Abstract
Objectives
The objective of this study was to investigate the potential anti-cancer effects of Micromeria fruticosa plant infusion on uterine cervix cancer cells, aiming to explore its potential as an alternative or supportive therapeutic approach to conventional treatments.
Methods
Micromeria fruticosa plant infusion was prepared and administered to uterine cervix cancer cells in vitro. Various assays were conducted to assess its impact on cell migration, proliferation, and tumorigenicity. Additionally, the phytochemical composition of the infusion was analyzed by using LC-MS to identify major functional compounds.
Results
The study revealed that Micromeria fruticosa plant infusion exhibited significant anti-migratory, anti-proliferative, and anti-tumorigenic effects on uterine cervix cancer cells. Analysis of the infusion identified several major functional phytochemicals, which likely contribute to its anticancer properties.
Conclusion
This research suggests that Micromeria fruticosa plant infusion possesses promising anti-cancer properties against uterine cervix cancer cells. The identified phytochemicals present in the infusion highlight its potential as a source of functional ingredients with anticancer effects. These findings support further exploration of Micromeria fruticosa as a potential therapeutic agent in both nutraceutical and pharmaceutical applications, offering new avenues for the development of alternative or supportive treatments for cervical cancer.
Introduction
Micromeria fruticosa L (Lamiaceae) is an aromatic herb, widely used in different Mediterranean regions for various medicinal and traditional uses as herbal infusion due to its pleasant minty fragrance.1,2 It also provides a sensation of coolness in the hot summer. Extracts of M. fruticosa leaves were used for respiratory system disorders, skin infections, wounds, fever, and eye redness. In Palestine, the infusion of M. fruticosa areal parts is used in treatment of diabetes, cough, headaches, and urinary diseases beside possessing anti-microbial and anti-oxidant activities. 2 Recently, the volatile oil and the aqueous extract of M. fruticosa were reported to have significant anticancer activities against human colon and breast tumor cells.3,4 Besides, the toxicity study of the aqueous extract of M. fruticosa has been evaluated in vivo on mice and showed safe up to 5 g/kg. 5 Regarding the essential oils of M. fruticosa, pulegone, a significant constituent, has been recognized by the European Medicines Agency (EMA) for its hepatotoxic effects. In light of this, the EMA suggests a daily intake of pulegone for a person weighing 60 kg to be around 2.3 mg/kg body weight. Additional in vivo research is necessary to explore the potential pharmacological effects and to evaluate the safety and toxicity of the plant extract. The major constituents of M. fruticosa volatile fraction were monoterpenes, and sesquiterpenes. 2 Recently, the polar extracts of M. fruticosa were shown to comprise a wide array of phytochemicals such as: phenolic acids, flavonoids, and their glycosides in addition to terpenoids and sulfate derivatives. 6 Phenolics are plant metabolites with well-known protective action against various health diseases. 7 Although the plant infusions and extracts are used extensively in the folk medicine, little information is available in the literature about the major phyto-compounds responsible for M. fruticosa therapeutic uses. 6
Cancer disease is a major cause of mortality worldwide. An approximate estimation of cancer deaths in 2020 accounts for nearly 10 million in the globe. 8 While radiotherapy and chemotherapy are considered as the most active approaches to treat different cancer types, they cause adverse side effects on patient's health including nausea, hematologic toxicity and liver dysfunction. 9 Moreover, patients suffering from recurrent cancer may have limited treatment choices and a poor prognosis. 9 Therefore, it is primordial to search for alternative drug options to fight against cancer. The present work aimed to identify the main composition and to investigate the effect of M. fruticosa infusion uterus cervix cancer cell growth and migration.
Materials and Methods
Chemicals
All chemicals utilized were of analytical reagent grade and employed without further purification. Double-deionized water was obtained using a Milli-Q system (Millipore, Bedford, MA). Analytical grade acetic acid was procured from Fluka (Buchs, Switzerland), while HPLC-grade acetonitrile and methanol were sourced from Labscan (Dublin, Ireland).
Micromeria Fruticosa Plant Extraction
The samples of Micromeria fruticosa were botanically identified by Dr N. Jaradat, Department of Pharmacy at An-Najah National University. The voucher specimen is labeled as Pharm-PCT-1575. Micromeria leaves were air-dried, and their aqueous extract was prepared by the infusion method which analogues the traditional preparation method. Thus, 150 mL of boiled water was added to the plant (3 g) then left to stand for 15 min, then filtered and the filtrate was lyophilized. The obtained residue (extract) was stored in the freezer until analysis. Micromeria fruticosa extract's stability over time depends on controlled storage temperatures (typically refrigerated or frozen), protection from light and oxygen, and maintenance of proper pH and humidity levels. Environmental factors such as temperature fluctuations, light exposure, and humidity can alter the extract's chemical composition and efficacy, emphasizing the need for careful handling and storage practices to preserve its quality and effectiveness.
