controls the expression of GSH-Px, which uses GSH as a cofactor to detoxificate peroxide-oxidize and it was oxidized to glutathione (GSSG) in the process. Nrf2 plays a key role in the regulation of the redox system. Several studies have proven that the Nrf2-keap1 pathway plays a crucial role in defending against various diseases, including cancers, neurodegenerative diseases, acute lung injury, cardiovascular diseases, kidney diseases, liver diseases, and inflammation.12–15 Several researchers have studied the Nrf2-keap1 pathway for its irreplaceable biological functions.
Tea has antioxidant and antitumor properties that provide defense against cardiovascular diseases and other health functions. 16 Tea polyphenols are the main active components, accounting for 15% to 30% of the dry weight of tea. Numerous studies have shown that tea polyphenols have a good antioxidant effect.17,18 Polyphenols are a highly diverse chemical class of molecules, ranging from simple catechols to complex molecules. They can affect biomarkers associated with oxidative stress; however, the relationship between polyphenols and oxidative stress is highly complex and poorly understood. 19 Furthermore, oxidative stress is widely recognized to serve either a causal or consequential role in several disease states, including pulmonary fibrosis, epilepsy, hypertension, atherosclerosis, Parkinson's disease, and drug-induced hepatocellular injury. 20 Several researchers have proved the antioxidant activity and hepatoprotective effect of tea polyphenols. Tea has always been an extremely potent candidate in the nutritional and medical fields. The antioxidant activity and hepatoprotective effect of tea polyphenols have been proved by several researchers. For example, its protective effects against carbon tetrachloride intoxication, sulfur dioxide inhalation, and APAP-induced liver injury.21–23
Several studies have shown that natural products can resist oxidative stress by regulating the Nrf2-keap1 pathway.24–26 The Nrf2-keap1 pathway has become an important target for the treatment of oxidative stress-related diseases. As a master regulator of antioxidative stress, Nrf2 controls the expression of antioxidant genes and phase II detoxifying enzymes such as NADPH quinone oxidoreductase 1 (nqo-1) and gclc that counteract oxidative stress by enhancing the removal of reactive oxygen species (ROS). Keap1 is an actin-binding protein, binding to Nrf2 to keep it in the cytoplasm. Oxidative stress causes oxidation of cysteine residues on keap1. This would disrupt the Nrf2-keap1 interaction. Restraint of keap1 leads to the release of Nrf2, which translocases to the nucleus and activates target gene expression. Molecular interactions play a fundamental role in all biological processes. 27 Docking calculation is an important tool to investigate the active site of proteins and DNA and elucidate the interactions and the binding affinity between the ligand and the biological molecule. It can provide essential information about the binding conformation of inhibitors at the active site and is the best tool for ranking inhibitors based on their binding free energy.
Phytochemicals have contributed enormously to drug discovery in recent decades. 28 Phytochemicals, especially phenolic compounds, could suppress oxidative stress as antioxidants.29,30 Green tea extract represents the richest source of natural polyphenols. 31 Tea has been a traditional Chinese drink for thousands of years, and it is popular worldwide. Hangzhou West Lake Longjing green tea is one of the most beloved green teas among tea lovers. It is primarily composed of catechins, theaflavins, and thearubigins. Epigallocatechin gallate (EGCG), epicatechin gallate (ECG), epigallocatechin (EGC), epicatechin (EC), and catechin (C) are predominant active monomer polyphenols in green tea. 32 Green tea polyphenol (GTP) has better antioxidant activity than vitamin C in vitro. 33
The potential and possibly mechanisms of green tea and its polyphenols in preventing certain types of liver injury have been proposed. 34 It has been confirmed that tea polyphenols have antioxidation and antitumor properties that could help fight against cardiovascular and other diseases.35–37 In addition, it could activate different detoxification enzymes, including glutathione S-transferase, ugt, GSH-Px, superoxide dismutase (SOD), and CAT. Polyphenols can affect biomarkers associated with oxidative stress. However, the relationship between polyphenols and oxidative stress is still poorly understood due to its highly complex relationship.
