Abstract
Introduction
The incidence and mortality rates of cancer have been on the rise according to the Global Burden of Disease Study, resulting in an increased economic burden for cancer research. Although age-standardized cancer mortality has declined in all types of cancer, new drugs account for only a small part of the improvement. Conventional chemotherapeutic agents are limited by chemotherapy resistance, and the survival quality of patients has decreased. Therefore, new drug therapies are urgently needed.
Natural agents have shown great potential in the field of anticancer therapy, as they can target different processes, including apoptosis, cell cycle, autophagy, and redox balance, with low cytotoxicity to normal cells. The genus Podocarpus has been traditionally used for treating fevers, asthma, inflammation, and arthritis. 1 Our research isolated CXX-3, a norditerpene lactones, from Podocarpus nakaii. CXX-3 has been reported to have cytotoxic activities against P388 murine leukemia cells. 2 However, the molecular mechanism remained to be elucidated.
Nuclear factor E2-related factor 2 (NRF2) is a transcriptional regulator of genes that regulates oxidative/xenobiotic stress response. 3 The primary function of NRF2 is to carry out antioxidation in cells, which can stabilize the inner cellular environment. Cancer cells are characterized by elevated levels of proliferation and imbalanced redox consumption and signaling. 4 Recent reports pointed out that some cancer cells can acquire the homeostatic capacity from NRF2, which enhances their adaptability and promotes tumor development. 5 Also, NRF2 plays a significant role in cancer cell resistance to oncology drugs.6-8 For instance, NRF2 can lead to cancer cell resistance to ferroptosis, 4 which can be reversed by inhibiting NRF2. 9 The NRF2-KEAP1-CUL3 complex is the major form of NRF2 in cells. KEAP1 acts like a sensing lock for NRF2, preventing it from entering the nucleus and balancing it by facilitating its degradation. When cells are under stress, the KEAP1 sensing lock opens, allowing NRF2 to accumulate and translocate into the nucleus to activate cytoprotective gene expression.3,10 Therefore, inhibiting NRF2 can be a promising therapeutic target in cancer treatment.
Therefore, this study aims to evaluate the biological activities of CXX-3 in vitro and explore its molecular mechanisms, with a focus on the reactive oxygen species (ROS) pathway. The results revealed that CXX-3 inhibits the proliferation of glioma strain U251 and colon carcinoma strain HCT-116 in vitro by promoting cell cycle arrest and increasing ROS levels. CXX-3 also interferes with the function of the NRF2/KEAP1 complex, resulting in the inability to activate cytoprotective gene expression as usual. Furthermore, we also found that CXX-3 may have a separate way to disable the function of NRF2 in U251 and HCT-116. Thus, our study suggests that CXX-3 is a pleiotropic compound and provides a novel theoretical basis for further in-depth investigation of CXX-3.
Methods and Materials
Cell Lines and Culture Condition
Human glioma cell line U251 and human colon cancer cell HCT-116 were used in this experiment. U251 cells were cultured in high glucose DMEM medium (whole medium) containing 10% newborn calf serum, and then penicillin and streptomycin were added. HCT-116 cells were cultured in RPMI-1640 complete medium containing 10% FBS and 1% double-antibody. Both cells were cultivated in an incubator with 5% CO2 at 37 °C.
Cell Viability
U251 cells or HCT-116 cells in the logarithmic growth stage were taken and made into a single-cell suspension using a culture medium. The cells (1 × 104-5 × 104 cell/mL) were inoculated into 96-well plates, with a volume of 100 μL per well. After the cells were placed in the incubator with 5% CO2 at 37 °C for 24 h, 10 μL of the drug with a concentration gradient was added. Each gradient had 4 multiple holes and the contrast hole was set. The cells were incubated in 5% CO2 at 37 °C for 48 h, and the changes in cell state were observed at different time points using a microscope. After reaching the measurement time, 20 μL of 5% MTT solution was added to each well and cultured for 4 h. Then, 200 μL of the triplex solution was added to each well with a pipette gun and placed in a constant temperature incubator at 37 °C for another 14 h. Finally, the enzyme-labeled instrument was applied to measure the OD value at 570 nm and the data were recorded.
