Abstract
Introduction
Botanical nature-identical ingredients are substances that originate from a component of a plant, from the plant's essential oils, or from plant molecules that have been chemically synthesized. These substances are known to have antibacterial and immunomodulatory effects. 1 The Extracts from natural products are rich sources of bioactive chemicals and do not harm the environment (eco-friendly). As a consequence, the investigation of natural products in the pursuit of identifying novel bioactive metabolites has become an essential part of a significant number of drug development activities. 2 It is estimated that more than 70% of approved pharmaceuticals were either derived from or structurally related to chemicals that may be found in nature. 3 This suggests that a fast method for identifying novel plant extract chemicals is needed. However, screening biological matrices for novel modulators still needs to be improved. Nature is full of phytochemical chemicals that benefit human health. 4 The primary biologically active components of these naturally occurring products are typically present, and they may take the shape of tiny molecules, macromolecular polysaccharides, or other similar compounds. 5
These naturally occurring substances can be isolated from the byproducts of fermentation produced by bacteria or secondary metabolites produced by plants. 6 Extraction, separation, and purification methods are necessary for successfully isolating bioactive chemicals. 7 To ascertain their identities, these isolated components are analyzed using various analytical techniques, such as GC/MS, LC/MS, IR, NMR, etc. In vitro, in vivo, and even clinical tests are carried out to ascertain whether or not the identified targets have specific physiological effects to provide references for the prospective future creation of medicines, including healthcare compounds.6–8 The discovery of bioactive chemicals ushered in an exciting new era in the biotechnology field, which has important implications for people's health. 9 Because the primary goal of natural health products is to prevent or alleviate a variety of chronic and degenerative diseases, the therapeutic potential of natural health products could be proved by in vitro and in vivo biological activities. 10 Natural extracts, including byproducts, have the potential to serve as valuable substrates for the production of new products. 11 Bioavailability is one of their critical limiting variables through the in vitro and in vivo correlations for the actions (antioxidant, anti-inflammatory, or antibacterial) these compounds exhibit. 12 They ameliorate chronic disorders, and the administration of these medicines could have powerful benefits (for conditions such as type 2 diabetes, cardiovascular dysfunctions, or neurodegenerative diseases). 13 They are also capable of producing several secondary metabolites that have antimicrobial characterization, such as phenols, phenolic acids, flavonoids, alkaloids, tannins, quinones, coumarins, saponins, terpenoids, triterpenoids, glycosides, and organic acids. These antimicrobial properties are associated with the fact that they can manufacture these secondary metabolites. Creating and identifying phytochemicals and determining the physiological pathways involved in the health advantages they provide. 14
Numerous studies have found that the geographical, climatic, and grape types have a significant impact on the phenol content of grapes. 15 Polyphenols, composed of flavonoids (catechin and epicatechin), tannin acid, ellagitannins, dimeric, trimeric, and polymeric procyanidins, and acids of phenols like ellagic and Gallic acid, are some of the antioxidant-active substances that can be found in grape leaf. In addition, a range of other compounds. 16 Grape seeds also contain antioxidants such as catechin and epicatechin that reduce lipid and protein demolition and modify the state of the antioxidant enzymes tested. 17 phenolic ingredients and the positive effects of V. vinifera leaf contribute directly to the preventative impact on the mouse brain. In addition, it has been used in the therapy of a wide variety of disorders, including hypertension, 18 diarrhea, 19 bleeding, varicose veins, and inflammatory upheaval, 20 as well as to lower blood glucose levels. Additionally, it has been demonstrated that the leaves of V. vinifera provide hepatoprotection against the hepatic DNA damage generated by acetaminophen. 21 Their impact on the sporulation and survivability of E. papillate oocysts in vitro and in mice that have been infected. 22
The study aims to assess the bioactive components in V. vinifera leaf extracts and their possible in vitro protective effect against Eimeria columbae oocysts.
Materials and Methods
Obtain of Herbal Material
Fresh leaves from the herbal plant (V. vinifera leaf) were obtained from the wild areas of Riyadh, Saudi Arabia. Voucher specimens were deposited in the herbarium of the Botany and Microbiology Department, King Saud University, to verify their botanical identity.
Preparation of Extracts
Following the technique that was presented by Manikandan et al (2008). 23 The collected leaves were thoroughly washed to get rid of any undesired particles. Then, they were allowed to dry at room temperature for the subsequent examinations and grounded utilizing an electric blender. Then, 400 mL of methanol (25 mg/mL) was used to extract 10 g of powdered leaves, which were shaken for 24 h at room temperature. Whatman filter sheets were used to filter the leaf extract. Subsequently, the extracted material was dried and concentrated at 40 °C under reduced pressure using a rotating vacuum evaporator (Yamato RE300, Tokyo, Japan) for future use.
