Abstract
Keywords
Introduction
Diabetic kidney disease (DKD) is a common complication of diabetes mellitus. As the incidence of diabetes mellitus gradually rises, the number of patients with DKD also increases gradually, which has a great impact on the physical and mental health of patients. With the continuous development of DKD, end-stage renal disease will be formed. At present, haemodialysis is an effective means of treatment, however, haemodialysis will have an impact on the physical health of patients. 1 Secondly, treatment by some antihypertensive drugs, such as angiotensin-converting enzyme inhibitors (ACE-I), angiotensin receptor blockers (ARBs), and dihydropyridine calcium channel blockers (CCBs), has good efficacy in DKD, however, it has more adverse effects, including kidney damage and gastrointestinal dysfunction. 2 Therefore, finding a drug with significant efficacy and fewer adverse effects has become a key goal in the treatment of DKD.
Shengjiang Powder (SJP) is composed of four Chinese herbs, including Bombyx batryticatus (JC), Cicadae periostracum (CT), Curcuma longa L. (JH) and Rheum officinale Baill. (DH). 3 It has the efficacy of elevating Qing and lowering turbidity and dispersing wind and clearing heat, and is commonly used clinically in the treatment of DKD. 4 The composition of TCM compounding is very complex, and quality control of a single drug cannot replace the composition of the entire formula, so it is necessary to analyse the chemical composition of the entire TCM compound. It is not clear what are the main active ingredients in the SJP formulation, how these ingredients enter the bloodstream to exert their effects in the treatment of DKD, and which specific targets they act on.
In recent years, UHPLC-Q-TOF-MS has provided a powerful method for the efficient separation and structural characterisation of traditional Chinese medicines due to its high resolution and sensitivity. 5 Q-TOF-MS is able to obtain accurate mass numbers of precursor ions in the full MS scans and fragment ions in the MS high-energy scans at the same time, which increases the credibility of the analysis results.6,7 In addition, Waters’ UNIFI software is a versatile automated data processing platform. The software integrates scientific libraries into a streamlined workflow that combines data acquisition, library retrieval, mass spectrometry fragment matching, and report generation, easing the workload of dealing with large amounts of mass spectrometry data and enabling rapid analysis of chemical compositions. 8 Cyberpharmacology is a biological method that can be used to analyse biological methods with multi-target and multi-pathway characteristics. Since the characteristics of Chinese medicines are in line with this, network pharmacology is currently being applied by more and more Chinese medicine research institutes, through which it is possible to connect the components of Chinese medicines with their mechanisms of action, and to elucidate the multi-component and multi-target as well as multiple efficacy aspects of Chinese medicines. 9
Currently, SJP has good therapeutic efficacy against DKD, but its pharmacodynamic material basis and mechanism of action are still unclear, which limits its clinical application. In this study, we established a comprehensive analytical strategy based on UHPLC-Q-TOF-MS and network pharmacology. Through analysing and identifying the migratory components in blood, establishing the “component-target-pathway” network, and combining with molecular docking technology for corresponding validation, the pharmacodynamic basis and mechanism of action of SJP in the treatment of DKD were revealed, and the theoretical basis was laid for its clinical application.
Materials and Methods
Materials and Reagents
JC (batch no.: 200701) was purchased from Anhui Zehua Chinese Medicine Drinking Tablets Co., Ltd, CT (batch no.: 191201) was purchased from Anhui Jishantang Traditional Chinese Medicine Science and Technology Co., Ltd, JH (batch no.: 200201) was purchased from Anhui Daoyuantang Traditional Chinese Medicine Drinking Tablets Co., Ltd, and DH (batch no.: 20080803) was purchased from Anguo Guangjitang Pharmaceutical Limited Liability Company. These herbs have all been authenticated by Professor Wang Jinhui of Shihezi University and are in accordance with the 2020 edition of the Chinese Pharmacopoeia. Standards, including quercetin, epicatechin and butyric acid, were purchased from Shanghai Amperexperiment Technology Co., Ltd and epicatechin gallate, kaempferol and leucovorin were purchased from Chengdu Mansion Bio-technology Co., Ltd and salicylic acid and gallic acid were purchased from Dr Ehrenstorfer. Our laboratory isolated the monomer compounds of Cicadellia and Stiffypods with purity over 98% by HPLC, and the specific information of the monomer compounds is shown in Table S1.10–16
Acetonitrile and methanol (LC-MS grade) were purchased from Fisher (Honeywell, USA). Formic acid was purchased from Thermo Fisher Scientific (China) Co. Ultrapure water was purified using Milli-Q water purification system (Millipore, USA).
Preparation of SJP
According to the ratio of each herb of SJP as noted in volume 4 of “Articles on Typhoid Fever and Warm Epidemic”, JC: CT: JH: DH = 2:1:3:4, appropriate amount of single herb was weighed and mixed, and immersed in 8 times the volume of water for 1 h, then the solution was boiled for 30 min and filtered through gauze, and each step was repeated 2 times. The aqueous extract of SJP was obtained and the concentrate was made into lyophilised powder and stored.