Cell Cycle Analysis
Cell cycle analysis was performed to assess the effect of our products on the distribution of tumor cells in G1, S, and G2/M phases of the cell cycle. This test was conducted using flow cytometry after DNA staining to reveal the total amount of DNA. Approximately 1.5 × 106 K-562 tumor cells were cultured in the presence of the tested products using their respective IC50. After 24 h of incubation, cells were collected, washed with PBS, fixed with cold 70% ethanol, and conserved at −20 °C overnight. Next, 100 µL of RNase A (1 mg/mL) was added and cells were stained with a solution containing 10 µg/mL of propidium iodide after 30 min of incubation at 37 °C. The samples were analyzed using a Guava easyCyte HT flow cytometer (Millipore) and the Guava Clean 3.1 software.
ROS Assay
Inoculate 80 × 104 U251 cells or HCT-116 in the logarithmic growth stage into a 6 cm Petri dish. The control group and the CXX-3 group (CXX-3 group included 0.25 μmol·L−1, 1 μmol·L−1, 5 μmol L−1CXX-3) were set. After the cells were placed in an incubator with 5% CO2 at 37 °C for 24 h, the original medium was discarded, and corresponding concentrations of drugs were added into the medium. Each experiment was repeated three times. After culturing in the incubator for 24 h, the cells were washed with a blank medium twice and then treated with trypsin without EDTA. The cells were centrifuged at 1500 r min−1 for 5 min at 4 °C and then collected into EP tubes. 500 μL of 5 μmol L−1 blank medium containing DCFH-DA probe was added to each medium and incubated at 37 °C for 20 min in the dark. Following incubation, the cells were centrifuged at 1500 r min−1 for 5 min. Then, the original medium was poured out and the new blank medium was added, followed by washing twice and centrifugation. Next, a 200 μL blank medium was used to suspend the cells. After filtration by a membrane, flow cytometry was employed to detect and record the data.
Mitochondrial Membrane Potential Measurement
The mitochondrial membrane potential (MMP) changes were measured by MMP assay kit with JC-1 (Beyotime) according to the manufacturer's instructions. Briefly, treated cells were harvested and washed with cold PBS twice. Thereafter, the cells were resuspended in a mixture of 500 μL culture medium and 500 μL JC-1 staining fluid for 20 min protected from light at 37 °C. Subsequently, cells were washed with a cold staining buffer three times prior to flow cytometry. JC-1 exists either as a cytoplasmic JC-1 monomer or mitochondrial J-aggregates depending on the potential of the mitochondrial membrane. In healthy cells with high MMP, JC-1 spontaneously forms J-aggregates in mitochondria which emits red fluorescence. However, in unhealthy cells, the MMP declines and JC-1 is released from mitochondria and exists as a monomer in the cytoplasm, which yields green fluorescence. Thus, MMP can be indicated by the ratio of red to green fluorescence intensity. Carbonyl cyanide 3-chlorophenylhydrazone (CCCP), which disrupted mitochondrial integrity and induced the complete loss of MMP, was used as a positive control.
Nucleocytoplasmic Separation
Cells were separated with 150 μL of nuclear material (protein extracted by adding PMSF), suspended again, and broken on ice for 5 min, 10 min, and mixed upside down every 1 min. During centrifugation at 4 °C and 12 000 r/min for 15 min, 120 μL of supernatant was taken as a cytoplasm sample and precipitated as the nucleus. For the nucleoprotein, the nucleus was washed three times with PBS. The 150 μL of strong lysate was added and resuspended until no flocculent precipitation was evident. The mixed lysate was left on ice for 30 min, followed by centrifugation at 12 000 r/min and 15 min. Then, 120 μL supernatant was centrifuged for 10 min. The samples were subsequently treated according to the experimental steps of Western Blotting.
Western Blotting
Cells (5 × 105 cell/well) in the logarithmic growth phase were inoculated into 6-well plates. The total protein was extracted by RIPA lysate and detected by the BCA method. The supernatant was obtained after centrifugation at 12 000 r/min for 15 min. A 10% SDS-PAGE Gel was prepared, and the protein was electrophoresed and transferred to a PVDF membrane. After blocking for 1 h at room temperature, the protein was rinsed with TBST and incubated with primary antibodies as β-actin, P27, and Cyclin D1 overnight at 4 °C. After washing with TBST, HRP-IgG was added and incubated at room temperature for 2 h. The relative expression of proteins was calculated after washing with TBST 3 times and light exposure with several internal references.