FT-IR Analysis of Leaf Extract
The phytochemical ingredients in V. vinifera leaf extract were determined by employing FT-IR using the protocol proposed by Kanthal et al (2014). 24 Once the processing stages were finished, a small amount of the material was homogenized with excessive potassium bromide powder (1: 99 wt%). After a coarse crushing process, the material was fed into a pellet-forming die. The NICOLET 6700 Fourier-transform infrared spectroscopy (FT-IR) optical spectrometer from Thermo Scientific was used to investigate the infrared spectrum. As a result, the most likely constituent classes might be predicted. At 25 °C, the FTIR spectra were acquired with a 4 cm−1 precision.
Leaf Extract GC-MS Analysis
The phytochemical ingredients of V. vinifera were identified utilizing GC-MS (Agilent Inc., Palo Alto, CA, USA). It was linked to a 5973 MSD operating in electron impact mode at 70 eV ion source energy. A DB-5MS GC column (30 m length, 0.25 μm inner diameter, and 0.25 m film thickness) was connected to the gas chromatograph (GC). Helium gas was used as a carrier at a constant flow of 1.0 mL min−1. Initial programming for the oven sets the temperature at 40 °Cd, with a two-minute hold. After that, the temperature was brought up to 200 °C at a pace of 5 °C each minute. After that, the temperature was raised to 300 °C at a rate of 5 °C per minute, where it remained for another two minutes. The temperature of the injector was set to 250 °C, while the detector's temperature was set to 275 °C. The total time the sample was allowed to run was 40 min. As the carrier, helium gas was utilized, and its flow rate was set at 1 mL/min, while the split flow rate was set at 25 mL/min, and the electron ionization energy was set at 70 eV. Utilizing the match percentages and commercial libraries provided by the National Institute of Standards and Technology (WILEY ninth edition, NIST-08 MS library, Gaithersburg, Maryland, United States), the presence of phytochemical components in the leaf extract was determined. The relative percentage amount for each element was calculated using the retention time index, and its average peak area was compared with the total peak areas. 24
A Potential Compound in Vitis vinifera Leaf Extract
The V. vinifera leaf extract's FT-IR and GC-MS analyses have identified 5 potential compounds that warrant further investigation and characterization for publication: Catechin is a flavanol compound with potent antioxidant properties. The catechol group in catechin is responsible for the strong absorption bands at 1209.1 cm1, which correspond to C-O tertiary alcohol stretching, according to FT-IR analysis. Gallic acid is a phenolic acid with anti-inflammatory, antimicrobial, and anticancer activities. A robust and wide absorption band was seen in the FT-IR spectrum at 1045.6 cm1 for CO-O-CO anhydride stretching. This could be linked to the carboxylic acid group in gallic acid. Quercetin is a flavonol compound with neuroprotective, cardioprotective, and anticancer effects. It was found that the FT-IR data had a strong absorption at 1510.70 cm1 for the stretching of the C = C aromatic ring. This is typical of the conjugated ketone and aromatic rings found in quercetin. Resveratrol is a stilbenoid with antioxidant, anti-inflammatory, and cardioprotective properties. An essential band in the FT-IR spectrum was seen at 1632.20 cm1 for C = C alkene stretching. This band might be related to the alkene group in the resveratrol molecule. Kaempferol is a flavonol compound with anti-inflammatory, antioxidant, and anticancer activities. The keto-enol tautomerism in the kaempferol structure could explain the medium absorption at 3382.1 cm1 for N-H stretching, as revealed by the FT-IR analysis.
Assessing of the Phenolic Content
The Mwamatope et al. (2020) Method was applied to estimate the total phenolic content (TPC) of the leaf extract, 25 100 µL of the sample with 100 µL of the Folin–Ciocalteu reagent, and 300 µL of a solution containing 20% sodium carbonate were mixed. After that, the sample was allowed to sit for 30 min at room temperature in a completely dark incubator. Utilizing a UV-visible spectrophotometer (SHIMADZU, UV-1800), it was determined that the absorbance was 765 nm. It was determined that the linear equation (y = 0.0021x + 0.0021 had an R2 value of 0.9995) for calculating the total phenolic content of the samples. To create a standard curve using gallic acid in values ranging from 25 to 400 µg/mL. The total amount of phenolic compounds was expressed as mg GAE/g DW for TPC.
Assessing the Tannin Content
The methodology that was presented by 26 was applied to determine the total Tannin content (TFC) of plant materials. 0.1 mL of the extracted sample was added to 1.5 mL of Milli-Q water and 0.1 mL of the Folin–Ciocalteu phenol reagent for 8 min. Then, 0.3 mL of a sodium carbonate solution that was 35% was added to the mixture so that it could be neutralized. Then, the ingredients were thoroughly combined and placed in a dark, room-temperature area for 20 minutes. The absorbance was measured at 700 nm. A calibration curve was developed using varying quantities of tannic acid standard to determine the total tannin content in the leaf extract, and the following equation (Y = 0.0013x + 0.0052 with R2 = 9937) was utilized. The total tannin concentration was determined and represented in mg TAE/g DW for TTC.