Phytochemicals’ Analysis by LC-MS (RRLC-DAD-ESI/MS)
Phytochemical compounds were analyzed using rapid resolution liquid chromatography-diode array detection/electrospray ionization tandem mass spectrometry (RRLC-DAD-ESI/MS). 10 Sumac samples were analyzed using an Agilent-1100 series high-pressure liquid chromatograph equipped with a Zorbax C18 reverse-phase column (10 mm × 4.6 mm, particle size 5 µm; Agilent Technologies, Palo Alto, CA, USA). The mobile phase consisted of water with 1% formic acid (A) and methanol (B), with a gradient elution profile as follows: 5%–15% B (0-5 min), 15%–25% B (5-7.5 min), 25%–50% B (7.5-25 min), 50%–85% B (25-33 min), followed by a 3 min post-run after each analysis. The column temperature was maintained at 25 °C, and the flow rate was set at 0.2 mL/min. Detection was performed using a diode array detector (DAD) for multi-wavelength detection within a range of 190–580 nm, interfaced with an AB Sciex API-5000 MS equipped with a Turbo V ESI source (Foster City, CA). The mass spectrometer operated in full scan mode under negative ionization conditions, with source and capillary voltages set at −10 V and 4.0 kV, respectively, and a capillary temperature of 270 °C. N2 was employed as the sheath gas at a flow rate of 8 L/min, and nitrogen was utilized for collision-induced dissociation at a normalized collision energy of 50%.
Culture of Hela Carcinoma Cells
HeLa cells are immortalized human uterine cervix epithelial cell lines mutated by human papilloma virus 18 (HPV18). Cells were cultured in a RPMI 1640 media complemented with 10% fetal calf serum (FCS), penicillin/streptomycin (1%), L-glutamine (1%). Cells were maintained in an incubator at a temperature of 37 °C in an atmosphere of 5% CO2.
Cell Proliferation Assay
26 000 cells were seeded per well in a 96-well plate. Cells were cultured with 0, 250, 500, 1000, 2000, 4000 and 8000 μg/mL of M. fruticosa extract for 24 h. The viability of cells was evaluated by CellTiter 96® Aqueous One Solution Cell Proliferation Assay (MTS) in accordance with the manufacturer's guidelines (Promega Corporation, Madison, WI). Eventually, 20 μL of MTS solution /100 μL of culture medium was added to cells in each well and incubated for 2 h at 37 °C. The wavelength of 490 nm was used to determine absorbance.
Wound Healing Migration Assay
24-well plates were utilized to incubate HeLa cells until confluence. The monolayer of confluent cells was scratched with a plastic sterile pipette tip in arrange to create a vertical wound across each well. All conditions were performed in triplicate wells in each experiment. Afterwards, cells were rinsed with PBS then incubated in the presence of a vehicle or of specified concentrations of M. fruticosa extract during 24 h in RPMI-1640 complete medium at 37 °C. 11
Migration Capacity Analysis
Wound length in the vertical diameter of the well was photographed using an inverted microscope (LaboMed- ARCO Med TCM-400 microscope - https://www.laboamerica.com/products/life-materials-sciences/tcm 400#specifications) at 40× of total magnification (10×/22 mm eyepiece * objective lens 4X). Three wells were considered for each condition. Three pictures were taken per well. Totally, 9 reads for each condition were considered per experiment. ImageJ software and MRI Wound Healing Tool (http://dev.mri.cnrs.fr/projects/imagej-macros/wiki/Wound_Healing_Tool) were used to determine the wound area at 0 h and 24 h. Wound area invaded by cells after 24 h was measured as follows: (Area of wound at time zero (0 h) – Area of wound at 24 h). Ultimately, the invaded area for each treated condition is expressed as a percentage of the control values.
Three-Dimensional Spheroids’ Formation
Spheroids/clusters formation was carried out using Hanging drop technique similarly as detailed previously. 12 Briefly, HeLa cells were cultured to confluence. Cell count was adjusted to 2.5 × 106 Cell/mL. Drops of about 10 μL of cells was deposited on the lid of a 60 mm tissue culture plate and 5 mL of PBS were placed in the bottom of the plate. The lid was inverted onto the PBS-filled bottom chamber and incubated at 37 °C and 5% CO2. Spheroid formation was monitored and images were taken after 72 h using an inverted microscope.
