Abstract
Coldenia procumbens L. (family Boraginaceae, genus Coldenia) is a medicinal plant traditionally used to treat various ailments. It has been indicated in the indigenous system of medicine for treating rheumatic swellings, boils, pains, immature abscesses, wounds, menorrhagia, and snake bites. This study aims to summarize up-to-date botany, traditional uses, phytochemical compounds, pharmacological activities, and toxicology. The relevant information about the C. procumbens plant, such as ethnobotany, phytochemistry, pharmacology, and toxicology, was collected via several scientific databases (Web of Science, Scopus, Google Scholar, ScienceDirect, SpringerLink, PubMed) and online databases of botanicals (Plants of the World Online, Flora of China, eFlora of India, Biodiversity in India, World Flora Online, Synonymic Checklist and Distribution of the World Flora). Modern pharmacological studies have revealed that C. procumbens exerts antioxidant, antimicrobial, anti-diabetic, anti-inflammatory, anti-arthritic, analgesic, anti-cancer, anti-hyperlipidemic, and hepatoprotective activities. More than 100 phytoconstituents have been discovered, including terpenes, sterols, coumestan, nitrile/cyano glucosides, fatty acids, and other compounds. This study has attempted to comprehensively describe C. procumbens in terms of ethnobotany, phytochemical profile, and pharmacological effect. These specific and valuable suggestions warrant further investigation to utilize their inherent potential for novel product development and immense utilization in medicine, cosmetics, and health supplements.
Introduction
Coldenia L. is a genus of the Boraginaceae family, represented by plant species of medicinal importance due to their benefits in traditional and modern medicine systems. The Coldenia genus is predominantly tropical and subtropical in the Old World and presents only one species, namely Coldenia procumbens L. 1 C. procumbens, known as “Creeping Coldenia”, is an annual herb that grows primarily in the seasonally dried tropical biome and is widely distributed in Asia, Africa, Australia, North America, and South America.1,2 It can now be found in many countries, such as China, Cambodia, India, Indonesia, Malaysia, Pakistan, Sri Lanka, Thailand, and Vietnam.2–4
Traditional medicine systems in some countries, especially Ayurveda in India and the Chinese system of medicines have used the C. procumbens plant to treat various diseases, such as rheumatic swellings, boils, pains, fever, piles, leucorrhoea, and menorrhagia.5–9 In particular, this plant is also used as an antidote for snake bites and scorpion stings.5–9 All these effects of the C. procumbens plant on neglected various diseases are significant for traditional medicine systems, which are traditional herbal medicines that have been used for a long time. The herb has been evaluated and investigated more profoundly in modern medicine through phytochemistry, pharmacology, and toxicity studies. It has been reported that the leaves and whole plants are important natural sources of molecules with therapeutic properties, including hydrocarbons, triterpenoids, steroids, coumestan, nitrile/cyano glucosides, tocopherols, fatty acids and ester derivatives, indole alkaloids, and amides, which support their use in traditional medicine for treating various diseases.8–13 Depending on the plant part and collection site, phytochemical analysis revealed different chemical compositions among several published studies. Moreover, C. procumbens has been explored for its pharmacological effects using crude extracts of different parts of this plant through in vitro and in vivo assays. Almost all of the parts of C. procumbens, mainly including the leaves, aerial parts, or whole plants, have been shown to have multiple pharmacological effects, such as antioxidant, antimicrobial, anti-diabetic, anti-inflammatory, anti-arthritic, analgesic, anti-cancer, anti-hyperlipidemic, and hepatoprotective effects.8,10 Although there is some published scientific literature on this traditional herb, scientific evidence concerning the pharmacological effects, phytochemistry, toxicity, and potential use of the C. procumbens plant is scarce. Therefore, this is the first study highlighting the recent pharmacological evaluations and summarizing the phytochemistry of the C. procumbens plant and its promising applications in modern medicine.
The current review aims (1) to provide updated information on the botany and traditional uses of C. procumbens; (2) to discuss the potential of C. procumbens as a source of phytoconstituents that can be applied in the treatment of microbial infections and inflammation-related diseases and the importance of this plant application in the pharmaceutical and cosmetic fields; and (3) to discuss toxicity and safety issues and proposals for conservation, cultivation, and development of this plant.
Research Methodology
The relevant information about C. procumbens was collected via major scientific databases such as the Web of Science, Scopus, Google Scholar, ScienceDirect, SpringerLink, and PubMed. The scientific names of the plants reported in the studies were confirmed with names available in botanical databases, including the Plants of the World Online (https://powo.science.kew.org/), the Flora of China (http://www.efloras.org/), the eFlora of India (https://efloraofindia.com/), the Biodiversity in India (https://indiabiodiversity.org/), the World Flora Online (https://www.worldfloraonline.org/), and the Synonymic Checklist and Distribution of the World Flora (https://www.worldplants.de/). Some terms or keywords used for collecting the published articles are “Coldenia procumbens”, its synonyms, traditional uses of C. procumbens, pharmacology, phytochemistry, and toxicological studies of C. procumbens. The published articles were retrieved from 1753 to April 2024. After collecting about 496 published articles, the results were analyzed and selectively grouped according to the theme, with 44 documents used for this review. The rest of the publications were excluded, considering the validity of the relevant content and the scientific proof.
The phytocompounds of C. procumbens were rechecked via the PubChem database (https://pubchem.ncbi.nlm.nih.gov/) and SpectraBase (https://spectrabase.com/). Their chemical structures were drawn using ChemDraw Professional 16.0.
Results and Discussions
Botany
Scientific Classification
C. procumbens L. is part of the Boraginaceae family. The genus Coldenia L. belongs to the Boraginaceae family, the Boraginales order, the Magnoliidae subclass, the Equisetopsida class, the Tracheophyta phylum, and the Plantae kingdom. According to synonym nomenclature, C. angolensis Welw., Lobophyllum tetrandrum F.Muell., and Waltheria microphylla Miq. ex C.B.Clarke are considered to be heterotypic synonyms of C. procumbens.1,14
Morphological Features and Geographical Distribution
Morphological description (Figure 1): Morphologically, C. procumbens is an annual herb, usually lying quite flat on dry ground. Stems and leaves are softly white and villous. Stems branched from base; branches often numerous, prostrate or ascending, 15–40 cm, densely spreading strigose. Lower stem leaves short petiolate, oblong to obovate or subpinnatifid, 1(1.5)–2(3) × 0.5–1.5 cm, asymmetrical, scabrous, strigose, margin coarsely crenate-lobulate or apex emarginate; lateral veins 4–6 pairs, prominent abaxially, concave adaxially; upper leaves sessile. Flowers are solitary and submissive. Calyx 4-lobed, ca. 1.5–2.0 mm; lobes lanceolate to ovate-lanceolate or lobes ovate-acute, ciliate. Corolla white, ca. 2.0–2.5 mm, glabrous, tube ca. 1.0 mm long; 4-lobes orbicular, ca. 0.3 mm. Stamens 4-included; filaments ca. 0.3 mm, glabrous, inserted at the middle of the corolla tube; anthers rounded to broadly ovate, ca. 0.2 mm. Ovary fastigiate, 4-sulcate; stigma obscurely 2-cleft. Fruit broadly triangular, drupe of 4 subconnate pyrenes, 3.0–4.0 mm, glandular-pubescent; mericarps wrinkled, spinescent-tuberculate. Flowering in April, fruiting in June.2–4,9,15

The morphological features of C. procumbens (Photos by Nguyen Hoang Nam).