Assessing the Flavonoid Content
The method 27 was used to determine the total flavonoid content (TFC) in plant material V. vinifera. Methanol extract of 0.5 mL was mixed with a water solution containing 2% AlCl3 and having the same volume. The absorbance was measured at 420 nm at 25 °C after 2 h. The TFC was calculated using a calibration curve constructed using different concentrations (50-0400 g/mL) of quercetin standard and the following equation (y = 0.0172x + 0.0507 with R2 = 0.995). The curve was generated using the data from the previous step. The calculated TFC has been represented as quercetin (mg QE/g DW for TFC).
Cytotoxicity Evaluation (MTT Assay)
Cell Culture
The Hep-G2/2.2.15 Human Hepatoblastoma Cell Line was obtained from Sigma-Aldrich Chemie GmbH (Taufkirchen, Germany). The cell lines were grown in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% Fetal Bovine Serum (FBS) in a humid atmosphere with 5% CO2 at 37 °C.
An MTT test (3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide, cat#475989-1GM, Sigma-Aldrich, Germany) was used to check the plant extract V. vinifera's ability to kill cells. Briefly, at a 5 × 104 cells/ml density, cells were plated in a 96-well culture plate and left to grow for 24 h. Cells were then treated with extract concentrations of 15.625, 31.125, 62.5, 125, 250, and 500 μg/mL. Additionally, doxorubicin was utilized as a positive control. 10 μL of MTT solution, with a concentration of 5 mg/mL in phosphate-buffered saline (PBS), was added to each well after the incubation lasted 48 h. After that, 100 µl of acidified isopropanol was introduced into every well to dissolve the formazan product. The plate was then placed in a shaker for 10 min. Using a microplate reader (BioTek in the United States), the absorbance was determined at 570 nm.
28
The cell viability % was calculated as follows:
DPPH Radical Scavenging Assay
The 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay was used to test the antioxidant potential of the leaf methanolic extract of V. vinifera. In total, 1 mL of leaf extract ranging from 31.25 to 500 μg/mL was combined with the same volume of DPPH dissolved in methanol at a concentration of 0.135 μg/mL. After a vigorous round of vertexing, the ingredients were allowed to sit undisturbed at room temperature for half an hour. At a wavelength of 517 nm, the optical density of the extract and the control mixture (1 mL DPPH and 1 mL methanol) was measured. 29 Using the following formula, the percentage of DPPH scavenging activity possessed by the extract or standard was estimated:
DPPH scavenging activity (%) = [(absorbance of control absorbance of the sample)/absorbance of control] *100, the absorbance of control is the absorbance of methanol plus DPPH and the absorbance of the sample is the absorbance of DPPH radical plus extract.
Evaluation of the Entire Antioxidant Capacity
The total antioxidant activity, abbreviated as TAC, was determined by applying the methodology outlined by Prieto et al. (2018).
30
In a nutshell, 0.15 mL of leaf extract was combined with 1.5 mL of reagent solution with 4 mM of ammonium molybdate, 28 mM of sodium phosphate, and 0.6 M of sulfuric acid. After that, the reaction mixture was kept warm for 90 min at 95 °C. 695 nm was found to be the samples’ optical density when measured. The total antioxidant capacity was given the unit of measurement of milligrams of ascorbic acid equivalence (AAE) per gram of dry weight (DW).
A control = Absorbance of negative control at the moment of solution preparation. A sample = Absorbance of the sample after 6 min.
The EC50 values were derived from the graph, which depicts the sample concentration necessary to scavenge 50% of the ABTS or DPPH free radicals. These values were determined. The EC50 value frequently represents the number of extracts required at a specific concentration to scavenge 50% of the free radicals. ABTS and DPPH were both expressed in milligrams of GAE/L.
Antiparasitic Activity
Test of Eimeria columbae Oocysts
The E. columbae parasite was isolated from wild pigeons that were usually infected. The parasite's passage through pigeons in the parasitology laboratory preserved its vitality. Dishes were incubated 1 × 103 oocysts of E. columbae in 2 mL of potassium dichromate consisting of 10, 25, 50, 100, 150, and 200 mg/mL of VVLE. The potassium dichromate solution (K2Cr2O7) was used as the control, and toltrazuril (Veterinary Products Company (VAPCO), Amman, Jordan) as the reference drug group. Three separate samples were used for each treatment in the experiment. The dishes (six-well plates) were incubated at 28 °C with a relative humidity of 70%. Additionally, all plates were semi-covered and shaken at regular intervals,
28
and sporulated and non-sporulated oocysts were recorded at 48 h, 72 h, and 96 h.
29
After 50 and 100 h, 5 µL of solution was inspected under a light microscope (BX51TF, OLYMPUS, Tokyo, Japan) at a magnification of 40x. This examination was performed on each group. The sporulate and inhibitory oocyst were assessed using a McMaster chamber and a light microscope. The percentages of oocysts were computed utilizing the formulas:
Statistical Analysis
The SPSS software, namely IBM SPSS Version 23, was utilized for statistical analysis, and a two-way analysis of variance (ANOVA) model was used to analyze the Tukey technique and variables for the in vitro investigation.