Data Analysis
Graphs’ design was carried out utilizing Graph pad Prism version 7. Data were reported in SEM; n = 3. ns stands for non-significant. *(P < .05), **(P < .01) signifies a statistical difference from the control at each time point (Unpaired t-test).
Results
Phytochemicals Identification by Liquid Chromatography–Mass Spectrometry (LC-MS)
The phenolics found in M. fruticosa leaves extract were identified utilizing DAD spectra and MS2 acceptable data. Figure 1 shows the characteristic UV chromatographic profile MF aqueous extract. In Table 1, a list of the key identified chemical compounds is outlined, along with their UV maximum absorption and MS2 fragmentation pattern in the negative ESI ionization mode, and by comparison with data available in the literature. The identified compounds were numbered in accordance with their elution order.

UV spectrum of the major identified compounds in Micromeria fruticosa.
List of major Compounds Identified in Micromeria fruticosa Leaves Infusion.

Peaks
It's worth noting that Peak
Micromeria fruticosa Extract Possesses an Inhibitory Impact on Cervical Carcinoma Migration
Cancer migration and invasion constitute together a way for cancer cells to escape the primary tumor in order to metastasize to distant organs and tissues. Metastatic events play major role in increasing cancer mortality. Interestingly, modulating cancer migratory capabilities is a powerful preventive/therapeutic approach. Thus, we investigated the effect of M. fruticosa extract on the migration of cervical cancer cells. We found that M. fruticosa significantly decreases the migration capacity of HeLa cells after 24 h of treatment (Figure 2). When cells were treated with 500 or 750 mg/mL of M. fruticosa extract, we observe an inhibition of the migration capacity by around 40% compared to non-treated cells. These results verify that M. fruticosa extract (infusion) possesses an inhibitory impact on uterine cervix cancer cells migration.

Micromeria fruticosa extract possesses an inhibitory activity on cervical carcinoma migration.
Micromeria fruticosa Extract Possesses an Inhibitory Activity on Cervical Carcinoma Viability
To determine the effect of M. fruticosa extract on HeLa cancer cells viability, we incubated HeLa cells with 0, 250, 500, 1000, 2000, 4000 and 8000 mg/mL of M. fruticosa extract during 24 h and we evaluated the viability of these cells compared to non-treated HeLa cells. We found that 4000 mg/mL and 8000 mg/mL of M. fruticosa extract have reduced the proliferative activity of HeLa cells of about 40% to 80% compared to non-treated cells (Figure 3). Our data show a significant anti-proliferative effect of M. fruticosa extract on cervical carcinoma viability.

Micromeria fruticosa extract possesses an inhibitory activity on cervical carcinoma viability.
Micromeria fruticosa Extract Hinders Cervical Cancer 3D Aggregates Formation
As a significant advancement over 2D culture methods, innovative three-dimensional (3D) in vitro models recapitulating tumor architecture and behavior within its microenvironment have been developed. Using three-dimensional models provides more accurate prediction approaches for personalized medicine, targeted therapies screening, and preclinical investigation.16,17 The capacity of cancer cells to form spheroids reveals an increased chemoresistance and cancer stemness.15,18 Therefore, we investigated whether M. fruticosa extract can perturb HeLa cancer cells’ ability to form spheroids. HeLa cells were able to form mainly a large cluster when incubated in complete media without any treatment. Once treated with M. fruticosa extract, HeLa cells clumped together in a smaller and more distorted cluster (Figure 4a). Clusters area and circularity were analyzed (Figure 4b, c and d). Our data show significant reductions in clusters area and roundness in M. fruticosa treated condition compared to Control. Together, our investigations demonstrate M. fruticosa extract ability to hinder cervical cancer 3D aggregates formation.