Distribution and habitat: C. procumbens is an annual and grows primarily in the seasonally dried tropical biome at altitudes up to 750 m. Particularly, it grows in sandy places, beaches, and dry cultivated ground. C. procumbens is found in the Andaman Islands, Angola, Assam, Bangladesh, Benin, Borneo, Burkina, Cambodia, Cameroon, Cape Verde, Central African Republic, Chad, Djibouti, East Himalaya, Egypt, Ethiopia, Gambia, Ghana, Guinea, Guinea-Bissau, Hainan, India, Jawa, Kenya, Laos, Lesser Sunda Islands, Madagascar, Malaya, Mali, Maluku, Mauritania, Mozambique, Myanmar, Nepal, New Guinea, Niger, Nigeria, Northern Provinces, Northern Territory, Pakistan, Queensland, Senegal, Somalia, Sri Lanka, Sudan, Taiwan, Tanzania, Thailand, Uganda, Vietnam, West Himalaya, Western Australia, Zambia, Zaïre, and Zimbabwe.1,2
Ethnobotanical uses
C. procumbens is commonly known as “Creeping Coldenia” in English but has its name in each region, 8 where the Vietnamese and Chinese names are “Cáp Điền” 9 and “双柱紫草” (Shuang Zhu Zi Cao), 2 respectively, whereas vernacular names in India include “Tripakshee” (Sanskrit), “Tripungkee” (Hindi), “Tripunkhi” (Assamese), “Hamsapadu” (Telugu), and “Seruppadai” (“செருப்படை”), “Cherupadi” (Tamil), “Cherupulladi”, or “Nilamparanda” (Malayalam).3,7,8
C. procumbens has a history of medicinal use in various regions, particularly in India. Siddha properties and action of C. procumbens were described as pungent in taste, hot in nature, and stimulant action. 10 The whole plant of C. procumbens can be used medicinally in alternative medicine for a wide variety of diseases, such as rheumatic swelling, immature abscesses, leucorrhoea, menorrhagia, anti-diabetic, anti-arthritic, and hypertensive. 16 In India and Vietnam, a poultice of the C. procumbens fresh leaf is used for rheumatic swellings.8,9,17 Additionally, the dried powder of the plant is crushed, mixed with seeds of Fenugreek (Trigonella foenum-graecum L.) or seeds of Purple Fennel (Foeniculum vulgare Mill.), with equal parts, and applied in poultices to treat boils as well as to relieve pains. The fresh leaf decoction and the mixture extracts (Centella asiatica (L.) Urb., Madhuca longifolia (L.) J.F. Macbr., and Ixora coccinea L.) are utilized to treat wound healing. The aqueous (Aq) extract of this plant in the form of a decoction is also utilized for treating fever, piles, leucorrhoea, and menorrhagia.5,6,8,9 Similarly, a powerful polyherbal Siddha preparation, named “Karisalai Karpa Chooranam” (Eclipta alba (L.) Hassk. or E. prostrata (L.) L., Wedelia chinensis (Osbeck) Merr., Indigofera tinctoria Chapm., Sphaeranthus indicus Kurz, C. asiatica, Acalypha indica K.Schum. & Hollr., and C. procumbens), is indicated for anemia, jaundice, hepatomegaly, generalized edema, and skin diseases and helps to enhance the immune system.18,19 Moreover, C. procumbens leaf juice has been used as an antidote for snake bites by Yanadi tribes in South India. 7 Particularly, C. procumbens is described as Serupadai or Serupadi in Siddha literature in an ancient Indian traditional treatment system (Traditional Tamil System of medicine), which means it is consumed either as “kudineer” or as powder.20,21 The “kudineer” is prescribed by Indigenous or traditional healers, and the usual dose is 3 to 6 g per day. 20
In Thailand, the vernacular name for C. procumbens is “Yah Teen Tukkae.” Many online websites have used images of Tridax procumbens L. (Asteraceae) to accompany articles about “Yah Teen Tukkae”. Alternatively, the scientific name of C. procumbens is sometimes paired with images of T. procumbens; these misunderstandings may be due to their shared vernacular name in Thai and the specific epithet “procumbens”. However, it's important to note that they are different species and belong to different families. This phenomenon raises an urgent issue in the application of identification and analytical methods for quality control and distinguishing counterfeit plants, such as the macro-morphological and micro-morphological methods, deoxyribonucleic acid barcode techniques, and chromatographic techniques (high-performance liquid chromatography (HPLC), gas chromatography-mass spectroscopy (GC-MS), liquid chromatography-mass spectrometry), etc.
Phytochemical composition
Various methods were utilized to extract, isolate, purify, and identify the phytochemicals from C. procumbens. In previous studies, the preliminary phytochemical analysis showed the presence of glycosides, alkaloids, steroids, terpenoids, proteins, amino acids, flavonoids, and phenols in the leaf extract of C. procumbens.11,12,15,22,23 A total of 110 phytoconstituents have been determined from this plant. The secondary metabolites mainly include hydrocarbons, triterpenoids, steroids, coumestan, nitrile/cyano glucosides, tocopherols, fatty acids and ester derivatives, indole alkaloids, amides, and other compounds.24–30 A list of the phytocompounds isolated and identified from C. procumbens is summarized and presented in Table 1 and Figures 2-8.

The structures of coumestan and nitrile glucosides isolated from C. procumbens.
Phytoconstituents from Different Parts of C. procumbens.

Coumestan
From 2011 to now, one coumestan derivative, namely wedelolactone
Nitrile glucosides (Cyano glucosides)
In 2013, three nitrile glucosides compounds, including ehretioside A1
Triterpenoids
Two pentacyclic triterpenoids, named α-amyrin

The structures of triterpenoid compounds isolated from C. procumbens.
Steroids
Interestingly, steroids and their derivatives are common chemical constituents of this species, and C. procumbens contains about nine steroids isolated and identified from the aerial parts and leaves of the plant, including β-sitosterol

The structures of steroid compounds isolated from C. procumbens.
Hydrocarbons
In 2020, Rani and colleagues
28
investigated the chemical profiling of C. procumbens leaves using GC-MS analysis, revealing the identification of thirteen phytoconstituents, namely 9-octadecene

The structures of steroid compounds isolated from C. procumbens.
Fatty acids and ester derivatives
In the leaves and aerial parts of C. procumbens, four fatty acids, named linoleic acid
Additionally, thirty-five fatty acid esters and their ester derivatives have been found in the leaves and aerial parts of C. procumbens. These fatty acid esters and their derivatives include methyl tetradecanoate

The structures of fatty acids and ester derivatives identified from C. procumbens.
Tocopherols
Two tocopherols, namely β-tocopherol

The structures of tocopherols, indole alkaloids, and amides identified from C. procumbens.

The structures of other components identified from C. procumbens.
Indole alkaloids and Amides
Two indole alkaloids (named tryptamine
Others
The presence of other phytocomponents from the MeOH extract of C. procumbens aerial parts, including 2,2,7,7-tetramethyl-3,6-octanedione
Moreover, Revathi et al (2023)
29
analyzed the Aq extract of C. procumbens leaves growing wild in Tamil Nadu (India) and identified other phytocompounds, including N-[3-methylaminopropyl]aziridine
In particular, using the GC-MS method, Rani and colleagues demonstrated that hydrocarbons, sterols, saturated fatty acids, and unsaturated fatty acids accounted for 33.45%, 61.06%, 49.43%, and 50.57%, respectively, as the predominant phytoconstituents identified in the crude n-Hex extract of C. procumbens leaves. 28 Moreover, the sterol concentration in the lipoidal matter was higher than in the hexane extract of C. procumbens leaves. In contrast, the linoleic acid concentration was high in the crude extract and the lipoidal matter. 28
Pharmacological effects
Traditional knowledge regarding C. procumbens utilization is based solely on observations and practical experiences passed on through generations with little documented scientific evidence. Therefore, numerous studies have been conducted to understand the current relevance of traditional uses based on scientific evidence of biological effects.