Results
The Phytochemical Analysis of the Extract
Table 1 displays the results of the spectroscopy investigation on V. vinifera leaf extracts using an FT-IR spectrometer. The results indicate the presence of influential bands at 3382.1 cm−1, 2119.8 cm−1, 1632.20 cm−1, 1510.70 cm−11, 1420.51 cm−1, and 1030.32 cm−1, respectively. With the absorbance at 400‒4000/cm−1, the active phytochemicals found are N-H stretch, N = C = S stretch, C = C stretch, C-Otertiary stretch, CO-O-CO stretch, and C-H bend (Figure 1).
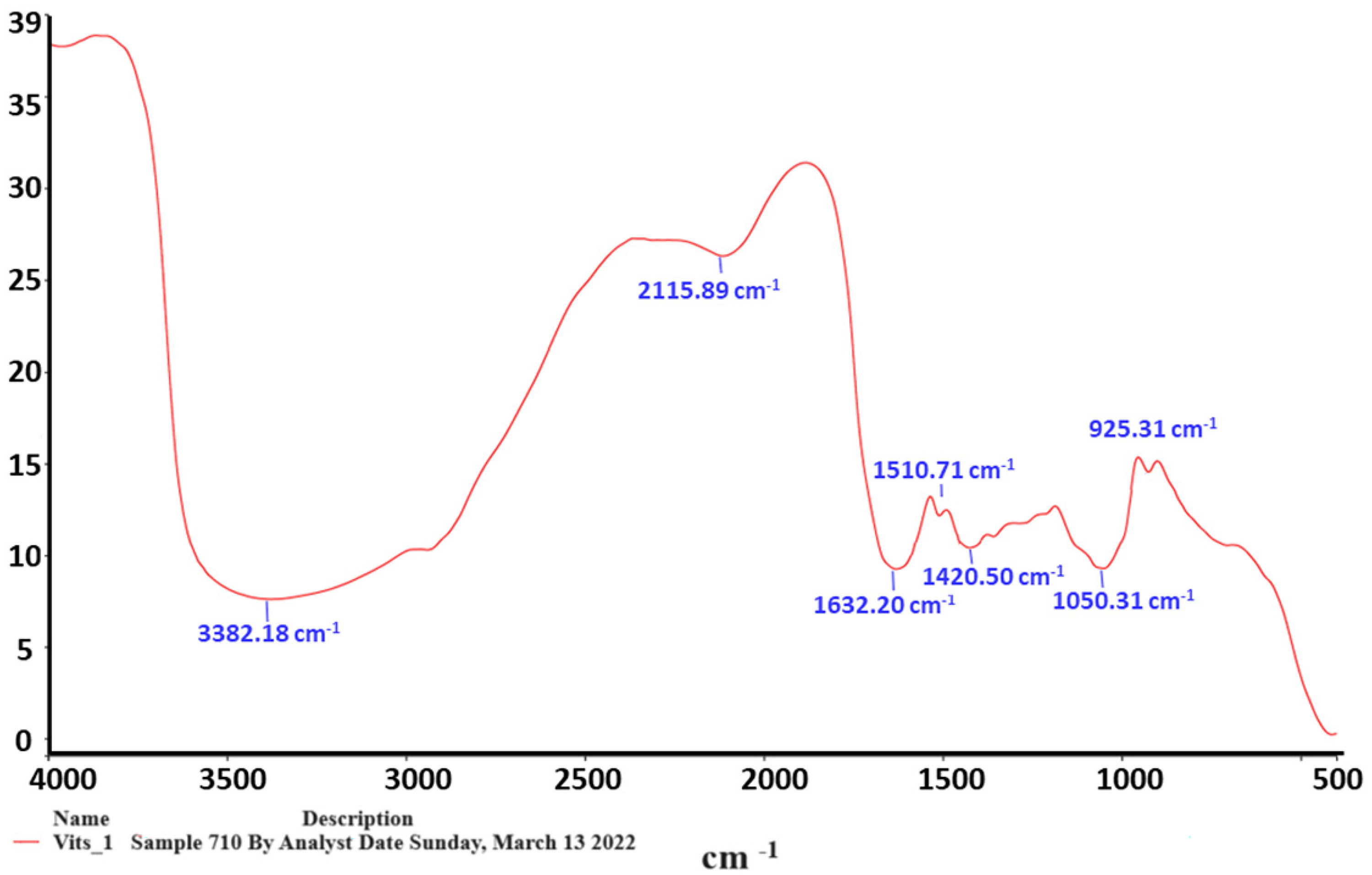
The findings of the Vitis vinifera leaf extract samples’ infrared spectroscopic study. Using a Nicolet 6700 FT-IR spectrometer and examining the data in the 400-4000/cm−1 range, the conclusions were reached. Fourier-transform infrared spectrometer, or FT-IR.
IR spectrum of Vitis vinifera leaf extracts by frequency range.