Antibodies CDK2, Cyclin D1, C-myc, P27, β-actin, KEAP1, NRF2, HO-1, GCLM, H3, and β-Tubulin were used in the detection.
Statistical Analysis
All experiments were performed at least 3 times, and the results were analyzed using SPSS 17.0. Data are presented as means ± SD. Two groups of quantitative data were compared using an independent sample t-test. Statistical significance was determined at *p < .05; **p < .01; ***p < .001.
Results
CXX-3 Triggers Cell Death and Promotes Cell Cycle Arrest in S-Phase in Glioma and Colon Cancer Cells
The structure of CXX-3 is displayed in Figure 1(a). To assess its cytotoxicity and inhibitory effect, CXX-3 with different concentrations was administered to U251 and HCT-116 cell lines. The results of MTT assays revealed that CXX-3 could inhibit the viability of both cell lines (Figure 1(b)) at low concentrations, with greater efficacy observed as the concentration escalated. IC50 for U251 and HCT-116 cells at 48 h were 0.55 ± 0.10 μM and 1.12 ± 0.13 μM.

CXX-3 inhibits the viability of glioma and colon carcinoma cells in vitro. Cell cycle was induced by CXX-3 in glioma and carcinoma cells. (a) The structural formula of CXX-3((1aR,1bS,1b1R,3aR,4R,4aR,5aS,5bS,9R,9aR)-4-hydroxy-3a,5b-dimethyl-9-vinyl-1a,1b,1b1,3a,4,4a,5a,5b-octahydro-3H,7H,9H-oxireno[2,3-i]oxireno[2’,3':5,6]isobenzofuro[7,1-fg]isochromene-3,7-dione). (b) The viability of U251 and HCT-116 were inhibited after treatment with different concentrations of CXX-3 for 48 h, the viability of both cell lines was estimated by MTT assay. All experiments were repeated 3 times. The bars represent mean ± SD of three independent experiments. *p < .05, **p < .01, ***p < .001. CXX-3 inhibits the viability of glioma and colon carcinoma cells in vitro. The cell cycle was induced by CXX-3 in glioma and carcinoma cells. (c) U251 and HCT-116 were treated with different concentrations of CXX-3 (0 μM, 0.25 μM, 0.5 μM, 1 μM for U251; 0 μM, 1 μM, 2 μM, 4 μM for HCT-116) for 24 h. The rates of cells in the cell cycle phase were estimated by flow cytometry. (d) The expression of CDK2, Cyclin D1, C-myc, and p27 protein in untreated cells (control) and CXX-3 was measured using western blotting. All experiments were repeated 3 times. The bars represent the mean ± SD of three independent experiments. *p < .05, **p < .01, ***p < .001.
To further investigate whether CXX-3 inhibited cell viability by inducing cell cycle arrest, flow cytometry was utilized to estimate the number of cells in different phases. The results indicated that CXX-3 promoted S-phase arrest, while the percentages of cells in the G0/G1 and G2/M were diminished in both U251 and HCT-116 (Figure 1(c)). These were validated by detecting the expression of Cyclin D1 and CDK2, which are critical for the S phase. The expression of CyclinD1 and CDK2 proteins was lowered after treatment with CXX-3 (Figure 1(d)). Taken together, the findings suggested the inhibition of the viability of U251 and HCT-116 and the arrest of the cell cycle after CXX-3 treatment, as well as the downregulation of cyclin D1 and CDK2.
CXX-3 Upregulates ROS Level and Induce MMP Decline in Glioma and Colon Cancer Cells
It is well known that the cell cycle is regulated by numerous signals.11,12 ROS has been shown to have an impact on these signals and thus can indirectly affect cell cycle progression.13,14 To explore whether CXX-3 can influence the level of ROS in cells, a DFCH-DA dual staining assay was performed using flow cytometry. The results suggested that after CXX-3 treatment, ROS levels were significantly elevated in U251 and HCT-116 in a concentration-dependent manner and we also fine CXX-3 can induce MMP decline at the same time (Figure 2(a)).