Animal Experimentation
Male SD rats (200 ± 20 g) were purchased from Xinjiang Medical University Laboratory Animal Centre. The rats were placed under controlled environmental conditions for 12 h of light-dark cycling. During the experiments, rats had free access to water and food. All animal experiments were approved and guided by the Ethics Committee for Animal Experiments of the First Affiliated Hospital of Shihezi University Medical College (ethical approval number: A2023-165-01).
All rats were fasted for 12 h before the administration of the drug and were given free access to water. The SJP extract was administered by gavage at a dose of 40 g/kg, the blank group was given the same volume of saline, and the administered group was given 3 ml of blood via the abdominal aorta of the rats 1 h after the administration, the blood was placed in heparinised centrifuge tubes and centrifuged at 4000 rpm/min for 10 min to obtain the plasma, and the plasma was obtained by taking 96-well plate HLB columns, which were pre-activated with 1 mL methanol and water balanced in 1 mL, and then each supernatant sample was taken separately 1 mL of supernatant sample, 1 mL of water drench, eluted with 1 mL of methanol, the eluent was collected and 2.0 µL was used for UHPLC-Q-TOF-MS analysis.
Instruments and Experimental Conditions
The separation of the samples was performed on a Waters Acquity H-Class Ultra High Performance Liquid Chromatograph (FTN autosampler, quadruple liquid chromatography pump) on an ACQUITY UPLC® BEH C18 column (1.7 μm, 2.1 mm × 100 mm) at a flow rate of 0.4 mL/min, with the injection volume of 2.0 μL and the temperature of the column set to 40 °C. The mobile phases A (acetonitrile) and B (0.1% formic acid-water solution) were used. The mobile phases A (acetonitrile) and B (0.1% formic acid-water solution) were used. The gradient distribution was as follows: 0-2 min, 5%-40% A; 2-5 min, 40%-65% A; 5-7 min, 65%-70% A; 7-8 min, 70%-80% A; 8-10 min, 80%-95% A;. The results were as follows.
A Waters Xevo G2 Q-TOF mass spectrometer (Waters Corporation, Milford, USA) equipped with an electrospray ionisation (ESI) source operated in both positive and negative ion modes was connected to the UHPLC with the ion source temperature at 110 °C, and the desolventising gas was nitrogen at a flow rate of 650 L h−1 and a temperature of 450 °C. The molecular weights were scanned in the range of 50-1200 Da. The energies of both positive and negative ions were 20-50 eV. The accurate mass numbers were calibrated with leucineenk ephalin (LE) solution at a concentration of 200 pg/mL. 556.2771 in positive ion mode and 554.2615 in negative ion mode to ensure accurate MS analysis.
Statistical Analysis
The data were collected using MassLynx V4.2 software and processed using UNIFI software. The acquisition time was from 0 min to 10 min. The mass detection range was 50-1200 Da, and the mass error was within 5 ppm. Addition ions were + H, +Na (positive mode) and -H, +HCOO (negative mode). After all the data have been analysed and initially processed by the software, manual checking and double-checking are still required to study the chemical compositional cleavage patterns and to improve the accuracy of the characterisation data.
The Construction of Network Pharmacology
Using the blood-entry components of SJP as candidate compounds, the sdf files corresponding to the 2D structures of the compounds or SMILES numbers were retrieved from the PubChem database, and the Swiss Target Prediction database (http://www.swisstargetprediction.ch/) was used to predict the potential relevance of each component in the the potential relevant targets of each ingredient in vivo. The keyword “diabetic kidney disease” was used to predict the potential targets of each component in vivo by GeneCards ((http://www.genecards.org/), DisGeNET (https://www.disgenet.org/), OMIM (http://www.omim.org/) databases to search for targets related to DKD. The potential targets of action and disease targets of active ingredients were uploaded to create a Venny diagram. 17
The obtained intersections were potential targets of action of SJP for the treatment of DKD. The obtained targets were uploaded to the STRING database (https://www.stringdb.org/) and imported into Cytoscape software to construct a protein-protein interaction (PPI) network to obtain key potential targets.
The key targets were enriched using gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) based on the metscape database, with the threshold value of P set at .01. The top 20 enriched pathways were screened, and the three-in-one (BP, CC, and MF) histograms and KEGG enriched bubble diagrams were established. Finally, Cytoscape software was used to build a “component-target-pathway” network.
Molecular Docking Validation
The structures of the target proteins screened by cyberpharmacology were downloaded from the RCSB PDB database (http://www.rcsb.org/), the mol2 format files of the active molecules of SJP were saved by chemdraw 3D, molecular simulation of docking was carried out by using Discovery Studio software, and the LibDockScore was used to evaluate the binding effect of SJP in the The binding effect of the main active ingredients with potential targets was evaluated by LibDockScore.
Results
Component Analysis of SJP
The chemical composition of each herb in SJP was collected and a database of SJP composition was established using UNIFI software. UPLC-Q-TOF-MS was used to detect the composition of SJP, and the base peak chromatogram (BPC) was obtained in positive and negative ion modes, and the major peaks detected were labelled with numbers, as shown in Figure 1A and B.