Table 2 and Figure 2 show the more bioactive phytochemical components of V. vinifera extracts (VVE), which contained 12 elements; the main components in the chemical group were flavonoids, Furanic aldehyde, pyranose, Fatty acid, diterpene Polyunsaturated fatty, acids (PUFA), Monounsaturated fatty acids (MUFA), Saturated fatty acids, Adipic acid ester, Glycidyl ester, and Fatty acyl chlorides.

GC-MS Chromatogram of aqueous leaf extract of Vitis vinifera leaf extracts (Murshed et al., 2023), 22 .
Determination of Phytochemical Ingredient by GC-Mass in Vitis vinifera Extract.

Murshed et al (2023). 22
Assessing the Phenolic, Tannin, and Flavonoid Content
In this study, the total phenolic content (TPC), total tannin content (TTC), and total flavonoid content (TFC) in the leaf extract of V. vinifera leaf extracts were estimated (Figure 3). The obtained results demonstrated that the methanolic extract had the highest concentration of TPC (160.217 0.652 mg/g DW), followed by TTC (65.872 0.263 mg/g DW), and TFC (34.0463 0.545 mg/g DW).

Flavonoids, tannins, and total Phenols in the leaves methanolic extract of the Vitis vinifera leaves extracts plant.
UV-vis Analysis
Figure 4 shows the UV-Vis spectrum of the V. vinifera extracts. As shown, the absorption peaks of leaf extracts were assigned at around 326 nm. This change indicated the existence of nucleic acids and aromatic chemicals.

The UV-Vis spectrum of Vitis vinifera leaves extracts.
Cytotoxicity Evaluation
Hep-G2/2.2.15 Human Hepatoblastoma Cell Line were seeded with different serial concentrations (15.625, 31.125, 62.5, 125, 250 and 500 μg/mL) of methanolic plant extract (VVLE) for 48 h. The IC50 of V. vinifera leaf extracts was obtained at (13.777 ± 0.337 µg/ml) μg/ml for the Hepatoblastoma Cell (Hep-G2/2.2.15) cell line (Figure 5).