Given the potent antioxidant activity of Longjing green tea polyphenol (GTP) and the importance of the Nrf2-keap1 pathway in antioxidant defense, we explored whether the Nrf2-keap1 pathway could be activated by GTP in C57BL/6J mouse liver and attempted to elucidate the activation mechanism.
Materials and Methods
Chemicals and Reagents
Yipinxuan teahouse (Shenzhen) provided the Longjing green tea. Abcam supplied the rabbit polyclonal anti-mrp2 antibody, rabbit monoclonal anti-p-gp, anti-ugt1a6, anti-gapdh, anti-Nrf2, anti-gclc, and anti-nqo-1. R&D systems provided the mouse anti-keap1 monoclonal antibody. Abcam produced the goat antirabbit immunoglobulin G (IgG). Sigma-Aldrich sold the gallic acid (GA), caffeine (CAF), gallocatechin (GC), catechin gallate (CG), EGCG, EGC, EC, ECG, C, gallocatechin gallate (GCG), theaflavin (TF), theaflavin-3-gallate (TF-3-G), and theaflavin-3′-gallate (TF-3′-G). Nanjing Jiancheng Bioengineering Research Institute supplied commercial assay kits for CAT, GSH-Px, and SOD. All other chemicals were of the highest grade from commercial sources.
Preparation of Green Tea Polyphenol
GTP was extracted from Longjing green tea according to the Chinese national standard GB/T8313-2018 with slight changes according to the laboratory conditions. Longjing green tea leaves were first powdered in an electronic grinder. Twenty grams of Longjing green tea powder was mixed with 500 mL of preheated 70% methanol (w/v = 1:25) in a 1000 mL round bottom flask. The mixture was kept in a water bath at 70 °C for 30 min. Afterward, the solution was filtered. Methanol was separated and stored in another flask. The same tea powder was mixed with 500 mL of 70% methanol again, incubated in a 70 °C water bath for 30 min and methanol was filtered from the mixture. This extraction process was repeated 3 times. The combined filtrates were then concentrated between 15% and 20% of their original volume (ensuring that methanol was completely removed). Chloroform (v/v, 3:1) was used for liquid–liquid extraction. The chloroform layer was collected and condensed to 5% of the original volume. Subsequently, concentrates were dissolved in ultrapure water and centrifuged at 10,000 r/min for 15 min. The supernatant was freeze-dried and stored at room temperature in a light-proof drying tank.
Total polyphenol content was determined using the Folin-Ciocalteu method using gallic acid as a reference substance. 38 Briefly, 0.2 mL sample or standard solution, 0.2 mL Folin-Ciocalteu reagent, and 1.5 mL saturated Na2CO3 solution were mixed and kept for 60 min at room temperature. Then the absorbance was measured at 760 nm. The method had good linearity in the range of 0 to 50 μg/mL, A = 0.0051C + 0.0415 (r2 = 0.9994). The polyphenol content of GTP was 49.49%.
The content of GA, CAF, GC, CG, EGCG, EGC, EC, ECG, C, GCG, TF, TF-3-G, and TF-3′-G was determined by high-performance liquid chromatography (HPLC) analysis. The chromatographic column Agilent ZORBAX Eclipse Plus Phenyl-Hexyl(4.6 mm × 250 mm, 5 μm). The analysis temperature was 35°C. The detection wavelength was 280 nm. The injection volume was 10 μL. The mobile phase consisted of acetonitrile (A) and 0.15% formic acid (B) at a flow rate of 1 mL/min. The gradient program was as follows: 0–5 min, 3% A; 5–10 min, 3%–7% A; 10–25 min, 7%–10% A; 25–30 min, 10%–12% A; 30–45 min, 12%–15% A; 45–50 min, 15% A; 50–70 min, 15%–30% A; 70–75 min, 30%–3% A; 75–80 min, 3% A. 39
Molecular Docking Analysis
The 3-dimensional (3D) conformers of 9 major compounds of GTP were downloaded from the PubChem library in the SDF format. The crystal structure of keap1 was obtained from the Protein Data Bank database (PDB code 4 l7b). For receptor and ligands preparation before docking, any existing ligands and water molecules were removed from 4 l7b; REDUCE was used to add hydrogens to the receptor and ligands. 