Abstract
Fast inverted, oil-in-water (o/w) emulsions, also known as SWitch-Oil-Phase (SWOP) emulsions, express the performances of both o/w and water-in-oil (w/o) emulsions during application to the skin, favoring their use as cosmetic carriers in sunscreen products. The objective of this study was to investigate the antioxidant potential (by 2 different methods) and the ultraviolet (UV) absorption ability of SWOP emulsion (S) with incorporated plant-based antioxidants dihydroquercetin (DHQ) and β-carotene (βC), using quercetin (Q) in a reference emulsion, in addition to the evaluation of their physicochemical properties and stability. A new biochemical extracellular model for in vitro assessment of antioxidative properties for the SWOP emulsions (S, SQ, SDHQ, and SDHQβC) was developed and compared with the results of 2, 2-diphenyl-1-picrylhydrazyl (DPPH) assay. The analyses were performed at 20 °C and 37 °C, and oxidative stress parameters were monitored and statistically analyzed. The sun protection factor (SPF) of the samples was determined in vitro. Q and DHQ incorporated into the SWOP emulsion exhibited a strong DPPH radical scavenging ability. Neither incorporated nor pure βC showed DPPH radical scavenging ability at the tested concentrations. Contrary to that, in the bioenvironment conditions, SDHQβC showed minor antioxidative effects increase and also a significant decrease in exogenous pro-oxidative effects, caused by pro-oxidant, when compared to SDHQ. The obtained SPFs of SDHQβC, SDHQ, and SQ were 5.19, 4.65, and 3.35, respectively. The physicochemical stability of the emulsions was satisfactory during 1 month storage. The presented results demonstrated that the SWOP emulsion is a suitable carrier for antioxidants with a photoprotective ability. The novel biochemical approach could be used in addition to DPPH assay with several advantages, relevant for the testing of antioxidant activity of potential active ingredients in cosmetic products.
Keywords
Introduction
As the largest organ in the human body, skin is exposed to numerous exogenous environmental factors, but ultraviolet (UV) radiation is recognized as an essential risk factor affecting skin physiology. Based on photon wavelength, the UV radiation from the sun consists of UVA (315-400 nm), UVB (280-315 nm), and UVC (100-280 nm) bands. 1 Chronic exposure to UV radiation leads to many side effects on the skin (thickening of the stratum corneum, epidermis, and dermis, 2 wrinkling of the skin and photoaging, 3 that are directly related to the formation of reactive oxygen species (ROS), 4 supporting the rational use of antioxidants in topical formulations. 5 The use of antioxidants in skin care is further supported by the reported efficacy in the enrichment of the endogenous cutaneous protection system after topical administration. 5 Additionally, the use of natural ingredients incorporated in innovative and efficient formulations becomes a world tendency along with cosmetic product development, especially in the photoprotection market, reinforced by population acceptance and the media. Active ingredients from natural products containing chromophores, aromatic rings, and compounds with antioxidant properties provide novel possibilities for the treatment and prevention of UV-caused oxidative damages. 6
Flavonoids, a group of polyphenolic compounds originating from plants, are interesting for the pharmaceutical, cosmetic, and food industries because of their variety of possible beneficial properties. Antioxidant activity is one of the most important properties of flavonoids. 7 Besides being scavengers of free radicals, flavonoids reduce their production due to UV absorption ability. Flavonoids cannot replace traditional synthetic sunscreens; however, in combination with commercial sunscreens, they could significantly improve the sun protection factor (SPF), the UVA protection factor (UVA-PF), and the photostability of formulations due to their additive and synergistic effects. 2
Dihydroquercetin (DHQ) is a flavonoid that acts as a powerful antioxidant having a similar antioxidant activity profile to that of α-tocopherol. 8 It is known that DHQ absorbs over a wide range of the UV spectrum, showing maximum absorption at 225 nm and 325 nm and helps prevent skin damage caused by external agents, alleviates skin inflammation, soothes irritated skin, 9 and even suppresses UV-induced skin carcinogenesis. 3 These properties of DHQ, as well as its strong safety profile, indicate that it could be interesting for further investigations, especially in combination with other UV filters and/or antioxidants incorporated into a suitable cosmetic carrier.
