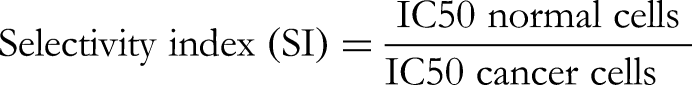Abstract
Background
Breast cancer poses a major threat to female health worldwide. One of the medicinal plants used in traditional Saudi medicine is the Juniperus procera (Arar) plant, which is used to cure various illnesses, including cancer.
Objectives
The cytotoxic properties of Saudi J. procera extracts and their regulatory mechanisms against tumorigenesis in breast cancer were investigated.
Methods
The antioxidant capacities of the fruit and leaf extracts were detected by the 2,2′-azino-bis 3-ethylbenzothiazoline-6-sulfonic acid (ABTS) assay and 2,2-diphenyl-1-1picrylhydrazyl (DPPH) assay. The cell viability of the extracts was determined by 3-(4,5-dimethylthiazoyl)-2,5-diphenyl-tetrazolium bromide assay. The cell migration was detected by a wound healing assay. The cell cycle and cell apoptosis were analyzed by flow cytometry. The reactive oxygen species (ROS) was evaluated using a DCFDA-cellular (the cell permeant reagent 2',7' –dichlorofluorescein diacetate) reactive oxygen species detection assay. The protein and gene expression levels were estimated by western blotting and Reverse transcription polymerase chain reaction (RT-PCR), respectively.
Results
Phytochemical analysis revealed a high content of phenolic and flavonoids in fruit and leaf extracts. Relative to leaf extract, the fruit extract showed higher antioxidant activities and exhibited more potent cytotoxic effects against two breast cancer cell lines: MCF-7 and MDA-MB-231. The fruit extract arrested the cell cycle of the tested cancer cell lines in S phase and G1 phase, and downregulated PI3K (phosphoinositide 3-kinase-protein kinase)/Akt (protein kinase B)/mTOR (Mammalian target of rapamycin) pathway. It also suppressed aromatase expression in MCF-7 but upregulated it in MDA-MB-231.
Conclusion
J procera fruit and leaf extracts have significant antitumor activities against MCF-7 and MDA-MB-231 breast cancer cell lines.
Introduction
Cancer is a leading cause of mortality and morbidity globally.1,2 Breast cancer (BC) is highly heterogeneous and the most diagnosed cancer in females (11.7% of total cases), which poses a major threat to female health worldwide. Its incidence and fatality rates are predicted to rise sharply in the coming years. 3 Breast cancer is the most common type among Saudi women (29%), 2 which necessitates intensive research for finding effective treatments and thus reduce its spread among patients.
The two metabolically distinct breast cancer cell lines, MCF7 and MDA-MB-231, represent a striking example of invasive ductal/breast carcinoma cells and have several phenotypic as well as genetic differences. The MCF7 cell line is hormone-dependent, estrogen receptor (ER) and progesterone receptor (PR) positive and is widely studied epithelial cancer cell line derived from breast adenocarcinoma. However, MDA-MB-231 is a triple-negative breast cancer cell line, greatly invasive, and aggressive. It is a poorly differentiated cell line because the lack of PR expression, ER as well as HER2 amplification.4,5 The lack of an ER rendered MDA-MB-231 insensitive to antiestrogen therapies that are widely used in breast cancer chemoprevention and as an adjuvant to treat the primary disease. 4 Normal breast epithelial cells (MCF-10A) are used as normal controls since they are untransformed nontumorigenic epithelial cells similar to those found in normal human breast tissue. 6
Many pharmacological components commonly used as anticancer agents were first isolated from plants. Recently, utilization of plant-derived bioactive compounds has emerged as a promising tool to control cancer cell division. 7 Since plant-derived anticancer are natural, safer, and have lower rates of adverse effects than synthetic anticancer chemicals, they are frequently employed in chemotherapy as possible source of anticancer and anti-proliferative substances. 7 Co-Application of medicinal plants derived bioactive compounds and synthetic anticancer chemicals can significantly reduce the chemotherapy-related harmful side effects via increasing cancer-killing effectiveness or strengthening the patient's immune system. 7 In cancer therapy, the anticancer drugs related genotoxicity and cytotoxicity form a major challenge as they impose the risk of induction of secondary malignancies. 8 A number of studies suggested that plants-derived therapeutic agents have high potential of inhibiting and/or reversing the advancement of cancer.7,8
One potential Saudi medicinal plant species is Juniperus procera (J procera). It is an evergreen tree from the Cupressaceae family.9,10 In Saudi Arabia, it is known as Arar with a variety of medicinal uses in local traditional medicine. 11 Numerous compounds found in J procera are effective in treating a variety of diseases, including hyperglycemia, ulcers, intestinal worms, wounds, liver diseases. Moreover, it was shown to be effective in the management of various skin conditions like eczema and skin rashes, as well as a variety of respiratory tract conditions eg, asthma, common cold, cough, bronchitis, throat inflammation, tuberculosis, and pneumonia.12,13 Several studies reported the cytotoxic efficiency of J procera and other Juniperus species against a variety of cancer cell lines. These anticancer activities were reported against human oral squamous cell carcinoma cell lines (SCC9), 11 colon carcinoma cell line (Caco2),14,15 melanoma tumorigenesis 16 and HeLa cervical cancer cell growth. 16
Understanding the mechanisms of cancer development and progression is critical for evaluating the effectiveness of the different anticancer compounds. Phosphoinositide 3-kinase-protein kinase/Protein kinase B/mammalian target of rapamycin (phosphoinositide 3-kinase-protein kinase [PI3K]/AKT/mTOR) signaling pathway for apoptosis comprises an important target for treatment of breast cancer. 17 PI3K signaling pathway diverges at many points and has a variety of functional consequences that play a dominant role in certain tumor-related outcomes. 18 Aromatase (estrogen synthase) belongs to cytochrome P450 enzymes and catalyzes the conversion of androgen C (19) to estrogen C (18). Breast tissues demonstrate aromatase activity in vitro, with upregulation on aromatase expression at or near the breast tumor site. Therefore, local regulation of aromatase using either endogenous or extrinsic agents affects estrogen levels for breast cancer growth. Inhibition of aromatase enzyme is linked to the prevention of breast cancer growth.19–21
The aim of the current study was to test the potential role of Saudi J procera fruits and leaf extracts, if any, in inducing apoptotic pathway and inhibiting aromatase enzyme activity using two breast cancer cell lines (MCF7 and MDA-MB-231).