Cytotoxicity (MTT) assay for tested Vitis vinifera at different concentrations (µg/mL) against Hepatoblastoma (Hep-G2) after 48 h of incubation. IC50 indicates the dose of tested plant extract, inducing 50% Hep-G2 (13.777 ± 0.337 µg/ml), Hepatoblastoma cell growth inhibition.
Radical Scavenging Test Utilizing DPPH
Using 2,2-diphenyl-1-picrylhydrazyl (DPPH), the antioxidant activity of the methanolic extract of V. vinifera leaf was evaluated for this current study. The findings showed that the ability to scavenge DPPH radicals was discovered to increase with the increase in the concentration of leaf extract (Figure 6). This was proved by the findings that were obtained. It was found that the radical scavenging activities of phenolic components such as polyphenols, flavonoids, tannins, and phenolic terpenes are primarily responsible for the antioxidant effect that plant products have.
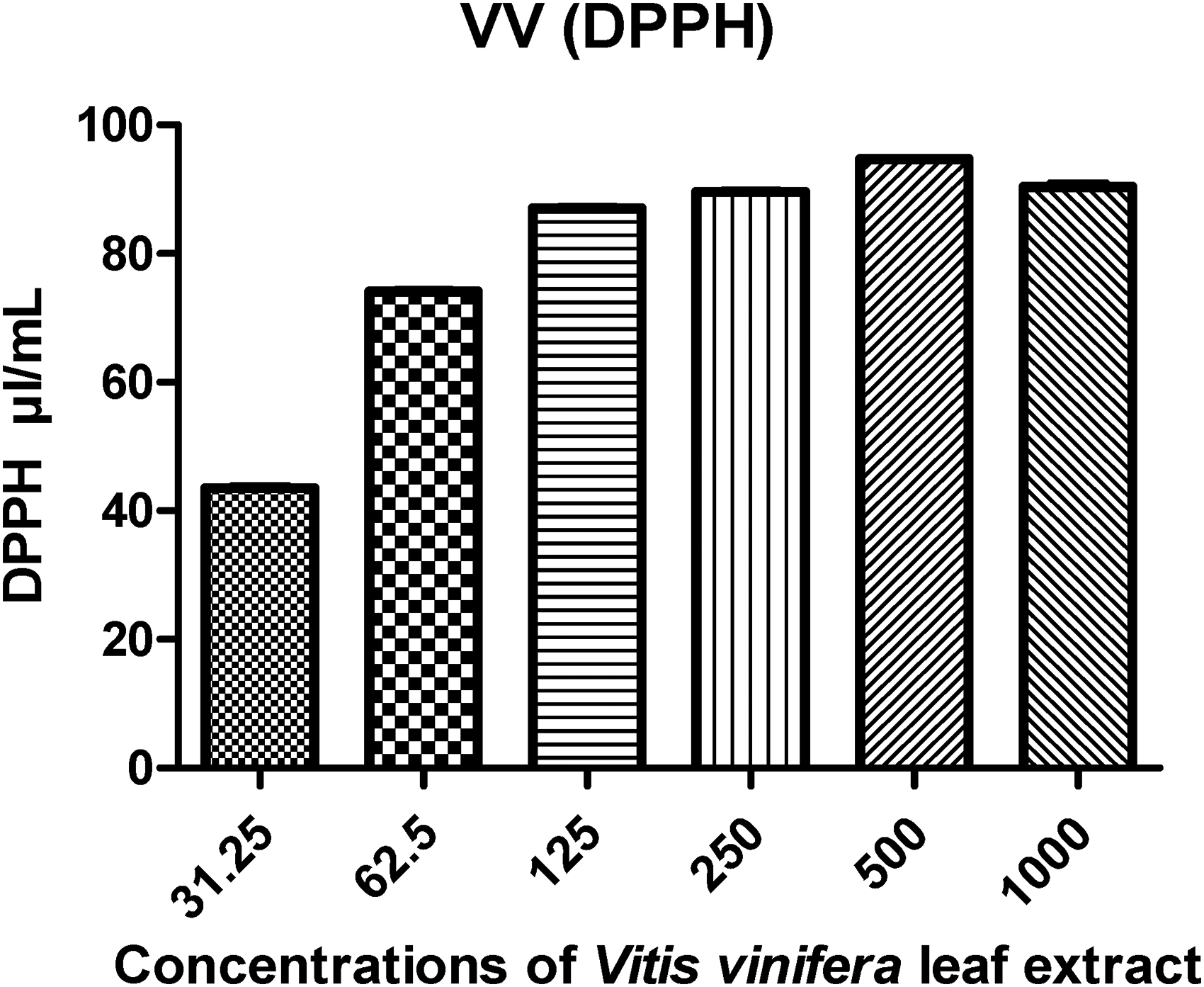
DPPH estimation: the data that were reported are the mean values based on three replicates ± SD.
Evaluation of the Entire Antioxidant Capacity
ABTS radical scavenging activity was positively correlated to the concentration of the methanolic extracts. In this analysis, the concentrations highest samples showed higher radical scavenging activity of ABTS than the lower concentrations (Figure 7).
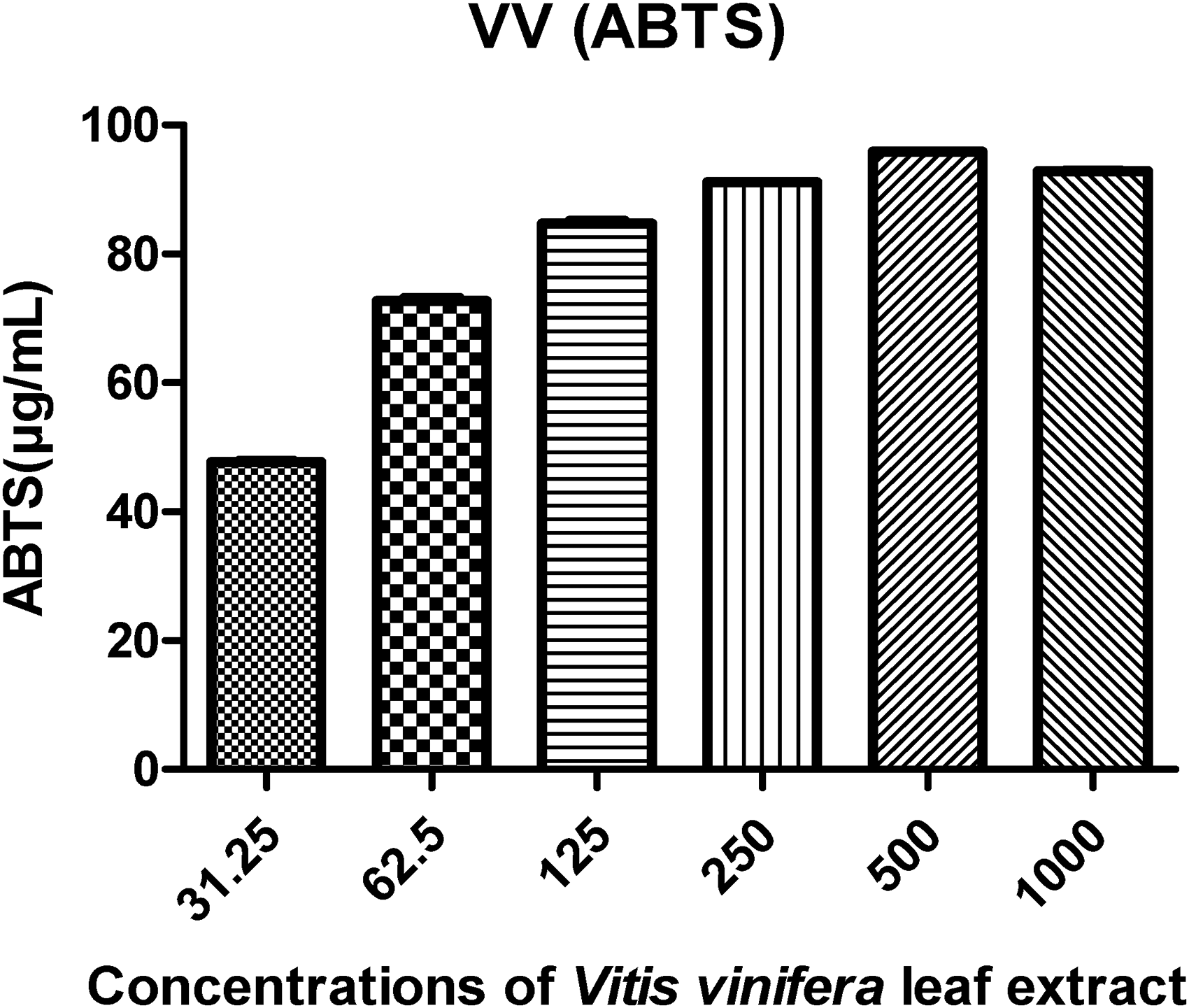
The significances are the average of three tests ± DS, and they are expressed as EC50 in mg GAE/L dry basis.