Vitamins A, C, E, carotenoids, and various enzymes, such as glutathione peroxidase (GPx), catalase (CAT), and superoxide dismutase (SOD), are the most common antioxidants in human skin. 10 Applied topically, these antioxidants are able to enrich the defense potential of the upper layers of human skin against the action of environmental hazards and irradiations. 11 Additionally, antioxidants are added to cosmetic products to increase their stabilization and protect active ingredients from oxidation/degradation. 5
Various in vivo, ex vivo, and in vitro methods have been developed for the evaluation of antioxidant activity, but many of them cannot be used in the cosmetic industry. Whenever possible, it is recommended to use noninvasive instrumental methods or in vitro tests according to the EU Regulation (EC) No. 1223/2009 and this provision is accepted by many other countries worldwide. Among in vitro free radical scavenging methods, the 2, 2-diphenyl-1-picrylhydrazyl (DPPH) assay is the most commonly used. However, standard in vitro tests cannot provide data on activity in contact with a bioenvironment and do not reflect the actual biological processes in skin cells.12, 13
Classical sunscreen formulations include the most popular, worldwide, emulsion products such as creams/gel creams and lotions. Water-in-oil (w/o) emulsions are preferred for sunscreens since they can provide high SPF and water resistance. Despite many desirable characteristics, w/o emulsions generally contain more oils than water and their greasiness and a heavier feel makes them less favored in comparison with oil-in-water (o/w) emulsions. 14 Therefore, fast inverted o/w emulsions, also known as SWitch-Oil-Phase (SWOP) emulsions, which invert into w/o emulsions during application to the skin and form a lipophilic, water-resistance layer, are especially suitable as carriers in sun protection products. 15
The aim of this study was the preparation of SWOP emulsions with incorporated plant-derived antioxidants (DHQ and β-carotene [βC]) and the investigation of their antioxidant potential and UV absorption ability in addition to physicochemical properties and stability. For these purposes, a comparative SWOP emulsion with the well-known plant-derived antioxidant quercetin (Q) was used as a reference emulsion. Antioxidant potential of the incorporated and pure plant-derived antioxidants was determined using the DPPH radical scavenging assay. To estimate and evaluate the antioxidant activity in a bioenvironment, a new in vitro extracellular biochemical model was developed. Antioxidant properties of different plant-based ingredients were monitored regarding their influence on oxidative stress parameters and the parameters of antioxidative protection. The UV absorption ability of the SWOP emulsions was estimated by an in vitro spectrophotometric method.