Chromatograms of the basal peaks in the positive (A) and negative (B) ion modes of SJP and the basal peaks in the positive (C) and negative (D) ion modes of SJP plasma.
A total of 162 constituents were identified in SJP, of which 60 belonged to DH, including anthraquinones, anthraquinones, tannins, and stilbenes; 42 belonged to JH, including curcuminoids and sesquiterpenes; 18 belonged to CT, including acetyl dopamine and amino acids; and 21 belonged to JC, including amino acids, steroids, and organic acids. Table S2 provides specific information on these compounds. A brief description of the cleavage pathways for each class of compounds in SJP is given below.
Curcuminoids
Twenty-six curcumin analogues were detected from SJP. The cleavage pattern was a single bond break between the carbonyl group and the double bond on the heptane carbon chain. 18 By comparing the cleavage pattern with the literature, peaks 134, 132, and 130 were identified as curcuminoids, demethoxycurcuminoids, and demethoxycurcuminoids. We studied the MS cleavage pattern of curcumin in detail to facilitate the characterisation of these curcumin analogues.
The quasimolecular ion peak of m/z 367.1187 [M-H]− in the negative ion mode produced m/z 149.0505 [M-H-C12H10O4]− and m/z 217.0349 [M-H-C9H10O2]− at high collision energies, which were compared with the literature, 19 and the compound was presumed to be curcumin. Figure 2 was used to characterise its cleavage pathway . In addition, UNIFI software automatically matched the database with the collected mass spectrometry data and analysed the compounds. In the negative ion mode, peak 132 quasi-molecular ion peak m/z 367.1187 [M-H]− produced fragment ions at m/z 187.0272 and 119.0420 at high collision energies, and the matching peak 132 of UNIFI was demethoxycurcumin, which was manually reviewed, and it was presumed that the [M-H]− stripped off C9H10O2 and C12H10O4 to form the m/z 187.0272 and 119.0420 fragment ions. Therefore, the compound was identified as demethoxycurcumin. Similarly, peak 130 showed demethoxycurcumin by UNIFI and manually verified matching results.

Secondary mass spectra and cleavage pathway of curcumin.
Anthraquinones
Twenty-two anthraquinones were detected from SJP, and eight free anthraquinones and 14 anthraquinone glycosides were identified based on MS database matching. Anthraquinones have strong mass spectral signals in negative ion mode, and a series of characteristic cleavage fragments of CH3, CO and H2O are easily lost under collision voltage. 20
Rhein is the major anthraquinone compound in SJP, and Figure 3 to characterise its cleavage pathway. Rhein shows its quasimolecular ion peak m/z 283.0248 [M-H]− in negative ion mode, and the loss of CO2 and CO neutral molecules generates fragment ions m/z 239.0181 [M-H-CO2]− and m/z 211.0244 [M-H-CO2-CO]−, and the further loss of CO generates m/z 183.0322 [M-H-CO2-CO-CO]−. Aloe emodin and Emodin are isomers with m/z 269.0455 [M-H]−, and at high collision energies, Emodin displays m/z 241.0266 [M-H-CO]− and m/z 225.0399 [M-H-CO-O]−, Aloe emodin can be cleaved by the characteristic cleavage behaviour of loss of CHO to produce m/z 240.0324 [M-H-CHO]− to distinguish it from Emodin. Physcion has a quasimolecular ion peak of m/z 283.0612 [M-H]−, and the loss of CO yields m/z 255.0208 [M-H-CO]− as well as the continued loss of CH3 yields m/z 240.0229 [M-H-CO-CH3]−. Chrysophanol had its paramolecular ion peak m/z 253.0506 [M-H]− and only one fragment ion m/z 225.0338 [M-H-CO]−.

Secondary mass spectra and cleavage pathway of rhein.
For anthraquinone glycosides, the identification of glycogen ions was based on the MS cleavage behaviour of free anthraquinone. Taking peak 19 as an example, its quasimolecular ion peak, m/z 445.0776 [M-H]−, produced m/z 283.0037 [M-H-162 Da]− by elimination of glucose residues and further loss of CO2 and CO produced 239.0149 [M-H-CO2]− and m/z 211.2000 [M-H-CO2-CO]−, The peak 53 was identified as 8-O-β-D-glucoside of Rhein, which has the same cleavage behaviour as that of Rhein. According to this cleavage pattern, other anthraquinone glycosides were deduced.
Stilbenoids
Eighteen stilbenes were detected in SJP, and among the stilbenes, the glycosides were easily hydrolysed to obtain glycosides, and the benzene rings on their glycosides were easily cleaved to remove acetylenic and acetylenic alcohols. 21
Taking peak 26 as an example, its quasimolecular ion peak m/z 243.0246 [M-H]−, m/z 225.0364 is the [M-H-H2O]− fragmentation ion formed by the removal of one molecule of water at m/z 243.0246, and m/z 201.3621 is the [M-H-C2H2O]− fragmentation ion obtained by the removal of one molecule of acetylenic alcohol at m/z 243.0246, and m/z 225.0364 is the fragmentation ion obtained by the removal of one molecule of acetylenic alcohol at m/z 243.0246. The chromatographic and mass spectrometric behaviours of peak 26 were consistent with those of the control, and peak 26 was identified as piceatannol. The cleavage pathway was characterised in Figure 4. Other stilbenes showed similar cleavage patterns.