40 Finally, PDBQT files with added atom types and partial charges were created using the MGLTools (http://mgltools.scripps.edu/) 1.54 scripts prepare_receptor4.py (receptors_pdbqt) and prepare_ligand4.py (ligands_pdbqt) for receptor and ligands, respectively. The docking of compounds into the keap1-binding pocket was conducted by AutoDock Vina. 41 To restrict the docking area at the Kelch domain pocket, the grid box was set to 50 Å × 50 Å × 50 Å dimensions with X, Y, and Z coordinates of −3.7, 2.4, and −27.5, respectively. All other parameters were defaulted, except for the num_modes value set to 20. To select the best pose from 20 protein–ligand poses of each compound generated by AutoDock Vina, we used the scoring function ΔvinaRF, 42 which employs 20 descriptors in addition to the AutoDock Vina score and could achieve excellent screen performance, to rescore and rank all poses for each ligand. Finally, the top-ranked poses were imported into the Protein–Ligand Interaction Profiler (PLIP) to characterize the detailed interactions of each protein–ligand complex. 43
Animals and Treatment
Eight-week-old male C57BL/6J mice were purchased from the Laboratory Animal Centre of Southern Medical University. The animal license number was SCXK (Guangdong 2016-0041). The experimental protocol was approved by the animal ethics committee (approval no. 2020-508). All procedures were performed following the Regulations of Experimental Animal Administration issued by the Ministry of Science and Technology of the People's Republic of China.
All mice were acclimatized to the laboratory environment for 2 weeks before the experiment. Animals were maintained under controlled conditions (23 °C ± 1 °C, 55% ± 5% humidity, and 12 h light–dark cycle) with free access to standard rodent chow and water. Then they were randomly divided into 4 groups (n = 30): control group, 37.5 mg/kg GTP, 75 mg/kg GTP, and 150 mg/kg GTP groups. GTP was dissolved in sterilized ultrapure water. Mice in the control group were administered with physiological saline every 8 h by intragastric administration. The remaining mice were administered 37.5, 75, and 150 mg/kg GTP every 8 h by intragastric administration. Ten mice in each group were sacrificed at the indicated time points (4, 12, and 20 h) after the first dose of GTP or physiological saline. Liver tissues were collected for further analysis.
Biochemical Assessment
Liver samples (0.2-0.5 g) were homogenized in 0.9% ice-cooled (4 °C) physiological saline (m:v = 1:9) in a FastPrep-24 automated homogenizer. Next, the mixture was vortexed and centrifuged at 3000 r/min for 15 min at 4 °C. The supernatant was used to detect the levels of GSH-Px, SOD, CAT, and malondialdehyde (MDA). GSH-Px Assay Kit (A005-1-2), MDA Assay Kit (A003-4-1), SOD Assay Kit (A001-3-2), and CAT Assay Kit (A007-2-1), which were acquired from Nanjing Jiancheng Bioengineering Institute, were used to determine liver GSH and MDA contents, and SOD activity. All assays were conducted according to the manufacturer’s instructions.
Western Blotting
Traditional western blot analysis was performed according to our previous method. 44 In brief, mice liver tissues were lysed in NP-40 lysis buffer (1% Tris–HCl, 150 mm NaCl, 10% glycerol, 1% NP-40, 2 mm EDTA, and 1% proteinase inhibitor). Minute™ cytosolic and nuclear extraction kit for frozen/fresh tissues kit (NT-032) was purchased from Invent Biotechnologies, Inc., which was used to extract the nuclear protein in mice liver tissues. The assay was conducted according to the manufacturer's instructions. The total protein concentration was determined with a BCA protein assay kit. Equivalent amounts of protein extracts (5 mg/mL) were separated in 8% to 15% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and electrophoretically transferred onto polyvinylidene fluoride membranes (Millipore). After blocking with 5% nonfat dry milk in Tris-buffered saline, the membranes were incubated with primary antibodies overnight. Specific bands were detected using an electrochemiluminescence (ECL) detection kit (Engreen Biosystem) and exposed to an x-ray film (GE Healthcare). The intensity of protein bands was analyzed using Quantity One software (Bio-Rad Laboratories). For protein simple analysis, equivalent amounts of protein extracts were adjusted to 1 mg/mL with their sample buffer. The expression of ugt1a6, mrp2, Nrf2, nuclear Nrf2, keap1, gclc, and nqo-1 was analyzed by Wes (ProteinSimple) according to the manufacturer's protocol. P-gp was analyzed using the traditional western blotting method. Gapdh and histone3 were used as the loading control. The antibody concentrations are shown in Table 1.