Materials and Methods
Materials
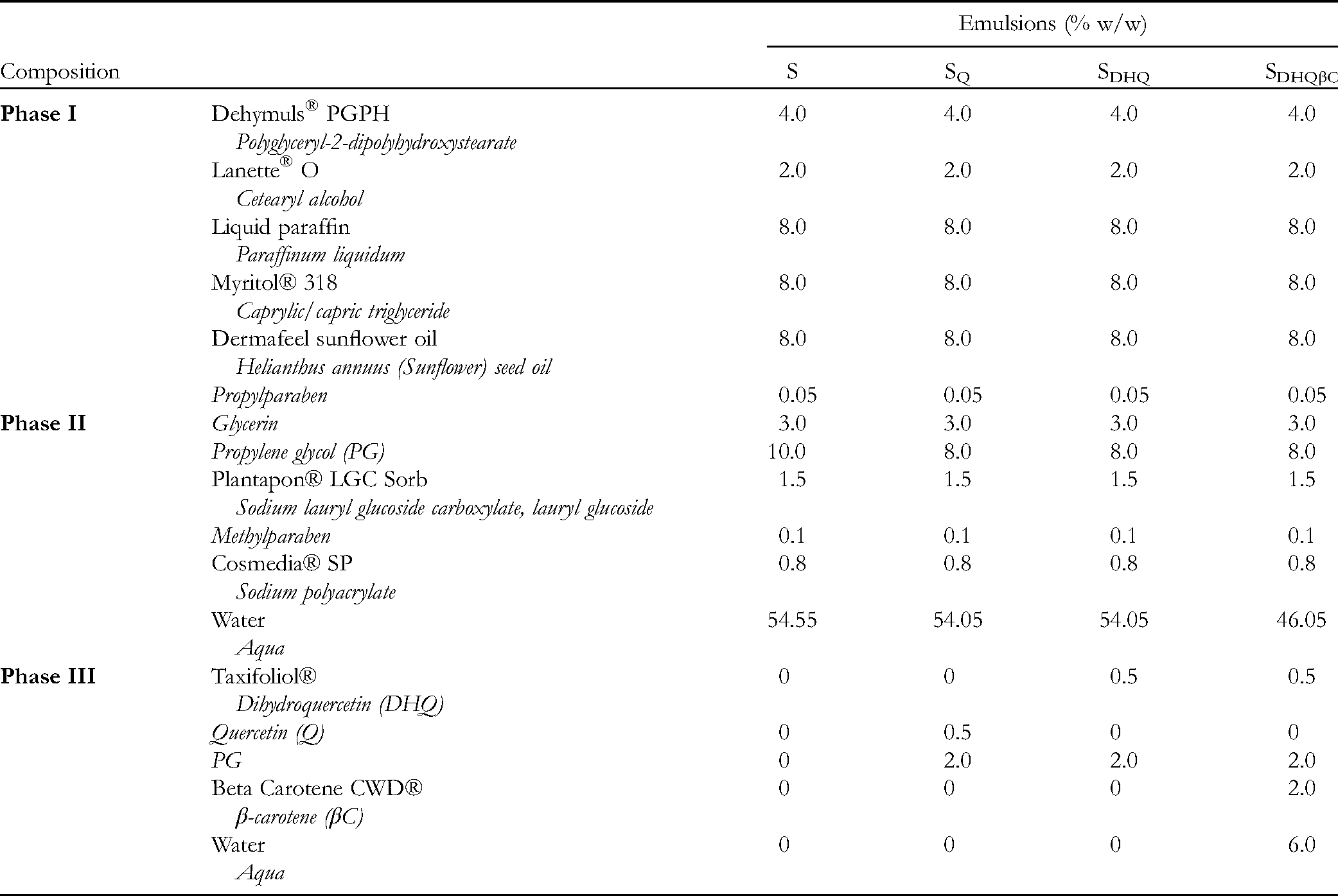
SWOP emulsion base (labeled as S) and SWOP emulsions with incorporated Q, DHQ, and a combination of DHQ and βC (labeled as SQ, SDHQ, and SDHQβC, respectively) were prepared according to the compositions presented in Table 1. As stated in our previous works,16, 17 the SWOP emulsions were stabilized with a combination of the nonionic emulsifier polyglyceryl-2 dipolyhydroxystearate (Dehymuls® PGPH, BASF, Germany), an anionic surfactant that is a mixture of lauryl glucoside and sodium lauryl glucose carboxylate (Plantapon® LGC Sorb, BASF, Germany), and the polymeric stabilizer sodium polyacrylate (Cosmedia® SP, BASF, Germany).
The oil phase (Phase I) consisted of liquid paraffin (R
All reagents used for antioxidant capacity measurement were of the highest purity available and obtained from Sigma Aldrich (Dorset, United Kingdom).
Methods
Preparation of the Emulsions
The SWOP emulsions, labeled as S, SQ, SDHQ, and SDHQβC, were prepared by hot–hot emulsification, as previously reported for SWOP emulsion base.16, 17 The active substances Q, DHQ, and βC, were dispersed in an appropriate solvent (Table 1, Phase III), and added to the emulsions while stirring at 40 °C. All the prepared samples were left for 48 h to equilibrate before further testing.
Physicochemical Properties of Prepared Emulsions
Freshly prepared emulsions were investigated by visual monitoring (general appearance, color, consistency of the emulsions, homogeneity of the emulsions, and occurrence of phase separation).
pH and conductivity measurements, rheological characterization, and differential scanning calorimetry were performed in a similar manner, as previously described.16, 17 pH and conductivity measurements were performed in triplicate at room temperature (20 ± 2 °C). Continuous flow tests for the prepared emulsions (S, SDHQ, and SDHQβC) were performed in triplicate after 48 h and 30 days’ storage at room temperature (20 ± 2 °C). Finally, the samples analyzed by differential scanning calorimetry (DSC) were heated from 25 °C to 130 °C, at a heating rate of 5 K min−1 to compare the endothermic transition enthalpies of the test and reference samples. All the samples were measured in duplicate at least. 17
Having applied a comprehensive analysis that included the aforementioned methods, alongside contact angle measurements and polarization microscopy, the colloidal structure, the phase inversion, and the sensorial properties of SWOP emulsions were discussed and explained in our previous papers.16, 17
In Vitro Determination of Antiradical Activity by DPPH Assay
The radical scavenging ability of Q, DHQ, βC, and SWOP emulsions containing Q (SQ), DHQ (SDHQ), and DHQ-βC (SDHQβC) was tested by DPPH assay.