Secondary mass spectra and cleavage pathway of piceatannol.
Tannins
Fourteen ellagitannins were identified from SJP, the ellagitannins are polyphenols consisting of gallic acid glucose (polyol) esters or their derivative polymers, the cleavage pattern is that the compounds are stripped of galloyl and gallic acid fragments one by one, leading to the production of a series of daughter ions differing from the quasi-molecular ions by 152 Da and 170 Da, and the resulting catechins further undergo the 1,3 bond breakage and 1,4 bond breakage on the C ring. 22
Taking peak 57 as an example, its quasimolecular ion peak m/z 441.0827 [M-H]−, one molecule of galloyl group was removed to produce m/z 289.0480 [M-H-152 Da]−, and one molecule of gallic acid could be removed to produce m/z 271.0393 [M-H-170 Da]− on the basis of the quasimolecular ion peak, which could be further broken to obtain m/z 125.0145 by breaking of 1,4 bonds of the C ring, m/z 169.0004 is its characteristic ion, and was clearly identified as epigallocatechin gallate by comparison with the control, peak 57 was clearly identified as epigallocatechin gallate. The cleavage pathway was characterised in Figure 5. Similar cleavage patterns were observed for other tannins.

Secondary mass spectra and cleavage pathway of epicatechin gallate.
Others
In addition to curcuminoids, anthraquinones, stilbenes, and tannins, we detected a variety of compounds in SJP, including organic acids, which contain α-linolenic acid, chlorogenic acid, and palmitic acid, among others. Sesquiterpenes, in turn, included curcuminoid diketones, curcuminoid enol, and gimarone.
Characterisation of SJP in Plasma
Twenty-eight components were identified in rat plasma after administration. The BPC corresponding to positive and negative ion patterns were obtained, as shown in Figure 1C and D. Thirteen constituents belonged to JC, including steroids, organic acids and amino acids; seven constituents belonged to CT, including acetyl dopamine and amino acids; three constituents belonged to JH, including sesquiterpenes; and seven constituents belonged to DH, including anthraquinones and their anthraquinone glycosides, with detailed information shown in Table 1.
Absorbed Prototype Components.

Abbreviations: JC, Bombyx batryticatus; CT, Cicadae periostracum; JH, Curcuma longa L.; DH, Rheum officinale Baill.
Network Pharmacological Analysis
Screening of Intersection-Targets
Firstly, we screened the targets of active ingredients in the plasma of the administered SJP rats, and imported the targets of active ingredients in the plasma into Swiss target prediction platform for target prediction, and obtained 526 relevant targets. Secondly, the disease-related targets of DKD were predicted and 2123 disease-related targets of DKD were obtained from Gene Cards, DisGeNET, and OMIM databases. Finally, 526 component-related targets were intersected with 2123 disease-related targets to establish a Wayne diagram, as shown in Figure 6A. and 224 potential targets for the treatment of DKD by SJP were screened. (Table S3).

Network pharmacological analysis of potential pharmacological mechanisms of SJP for DKD. A: Wayne diagram of intersecting targets of SJP and DKD; B: screening process of 39 key potential targets; C: bar diagram of GO enrichment analysis; D: bubble diagram of KEGG enrichment analysis; E: diagram of component-target-pathway (C-T-P) network construction of SJP for DKD. Yellow rectangles represent targets on the pathway, green dart shapes represent the pathway, red hexagons represent single herbs in SJP, and diamond shapes of different colours represent active compound IDs in SJP.Specific compounds corresponding to said compound IDs are listed in Table S4.