Description of Primary and Secondary Antibodies for Western Blotting.

Abbreviations: NFDM, nonfat dry milk; Con, antibody concentration; Tm, incubation temperature; T, incubation time; ON, overnight; RT, room temperature; Nrf2, nuclear factor erythroid-2-related factor 2; keap1, Kelch-like ECH-associated protein 1; p-gp, p-glycoprotein; gclc, glutamate-cysteine ligase catalytic subunit; nqo-1, NADPH quinone oxidoreductase 1; mrp2, multidrug-resistant protein 2.
Statistics Analysis
The obtained data are presented as mean ± standard deviation (SD) and analyzed with SPSS 17.0 using one-way analysis of variance (ANOVA), followed by Duncan's test. P < 0.05 was considered statistically significant.
Results
Molecule Docking Result of Major Monomer Phenol in GTP
The total polyphenol content in GTP was 49.49%. HPLC resolution was acceptable, and the linearity of the standard compounds was good (Supplemental Figure S1 and Table S1). The following compounds were detected: GA (1.37 μg/mg), CAF (21.69 μg/mg), GC (4.95 μg/mg), CG (5.99 μg/mg), EGCG (2.88 μg/mg), EGC (57.74 μg/mg), EC (21.14 μg/mg), ECG (42.08 μg/mg), C (32.22 μg/mg), and GCG (9.47 μg/mg), respectively. And TF, TF-3-G, and TF-3′-G were not detected. C, EGCG, EGC, EC, and ECG—the 5 main active monomer phenols—comprised 92.86% of the total polyphenol.
To study the effect of GTP on the Nrf2-keap1 pathway, docking calculations were adapted to study the detailed molecular interactions between dominant compounds in GTP and keap1 protein, which plays a central role in the precise regulation of Nrf2 within cells. 45 As shown in Table 2, molecular docking analysis clearly revealed that most compounds have a strong binding affinity with keap1, except for caffeine, which did not show any hydrogen binding or hydrophobic interaction with the keap1 Kelch domain. It was worth noting that the predominant compound of GTP, namely EGCG showed the best binding affinity score and had the most hydrogen-bond interactions with keap1 (Table 2). The hydrogen-bound interactive amino acids, including SER-363, GLY-364, ASN-414, ARG-415, ILE-461, ARG-483, SER-508, GLN-530, SER-555, ALA-556, and GLY-603. According to previous reports, the binding cavity of keap1 can be divided into 5 subpockets, including 3 polar subpockets p1 (Ser-508, Phe-478, Ile-461, Arg-483, Arg-415, and Gly-462), p2 (Ser-363, Arg-380, Asn-382, and Asn-414), and p3 (Gly-509, Ala-556, Ser-555, Ser-602, Gly-603, and Gly-571) and 2 hydrophobic subpockets. 46 The docking analysis showed that EGCG could fully occupy all 3 polar subpockets and make extra strong hydrophobic interactions with ALA-556 and TYR-572, indicating that EGCG could be a novel high-activity noncovalent keap1-Nrf2 protein–protein interaction (PPI) inhibitor. The binding affinity score from high to low was EGCG > ECG > C > GC > EGC > EC > EGC > CAF > GA. In summary, we speculated that GTP could directly target keap1 to activate Nrf2 by inhibiting keap1-Nrf2 PPI. Then we conducted the in vivo experiment to verify these results (Figure 1).

Schematic diagram of the regulation of the Nrf2 activity and binding of EGCG in the Kelch domain of keap1. (A) Under normal conditions, Nrf2 can use the ETGE/dLG motif to bind the keap1 Kelch domain. This protein-protein interaction causes the ubiquitination of Nrf2. Thus, a small molecule that inhibits keap1-Nrf2 PPI can act as an Nrf2 activator. (B) The binding pocket surface of keap1 and EGCG. (C) Detailed binding model of EGCG, the hydrogen bonds are represented as solid blue lines.