18
Different aliquots of stock solutions of pure flavonoids Q and DHQ (0.25 mg mL−1), βC (1 mg mL−1), and SWOP emulsions (5 mg mL−1) in PG (or water in the case of βC and SWOP emulsion containing βC) were diluted to 2 mL with 99.9% ethanol, and then 0.5 mL of 0.5 mM DPPH solution in ethanol was added to each dilution. Mixtures were vigorously shaken and left in the dark for 30 min. Absorbances were measured at 517 nm (and additionally at 540 nm for pure βC and SWOP emulsion containing βC) using corresponding mixtures of ethanol and PG (or water) as blanks. One mL of 0.5 mM DPPH solution diluted with 4 mL of the corresponding solvent mixture was used as a negative control. Scavenging of DPPH radical, S(%), was calculated using the following equation:
In Vitro Antioxidant Activity of the Tested Emulsions in a Bioenvironment
Serum samples were collected from healthy individuals after regular daily work in a clinical chemistry laboratory of the Military Medical Academy (Belgrade) in order to make the serum pool. Healthy volunteers, who had attended regular medical check-ups and had given written approval that any serum leftover after biochemical analyses ordered by physicians could be used for our antioxidative potency investigation, were included in this study. The members of the study team did not use any of the data connected with subjects whose serum leftovers were used. The Ethical Committee (decision No. 797/2, May 5, 2012. University of Belgrade – Faculty of Pharmacy) has verified this procedure for sample collection. Samples were selected according to basic biochemical parameter results, ie, those whose metabolites were within reference ranges were used, as an additional confirmation about the subject's health status. Serum samples were pooled and aliquoted, then frozen at −80 °C and used for redox status analytics no more than 2 weeks after the collection. 19
The test samples were prepared by diluting each emulsion (S, SQ, SDHQ, and SDHQβC) to 1% in PG, and dissolving antioxidants (Q and DHQ) to 0.005% in PG. Serum volume was 450 μL and the added volume of each previously prepared test sample was 50 μL (1:10 ratio), ie, the exogenous addition did not exceed 10% of the total sample volume, which is of the utmost importance to samples’ bio-matrix quality provision. For probes with the well-known pro-oxidant tert-butyl-hydroperoxide (TBH, conc. 0.25 mmol L−1), 25 μL of each previously described sample (prepared in double concentration) and 25 μL TBH were added to 450 μL serum samples. All the samples were incubated in duplicate for 2 h, at either 20 °C or 37 °C. This model enabled us to estimate antioxidative potency of carotenoid and polyphenolic compound-enriched emulsions in a bioenvironment by measuring several redox status parameters: pro-oxidants (total oxidant status—TOS, pro-oxidant-antioxidant balance—PAB, and advanced oxidation protein products—AOPP) and antioxidants (total antioxidant status—TAS, serum paraoxonase-1—PON1, and total protein sulfhydryl groups—SHG), to some extent, similar to human skin.
The colorimetric assay based on the oxidation of ferrous ion to ferric ion in an acidic medium in the presence of different oxidant species such as H2O2 and lipid hydroperoxides, was used for TOS measurement. 20 AOPP were determined by the Witko-Sarsat method, in a reaction with glacial acetic acid and potassium iodide. 21 The assay used for measurement of PAB is based on complex reactions of 3,3′, 5,5′-tetramethylbenzidine and its cation used as a redox indicator, which is simultaneously developing. 22 TAS was determined with a novel automated colorimetric method with ABTS cation developed by Erel. 23 PON1 activity was measured as the rate of paraoxon (POase) hydrolysis, according to Richter and Furlong. 24 SHG in serum (mmol/L) was determined by the method of Ellman, 25 using 5,5′-dithiobis (2-nitrobenzoic acid) (DTNB). Values of the monitored parameters were analyzed using Kruskal–Wallis test and Mann–Whitney post hoc test, and the statistical significance was set at the 0.05 value.
Pro-oxidative score, antioxidative score, and oxy score (OS) were calculated from separate redox status parameters using Z score statistics. OS was calculated as the difference between pro-oxidative score (average value of Z scores of all measured pro-oxidants and their products, ie, TOS, AOPP, and PAB) and antioxidative score (average value of Z scores of all measured antioxidants, ie, TAS, PON1, and SHG). Z scores represent the difference between the original value and the control value divided by the SD of control values (or population means and SDs). A higher OS of substance means weaker antioxidative protection and higher pro-oxidants content and vice versa. 26
Spectrophotometric Determination of SPF
A quantity of 0.25 g of the emulsions S, SQ, SDHQ, and SDHQβC were weighed, transferred to a 25 mL volumetric flask, and diluted to volume with ethanol. Further, it was kept for ultrasonication for 5 min and then filtered through cotton, disposing of the first 10 mL. Then, a 2 mL aliquot (filtered through filter paper) was transferred to a 10 mL volumetric flask and the volume was adjusted with ethanol.
The absorption spectra of the prepared samples were obtained in the range of 290 to 320 nm at intervals of 5 nm using a UV/Visible spectrophotometer (UV-VIS Cintra 202, GBC Scientific Equipment, Australia). Three determinations were made at each point using ethanol as a blank.
SPF values were calculated using a mathematical equation (Eq. 2)
2
developed by Mansur et al
27
with an intention to substitute the in vitro method proposed by Sayre et al4, 28:
The values of EE × I, presented in Table 2, are constants and predetermined by Sayre et al 28
Abbreviations: EE, erythemal effect spectrum; I, solar intensity spectrum.