Antiparasitic Activity
In vitro studies, the main impacts of the periods of sporulation and Tested groups on sporulation (%), non-sporulation (%), and inhibition of E. columbae oocysts are shown. The results showed that the percentage of sporulation increased with the increase in the concentration ratio of the extract. On the contrary, the percentage of non-sporulating oocysts decreased with the concentration reduction (Figure 8). In addition, We have noticed that the sporulation inhibition rate increased significantly with increasing incubation time up to 72 h (p<0.05). Therefore, the sporulation inhibition rate did not differ significantly between 72- and 96-h exposure.
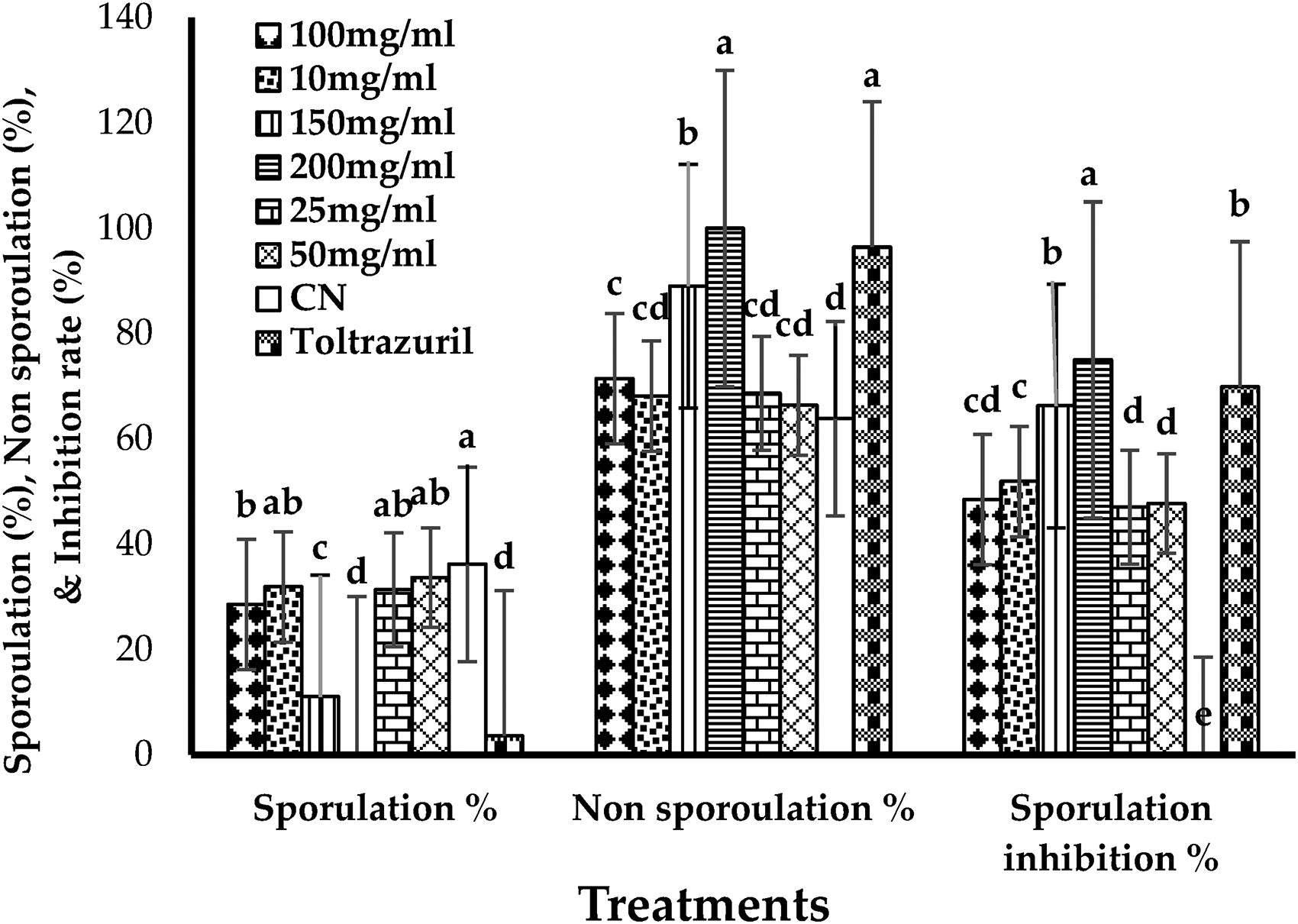
In vitro main impacts of Vitis vinifera on sporulation%, non-sporulation %, sporulation inhibition of Eimeria columbae oocysts at different concentrations at effects of treatments.