Molecular Interactions Between the Major Compounds of GTP and keap1 Protein.

Abbreviations: CAF, caffeine; C, catechin; CG, catechin gallate; EC, epicatechin; ECG, epicatechin gallate; EGC, epigallocatechin; EGCG, epigallocatechin gallate; GA, gallic acid; GC, gallocatechin.
Effect of GTP on Liver Nrf2 and keap1 Expression
To validate the docking results, suggesting that GTP can act as an Nrf2 activator by directly inhibiting the keap1-Nrf2 PPI, the changes of Nrf2 and keap1 protein expression levels in GTP-treated mice liver compared to the control groups were determined. GTP doses of 75 and 150 mg/kg significantly upregulated the expression of Nrf2 at 4 h (Figure 2, α-B). The GTP dose of 75 mg/kg significantly upregulated the expression of Nrf2 at 12 h (Figure 2, α-C). Three doses of GTP significantly upregulated the expression of Nrf2 at 12 h (Figure 2, α-D). There was no significant difference between the expression of keap1 among the GTP groups and the control group (Figure 2, β). For nuclear Nrf2, 3 doses of GTP significantly upregulated the expression of nuclear Nrf2 at 4 h (Figure 2,γ-B). Then 37.5 and 75 mg/kg GTP significantly upregulated the expression of nuclear Nrf2 at 12 and 20 h (Figure 2, γ-C/D). These findings suggested that GTP positively affected on Nrf2-keap1 pathway, consistent with our molecular docking results. Compounds in GTP, especially EGCG, can directly interact with the Kelch domain of keap1, disrupting the proper confirmation of the Nrf2-keap1 complex and weakening the ubiquitination modification and degradation of Nrf2. Ultimately, the newly synthesized Nrf2 bypasses keap1, translocates into the nucleus, and stimulates its target genes by binding to their promoters, such as antioxidative enzymes, phase II metabolic enzymes, and some transporters. Next, the dynamic activation of these downstream ARE regulated by Nrf2 was determined in the GTP-treated mice.

Effects of GTP on the expression of liver Nrf2 and keap1 protein expression. α: Nrf2; β: keap1;γ: nuclear Nrf2. Data are presented as mean ± SD (n = 10) * P < 0.05, **P < 0.01 (control vs GTP).
Effect of GTP on Antioxidative Enzymes
GSH-Px, SOD, and CAT are downstream antioxidant enzymes regulated by the Nrf2-keap1 pathway. GSH-Px is an important peroxidase that is widely present in the body. It is responsible for toxic catalytic peroxides into nontoxic hydroxyl compounds to protect the structure and function of cell membranes from peroxide interference and damage. GTP (37.5 mg/kg) increased the GSH-Px activity, 75 mg/kg GTP decreased the GSH-Px activity, and 150 mg/kg GTP did not affect the GSH-Px activity at 4 h (Figure 3, α-A). Three doses of GTP increased the GSH-Px activity significantly at 12 and 20 h (Figure 3, α-B/C). CAT is an enzyme in the peroxide system of cells and catalyzes the decomposition of hydrogen peroxide into oxygen and water. CAT is the marker of peroxisome enzyme, accounting for about 40% of the total peroxisome enzyme. It exists in various tissues in all known animals, especially in the liver. Three doses of GTP suppressed the CAT activity significantly at 4 h and 20 h compared with the control group (Figure 3, β-A/C). GTP could significantly upregulate the CAT level in mouse liver at 12 h and was negatively correlated with doses (Figure 3, β-B). Meanwhile, the total amount of CAT returned to baseline levels at 20 h of GTP treatment (Figure 3, β-C). Since no ARE (antioxidant response element) sequences were found in the CAT promoters and there was no evidence that Nrf2 can bind directly to the CAT promoters, 47 we speculate that this pattern was related to the negative feedback regulation of CAT by reduced cellular ROS level at 20 h GTP treatment. Previous studies have reported results in Nrf2-mediated regulation of CAT that are contrary to our observations. For example, a pleiotropic transcription factor involved in cellular defenses against oxidative stress, increases the expression of several antioxidant enzymes, including CAT. 48 Conversely, CAT expression was significantly lower in various cell types derived from Nrf2-mice than in wildtype control animals. 49 SOD is an antioxidant metalloproteinase existing in organisms that catalyzes the disproportionation of superoxide anion radicals to produce oxygen and hydrogen peroxide. It plays an important role in maintaining the balance of oxidation and antioxidation of organisms. It is closely related to the occurrence and development of several diseases. At 4 h, GTP does not affect the SOD activity in the mouse liver (Figure 3, γ-A). At 12 h, 3 doses of GTP increased the activity of SOD significantly (Figure 3, γ-B). At 20 h, 75 and 150 mg/kg GTP raised the SOD activity significantly, and in accordance with the dose (Figure 3, γ-C). MDA is a lipid peroxidation marker. Compared with the control group, 3 doses of GTP increased the content of MDA at 4 h, then decreased at 12 and 20 h (Figure 3, δ).