Results and Discussion
Evaluation of Physicochemical Properties of the SWOP Emulsions
Organoleptic properties and several parameters which include pH value, conductivity, and viscosity of the emulsions were regularly monitored in the previous researches of SWOP emulsions under normal storage conditions and gave the first insight into the structure and stability of an emulsion.16, 17 The prepared emulsions were shiny, smooth, and light creams, but varied in color (S—white, SDHQ—yellowish, and SDHQβC—yellow to orange) due to the color of the incorporated antioxidants. The emulsion SDHQβC had the most intense color, due to the high βC concentration, which was used to achieve a better antioxidant effect. The consistency of the SWOP emulsion S was soft semisolid, while the emulsions with incorporated antioxidants had lower viscosity due to the presence of flavonoid DHQ 29 and the method of antioxidant incorporation.
The measured pH values of the prepared emulsions stored under normal conditions slightly changed during 30 days; 48 h after preparation, pH values for S, SDHQ, and SDHQβC were 5.83, 5.82, and 5.82, respectively, while 30 days after preparation the alterations in these values were negligible (5.84, 5.80, and 5.78, respectively). The conductivity measurements demonstrated differences between the emulsions during the testing period. The initial conductivities were higher than 1000 μS cm−1 and in accordance with our previous findings. 16 The SWOP emulsion base S had higher and less changeable conductivities (1665 μS cm−1 after 48 h, ie, 1686 μS cm−1 after 30 days) than the emulsions with antioxidants: 1675 μS cm−1 after 48 h, ie, 1598 μS cm−1 after 30 days for sample SDHQ, and 1162 μS cm−1 after 48 h, ie, 725 μS cm−1 after 30 days for sample SDHQβC. Although the conductivity of sample SDHQβC decreased more significantly during the testing period, it was still about fifteen times higher compared to that of the conventional o/w emulsions (∼50 μS cm−1). The obtained results indicated changes in the structure of SWOP emulsion with both antioxidants (SDHQβC), which could affect its long-term stability.
The rheological profiles (Figure 1, Table 3) indicated “shear-thinning” flow behavior with a moderately pronounced thixotropy, ie, these emulsions are pseudoplastic non-Newtonian semisolid systems with hysteresis, which is of high importance for sunscreen formulations. Namely, the sunscreen formulations with a pseudoplastic flow produce a coherent protective film covering the skin surface with evenly distributed UV filters and this characteristic helps to promote a higher SPF. 30 Additionally, the formulations with lower hysteresis area (HA) are ideal for cosmetic actives that should stay on the skin surface, such as sunscreens. 31 It was noticed that the apparent viscosity (maximal—ηmax and minimal—ηmin) of the emulsions (S, SDHQ, and SDHQβC) decreased during the storage time (48 h and 30 days) at room temperature (Figure 1, Table 3). However, a pronounced decrease in rheological parameter values was observed for both SDHQ and SDHQβC, which could be an indicator of changes in the structure as a result of the addition of DHQ and βC, and also of stability of these emulsions that should be investigated in further research.

Flow curves of the investigated emulsions plotted with shear stress as a function of the increasing/decreasing shear rate, after 48 h and 30 days storage at room temperature (n = 3, all points in each flow curve have a coefficient of variation lower than 3%).
Values of Maximal (ηmax) and Minimal (ηmin) Apparent Viscosity, Yield Stress (σ0), and Hysteresis Area (HA) for the Investigated Emulsions During Testing Period.

Due to its sensitivity to thermal changes in materials, in the previous research, DSC was used as a tool for the investigation of colloidal structure and stability of the SWOP emulsion. 17
DSC curves of all the investigated samples exhibited similar behavior characterized by a series of intensive endothermic events ranging from 100 °C to 120 °C (Figure 2). Namely, the endothermic events were split into several sharp peaks suggesting intensive structural changes due to the gradual evaporation of water and PG. Loss of water at such high temperatures implies excellent thermal stability of all the investigated formulations, which could be ascribed to the strong interactions with emulsifiers and stabilizers.