Antioxidant effect
Several studies have determined the antioxidant potential of different extracts of C. procumbens, which is highly correlated to its total phenol and flavonoid contents. In 2010, Lavanya et al 31 reported that the MeOH extract of C. procumbens leaves exhibited 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging ability with a percentage inhibition of 76.26% at a concentration of 5000 μg/mL compared to quercetin (87.74%). However, this study showed that the ability to scavenge DPPH radicals was very weak, with a very high half-maximal inhibitory concentration (IC50) value of 750 µg/mL. 31 Moreover, the total antioxidant activity was found to be 0.2 mg equivalents of ascorbic acid. Meanwhile, the total phenolic content was 31.9 mg pyrocatechol equivalent per gram of extract. 31 When compared to quercetin (90.52% inhibition at 500 µg/mL), the C. procumbens MeOH leaf extract's reducing properties (88.82% inhibition at 500 µg/mL) were discovered to be dose-dependent. 31 In contrast, Beena and colleagues reported that the ethanolic (EtOH) extract from C. procumbens leaves showed moderate antioxidant activity in reaction with DPPH (IC50 = 121.3 ± 8.04 µg/mL) in comparison with the ascorbic acid standard (IC50 = 18.5 ± 1.48 µg/mL). 32 In addition, Beena and colleagues conducted another assessment of the antioxidant activity of this EtOH extract using nitric oxide radical scavenging activity. As a result of the study, this extract had weak antioxidant activity (IC50 = 313.5 ± 5.8 µg/mL) compared to the rutin standard (IC50 = 91.5 ± 3.3 µg/mL). 32
In another study, Ganesan et al (2014) 33 mentioned that the C. procumbens whole plant MeOH extract had significant antioxidant potential in the DPPH assay with a percentage inhibition of 53.60% at a concentration of 50 µg/mL (IC50 = 45 µg/mL) compared to the butylated hydroxyanisole standard (89.19%). 33 Additionally, Chellappandian et al 34 evaluated the antioxidant activity of the EtOH extract of C. procumbens aerial part through the DPPH assay. This extract showed more than 50% DPPH radical scavenging compared with the butylated hydroxytoluene (BHT)-positive control, which showed more than 90% DPPH radical scavenging. 34 Babu et al also reported that the good antioxidant capacity of the MeOH extract from C. procumbens aerial parts was found by the DPPH method. Its IC50 for scavenging DPPH radicals was 46.0 μL/mL, while the IC50 of gallic acid was 46.40 μL/mL. 35
Besides the DPPH assay, other antioxidant assays, including ferric-reducing antioxidant power (FRAP), 2,2′-azinobis(3-ethylbenzothiazoline 6-sulfonic acid) (ABTS), thiobarbituric acid reactive substances, and superoxide, were also determined by Ganesan et al (2014). 33 In the FRAP assay, the C. procumbens extract was shown to have 1000 µg equivalent to BHT/mg of sample. For the ABTS scavenging-radical assay, this extract showed weak ABTS scavenging activity with 90.18% inhibition at 500 µg/mL (IC50 = 200 µg/mL) compared to 90.72% inhibition for the gallic acid standard. At 500 µg/ml concentration, the superoxide scavenging abilities of BHT standard and C. procumbens extract (IC50 = 70 µg/mL) were observed at 92.91% and 72.77%, respectively. Similarly, C. procumbens extract was shown to have weaker anti-lipid peroxidation properties when compared to the naturally occurring antioxidant, vitamin E. Briefly, thiobarbituric acid reactive substances scavenging activity for Coldenia extract (IC50 = 1000 µg/mL) and vitamin E was 51.82% and 47.17% inhibition at 500 µg/mL concentration, respectively. Particularly since C. procumbens 100 µg/mL extract contains 12.0943 µg BHT equivalent of total phenol, it can be assumed that the possible secondary metabolite compounds that play a role in the antioxidant property of C. procumbens are due to phenolic compounds. 33
In a study by Suvarna et al (2017), 11 the MeOH extract of C. procumbens leaves collected from Telangana (India) was evaluated for its antioxidant capacities via the hydrogen peroxide radical scavenging assay and reducing power activity. The MeOH extract of C. procumbens leaves displayed an inhibition effect on hydrogen peroxide radical with inhibitory rates from 70.00 ± 0.282% to 75.00 ± 0.236% at different concentrations (20 µg/mL, 40 µg/mL, 60 µg/mL, and 80 µg/mL) and exhibited significant inhibition when compared with that of the positive control (74.10 ± 0.243%–80.00 ± 0.189%). 11 Similarly, the reducing power of this extract also depended on the concentration (20 µg/mL, 40 µg/mL, 60 µg/mL, and 80 µg/mL) with inhibitory rates from 27.90 ± 0.09% to 79.80 ± 0.266%. In contrast, the reducing ability of ascorbic acid had inhibitory rates from 21.9 ± 0.073% to 78.30 ± 0.261%. According to the results of this study, the MeOH extract of C. procumbens exhibited significant inhibition of reactive oxygen species, hydrogen peroxide, and reducing ability. 11
A study by Babu et al (2021) 35 also reported significant radical scavenging of MeOH extract of C. procumbens aerial parts, with IC50 values of 47.90 μL/mL for ferric reducing power, 49.20 μL/mL for ABTS radical cation scavenging, 46.90 μL/mL for superoxide scavenging, and 49.0 μL/mL for phenolic content, as compared to that of BHT (IC50 = 48.20 μL/mL), butylated hydroxyanisole (IC50 = 48.20 μL/mL), ascorbic acid (IC50 = 48.50 μL/mL), and vitamin C (IC50 = 48.50 μL/mL), respectively. 35
Recently, Revathi et al (2023) 29 showed that the antioxidant potential of the crude Aq extract from C. procumbens leaf was determined against DPPH, FRAP, and H2O2 free radicals. At 90 mg/mL concentration, this Aq extract exhibited DPPH, FRAP, and H2O2 radicals scavenging activity of 69.0%, 25.0%, and 73.0%, respectively. 29 The antioxidant activities of the positive controls, ie, tannic acid for the DPPH assay, BHT for the H2O2 assay, and ascorbic acid for the FRAP assay, were 99.0%, 98.0%, and 94.0%, respectively. 29
These antioxidant findings are further supported by previous studies, which revealed that this plant possesses different antioxidant activities that may be due to the assay method, extraction solvent, harvest season, location of sample collection, different parts of the plant, etc.
Antimicrobial effect
Numerous investigations on the antimicrobial effects of C. procumbens extract have lately been done in an endeavor to substantiate a few of the more common uses of this plant, such as white vaginal discharge caused by yeast and bacterial infections.36,37 The antimicrobial activity of C. procumbens extract was tested by the agar well diffusion method using fungi (Candida albicans, Aspergillus sydowii, A. raperi, A. terreus, and Fusarium sp.), Gram-negative bacteria (Aeromonas hydrophila, Proteus vulgaris, P. mirabilis, Enterobacter aerogenes, Escherichia coli, Klebsiella pneumoniae, Salmonella typhi, S. typhimurium, S. paratyphi A, S. paratyphi B, Shigella sp., Vibrio fischeri, and Pseudomonas aeruginosa), and Gram-positive pathogenic bacteria (Bacillus subtilis, Bacillus cereus, Streptococcus pyogenes, and Staphylococcus aureus).