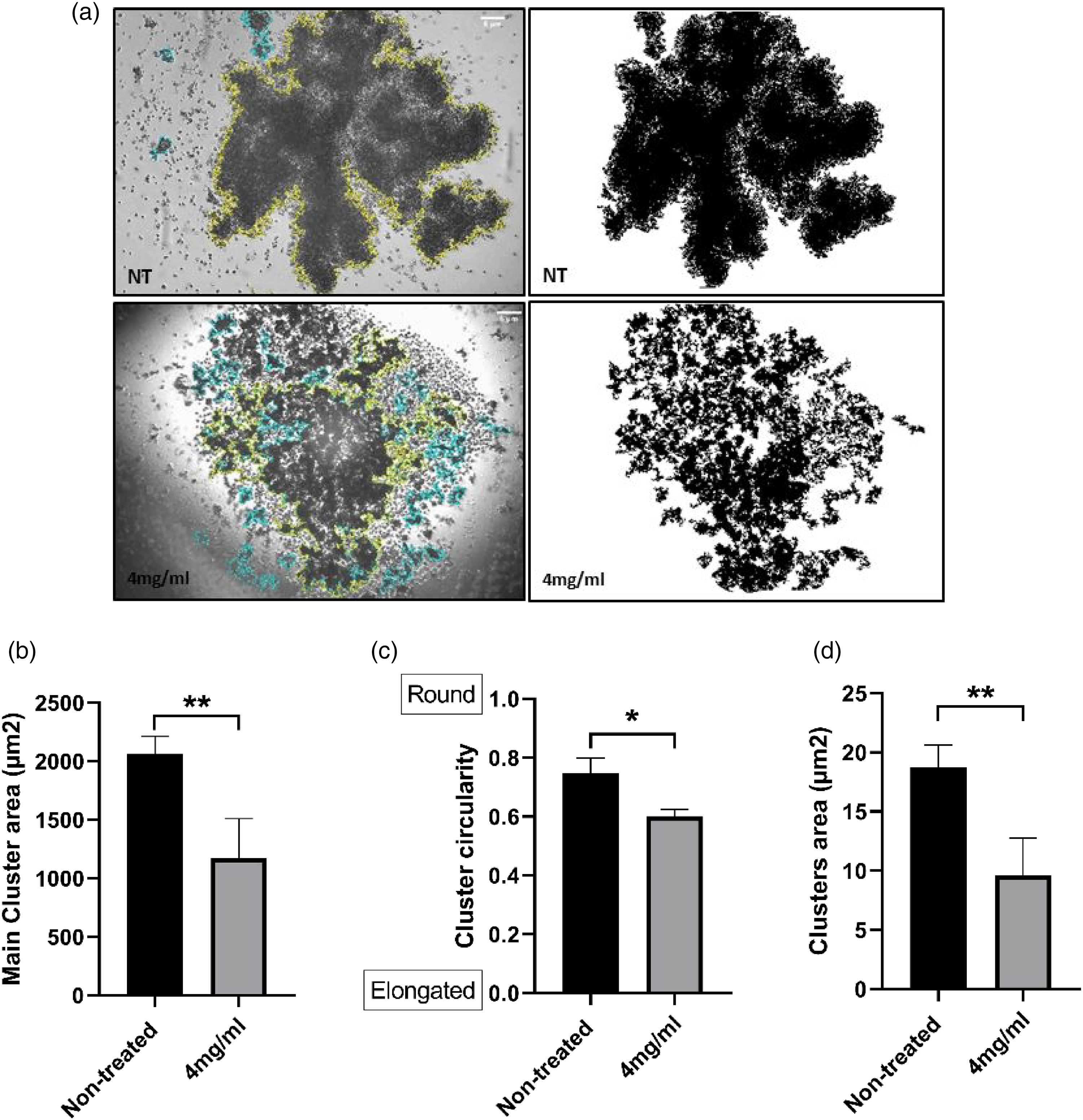
Micromeria fruticosa extract hinders cervical cancer 3D aggregates formation.
Discussion
Plants natural products and their phytochemical components are taking on greater importance in disease prevention and treatment. M. fruticosa is a Mediterranean medicinal plant that has been broadly used in folk medicine. Our study shows that M. fruticosa extract has an inhibitory activity on the 3D Spheroid formation, 2D cell migration and proliferative capacities. We are first to demonstrate that non-cytotoxic concentrations of M. fruticosa extract has an anti-migratory impact on uterine cervical cancer cells (Figure 2). In our prior studies, it was demonstrated that both Rhus coriaria and Nepeta curviflora exhibit a reduction in the migration capacity of cervical cancer cells.11,19 Variations observed in the effective concentrations are attributed to differences between the plant species.