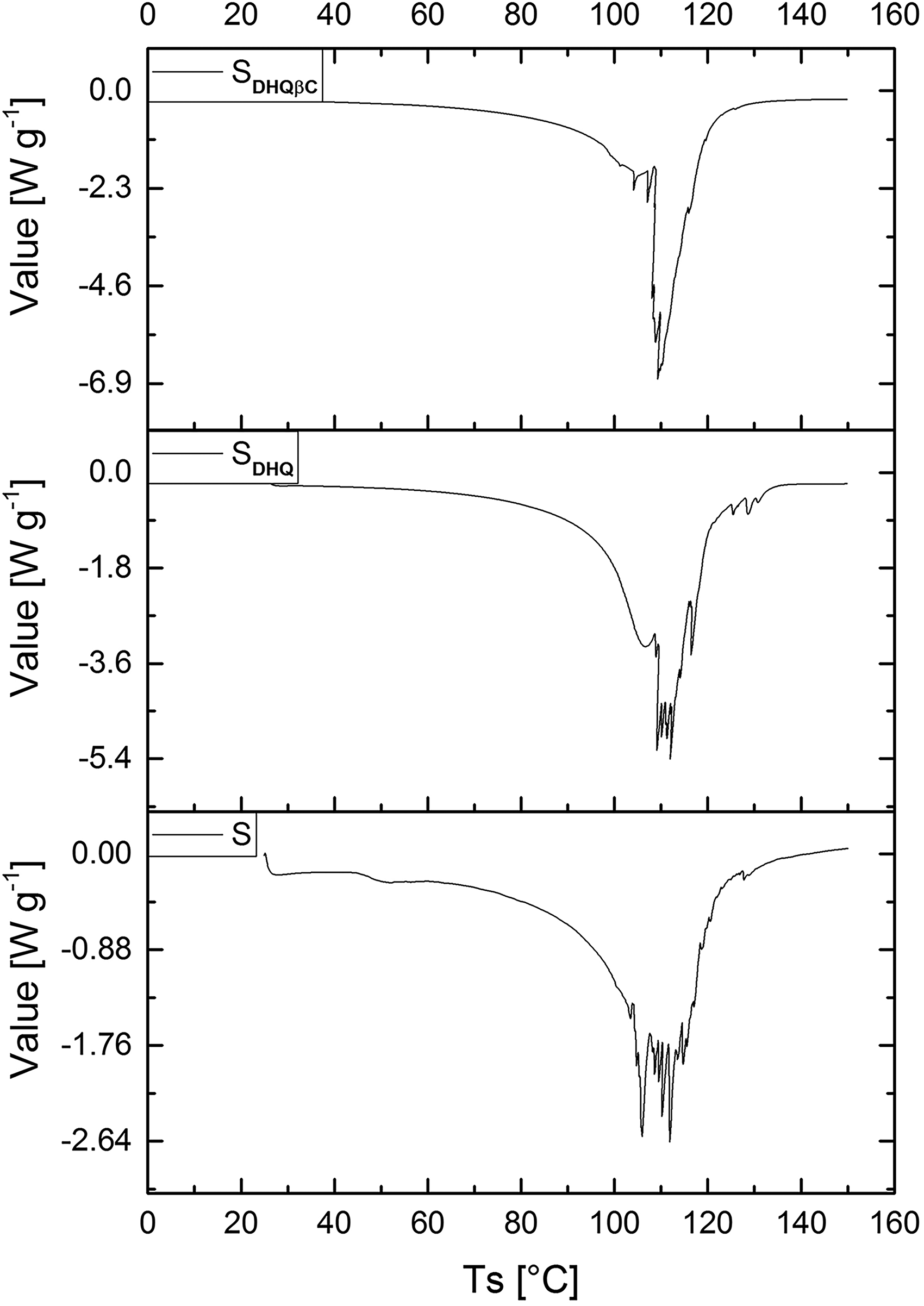
DSC curves of the investigated emulsions.
The enthalpies of these thermal transitions were −0.66·103 J g−1, −1.01·103 J g−1, and −0.9·103 J g−1 for S, SDHQ, and SDHQβC, respectively, implying a decrease in thermal stability upon addition of DHQ, which was in agreement with a previously reported decrease in rheological parameters of the emulsions containing flavonoids.16, 17 However, the effect of DHQ, and even βC, on the thermal stability of emulsions was negligible at temperatures below 80 °C.
In Vitro Antioxidant Activity and Determination of SPF
The DPPH test is a method for evaluation of the antioxidant potential indirectly by spectrophotometry used to measure the color change that occurs during the reaction between the purple, stable, free radical DPPH and the antioxidant. 12 DHQ and Q incorporated into SWOP emulsion exhibited strong DPPH radical scavenging ability, without significant statistical difference compared to the pure compounds. The SC50 values of incorporated and pure Q were 3.48 ± 0.10 µg mL−1 and 3.37 ± 0.03 µg mL−1, respectively, and the SC50 values of DHQ incorporated in SDHQ and SDHQβC, and of pure DHQ were 5.36 ± 0.27 µg mL−1, 5.06 ± 0.14 µg mL−1, and 5.02 ± 0.10 µg mL−1, respectively (Table 4). However, there was a statistically significant difference between the SC50 values of Q (pure and incorporated into SWOP emulsion) and DHQ (pure and incorporated into SWOP emulsion) indicating better antioxidant activity of Q. On the other hand, although known for its antioxidant activity, neither incorporated nor pure βC showed any DPPH radical scavenging ability at the tested concentrations (0.40-8.00 µg mL−1), which is in agreement with the findings of Müller et al. 32 The same results were also obtained when the measurements were made at 540 nm, to minimize the interference at 517 nm between the absorbance of DPPH and carotenoids. Therefore, DPPH assay is not suitable for the testing of antioxidant activity of βC incorporated into SWOP emulsion, since carotenoids are not able to scavenge the DPPH radical. 32
DPPH Radical Scavenging Activity of Pure Flavonoids and Flavonoids Incorporated in SWOP Emulsions Expressed as the Concentrations That Scavenge 50% of DPPH Radicals (SC50).