Effects of GTP on liver antioxidant enzyme system. α: GSH-Px, β: CAT, γ: SOD, δ: MDA. Data are presented as mean ± SD (n = 10) * P < 0.05, **P < 0.01 (control vs GTP).
Effect of GTP on Liver Phase II Enzymes Expression
Gclc and nqo-1 are phase II antioxidative enzymes regulated by the Nrf2-keap1 pathway. A 150 mg/kg GTP increased the expression of nqo-1 at 4 h and 12 h. After treatment with different doses of GTP, 37.5 mg/kg dose at 4 h and 150 mg/kg dose at 12 h upregulated the expression of gclc (Figure 4, β-B, C). The other doses at experiment time points did not affect its protein expression (Figure 4, β). Ugt1a6 is a phase II metabolite enzyme, which catalyzes the transformation of active phase I metabolites into more hydrophilic and stable products. Moreover, it is a target protein of the Nrf2-keap1 pathway. Three doses of GTP upregulated the expression of ugt1a6 at all experiment time points (Figure 4, γ).

Effects of GTP on the expression of liver phase II enzyme proteins. α: nqo-1, β: gclc, γ: ugt1a6. Data are presented as mean ± SD (n = 10) * P < 0.05, **P < 0.01 (control vs GTP).
Effect of GTP on Liver Transporter Expression
Metabolites of exogenous compounds can be excreted by efflux proteins, including mrps and p-gp. These transporters played an important role in protecting organs and tissues from damage by exogenous compounds through excretion. The protein expression of mrp2 was upregulated at 4 and 20 h by GTP (Figure 5, α-B/D). However, the expression did not change at 12 h (Figure 5, α-C). P-gp is a transmembrane glycoprotein that has an energy-dependent “drug pump” function. Three doses of GTP increased its expression at 20 h (Figure 5, β-D), with no influence at 4 and 12 h (Figure 5, β-B/C).