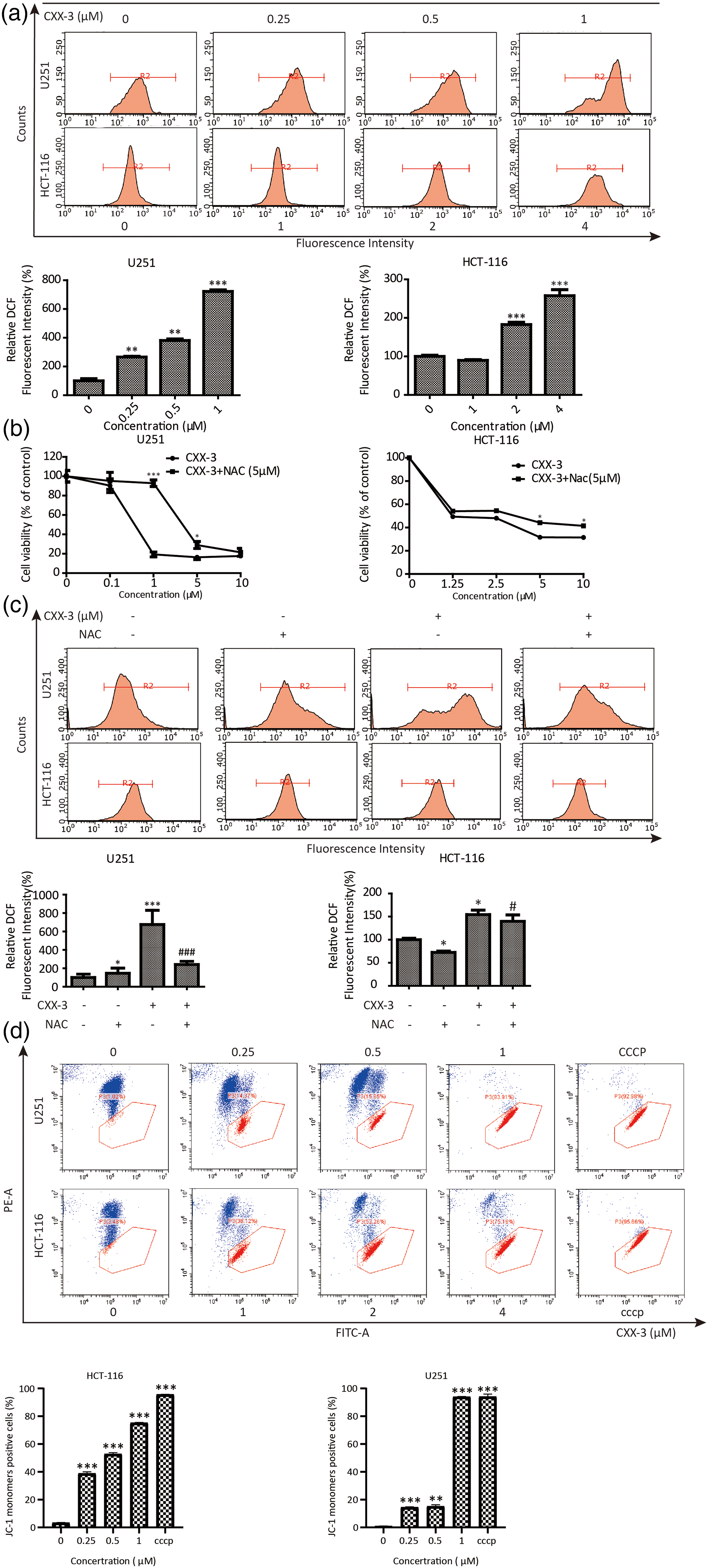
CXX-3 induces high levels of reactive oxygen species (ROS) and mitochondrial membrane potential (MMP) decline in U251 and HCT-116 cells. (a) U251 and HCT-116 were treated with CXX-3. DCF fluorescent intensity was performed by flow cytometry. (b) CXX-3 or CXX-3 combined with NAC (5 μM, 2 h) treatment on U251 and HCT-116. Cell viability was estimated using the MTT assay. All experiments were repeated 3 times. The bars represent the mean ± SD of 3 independent experiments. *p < .05, ***p < .001. #p < .05, ###p < .001. CXX-3 induces high levels of ROS and MMP decline in U251 and HCT-116 cells. (c) U251 and HCT-116 cells were treated with CXX-3 (1 μM for U251, 4 μM for HCT-116) and NAC (5 μM) or a combination ROS level at 24 h after drug treatment (*: vs control; #: vs CXX-3 + NAC). All experiments were repeated three times. The bars represent the mean ± SD of three independent experiments. *p < .05, ***p < .001. #p < .05, ###p < .001. CXX-3 induces high levels of ROS and MMP decline in U251 and HCT-116 cells. (d) U251 and HCT-116 cells were treated with CXX-3 (0, 0.25, 0.5, 1 μM for U251, 0, 1, 2, 4 μM for HCT-116) and cyanide 3-chlorophenylhydrazone (CCCP) (5 μM) red/green ratio at 24 h after drug or CCCP treatment(*: vs control). All experiments were repeated three times. The bars represent the mean ± SD of three independent experiments. *p < .05, ***p < .001.