Different letters in superscript indicate significant differences between the SC50 values (determined by one-way ANOVA, followed by Tukey's post hoc test, P < 0.05).
Abbreviations: DPPH, 2, 2-diphenyl-1-picrylhydrazyl; SWOP, Switch-Oil-Phase.
The SC50 values of the tested SWOP emulsions SQ, SDHQ, and SDHQβC were 0.70 ± 0.02 mg mL−1, 1.07 ± 0.05 mg mL−1, 1.01 ± 0.03 mg mL−1, respectively. Therefore, there was no significant statistical difference between the SC50 values of the two DHQ containing SWOP emulsions, ie, emulsion with and without βC (SDHQ and SDHQβC).
We presumed that the in vitro antioxidant activity of the tested emulsions in a bioenvironment could give a more realistic insight into the human in vivo reaction toward the investigated emulsions. The main way was to estimate the changes in antioxidant capacity, ie, the sum of all antioxidant activities of the serum (a human serum mixture containing many antioxidants) 33 in the presence of the investigated antioxidants. The reactions were performed at two different temperatures (20 °C and 37 °C) and five subgroups (Figure 3). Antioxidative score values at 20 °C were ranked as follows: Q/PG and SQ/PG −0.33 (−5.11–6.17) > SDHQβC/PG −5.50 (−14.67–3.67) > DHQ/PG, and SDHQ/PG −9.33 (−9.89– −7.50), and the difference between last two subgroups was insignificant (Figure 3A). At 37 °C, the rank order was the same: Q/PG and SQ/PG −7.34 (−14.50– −3.00) > SDHQβC/PG −33.50 (−45.67– −21.33) > DHQ/PG, and SDHQ/PG −37.33 (−39.00– −34.67), with a significant difference between the last two subgroups (Figure 3B). The comprehensive OS, reflecting the balance between pro-oxidants and antioxidants, was the lowest in Q containing samples, which indicates the superiority of this antioxidant compared to the other tested substances. The superiority of Q containing serum samples was also observed after incubation at 37 °C. A marginally significant difference between Q samples and DHQ-βC combination sample speaks in favor of the antioxidant capability improvement after the βC integration in the complex mixture of the skin protective emulsion (Figure 3B). The βC inclusion in the DHQ containing sample leads to the improvement of its antioxidative potential.

Range of pro-oxidative, antioxidative, and oxidative scores for different subgroups at (A) 20 °C and (B) 37 °C. Box plots range from the 25th to the 75th percentile, the 50th percentile is drawn inside the box, the whiskers present minimal and maximal values and squares presents mean values.
Nonparametric repeated measures ANOVA (Friedman's test) was used for the comparison of antioxidative potency of the investigated samples (Figure 4), in a native state and upon TBH influence. Antioxidative potency of the investigated samples, regarding the separate oxidative stress parameters (PON1, SHG, AOPP, and TOS) (Figure 4A-C and E), in a native state (labeled as 20 °C) was statistically different in comparison to the antioxidative potency under TBH influence (labeled as 37 °C + TBH). The parameter TAS did not show statistical difference under the introduced conditions (Figure 4D).

Presentation of separate oxidative stress parameters: (A) AOPP, (B) TOS, (C) PON1, (D) TAS, and (E) SHG for the investigated samples under introduced conditions (20 °C and 37 °C + TBH).
The comparison of the results for the samples SerEDP and SerEQP (containing the SWOP emulsion) and the samples SerDP and SerQP (without the SWOP emulsion) (Figure 4) imply that the SWOP emulsion is a suitable carrier for the investigated antioxidants and that it shows beneficial effects, especially in the state with exogenous pro-oxidant TBH.
In native bioenvironmental conditions and room temperature, the best overall antioxidant power was shown by SerEQP, followed by SerEDP, and the weakest was SerEDβC. However, upon the pro-oxidant challenge and body temperature (37 °C), ie, the conditions imitating damaging processes caused by external harmful influences with oxidative stress involvement, the sample with DHQ-βC combination became a stronger antioxidant compared to the sample with DHQ only. This implies that some level of this antioxidant mixture activation could be expected upon ROS confrontation, as well as the increase of its effect in contact with body temperature.