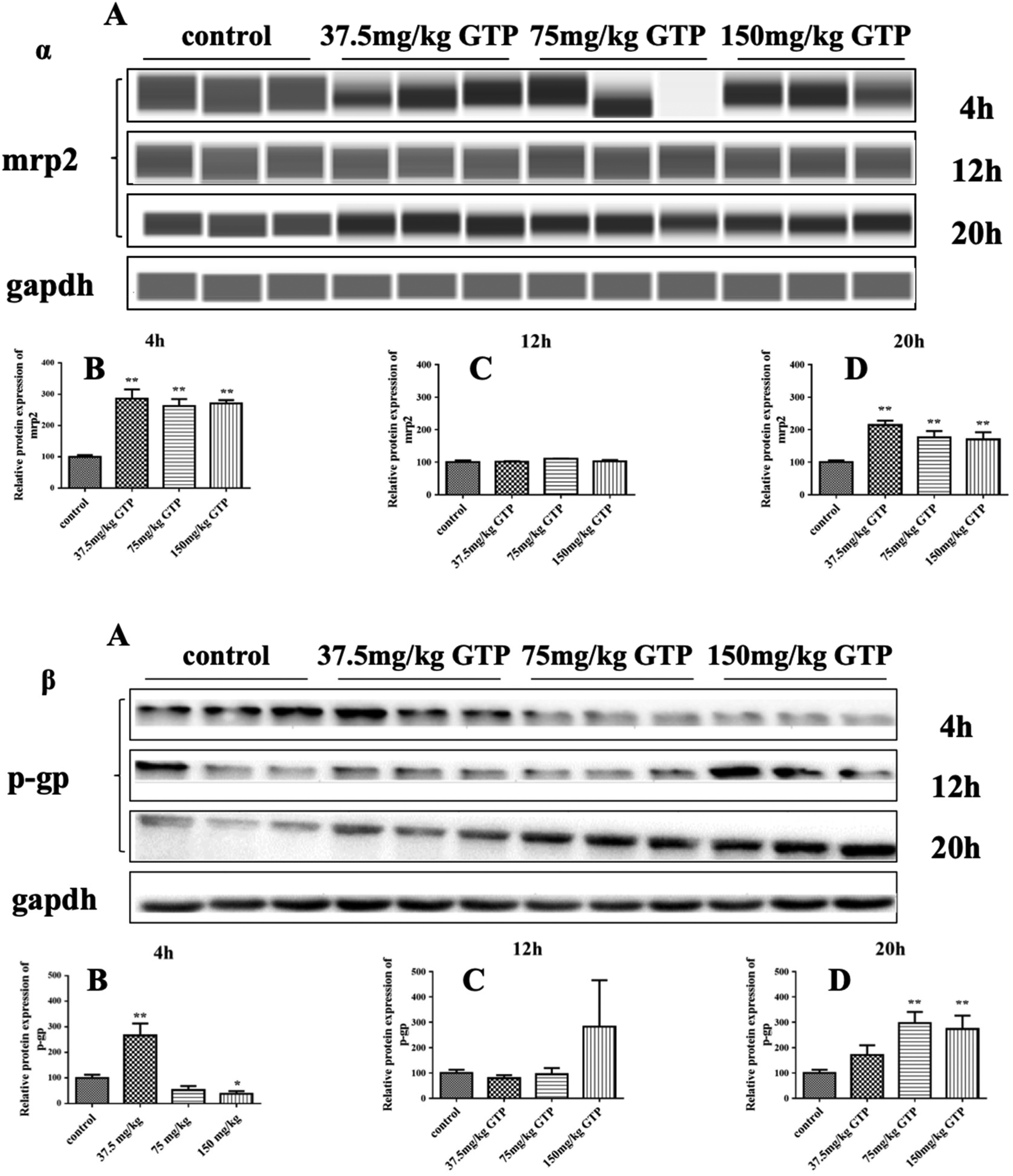
Effects of GTP on the expression of liver transporters. α: mrp2, β: p-gp. Data are presented as mean ± SD (n = 10) * P < 0.05, **P < 0.01 (control vs GTP).
Discussion
In the present study, docking calculations were used to explore the binding free energy of the primary monomer phenol in GTP with keap1. Except for CAF, all other monomer phenols have a strong binding affinity with keap1. EGCG shows the best binding affinity score and has the most hydrogen-bound interactions with keap1. Therefore, EGCG may be a novel, highly active noncovalent keap1-Nrf2 PPI inhibitor. EGCG is also the most abundant component in GTP. We speculated that GTP could directly target keap1 to activate Nrf2 by inhibiting keap1-Nrf2 PPI. We conducted the animal experiment to determine whether GTP could activate the Nrf2-keap1 pathway in vivo. Results showed that GTP upregulated the relative protein expression level of intracellular and nuclear Nrf2, in the meantime, cells maintained a stable protein level of keap1, which agreed with our docking analysis results. GTP deactivates the keap1 protein through noncovalent interaction with the kelch domain instead of directly regulating the protein level of keap1. Since keap1 can binds to Nrf2 and promotes its degradation by the ubiquitin-proteasome pathway, GTP deactivating the keap1 protein will lead to Nrf2 accumulation in the cytosol. Consequently, Nrf2 translocases to the nucleus and binds to ARE in the promoter region of many cell defense genes to activate their transcription.