To clarify the role of CXX-3-induced ROS accumulation in cell death, ROS inhibitor N-acetyl-l-cysteine (NAC) was utilized. As illustrated in Figure 2(b), treatment with NAC partially protected cells from CXX-3-induced cell death and high ROS levels caused by CXX-3. These findings showed that CXX-3 can lead to oxidative stress and result in the accumulation of ROS in both U251 and HCT-116 cells.
KEAP1/NRF2 Complex Can Be Incapacitated by CXX-3 in Different Ways
Previous studies indicated that ROS can be an important mechanism in mediating the cell cycle in some anticancer drugs.13-15 To investigate the target of CXX-3-induced cell oxidative stress, we detect the expression of the KEAP1/NRF2 complex and its client protein by Western Blotting. Interestingly, the expression of NRF2 was upregulated and the expression of KEAP1 was downregulated in both two cell lines after 24 h of treatment with CXX-3. However, these results could not explain the accumulation of ROS in the cells. Therefore, we assessed the expression of its client protein GLCM and HO-1, which are regulated by KEAP1/NRF2 and exert antioxidant effects. The results revealed that the expression of these two proteins was downregulated. Thus, we speculated that CXX-3 might undermine the functioning of NRF2.
It is reported that the reactive cysteine residues of KEAP1 will be directly modified, which reduces the ubiquitin E3 ligase activity of the KEAP1-CUL3 complex, leading to the stabilization of NRF2 when cells are exposed to ROS. The nascent NRF2 can then be translocated directly to the nucleus to activate client genes.3,6,10 To determine whether CXX-3 arrests the capacity of NRF2 in the nucleus or cytoplasm, the nucleocytoplasmic separation assay was carried out in both cells after 24 h of CXX-3 treatment. The results showed that the expression of NRF2 was upregulated in the nucleus in both U251 and HCT-116 cells but could not translocate into nuclear (Figure 3(b)). Thus, we speculated that in U251, CXX-3 mainly affects NRF2's binding to ARE/EpRE, while in HCT-116, CXX-3 may block the nuclear entry pathway of NRF2. These results suggested that CXX-3 upregulates ROS levels in U251 and HCT-116 cells by impairing the antioxidative capacity of NRF2, and it may achieve this effect in a unique way.

CXX-3 induces high levels of reactive oxygen species in U251 and HCT-116 cells by suppressing the KEAP1/NRF-2 signal pathway. (a) The expression of KEAP1, NRF-2, HO-1, and GCLM protein in untreated cells (control) and those treated with CXX-3 was examined by western blotting. (b) Analysis of NRF-2 expression in the nuclear and cytoplasmic protein of U251 and HCT-116 cells treated with CXX-3 for 24 h by western blotting.