Ramakrishnan et al (2011) 37 reported strong antibacterial activity of EtOH and Aq extracts of C. procumbens leaves against S. aureus and S. pyogenes, with relative inhibition zone diameters of 16-22 mm and 13-16 mm, respectively. The Aq extract of C. procumbens leaves displayed the lowest zone of inhibition (ZI) against S. typhi (ZI = 12 mm) and E. coli (ZI = 7 mm), while the EtOH extract of C. procumbens leaves showed no inhibition against S. typhi and E. coli. Furthermore, no antifungal activity was observed for the EtOH and Aq extracts of this plant. 37 This suggests that its efficacy against several tested bacteria may be limited.
In a study by Shakila et al (2017), 38 the EtOH extract of C. procumbens whole plant was evaluated for its antimicrobial properties against 13 pathogenic bacteria and fungi. The EtOH extract was less active at concentrations from 15.625 to 500 mg/mL with ZI of 10-27 mm (B. subtilis MTCC 441), 10-25 mm (E. coli ATCC 25922), 16-22 mm (K. pneumoniae NCIM 2957), 8-13 mm (P. aeruginosa NCIM 2945), 6-18 mm (S. typhimurium NCIM 2501), and 5-20 mm (C. albicans MTCC 227). It can be concluded that this concentration range is too high for the EtOH extract of C. procumbens whole plant to be considered active. Moreover, the EtOH extract of C. procumbens whole plant showed no inhibition against A. hydrophila (ATCC 7966), B. cereus (NCIM 2458), E. aerogenes (NCIM 5139), P. vulgaris (NCIM 2857), S. aureus (NCIM 5021), S. pyogenes (ATCC 19615), and V. fischeri (ATCC 7744). 38
The results of the above two studies demonstrated that the EtOH extract of the C. procumbens whole plant in the study by Shakila et al (2017) 38 had mild antibacterial activity against C. albicans (ZI = 5-20 mm), while the leaf extract of this plant in the study by Ramakrishnan et al (2011) 37 had no inhibitory effect against C. albicans (ZI = 0 mm). On the contrary, the leaf extract had mild activity against S. aureus (ZI = 16-22 mm) and S. pyogenes (ZI = 13-16 mm), whereas this result was not observed with the EtOH extract of the C. procumbens whole plant.
Previously, Babu et al (2018) 39 noted that various solvent extracts, namely acetone, EtOH, MeOH, and Aq extracts from C. procumbens whole plant, displayed a wide range of antimicrobial activity at concentrations of 25-100 μL against B. cereus (ZI = 1-9 mm), E. coli (ZI = 1-14 mm), P. aeruginosa (ZI = 1-9 mm), S. aureus (ZI = 1-7 mm), A. sydowii (ZI = 4-15 mm), A. raperi (ZI = 3-17 mm), A. terreus (ZI = 2-10 mm), and Fusarium sp. (ZI = 4-14 mm). 39
In 2022, Satyanarayana et al 40 evaluated the antibacterial activity of the MeOH extract from C. procumbens leaves against strains of E. coli, K. pneumonia, P. aeruginosa, S. aureus, B. subtilis, and E. faecalis. It was observed that this extract weakly inhibited growth at concentrations of 25-100 µg/mL against E. coli (ZI = 6 ± 0.1-12 ± 0.1 mm), K. pneumoniae (ZI = 2 ± 0.3-7 ± 0.5 mm), P. aeruginosa (ZI = 1 ± 0.2-7 ± 0.5 mm), S. aureus (ZI = 4 ± 0.5-7 ± 0.4 mm), B. subtilis (ZI = 3 ± 0.4-6 ± 0.7 mm), and E. faecalis (ZI = 2 ± 0.4-7 ± 0.5 mm). 40 Similarly, according to Revathi et al, 29 the antibacterial activity carried out in the Aq extract of C. procumbens leaves at concentrations of 20 and 50 mg/mL against the tested bacterial strains revealed that it had very low antimicrobial activity. Briefly, at concentrations of 20 and 50 mg/mL, E. coli was inhibited at 8 ± 0.3 mm and 10 ± 0.5 mm, B. subtilis at 6 ± 0.5 mm and 9 ± 0.4 mm, Shigella sp. at 6 ± 0.5 mm, S. paratyphi A at 2 ± 0.3 mm and 4 ± 0.3 mm, S. paratyphi B at 5 ± 0.2 mm and 7 ± 0.7 mm, S. aureus at 4 ± 0.3 mm and 6 ± 0.5 mm, P. mirabilis at 5 ± 0.6 mm and 6 ± 0.2 mm, P. vulgaris at 3 ± 0.4 mm and 18 ± 1.5 mm, and K. pneumoniae at 7 ± 0.3 mm. 29 This suggests that its effectiveness against the tested bacterial strains may have a very weak antibacterial effect when very high extract concentrations are used.
In 2018, the anti-dermatophytic activity of the EtOH extract of the aerial part of C. procumbens against various dermatophytes, including T. rubrum (TRCl-1), T. rubrum (TRCl-2), T. mentagrophytes (TMCl-1), T. mentagrophytes (TMCl-2), T. simii, T. tonsurans, and Microsporum gypseum, was reported by Chellappandian et al 34 based on in vitro anti-dermatophytic activity and cytotoxic activity using tests such as the determination of the minimum inhibitory concentration (MIC)/minimum fungicidal concentration (MFC) assay and the sulpho-rhodamine B assay on 3T3-L1 fibroblast cells, respectively. As a result of this study, the aerial part of C. procumbens extract showed maximum inhibition on all tested dermatophytes. Notably, C. procumbens displayed MIC and MFC values ranging from 62.5 to 500 μg/mL and 250 to 2000 μg/mL against the tested strains, compared to fluconazole (MIC = 1.0-16 μg/mL). In other words, prominent anti-dermatophytic activity against T. mentagropytes (TMCl-1) was shown by C. procumbens extract (MIC = 62.5 μg/mL and MFC = 250 μg/mL). Moderate activity against T. mentagropytes (TMCI-2) (MIC = 125 μg/mL and MFC = 500 μg/mL), M. gypseum (MIC = 125 μg/mL and MFC = 250 μg/mL), and T. tonsurans (MIC = 125 μg/mL and MFC = 500 μg/mL) was found in this plant. However, the anti-dermatophytic activity of C. procumbens was observed in mild activity against T. rubrum (TRCl-1) (MIC = 500 μg/mL and MFC = 100 μg/mL) and T. rubrum (TRCI-2) (MIC = 500 μg/mL and MFC = 200 μg/mL). The cytotoxic activity of the C. procumbens aerial part displayed good cell viability up to 80% against 3T3-L1 fibroblast cells at tested concentrations (50 to 500 μg/mL). However, the higher concentration (500 μg/mL) showed 50% cell death. Interestingly, C. procumbens extract showed significant inhibition of ergosterol synthesis in dermatophytic fungi at a concentration of 16.37 μg/mL with more than 80% cell death due to disruption of membrane integrity, compared to fluconazole (35 μg/mL). 34 It suggests that C. procumbens extract has a stronger ability to inhibit ergosterol synthesis than a well-known antifungal drug, fluconazole. Ergosterol, the major sterol of fungal membranes, is essential for defining membrane fluidity and regulating cellular processes.41,42 Particularly, ergosterol is the main target of antifungals (eg, allylamines, azoles, and morpholines), which have been known for decades. 43 Ergosterol, through its protective role against mechanical and oxidative stress, suggests ergosterol as an antioxidant molecule that prevents phospholipid oxidation in the cellular context.44,45 Therefore, ergosterol may be the most efficient molecule to both provide structural functions to the fungus plasma membrane and to protect it from oxidation.44,45 C. procumbens extract reduced ergosterol content in the tested Trichophyton spp. and Microsporum gypseum strains, indicating that interference with ergosterol synthesis caused cell membrane disruption (Figure 9). The result of the study shows that C. procumbens is a promising candidate for use in cosmeceutical technology as an alternative solution for controlling skin disease-causing fungus.