Nrf2 serves as an endogenous regulator that governs the expression of a battery of cytoprotective genes encoding intracellular detoxifying enzymes and antioxidant proteins responsible for antioxidant defense, conjugation, transport, and excretion of metabolites through ARE. 50 We explored the dynamic effects of GTP on the expression of major enzymes involved in antioxidant defense, phase II metabolite enzyme, and transporters targeted by the Nrf2-keap1 pathway in 20 h. We found that GTP upregulated the protein expression of phase II enzyme ugt1a6, efflux transporter p-gp, mrp2, and SOD, GSH-Px, and CAT activity in the mouse liver. This result revealed that GTP activates the Nrf2-keap1 pathway in mouse liver.
It was reported that animals deficient in Nrf2 were highly susceptible to organ injury from toxic stimuli such as APAP, 3,4-benzopyrene, and other oxidative stresses due to decreased antioxidant protection. 51 About 85% of APAP in humans is metabolized by ugts and sults. P-gp and mrp2 belong to the superfamily of ABC transporters. P-gp functions as a barrier to pump xenobiotics out of the cells. Thus far, it is considered the most important efflux transporter expressed in multidrug-resistant cancer cells and several organs such as the intestine, liver, kidneys, and the blood–brain barrier. Mrps can transport a wide range of organic anions, including glucuronic acid, glutathione, sulfate, or certain anticancer drugs. 52 mrp2 plays an important role in cholestasis, which can transport bile acid, glucuronic acid, and other toxic substances. 53 Their function contributes to preventing the accumulation of toxic drugs or potentially dangerous endogenous compounds in these tissue sanctuaries.
In summary, a combination of in silico and in vivo approaches was adopted to explore the underlying mechanism of GTP on the Nrf2-keap1 pathway. We speculated that the GTP could improve endogenous antioxidant defenses via Nrf2-keap1 pathway activation by directly inhibiting the binding of the keap1 Kelch domain to the Nrf2. Therefore, GTP is a promising natural ingredient to protect against oxidation and stress-induced liver injury. Moreover, molecular docking results showed that EGCG is expected to lead to the discovery of potent keap1-Nrf2 PPI inhibitors. This is worth further research.
Conclusion
In conclusion, our study demonstrated that the administration of GTP could modulate the Nrf2-keap1 signaling pathway. This study provided new insights into the potential use of GTP as an antioxidative agent to treat oxidation-related liver injury.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221139409 - Supplemental material for Activation of the Nrf2 Antioxidant Pathway by Longjing Green Tea Polyphenols in Mice Livers
Supplemental material, sj-docx-1-npx-10.1177_1934578X221139409 for Activation of the Nrf2 Antioxidant Pathway by Longjing Green Tea Polyphenols in Mice Livers by Le Lv, Haoyue Shu, Xiaoye Mo, Yongjing Tian, Hui Guo and Hai-Yan Sun in Natural Product Communications
Footnotes
Acknowledgments
The authors are thankful to thank the Shenzhen Stability Support Project for Colleges and Universities (No.20220814205518001), Characteristic Innovation Project and Marine Medicine Innovation Platform for the integration of Production and Education Project of Guangdong Provincial Education Department (No. 2020KTSCX295, 2021CJPT014),Shenzhen Institute of Technology Scientific Research Projects (2211003, 2214010), and Post-doctoral Foundation Project of Shenzhen Polytechnic (6020330012K0) for financial support of this study.
Authors’ Contribution
LL contributed to conducting experiments, data compilation, and manuscript writing. HS contributed to docking data recording and analysis. XM contributed to data recording and HPLC analysis. YT contributed to the determination of nuclear Nrf2. HG contributed to nucleoprotein extraction. HS contributed to the conceptualization and design of the experiments, data analysis, and paper writing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shenzhen Stability Support Project for Colleges and Universities, Characteristic Innovation Project and Marine Medicine Innovation Platform for the integration of Production and Education Project of Guangdong Provincial Education Department, Shenzhen Institute of Technology Scientific Research Projects, and Post-doctoral Foundation Project of Shenzhen Polytechnic (grant numbers 20220814205518001, 2020KTSCX295, 2021CJPT014, 2211003, 2214010, and 6020330012K0).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