Discussion
In recent years, there are increasing studies reporting the unique role of ROS in the steady state of animals and plants,16-18 where NRF2 is becoming a research hotspot. NRF2 is well-known for its antioxidant affection in cells,10,19 and the KEAP1/NRF2 system is considered the main defense mechanism of the human body against environmental pollution. 3 In normal cells, NRF2 and the KEAP1-CUL3 complex act as markers through the binding of ubiquitin E3 ligase, allowing the complex to be degraded by the proteasome.20,21 When the cell is in a state of oxidative stress or the levels of ROS in the cell increase, the activity of KEAP1-CUL3 ubiquitin E3 ligase decreases, and NRF2 is liberated from the previous complex, allowing NRF2 to be stable and accumulate in the cell, thereby entering the nucleus through nuclear translocation and activating the transcription of downstream antioxidant related genes. 22 However, NRF2 is a double-edged sword. In tumor cells, the antioxidative capability of NRF2 allows the cells to obtain better adaptability to environmental changes, which can be beneficial to their development and progression.6,23 Additionally, tumor cells may also develop resistance to antitumor drugs because of NRF2,24,25 making drug treatment of tumors more challenging. 26 As a result, NRF2 is increasingly considered a potential target for tumor prevention and therapy.9,27-29
CXX-3 is a norditerpene lactone extract from P. nakaii, which has been previously reported to exhibit strong pharmacological activity.30-32 In this work, we first investigated the toxicity of CXX-3 in two cell lines and found that it has a good inhibitory effect on tumor cells at a low concentration. This is the first report that the extract isolated from P. nakaii has an antitumor active. Then, we explored its impact on the cell cycle using flow cytometry. The results showed that CXX-3 treatment significantly increased the percentage of S-phase cells compared to G0/G1 and G2/M phases in both glioma U251 and colon carcinoma cell line HCT-116. Therefore, we detected CyclinD1 and CDK2, which are key proteins of S-phase arrest of the cell cycle. We observed that their expression was downregulated by CXX-3, which could explain the cell cycle arrest in S-phase. The accumulation of ROS in cells can affect the cell cycle by activating growth factor receptors and affect cell cycle regulators via phosphorylation and ubiquitination. 14 Therefore, we detected the intracellular ROS levels by flow cytometry and found that the intracellular ROS levels increased in a dose-dependent manner after CXX-3 treatment. Furthermore, the cell death and increased ROS levels caused by this effect can be partially rescued by the ROS inhibitor NAC. Considering that NRF2/KEAP1 complex plays a vital role in cell antioxidation, we detected the expression of NRF2 and its downstream client protein HO-1 and GCLM in two cell lines through western blotting. The results indicated that NRF2 increased in a dose-dependent manner, but the expression of KEAP1 client protein was lessened, suggesting that CXX-3 may block the effective pathway of NRF2. Moreover, the nucleocytoplasmic separation assay revealed that the expression of NRF2 was upregulated in the nucleus of U251. In contrast, in HCT-116, the expression of NRF2 was only elevated outside the nucleus and there was no trace of NRF2 in the nucleus. Therefore, we speculated that CXX-3 may interfere with transcription in the nucleus directly or affect the binding of NRF2 to ARE/EpRE in U251. Additionally, CXX-3 may disable the antioxidant capacity of the cells by blocking the nuclear translocation channel of NRF2. 33
In this research, we proved that CXX-3 can increase ROS levels in cells by disrupting the antioxidant capacity of NRF2 and arresting the cell cycle in the S phase. Furthermore, after investigating the expression of intranuclear and extranuclear proteins, we found that CXX-3 may affect NRF2 in various ways. Also, we observed the antitumor activity of the P. nakaii for the first time, which to a certain extent offers the possibility of its use beyond ornamental plants. Moreover, this study discussed for the first time the possible causes of NRF2 inactivation inside and outside the nucleus induced by natural compounds for the first time. CXX-3 has good pharmacological activity and targets KEAP1/NRF2, suggesting that it has the potential to be a precursor for tumor drug-resistant cells. The specific mechanism of CXX-3 interfering with NRF2 remains a complex issue and further studies are needed to investigate the effectiveness and safety of CXX-3 in animal experiments to obtain a potential tumor therapeutic drug. However, there are still two limitations in our research. First of all, this study focused on the efficacy and mechanism in vitro. Due to the limitations of the drug source and extraction process, we failed to carry out further experiments in animal models to verify the mechanism, effectiveness and safety of CXX-3 in vivo. Secondly, we failed to further clarify how CXX-3 affects the entry and exit of the KEAP1-NRF2 complex inside and outside the nucleus from the molecular mechanism. It is hoped that in future research, we can overcome these two difficulties to explore the potential of CXX-3.
Conclusion
This study supports that CXX-3 can inhibit the proliferation of U251 and HCT-116 cells in vitro by arresting the cell cycle and increasing ROS levels. In addition, CXX-3 can disable the KEAP1/NRF2 complex, leading to cancer cell death. In summary, our findings suggest that CXX-3 may be a promising compound for the treatment of cancer.
Footnotes
Authors’ Notes
Jingtao Yuan and Hang Gao contributed equally to this work.
Data Availability
All data used to support the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable to this article.
Funding
This work was financially supported by the National Natural Science Foundation of China (82104448), the Zhejiang Chinese Medical University Research Fund Project (2021ZR10) and the Scientific Research Fund of Zhejiang Provincial Education Department (Y202251034).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