All trials looked at, particularly, showed that the various plant extracts were effective against the different tested microorganisms. Extensive studies are needed to explore the antimicrobial action of various part extracts of C. procumbens and to identify the phytocomponents responsible for their antimicrobial properties.
Analgesic effect
Several reports have demonstrated the analgesic activity of C. procumbens extract.46,47 Senthamarai et al (2001) 46 investigated the analgesic activity of benzene, chloroform (CHCl3), acetone, and EtOH extracts from C. procumbens leaves collected in India using male Albino rats under in vivo conditions, such as the tail clip and hot plate assays. The results revealed that the extracts (200 mg/kg) exhibited marked analgesic activity compared to standard morphine sulfate (5 mg/kg) in both methods (p < 0.01). The basal reaction time was increased, similar to morphine sulfate. 46 This study identified that the possible mechanism may be due to the involvement of opioid receptors and the enhancement of thresholds for painful stimuli. 46
In a recent study, the MeOH extract of C. procumbens whole plant was also investigated for its possible analgesic activity on in vivo methods like hot plate, tail-flick, tail-immersion, and acetic acid-induced writhing methods at doses of 200 and 400 mg/kg body weight (b.w.). 47 In the hot plate, tail-flick, and tail-immersion assays, the studies were designed into 4 experimental groups (n = 6), including the normal saline control (10 mL/kg, i.p.), pentazocine (10 mg/kg, i.p.)-treated, C. procumbens (200 mg/kg, i.p.)-treated, and C. procumbens (400 mg/kg, i.p.)-treated groups. Meanwhile, in the acetic acid-induced writhing method, groups 1 and 2 included 0.6% acetic acid (10 mL/kg, i.p.) and aspirin (20 mg/kg, i.p.), respectively.
In the hot plate method, the results indicated that the MeOH extract of C. procumbens (at doses 200 and 400 mg/kg, i.p.) showed an increase in the mean basal time (p < 0.01). The highest nociception inhibition of stimulus exhibited by C. procumbens extract (200 mg/kg) was observed at 120 min (p < 0.01), which was also found to be effective with a reaction time of 5.6 ± 0.30 s. However, the 400 mg/kg dose (with a reaction time of 3.5 ± 0.42 s) was found to be ineffective in this analgesic model, compared to pentazocine (10 mg/kg, with a reaction time of 19.0 ± 0.85 s). 47
In the tail-flick method, this extract showed significant analgesic activity at a dose of 200 mg/kg (p < 0.01) with a reaction time of 5.9 ± 0.62 s at 15 min. However, the MeOH extract showed an increase in tail-flick twitch latency in mice for the 200 mg/kg dose after 60 min (with a reaction time of 5.2 ± 0.48 s) compared to the control group (with a reaction time of 3.6 ± 0.31 s) (p < 0.01). Moreover, a comparable result was observed in the C. procumbens-treated group (200 and 400 mg/kg) in the tail immersion method with a reaction time of 5.1 ± 0.31 s and 5.1 ± 0.48 s at 60 min, respectively, compared to the pentazocine-treated group (18.0 ± 1.29 s) and the control saline group (3.6 ± 0.31 s) (p < 0.01). 47
Additionally, the MeOH extract significantly reduced the number of abdominal constrictions induced by acetic acid (76.7% at 200 mg/kg and 75.0% at 400 mg/kg) compared to the standard aspirin (60.2% at 20 mg/kg) (p < 0.001). It can be concluded that C. procumbens can be used as an analgesic plant, with similar results to aspirin. In other words, at a single dose of 200 mg/kg b.w., the crude extract of C. procumbens may be an agent for giving very effective pain relief. 47
Anti-inflammatory and anti-arthritic effects
The aerial parts of C. procumbens have been traditionally used to treat pain and inflammation. Inflammation involves the proliferation of macrophages, neutrophils, and fibroblasts, which are the basic sources of granuloma formation. Mikami et al (1983) 48 reported earlier that the EtOH extract of C. procumbens has properties as an inhibitor of leukocyte migration and the formation of pleural exudates. 48 Moreover, it has also been shown to have anti-inflammatory effects. 49
In 2005, an in vivo study verified the EtOH extract of C. procumbens aerial part's ability to reduce inflammation effectively. The anti-inflammatory efficacy of this extract was assessed using carrageenan-induced acute paw edema and carrageenan-induced pleurisy in rat methods. The researchers divided 18 male Wistar albino rats into three groups (n = 6), including the normal saline control (1 mL, p.o.), indomethacin (10 mg/kg, p.o.)-treated, and C. procumbens (150 mg/kg, p.o.)-treated groups. After 1 h, the rats were injected subcutaneously (s.c.) and intrapleurally with 0.1 mL of 1.0% carrageenan. After 3 h, these rats were further tested to evaluate the anti-inflammatory effect depending on the experimental models.49,50 The EtOH extract of C. procumbens aerial parts showed that the inhibition percentage of paw edema, the inhibitory effect on leukocyte migration, and the reduction of pleural exudate were 59.5%, 0.48 ± 0.02 × 103 cells/mL, and 0.16 ± 0.002 mL, respectively, whereas these values for the standard drug (indomethacin) were 64.9%, 0.43 ± 0.08 × 103 cells/mL, and 0.12 ± 0.001 mL, respectively (p < 0.001). Particularly, the result of this study indicated that the EtOH extract also validated significant anti-inflammatory activity compared with the standard drug and untreated control (p < 0.001).49,50 Similarly, in the cotton pellet granuloma model, the animals were also divided into three groups, as described by Arul et al (2005).49,50 The animals were anesthetized with pentobarbitone (30 mg/kg, s.c.). An incision was made at the lumbar region of each rat. The EtOH extract of C. procumbens (150 mg/kg, p.o.) and indomethacin (10 mg/kg, p.o.) were used in each group for 7 days. The result showed that the granuloma weight was decreased significantly in rats of the EtOH extract-treated group (27.2 ± 1.19 mg) as compared with the control group (58.2 ± 2.04 mg) (p < 0.001). Meanwhile, treatment with indomethacin (10 mg/kg, p.o.) reduced a granuloma weight of 20.7 ± 0.65 mg (p < 0.001). The EtOH extract of C. procumbens and indomethacin inhibited granuloma tissue formation with percentage inhibitions of 53.3% and 64.4%, respectively.49,50
In a study by Lavanya and colleagues (2010), 31 the anti-inflammatory potential of the MeOH extract from C. procumbens leaves at concentrations of 31.25, 62.5, 125, 250, 500, 1000, and 2000 µg/mL was determined via the human red blood cell membrane stabilization method, using diclofenac sodium as a positive control. The MeOH extract of C. procumbens leaves possessed considerable anti-inflammatory activity at concentrations of 31.25, 62.5, 125, 250, 500, 1000, and 2000 µg/mL, which were found to be at 86.58%, 95.26%, 95.48%, 96.27%, 96.41%, 96.51%, and 98.09%, respectively, when compared with diclofenac sodium at concentrations of 250 µg/mL (82.74%), 500 µg/mL (88.39%), 1000 µg/mL (90.19%), and 2000 µg/mL (99.94%). 31 Similarly, the MeOH extract of C. procumbens leaves was reported in this study via an inhibition of protein denaturation assay. The MeOH extract of C. procumbens leaves (250 µg/mL) and diclofenac sodium (250 µg/mL) displayed significant anti-arthritic activity with percentage inhibitions of 52.84% and 94.22%, respectively. 31 Recently, Babu et al (2021) 35 showed significant anti-inflammatory properties of MeOH extract from C. procumbens leaves via in vitro egg albumin denaturation, human red blood cell membrane stabilization, and bovine serum albumin (BSA) denaturation activities, with IC50 values of 46.30, 46.70, and 47.40 μL/mL, respectively. In contrast, the standard drug, diclofenac sodium, had an IC50 value of 48.10 μL/mL. 35
According to relevant literature reports, previous research results have provided evidence supporting the traditional use of C. procumbens, thereby partly explaining the ethnopharmacology of this plant's “anti-rheumatic swelling” and “anti-arthritic” properties. C. procumbens extracts showed clear benefits in promoting anti-inflammatory processes. These promising pharmacological data prompted scientists to perform further studies to clarify the benefits of using C. procumbens in inflammation-related diseases.