PPI Analysis and Acquisition of key Potential Targets
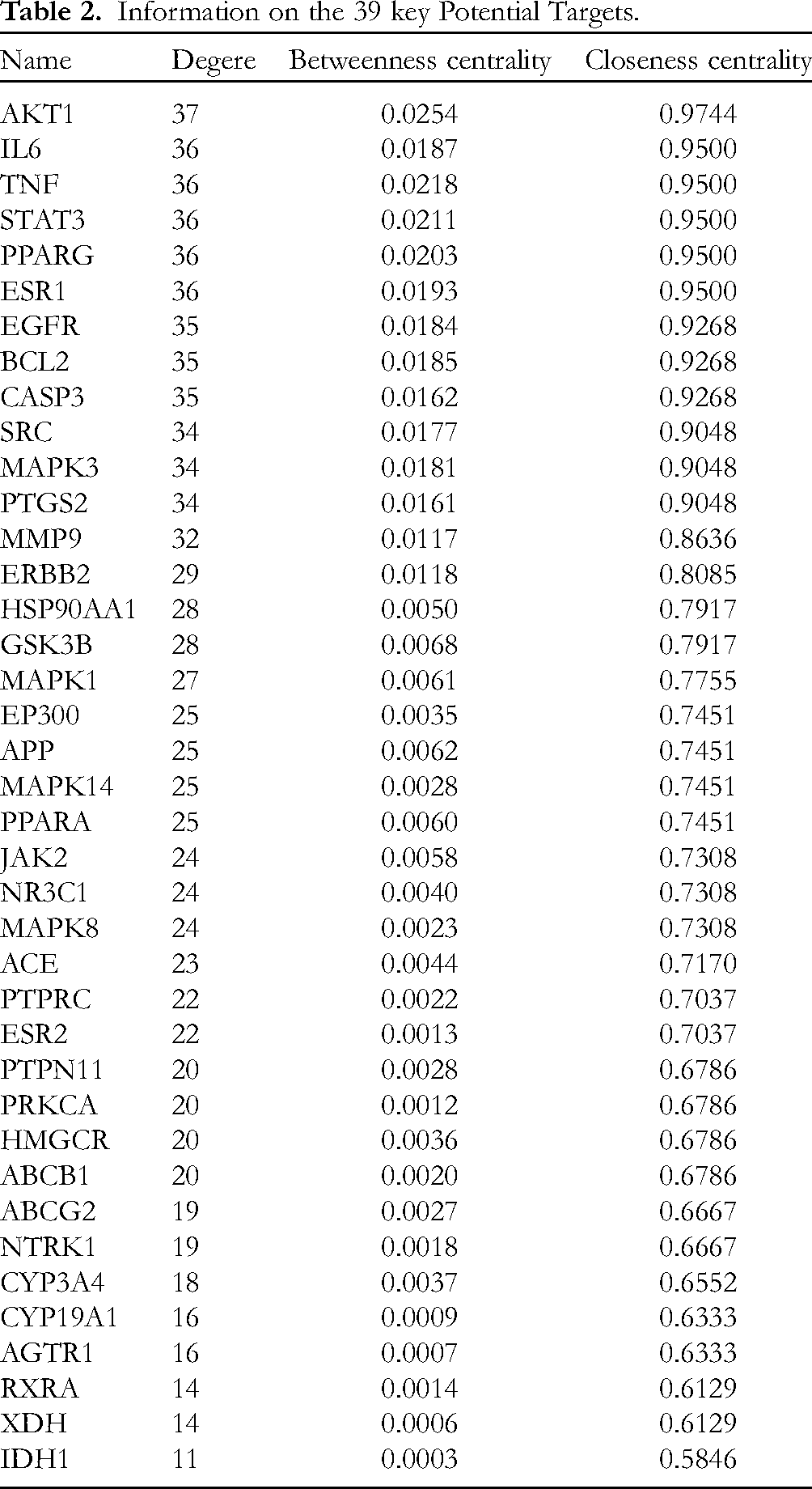
A comprehensive network of PPI relationships was obtained by analysing 224 potential targets in the String database. Cytoscape 3.7.1 software was used to further reveal the key potential targets of SJP for the treatment of DKD. Based on the screening principles of “Betweenness Centrality”, “Closeness Centrality” and “Degree” being greater than their respective medians, 39 key potential targets were screened, as shown in Table 2. The specific screening process is shown in Figure 6B.
Information on the 39 key Potential Targets.
Pathway Enrichment Analysis
Using the metscape database, GO functional enrichment analysis was performed for potential targets of SJP for the treatment of DKD. Screening the first 10 items, limited by P-value less than .01, BP analysis showed that the potential targets were enriched in the regulation of peptide-tyrosine phosphorylation, modification of peptide-tyrosine, and regulation of MAP kinase activity; CC analysis showed that the potential targets were associated with membrane raft, membrane microdomain, and membrane region CC analysis showed that the potential targets were related to membrane raft, membrane microdomain and membrane region; MF analysis showed that the potential targets were concentrated in protein tyrosine kinase activity, protein serine/threonine kinase activity and transmembrane receptor protein tyrosine kinase activity. activity, etc, and the mapping results are shown in Figure 6C.
Based on the metscape database, KEGG enrichment analysis was performed for potential targets of SJP for DKD. The top 20 pathways with the highest P values were selected to construct bubble and bar graphs, as shown in Figure 6D. It indicated that the potential targets were in Endocrine resistance, Lipid and atherosclerosis, PI3K-Akt signaling pathway, Insulin resistance, AGE-RAGE signaling pathway in diabetic complications, and HIF-1 signaling pathway were significantly enriched.