Multicellular tumor clusters have been identified in the systemic circulation, at tumor invasive fronts and invading distant organs.20-22 In many cancer types, tumor cell clusters found in circulation are associated with bad prognosis.23,24 In addition, tumor-derived spheroids are different in that they are intended for the enrichment of cancer stem cells (CSCs) or cells with stem cell-like features. 25 Cancer stem-like cells (CSCs) have been linked to tumor recurrence and the development of chemoresistance. 26 Remarkably, Cheung and coworkers have shown that inducing cancer cells to cluster increases their propensity to generate metastases by 500-fold compared to single cell metastasis. 27
We have also found out that M. fruticosa extract has an anti-proliferative effect on cervical cancer cells (Figure 3). Previously, it has been shown that M. fruticosa extract has an inhibitory activity on the cell proliferation and induces cell cycle arrest in mammary and colorectal carcinomas. 28 Differences in concentrations in comparison to our study may be related to extract preparation and cell type differences. Altogether, those results confirm the anti-proliferative activity of M. fruticosa extract and suggest extending these findings to other cancer cells’ types.
Our work manifests M. fruticosa extract capacity to inhibit the production of cervical cancer 3D spheroids (Figure 4). Previous studies showed plant extracts capacity to perturb tumor spheroids’ formation.29,30 In conjunction, those data prove for the first time M. fruticosa extract impact on aggregates formation indicating its anti-tumorigenic properties.
Recent studies revealed an anti-bacterial, antifungal and antioxidant effects of M. fruticosa extract. 2 ) In this paper, the study entails the phytochemicals’ characterization and the hindering abilities on cell proliferation, cell migration and three-dimensional spheroids’ formation capacity of the Micromeria fruticosa infusion which sheds light on the potential in vitro anti-cancer properties of M. fruticosa plant infusion against uterine cervix cancer cells. However, few limitations are noteworthy such as: The research primarily relies on in vitro experiments using HeLa cell line, offering valuable insights through applying two dimensional and three-dimensional settings but lacking the complexity of interactions within a living organism. Careful interpretation is needed before extrapolating to clinical settings. Several signalling pathways are involved in the progression of cervical carcinoma such as JAK/STAT, Wnt/Beta catenin, PI3 K/AKT and NFKB signalling pathways. 31 Interestingly, M. fruticosa was shown to induce a cell cycle arrest of breast and colon carcinomas via hindering the expression levels of survivin, cyclin dependent kinase 1 (cdk1) and cyclin B1. 28 In addition, M. fruticosa inhibited melanoma cell migration and reduced tumor growth in vivo through regulating MMP-9 and NFKB pathway. 32 Because of restricted funding, this study solely conducted nontargeted phytochemical identification without using commercial standards which comes with certain constraints. While major phytochemical compounds are identified, in-depth exploration of their mechanisms of action is not mentioned. Previous studies have validated the effect of M. fruticosa on melanoma tumor growth. We understand that an in vivo validation experiment for cervical cancer is essential for confirming observed effects in a physiologically relevant context. Further research is essential for advancing the understanding of Micromeria fruticosa's potential as a therapeutic agent for cervical cancer. Notably, it has been proved that multiplex chemotherapy is required and unavoidable once compared to single chemotherapeutic approach. Nevertheless, traditional chemotherapy lacks cell selectivity between normal and cancer cells, which can have major adverse effects. 33 (Wang J. Combination Treatment of Cervical Cancer Using Folate-Decorated, pH-Sensitive, Carboplatin and Paclitaxel Co-Loaded Lipid-Polymer Hybrid Nanoparticles. Drug Des Devel Ther. 2020 Feb 26;14:823-832. doi: 10.2147/DDDT.S235098. PMID: 32161442; PMCID: PMC7049774.) Altogether, these data may suggest M. fruticosa as a natural product that might be included in a combined therapy approach to reduce cell migration capacity and tumor growth.
M. fruticosa is an abundant source of functional compounds with anticancer properties which may encourage the frequent use of this plant as an enriched source of phytochemicals and can have promising use in the nutraceutical and pharma industries. Nevertheless, more research is still in need to reveal the other bioactivities and to separate the individual components to explore the functional and the bioactive compounds in charge of the anticancer and anti-migration activities.
Conclusion
Our findings show that M. fruticosa plant infusion holds an anti-migratory, anti- tumorigenic and anti-proliferative properties on uterus cervix cancer cells. The major functional components identified in the Micromeria infusion could be the main components responsible for this bioactivity. M. fruticosa represents a promising source of functional ingredients with anticancer properties, which may encourage the further use of this plant as an enriched source of phytochemical and might be promising for the nutraceutical and pharma industries’ use.
Footnotes
Acknowledgements
Authors contributions
SA developed ideas and concepts. SA designed and performed 3D Spheroids’ formation, cancer migration, proliferation experiments. AM performed proliferation experiments. IA designed, performed, analysed and wrote phytochemical analysis part. SA analysed data and wrote the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