Antidiabetic and anti-hyperlipidemic effects
Diabetes mellitus is a serious, lifelong metabolic disorder characterized by a person's blood glucose levels becoming too high, resulting in hyperglycemia.51–53 If not treated properly, diabetes can lead to several complications, such as coronary heart disease, stroke, cardiovascular disease, kidney disease, retinopathy, etc54,55 Recent data indicates that hyperlipidemia has been related to diabetes mellitus and its associated consequences, including diabetic retinopathy, in addition to its known association with cardiovascular disorders.56–58 In recent years, with the increasing rise in obesity, dyslipidemia, and diabetes, scientists have been constantly looking for herbal sources in the management and treatment of obesity, hyperlipidemia, and diabetes. Plant-based herbs have always been attractive to humans for treating hyperlipidemia and diabetes.
Patel et al (2007) 59 investigated the antidiabetic activity of the Aq extract of C. procumbens whole plant on alloxan-induced diabetic rats. In overnight-fasted rats, alloxan monohydrate (180 mg/kg b.w.) was injected intraperitoneally (i.p.) to induce diabetes. The oral administration of the Aq extract of C. procumbens whole plant in diabetic rats at a dose of 100 mg/kg reduced blood glucose levels from 394.17 ± 10.52 mg/dL to 152.83 ± 2.15 mg/dL in tested rats (p < 0.05) after 7-day treatment when compared with the diabetic control group of rats. Meanwhile, treatment with glibenclamide (5 mg/kg b.w.) also reduced blood glucose levels from 389.17 ± 2.21 mg/dL to 169.33 ± 2.81 mg/dL in tested rats (p < 0.05). The results differed significantly from the positive group (p < 0.05). Notably, the treatment with C. procumbens also reduced triglyceride (109.17 ± 1.74 mg/dL) and cholesterol (44.33 ± 1.96 mg/dL) levels significantly in the treated diabetic rats, compared to the positive and control groups (p < 0.05). The data showed that treatment with C. procumbens whole plant has antihyperglycemic and anti-hyperlipidemic effects. 59
Recently, Rethinam and colleagues (2020)
25
established that the MeOH extract from C. procumbens whole plant had lower blood glucose-reducing capability (246 ± 103 mg/dL at a dose of 200 mg/kg, b.w.) than glibenclamide (159 ± 48 mg/dL at a dose of 25 mg/kg, b.w.) in streptozotocin-induced Wistar albino rats. However, compared to the dose of 400 mg/kg, the blood glucose-lowering effect of this extract (160 ± 45 mg/dL) was equivalent to that of the glibenclamide group. In other words, the whole plant MeOH extract (200 and 400 mg/kg, per os/orally (p.o.)) was found to reduce blood glucose levels in both streptozotocin-mediated diabetic rats (246 ± 103 mg/dL and 160 ± 45 mg/dL) and normal control rats (133 ± 21 mg/dL). Moreover, the treatment with C. procumbens extract whole plant also reduced total cholesterol (57 ± 18 mg/dL and 59 ± 12 mg/dL, respectively, at 200 and 400 mg/kg) levels significantly in the treated diabetic rats, compared to the normal control group (61 ± 7.4 mg/dL), disease group (69 ± 5.0 mg/dL), and positive group (55 ± 9.4 mg/dL). Additionally, the study identified the major phytocompounds, namely wedelolactone
Hepatoprotective effect
Studies have shown that C. procumbens extract reported through different models, such as in vitro and in vivo studies, will cause hepatoprotective activity. Beena et al (2011) 32 conducted an in vitro study to assess the potential of the EtOH extract of C. procumbens leaves in hepatoprotective activity on the BRL-3A cell line model using antitubercular drugs, namely galactosamine HCl and the mixture of isoniazid, rifampicin, pyrazinamide (1:2:5) as a toxicant and silymarin as a standard drug by MTT ((3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide)) assay. As a result of this study, it was found that C. procumbens extract markedly increased the hepatoprotective effect at concentrations of 125, 250, 500, and 1000 µg/mL against anti-tubercular drugs by MTT assay, with protection percentages of 56.47%, 67.84%, 77.64%, and 80.0%, respectively, when compared with silymarin (96.47% protection at a concentration of 250 µg/mL) and the mixture hepatotoxicants of isoniazid, rifampicin, and pyrazinamide (500 µg/mL). Similarly, in comparison to silymarin (97.14% protection at a concentration of 250 µg/mL) and hepatotoxicant drug (40 µg/mL), the hepatoprotective activity of C. procumbens extract against galactosamine HCl by MTT assay showed the hepatoprotective percentages of 56.47% (125 µg/mL), 67.84% (250 µg/mL), 77.64% (500 µg/mL), and 80.0% (1000 µg/mL), respectively. 32
In an in vivo study, the CHCl3 extract of C. procumbens whole plant demonstrated the potential to protect the liver against diseases. 60 Administration of this extract at dosages of 200 and 400 mg/kg for 11 days in D-galactosamine (D-GalN)-induced acute liver damage rats reversed the levels of serum enzymes. They caused a subsequent recovery towards normalization; however, significantly increasing the total protein level in a dose-dependent manner. 60 Specifically, the levels of serum enzymes in D-galactosamine (D-GalN)-induced acute liver damage rats, namely aspartate aminotransferase (AST) (841 ± 5.75 U/L), alanine aminotransferase (ALT) (95.0 ± 5.19 U/L), alkaline phosphatase (ALP) (624.3 ± 31.25 U/L), total protein (8.48 ± 0.10 g/dL), albumin (2.95 ± 0.11 g/dL), globulin (5.23 ± 0.10 g/dL), total cholesterol (81.0 ± 6.92 mg/dL), and total bilirubin (1.43 ± 4.02 mg/dL), were noted. Meanwhile, the levels of serum enzymes in the C. procumbens-treated group at dosages of 200 and 400 mg/kg, including AST (134.33 ± 11.66 U/L and 130.66 ± 10.05 U/L), ALT (52.66 ± 3.74 U/L and 62.5 ± 3.41 U/L), ALP (513.5 ± 88.47 U/L and 433.16 ± 35.24 U/L), total protein (8.53 ± 0.42 g/dL and 8.36 ± 0.21 g/dL), albumin (3.03 ± 0.14 g/dL and 5.31 ± 0.10 g/dL), globulin (5.5 ± 0.46 g/dL and 5.2 ± 0.12 g/dL), total cholesterol (70.33 ± 5.22 mg/dL and 85.5 ± 5.85 mg/dL), and total bilirubin (0.48 ± 3.89 mg/dL and 0.35 ± 3.59 mg/dL) were significantly subsequent when compared with the D-GalN control (400 mg/kg) and the normal control (0.3% carboxymethyl cellulose, 5 mL/kg, b.w.) groups (p < 0.05, p < 0.01). 60 Furthermore, the results indicated that the C. procumbens extract (doses 200 and 400 mg/kg) displayed significant hepatoprotective activity via biochemical parameters and was comparable with the standard silymarin (100 mg/kg) with the levels of AST (171.83 ± 27.89 U/L), ALT (63.33 ± 9.93 U/L), ALP (389.33 ± 115.27 U/L), total protein (7.68 ± 0.33 g/dL), albumin (4.76 ± 0.14 g/dL), globulin (2.9 ± 0.28 g/dL), total cholesterol (59.33 ± 9.71 mg/dL), and total bilirubin (0.515 ± 4.06 mg/dL) (p < 0.05, p < 0.01). 60