Component-Target-Pathway (C-T-P) Network Construction
Meanwhile, we mapped 20 KEGG pathways and their corresponding targets and TCM compounds to illustrate the potential regulatory mechanisms of SJP on DKD. As shown in Figure 6E, the 39 key potential targets screened by the PPI network and the core targets screened by the “component-target-pathway” network according to the degree value were intersected to obtain five targets, namely, V-akt murine thymoma viral oncogene homolog 1 (AKT1), Mitogen-activated protein kinase 3 (MAPK3), Mitogen-activated protein kinase 1 (MAPK1), Mitogen-activated protein kinase 14 (MAPK14) and Mitogen-activated protein kinase 8 (MAPK8). According to the Degree value in the “Component-Target-Pathway” network, the top five components, (2R,3S)-2-(3′,4′-Dihydroxyphenyl)-3-acetamido-7-(N-acetyl-2″-aminoethyl)-1,4-piperoxane(CT-1), 3β,5α-dihydroxy-(22E,24R)-ergosta-7,22-diene-6-one(BJC-5), 7α- methoxy-(22E, 24R)-5α,6α-epoxyergosta-8(14),22-Diene-3β-ol(BJC-2), (2S,3S)-2-(3′,4′-dihydroxyphenyl)-3-acetylamino-7-(N-acetyl-2″-aminoethylene)-1,4-benzodioxane(CT-7), (2R,3S)-2-(3′,4′-Dihydroxyphenyl)-3-acetamido-6-(N-acetyl-2″-aminoethyl)-1,4-piperoxane(CT-2), were found to have a higher degree of efficacy in the treatment of DKD, and they should be the main active ingredients in the treatment of DKD by SJP, Its chemical structure is shown in Figure 7.
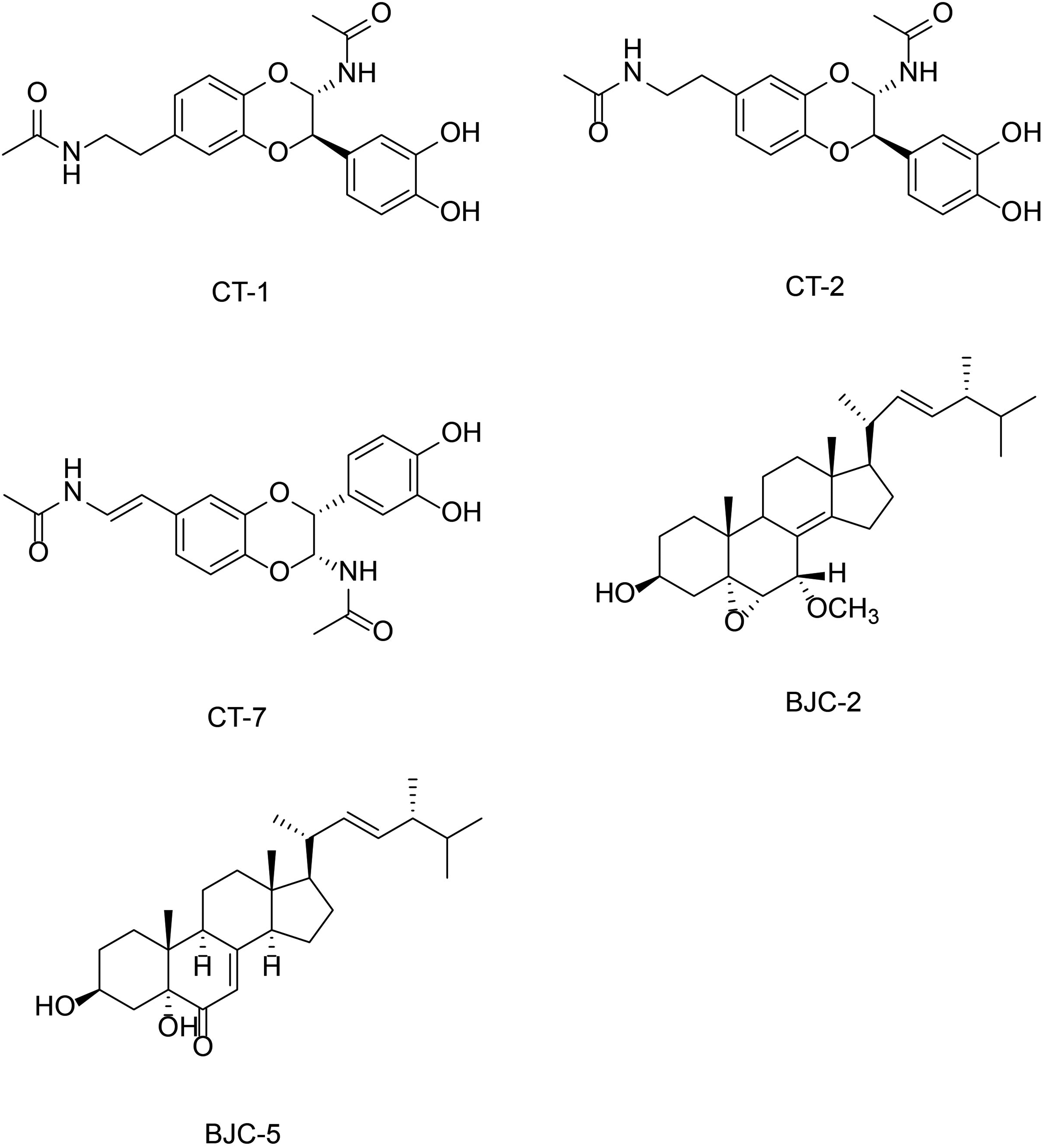
Chemical structure of the main active ingredients.