The sporulation times we interacted with the experimental groups’ concentrations on the sporulation and inhibition of E. columbae oocysts (Figure 9). Therefore, the sporulation of the oocysts increased as the incubation period increased, and the inhibition rate increased with the higher concentrations despite the increase in the incubation period until it reached 96 h.

In vitro main effects of Vitis vinifera on sporulation%, non-sporulation %, sporulation inhibition of Eimeria columbae oocysts at different concentrations at effects of contact time (24, 48, 72, and 96 h).
The study showed contemporary methodologies for phytochemical analysis and the identification of physiologically active components. This helps to illustrate further that isolated chemicals may have potential bioactivities.
Discussion
The bioactive substances derived from medical herbs are crucial to the production of drugs.30,31 It is still necessary to find new bioactive sources. As a result, academic and pharmaceutical organizations are constantly searching for new bioactive compounds in plants to find new products. 32 According to, 15 the leaf of the V. vinifera plant has been extensively utilized in traditional and current medical practices.
The phytochemical analysis of V. vinifera (grape) leaf extracts shows essential information about their chemical makeup and possible biological activities. Using Fourier-transform infrared (FT-IR) spectroscopy, the study found several influential absorption bands that show different functional groups. These findings suggest the presence of diverse phytochemicals with potential health benefits, such as flavonoids and fatty acids, which are known for their antioxidant properties. 33
The analysis indicated that the methanolic extract of V. vinifera leaves contains a rich array of bioactive compounds. The primary components identified include flavonoids, furanic aldehydes, and various fatty acids, including polyunsaturated fatty acids (PUFAs) and monounsaturated fatty acids (MUFAs). We measured the total phenolic content (TPC) at 160.217 mg/g dry weight, which was notably high, followed by tannins and flavonoids at 65.872 mg/g and 34.046 mg/g, respectively. Further analyses34,35 support the association of this high concentration of phenolic compounds with enhanced antioxidant activity.
The DPPH and ABTS radical scavenging assays evaluated the extracts’ antioxidant potential. The results showed that the antioxidant activity rose as the content of the leaf extract increased, suggesting that the phenolic components, such as polyphenols and flavonoids, are essential in neutralizing free radicals.36,37 Engaging in this exercise is crucial for reducing oxidative stress associated with various disorders.
The extracts were tested for cytotoxicity against the Hep-G2/2.2.15 human hepatoblastoma cell line and showed an IC50 value of 13.777 µg/ml. This showed that they had strong anticancer properties.38–40 Additionally, we evaluated the extracts’ in vitro antiparasitic activity,34,41 which successfully suppressed the sporulation of E. columbae oocysts, particularly at higher doses and more extended incubation periods. The findings demonstrated a significant decrease in oocyst sporulation, underscoring the potential use of V. vinifera extracts in parasitic disease control. This result is consistent with showed to the grape extract when testing E. papillata oocysts. 42
Conclusions
The methanolic extract of Saudi Arabia-grown V. vinifera revealed the presence of a wide variety of active ingredients that have the potential to be used in both traditional medicine and primary health care. The results confirm the possibility of using grape leaves to treat bird coccidiosis (E. columbae oocysts). That infects domestic pigeons. Therefore, the results of this study offer future solutions to combat avian coccidiosis.
Footnotes
Acknowledgments
Thank you to Researchers Support Project No. (RSPD2024R1081), at King Saud University, Riyadh, Saudi Arabia.
Author Contributions
The study was conceived and designed by Mutee Murshed. Jameel Al-Tamimi and Saleh Al-Quraishy analyzed the data. Mutee Murshed prepared the first draft of the manuscript. Mutee Murshed, Jameel Al-Tamimi, and Saleh Al-Quraishy collected and interpreted the data. The other researchers contributed the data and critically revised the work to update it with significant intellectual substance. The authors read the final version of the manuscript and were approved.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Does not apply to this article.
Statement of Human and Animal Rights
No research involving humans or animals is included in this article.
Statement of Informed Consent
This is not applicable because no human subjects are in this article.