In 2018, Ramachandran et al 61 conducted a study on the hepatoprotective activity of C. procumbens whole plant extract on 1,2-dimethylhydrazine (DMH)-induced liver damage in colon cancer male Wistar albino rats through antioxidant activity in liver tissue of this extract. The experiment was designed into 4 groups (n = 6), including the normal saline, DMH-treated, and C. procumbens (200 and 400 mg/kg)-treated groups. After the administration of DMH, the rats were given C. procumbens extract once daily for 30 weeks. 61 The results indicated that the dichloromethane extract of C. procumbens whole plant (at doses of 200 and 400 mg/kg, respectively) restored the levels of protein (0.5610 ± 0.01950 and 0.5633 ± 0.02186 mg/g tissue), catalase (CAT) (32.67 ± 0.2805 and 37.17 ± 0.2443 moles of H2O2 consumed/min/mg protein), glutathione (GSH) (99.53 ± 1.048 and 118.8 ± 3.627 g/mg protein), superoxide dismutase (SOD) (4.043 ± 0.06642 and 4.183 ± 0.1126 units/min/mg protein), glutathione peroxide (GPx) (189.9 ± 12.42 and 174.3 ± 8.486 g/mg protein), and lipid peroxidation (LPx) (85.17 ± 1.530 and 90.14 ± 1.609 MDA formed/mg protein) to the normal level in liver tissue when compared to the DMH-treated group (protein = 0.2700 ± 0.02646 mg/g tissue, CAT = 24.15 ± 0.5355 moles of H2O2 consumed/min/mg protein, GSH = 81.27 ± 4.816 g/mg protein, SOD = 2.437 ± 0.0581 units/min/mg protein, GPx = 73.74 ± 2.681, LPx = 68.14 ± 2.752 MDA formed/mg protein) (p < 0.001) and the normal control group (protein = 0.6433 ± 0.02906 mg/g tissue, CAT = 51.01 ± 1.324 moles of H2O2 consumed/min/mg protein, GSH = 130.1 ± 9.655 g/mg protein, SOD = 7.717 ± 0.1989 units/min/mg protein, GPx = 207.6 ± 5.485 g/mg protein, LPx = 92.15 ± 1.791 MDA formed/mg protein) (p < 0.001). 61 Moreover, the histopathological evaluation also showed that the dichloromethane extract had a protective effect on the liver due to the antioxidant properties of this plant. 61
A similar study by Rethinam et al (2020) 25 reported the hepatoprotective effects of MeOH extract from the C. procumbens whole plant (doses 200 and 400 mg/kg) in diabetic Wistar albino rats via tissue marker enzyme levels. The levels of serum ALT (163 ± 23 U/L) and ALP (467 ± 49 U/L) significantly increased in streptozotocin-induced rats. Whereas the groups treated with C. procumbens (105 ± 43 U/L for ALT and 248 ± 88 U/L for ALP at 200 mg/kg; 63 ± 19 U/L for ALT and 234 ± 79 U/L for ALP at 400 mg/kg) reduced considerably with positive group animals (64 ± 33 U/L for ALT and 210 ± 75 U/L for ALP) showing an almost two-to-three-fold decrease in serum ALT and ALP levels, compared to the normal control group (49 ± 16 U/L for ALT and 202 ± 85 U/L for ALP). 25
Based on the search results for publications related to this plant, the major components in the C. procumbens extracts have yet to be studied for their pharmacological effects. Therefore, the hepatoprotective activities of C. procumbens demonstrated in previous studies are still limited. Further study is required to validate the hepatoprotective properties of the major phytocompounds isolated from different parts of C. procumbens extracts.
Anticancer effect
The anti-cancer activity of the C. procumbens whole plant has been reported using DMH-induced colon cancer in male Wistar albino rats model. 61 The researchers divided 24 male Wistar albino rats into four groups of six rats each, including the normal saline, DMH-treated, and C. procumbens (200 and 400 mg/kg, b.w.)-treated groups. After the administration of DMH, C. procumbens extract (at doses of 200 and 400 mg/kg, p.o.) was used in each group for 30 weeks. The aberrant crypt foci and polyps, carcinoembryonic antigen, alpha-fetoprotein, and histopathological evaluations were recorded at the end of the 30th week. 61
The study demonstrated significant anticancer activity of the dichloromethane extract of C. procumbens whole plant against colon cancer in male Wistar albino rats at both tested doses, especially at the high 400 mg/kg dose. This extract (400 mg/kg, b.w.) was found to decrease the aberrant crypt foci and polyps (5.000 ± 0.5774), carcinoembryonic antigen (0.2177 ± 0.002404 ng/dL), and alpha-fetoprotein (0.4633 ± 0.01856 ng/dL) levels significantly in rats as compared with the DMH-treated group (p < 0.001). 61 Additionally, histopathological features also showed that the crude extract had a protective effect on the colon tissue in colon cancer rats due to the antioxidant properties of the plant via increased levels of SOD (3.93 ± 0.09849 units/min/mg protein), GSH (106.6 ± 12.89 g/mg protein), GPx (233.5 ± 14.17 g/mg protein), LPx (101.2 ± 1.273 MDA formed/mg protein), and CAT (28.41 ± 0.7664 moles of H2O2 consumed/min/mg protein) in the C. procumbens (400 mg/kg, b.w.)-treated group as compared with the DMH-treated group (p < 0.001), 61 suggesting that C. procumbens has promising application potential in promoting antitumor and anticancer activities. These results have guided researchers to investigate the anti-cancer mechanisms and activities of C. procumbens phytocompounds.
Toxicity and safety aspects
Previously, several studies have been performed to evaluate the toxicity of C. procumbens. The safety of the dichloromethane extract of C. procumbens whole plant at doses of 5.0 mg/kg, 50 mg/kg, 300 mg/kg, and 2000 mg/kg b.w. in Wistar albino rats was reported by Ramachandran and colleagues, and there was no mortality in the rats. 61
Interestingly, the various extracts of C. procumbens did not produce significant physical and behavioral changes (eg, motor activity, pupils, urination, salivation, skin color, etc) at doses of up to 2000 mg/kg of the extracts with no deaths, compared to the control group rats. 60 Particularly, in the subacute toxicity study of the MeOH extract from C. procumbens whole plant, there were no significant changes in organ weight of the three essential organs, including the liver, kidney, and pancreas, recorded in the treated groups when compared with the normal control groups. Moreover, the cytotoxicity of this extract via the microscopic structure of histopathology in Wistar albino rats was also recorded. The 400 mg/kg b.w. dose of MeOH extract from C. procumbens whole plant for 28 days did not provoke toxicity signs or normal polygonal cellular architecture with no abnormalities. 25 More studies could be conducted to evaluate the acute and chronic toxicity of various solvent extracts and used parts from C. procumbens in different experimental models.
In traditional and folklore medicine, C. procumbens has been widely used for its multiple pharmacological effects along with safety and non-toxicity properties, making it an effective remedy for various illnesses. Although these studies have yet to fully understand the mechanism, the current literature review could conclude that C. procumbens has important medicinal values and the potential for various treatments. Figure 10 illustrates phytochemical compounds, biological effects, and future research applications for different parts of C. procumbens.