Molecular Docking
To validate the binding ability between potential active ingredients absorbed in vivo by SJP and key targets of the network pharmacological screen, we performed molecular docking simulation analysis. Five targets, AKT1, MAPK3, MAPK1, MAPK14, and MAPK8, were selected as predicted receptors for SJP. Five active ingredients were selected as potential ligands for the above targets, namely CT-1, BJC-5, BJC-2, CT-7, and CT-2. It is generally believed that the more stable the conformation of the ligand and the receptor, the higher the LibDockScore, and the higher the possibility of their binding. The molecular docking LibDockScore results are shown in Table 3. The 3D simulation and 2D simulation of molecular docking are given in Figure 8, respectively, which clearly show the binding modes and 3D structures of various active ingredients with key targets.

Molecular docking simulation results of SJP active ingredients with key targets. A: BJC-5 and AKT1 molecular docking structure; B: CT-1 and MAPK1 molecular docking structure; C: CT-1 and MAPK3 molecular docking structure; D: CT-2 and MAPK8 molecular docking structure; E: CT-7 and MAPK14 molecular docking structure. (a) 3D molecular docking structure; (b) detailed 3D molecular docking structure; (c) 2D molecular docking structure.
Molecular Docking Analysis of SJP Active Ingredients with Candidate Target Proteins.

The results showed that the five active ingredients had strong binding ability to five targets, among which BJC-5, CT-2 and CT-7 had the best binding ability to AKT1. CT-1 and CT-7 had the strongest binding ability to MAPK1. CT-1 and CT-2 had the strongest binding ability to MAPK3 as well as MAPK8. CT-7 had the strongest binding ability to MAPK14. Since AKT1, MAPK3, MAPK1, MAPK14, and MAPK8 play key roles in DKD, the binding ability of the active ingredients in SJP to these targets suggests that its therapeutic mechanism may be closely related to these targets.
Discussion
SJP is a traditional Chinese medicine compound formula commonly used in the treatment of DKD. However, there is a lack of research on the material basis and mechanism of action of SJP for the treatment of DKD. Therefore, this study developed an integrated research strategy combining UPLC-Q-TOF-MS and network pharmacology to reveal the material basis and mechanism of action of SJP for the treatment of DKD.
The composition of SJP was analysed using UPLC-Q-TOF-MS. A total of 162 compounds were identified, including curcuminoids, anthraquinones, stilbenes, tannins, and so on. In addition, we analysed the components that entered the circulation after SJP administration to rats and obtained a total of 28 components. It is noteworthy that curcumin analogues account for a large proportion of the aqueous extract of SJP, yet their prototypical components are barely detectable in blood. According to the literature, 23 curcuminoids have a wide range of pharmacological effects but their bioavailability is extremely low, suggesting that the activity of curcuminoids is mainly related to their metabolites. Many studies have confirmed the involvement of inflammation in the pathogenesis of DKD and the progression of renal injury, 24 and acetyl dopamine polymers from CT have anti-inflammatory and antioxidant properties, 25 suggesting a therapeutic role in DKD. Some studies have shown that when taken for two consecutive months, the JC have a significant hypoglycaemic effect, resulting in a therapeutic effect on DKD, 26 in which the steroidal compounds from the JC are likely to be the potent substances. Curcuminol from JH has anti-inflammatory, antidiabetic, and anticancer pharmacological effects, 27 and curcuminodione has anti-inflammatory and antioxidant properties. 28 Anthraquinones from DH, such as rhein, can have a variety of pharmacological effects such as hypoglycaemic, renoprotective, lipid-regulating and anti-nephritic effects. 29 Emodin plays a renoprotective role in DKD by inhibiting iron metabolism and restoring Nrf2-mediated antioxidant capacity. 30 These components are essentially identical to the pharmacological effects of SJP, so it is hypothesised that they may be the pharmacodynamic substances of SJP. An “component-target-pathway” network was created using Cytoscape. There are 148 nodes and 851 edges in the network. Nodes with degree values greater than twice the median of such nodes are generally considered to be key nodes in the network.The degree values of the five components, CT-1, BJC-5, BJC-2, CT-7, and CT-2, were all more than four times the median. The results showed that CT-1, BJC-5, BJC-2, CT-7, and CT-2 had higher degree of action in the treatment of DKD, and should be used as the main active ingredients in the treatment of DKD by SJP. Through the PPI network and “C-T-P” network, the core targets were mainly AKT1, MAPK3, MAPK1, MAPK14 and MAPK8, which might affect the PI3K-Akt signaling. PI3K-Akt signaling pathway, Insulin resistance and AGE-RAGE signaling pathway in diabetic complications. In order to validate the relevant results of network pharmacology, we performed molecular docking simulations of the five potential active ingredients screened with five in vivo target proteins Molecular docking simulations were carried out, and the results showed that all five potential active ingredients had strong binding ability to AKT1, MAPK3, MAPK1, MAPK14, and MAPK8 targets, and exerted their therapeutic effects on DKD through the above targets.