The higher antioxidant potency of Q compared to DHQ, ie, the emulsion SQ compared to the emulsion SDHQ, could be explained by differences in the chemical structures of these antioxidants (Figure 5A and B). DHQ meets two of the three criteria for effective radical scavenging ability. The first criterion is the presence of the o-dihydroxy structure in the B ring responsible for its stability, and the second one is the 5- and 7-OH groups with a 4-oxo function in the A and C rings, responsible for a maximum radical scavenging potential. However, DHQ lacks the 2, 3 double bond in conjunction with the 4-oxo function in the C ring (Figure 5A), which makes it less potent than its oxidation product and planar hydrophobic equivalent Q, which contains this 2, 3 double bond.8 Even the addition of βC did not increase the antioxidative potency of DHQ enough to reach the one of Q. It is possible that a combination of another antioxidant with DHQ would have a more synergistic effect or that some multicomponent mixture of antioxidants at a low, physiological concentration would be more efficient, since it is known that there is a critical concentration at which the antioxidants can start to produce radicals, instead of acting as neutralizers. 11

Chemical structure of (A) dihydroquercetin (DHQ), (B) quercetin (Q), and (C) β-carotene (βC).
The SWOP emulsion containing a combination of DHQ and βC showed minor antioxidative effect amplification compared to SDHQ, which was not in agreement with the results obtained by DPPH radical assay. This disagreement could be explained by the lack of DPPH assay ability to test the antioxidant activity of βC incorporated into the SWOP emulsion. Additionally, this analysis indicated that SDHQβC showed a significant decrease in exogenous pro-oxidative effects caused by TBH, when compared to SDHQ, which could not be revealed using the DPPH radical assay test. Determination of the in vitro antioxidant activity of the antioxidants/emulsions tested in the bioenvironment has another advantage over the DPPH radical assay. Namely, using the former method, it is possible to monitor the influence of temperature (room temperature vs higher temperature corresponding to skin temperature) on the antioxidant power of the tested antioxidants/emulsions.
Spectrophotometric determination of in vitro SPF by applying the Mansur equation is a simple, easily reproducible, and economical, and it is usually performed as a screening method for a selection of the best photoprotective substance, ie, the best formulation. 4 SDHQ showed better UV absorption than SQ (SPF 4.65, ie, 3.35, respectively) (Figure 6) indicating that DHQ absorbed better in the UV spectrum than Q. The SPF of SDHQβC was the highest (5.19), probably due to the contribution of both active ingredients. Although βC is not a typical UV absorber, it probably protects DHQ from oxidation due to its antioxidant activity, and supports DHQ absorbance in the UV spectrum. The SPF of the emulsion base S was negligible (1.67) (Figure 6).

Absorbance spectra of the investigated emulsion samples.
The SPF of Q and rutin were also investigated using another method, ie, the transmittance method proposed by Diffey et al34, 35 Both Q and rutin (10%) formulations yielded similar SPF values (4.52 ± 0.38, ie, 4.72 ± 0.20, respectively). 35 In our study, the SPF values of the emulsions with Q, DHQ, and βC (SQ, SDHQ, and SDHQβC) were similar to the reported results, despite the different SPF determination methods and lower concentrations of flavonoids. Thus, these results indicate the good photoprotective potential of the selected antioxidants in combination with a prospective cosmetic vehicle such as SWOP emulsion.
Conclusions
The objective of this work was the investigation of SWOP emulsion base as a carrier for natural ingredients DHQ and βC, used in cosmetic products due to their antioxidant and UV protection activity. The results of the antioxidant effects of the tested samples revealed that the innovative biochemical approach could be used as a simple, economical, and easy achievable additional test to basic in vitro DPPH radical assay with a few advantages, especially when it comes to a testing of antioxidant activity in cosmetic formulations with complex structure such as emulsion carriers. Principally, this analysis could be more suitable for the evaluations of antioxidants and/or cosmetic products with antioxidants such as βC, since the DPPH assay was not suitable for the evaluation of its antioxidant activity. Furthermore, the analysis in a bioenvironment indicated that the emulsion with a combination of DHQ and βC exhibited a significant decrease in exogenous pro-oxidative effects. The SPF of the SDHQβC emulsion was the highest because of βC addition, which was expected. The physicochemical stability of the investigated samples was satisfactory during 30 days storage at room temperature. The presented results demonstrated that the SWOP emulsion could be used as a cosmetic vehicle for the antioxidants with UV protection ability such as DHQ and βC. However, the development of each new cosmetic product has to be accompanied by an assessment of its overall complex composition and stability.
Footnotes
Acknowledgements
This research was funded by the Ministry of Education, Science and Technological Development, Republic of Serbia through Grant Agreement with the University of Belgrade – Faculty of Pharmacy No. 451-03-68/2022-14/200161. The authors thank the Kingherbs Limited, China for the free samples of cosmetic active ingredients.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministarstvo Prosvete, Nauke i Tehnološkog Razvoja (grant number 451-03-68/2022-14/200161).