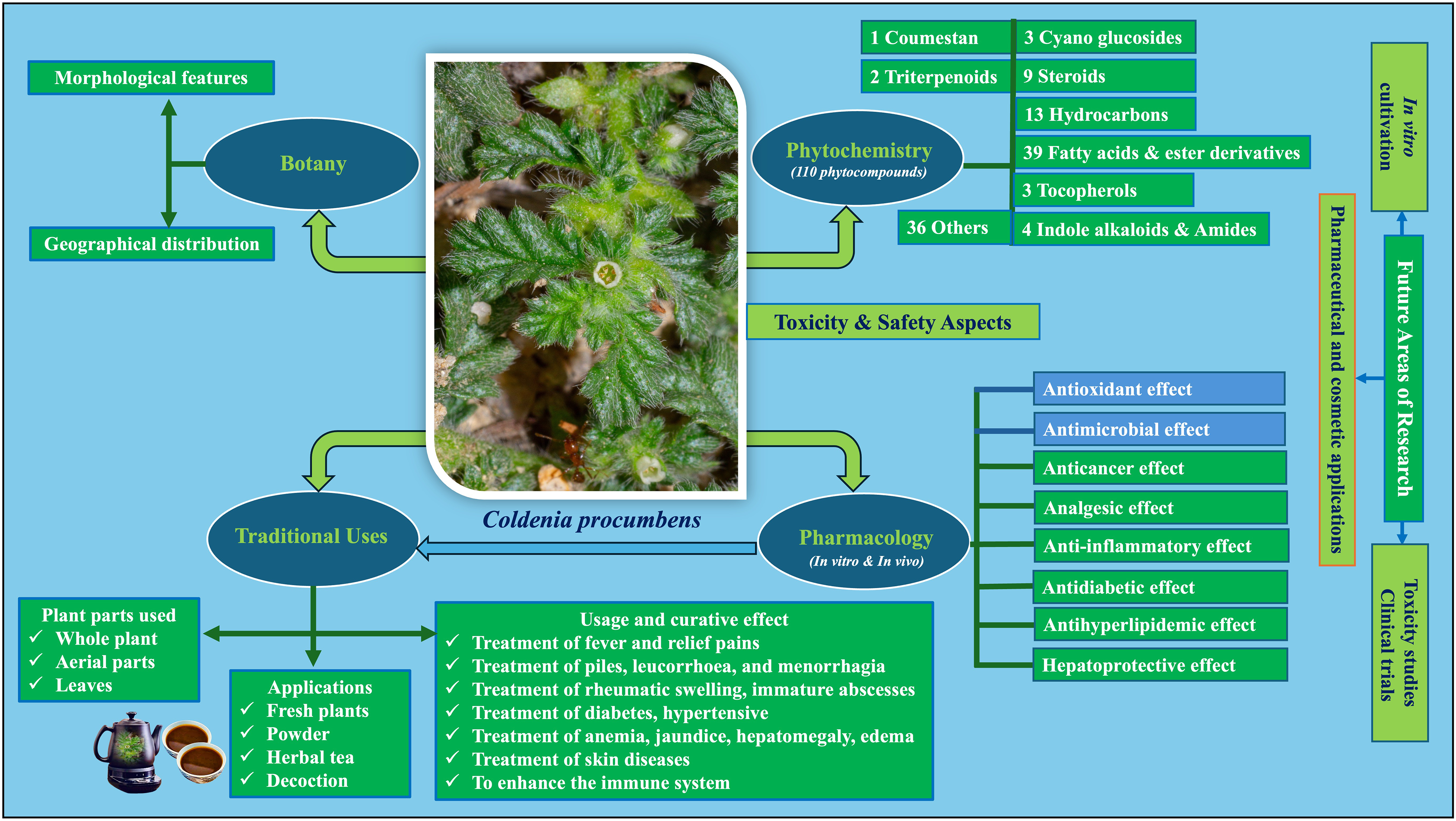
Ethnobotany, phytochemistry, pharmacological effects, and future studies of C. procumbens.
Limitations of the evidence and Recommendations
Searching for C. procumbens and its synonyms, 44 articles were identified. The results of these articles were not repeatedly confirmed. Therefore, this review shows the limitations of published studies and gives recommendations regarding C. procumbens.
Firstly, with such a limited number of studies, the medicinal values of C. procumbens parts are mainly accepted based on the traditional medicine perspective. Therefore, there is still a lack of significant evidence in the medical literature on this plant's pharmacological, pharmacokinetic, pharmacodynamic properties, and phytochemical profile. In another aspect, there are differences between the solvents used for extraction of different parts of C. procumbens (eg, n-Hex, CHCl3, dichloromethane, EtOH, MeOH, and Aq) in terms of phytochemical compositions, as well as pharmacological effects, and this partly explains the doubts about which solvent is best for preparing the extract. However, 110 chemical components have been identified from the different solvent extracts of C. procumbens parts. It doubts their significance, and coumestan, triterpenoids, and steroids are still considered the major bioactive constituents. In particular, some compounds, such as octadecanoic acid, benzyldimethylsilyl ester
Secondly, research reports on the chemical compositions and pharmacological effects of C. procumbens are still limited so far. Therefore, it is important to identify the active ingredients and clarify the mechanisms of action to confirm their pharmacological evidence.
Thirdly, there is no further evidence available in the literature reporting toxicity assessment except the reports of Ganesan et al (2013), 60 Ramachandran et al (2018), 61 and Rethinam et al (2020). 25 Thus, further research on the types of toxicity tests, such as acute and chronic toxicity, must be carried out to examine the therapeutic benefits and safety of C. procumbens. Clinical trials of C. procumbens are also needed to explore its efficacy and safety.
Finally, C. procumbens can be found in many countries (eg, India, Vietnam, Laos, Cambodia, etc) and is grown in various climates and soil conditions. Together with the available literature review, a study has yet to be conducted on the environmental factors affecting the active ingredients of this plant, so there is still a gap in research on cultivation techniques for C. procumbens. Therefore, researchers need to put more effort into studying environmental factors, growing area, and harvesting time because these factors can affect the bioactive ingredient content and phytochemical composition. Moreover, it is necessary to develop fingerprinting techniques that give insights into differences among cultivating conditions and main bioactive ingredients and provide the basis for herbal raw material quality control.
We believe that we can utilize the valuable medicinal values that traditional medicine has left for humans if we can thoroughly solve the above problems and further explore the medicinal potential of C. procumbens in traditional medicine systems. Additionally, we hope this evaluation highlights the significance of C. procumbens and provides some recommendations for the future development of this plant.
Conclusions
Medicinal herbs and their phytocompounds have been shown to significantly contribute to the prevention and treatment of several ailments with diverse pharmacological effects. Among them, C. procumbens has shown several promising pharmacological activities, including antioxidant, antimicrobial, anti-diabetic, anti-inflammatory, anti-arthritic, analgesic, anti-cancer, anti-hyperlipidemic, and hepatoprotective activities. These pharmacological effects are due to a variety of phytoconstituents, such as triterpenoids, steroids, coumestans, cyanoglucosides, and fatty acids. Encouragingly, ethnobotanical studies of C. procumbens have also reinforced the argument for the safe and effective use of this plant in treating several diseases through modern pharmacological evidence. Finally, the knowledge of C. procumbens summarised and presented here is a source of relevant information about this plant for application in functional food, pharmaceutical, and cosmetic fields.
Footnotes
List of Abbreviations
Acknowledgment
The authors thank Dr Van Thanh Tien Nguyen (Industrial University of Ho Chi Minh City, Vietnam) for his comments and corrections to this paper.
Authors’ Contributions
The research idea was provided by T.V.Chen. Analyzing, writing, and discussion were performed by T.V.Chen, N.T.Hieu, T.Boonma, and D.N.X.Lam. Reading and revising the manuscript were done by T.V.Chen and N.T.T.Hien. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the review research, authorship, and/or publication of this review article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