AKT1, also known as Protein Kinase B, is an important member of the PI3 K/Akt pathway, and activation of AKT1 promotes cell proliferation and inhibits apoptosis. It is an important player in the immunoinflammatory mechanism of DKD. 31 Hyperglycaemia consistently reduces the expression of the Akt activation inhibitor, the lipophosphatase PTEN, and activation of the Akt signalling pathway promotes thylakoid proliferation, while high glucose induces collagen production by thylakoid cells exacerbating nephropathy. 32 Mitochondrial dysfunction can be regulated by AKT1, which may play a key role in nephropathy and subsequent renal failure, and activation of mitochondrial AKT1 enhances the efficiency of oxidative phosphorylation and reduces respiratory uncoupling.In addition to local effects on renal function in diabetic nephropathy, phosphorylation of AKT1 directly promotes proliferation of mesangial cells, thickening of the basement membrane, tubular epithelial cell transdifferentiation and podocyte injury. The critical role in the regulation of systemic metabolism, insulin sensitivity and body composition. 33
MAPK1, also known as extracellular signal-regulated protein kinase, is a member of the MAPKs family, which contributes to enhanced cell proliferation and is involved in physiopathological processes such as cell metabolism, proliferation, apoptosis, differentiation and survival. It has been demonstrated that the MAPK pathway exerts apoptosis inhibiting, proliferation promoting and anti-inflammatory effects in DKD rats associated with MAPK1. 34 MAPK1 increases a variety of inflammatory and adhesion factors in glomerular cells, exacerbating the damage in the pathological state of DKD. 35 MAPK3/ERK1, a member of the MAPK family, is involved in growth factor signalling. It has been reported that activation of glomerular ERK pathway may have a significant effect on the progression of DKD. 36 Overactivation of MAPK8 plays a key role in the pathogenesis of insulin resistance and obesity, and MAPK8 knockout mice exhibit improved obesity and insulin sensitivity. 37 MAPK14, also known as p38α, is an important component of the p38MAPK signalling pathway. The p38MAPK signalling pathway is activated after high glucose stimulation, which promotes the release of inflammatory factors IL-6, IL-1, and TNF-α from podocytes and renal tubular epithelial cells, mediating renal microinflammation and fibrosis. 38
Our study showed that BJC-5, CT-2 and CT-7 had the best binding ability to AKT1. CT-1, CT-7 had the strongest binding ability to MAPK1. CT-1 and CT-2 had the strongest binding ability to MAPK3 as well as MAPK8. CT-7 had the strongest binding ability to MAPK14. The interactions of the above components with their targets validated the results of network pharmacology. Finally, we found that multiple active components of SJP acted on multiple targets in vivo and exerted synergistic effects, which may be hypothesised to be related to the reduction of renal mesangial cell proliferation, inhibition of inflammatory factors, and increased insulin sensitivity. In this study, the blood-entry components of SJP were explored only through network pharmacology, and the target of action and therapeutic mechanism of the formula were predicted. The results require further experimental verification.
Conclusion
In this study, we analysed the chemical composition of SJP using UPLC-Q-TOF-MS and identified 162 compounds, including curcuminoids, anthraquinones, stilbenes and tannins. In addition, we analysed the components that entered the blood circulation after administration of SJP to rats, and a total of 28 components were obtained. Then we conducted a network pharmacological study and found that the potential active ingredients of SJP for DKD treatment included CT-1, BJC-5, BJC-2, CT-7, CT-2, and the key targets were mainly AKT1, MAPK3, MAPK1, MAPK14, and MAPK8, which might affect the PI3K-Akt signaling pathway, Insulin resistance, AGE-RAGE signaling pathway in diabetic complications and so on. In order to validate the results of network pharmacology, we performed molecular docking simulations of the five potential active ingredients screened with five target proteins in vivo, and the results showed that SJP mainly acted on AKT1 and MAPK-related targets via CT-1, BJC-5, BJC-2, CT-7, CT-2, and SJP may exert its therapeutic effect on DKD through mechanisms related to reducing renal mesangial cell proliferation, inhibiting inflammatory factors, and increasing insulin sensitivity.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241277543 - Supplemental material for Compositional Analysis and Network Pharmacological Strategy for the Treatment of Diabetic Kidney Disease by Shengjiang Powder Based on UHPLC-Q-TOF-MS
Supplemental material, sj-docx-1-npx-10.1177_1934578X241277543 for Compositional Analysis and Network Pharmacological Strategy for the Treatment of Diabetic Kidney Disease by Shengjiang Powder Based on UHPLC-Q-TOF-MS by Yongzhi Yin, Lu Yang, Ke Zhang, Silin Yu, Jinfa Liao and Jinhui Wang in Natural Product Communications
Footnotes
Acknowledgments
This research was funded by the National Natural Science Foundation of China, grant number 81860725.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, (grant number 81860725).
Ethical Approval
The study was approved by the Ethics Committee for Animal Experiments of the First Affiliated Hospital of Shihezi University Medical College (ethical approval number: A2023-165-01).
Statement of Human and Animal Rights
All animal experiments were approved and guided by the Ethics Committee for Animal Experiments of the First Affiliated Hospital of Shihezi University Medical College.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