Material and Methods
Plant Collection and Identification
Plants J procera samples including leaves and fruits were collected from Raghadan forest (latitude 20.0214399; longitude 41.44405372; Al Bahah Region, Kingdom of Saudi Arabia) in December 2019. The plant material was identified by Professor Ahmed M. Abd-ElGawad, Department of Plant Production, College of Food & Agric. Sciences, King Saud University P.O. Box 2460 Riyadh, Saudi Arabia. 11451.
Phytochemicals Extraction
Shade-dried fruits and leaves (250 g) were ground into a fine homogenous powder and extracted thrice in 96% ethanol. The homogenates were then filtered through Whatman's no.1 filter papers. The filtrates were evaporated to dryness using a rotary evaporator. The fruit and leaf residues were then resuspended in methanol (2 mg/mL), kept at −20 °C, and used for downstream analyses.
Determination of Total Flavonoids and Total Phenolics
The J procera fruits or leaves were extracted in methanol (2 mg/mL) and the total flavonoids and phenolics content in the methanolic extracts were determined spectrophotometrically at 415 and 630 nm, respectively, as described previously22,23 The concentration of total flavonoids was then determined using rutin standard curve whereas the level of total phenolics was calculated using gallic acid standard curve. All measurements were performed in triplicate and the mean ± SD of the three measurements were expressed as µg/g dry tissue.
ABTS Assay
The ABTS assay was carried out as described by Arnao et al. 24 with slight modifications. Briefly, the ABTS reagent was freshly prepared by mixing 1 mL of ABTS solution (192 mg in 50 mL distilled water) with 17 µl potassium persulphate (140 mM) and the mixture was incubated in the dark for 24 h. One mL of the mixture was then mixed with 49 mL methanol. Subsequently, 10 µl of the samples were distributed into 96-wells plate (n = 6) and 190 µL of the ABTS reagent were mixed with each sample, mixed well, and the plates were incubated in the dark for 120 min at 28 °C. Afterward, the decline in the optical density of ABTS reagent was recorded at 734 nm and the percentage of decolorization was calculated using the following equation.
Determination of the Free Radicals Scavenging Activity of J procera Fruits and Leaves Extracts
The free radicals scavenging activity of fruits and leaf methanolic extracts was carried out using 2,2-diphenyl-1-1picrylhydrazyl (DPPH) assay (Cat. No. D9132-1G, Sigma-Aldrich) according to. 25 Aliquots (200 µL) of the fruit and leaf extracts, as well as, ascorbic acid were mixed with 1000 µl of DPPH, shaken well, and the mixtures were incubated for 30 min in the dark at 28 ˚C. The optical density of the developed color was determined using spectrophotometer (BioTek Instruments, Inc.) at 517 nm. The percentage of the radical scavenging activity of the tested extracts, relative to ascorbic acid, was calculated using the following equation.
where
Cell Lines, Maintenance, Reagents, and Preparation
The normal breast cells MCF-10A, and both human breast cancer MCF7 and MDA-MB-231 cells were obtained from Nawah Scientific Company. These cells were maintained in a complete DMEM medium with high glucose (BioWhittaker™) supplemented with 10% Fetal Bovine Serum, + 1% 100 IU/mL penicillin/streptomycin (100 µg/mL), Lonza, 17-602E). Adricin (Sigma-Aldrich) was dissolved in dimethyl sulfoxide (DMSO, Cat. No. 20385.02, Serva) and kept at −20 °C. The cells were incubated at 37 °C in a 5% CO2 atmosphere.
Initial Screening and Cell Viability by 3-(4,5-Dimethylthiazoyl)-2,5-Diphenyl-Tetrazolium Bromide Assay
Cell viability was monitored as described by El-Senduny et al. 26 Antiproliferative activity of fruit or leaf extracts against MCF-7 cells and MDA-MB-231 cells were tested. The 3-(4,5-dimethylthiazoyl)-2,5-diphenyl-tetrazolium bromide (MTT) formazan dye was dissolved in DMSO and detected using a plate reader (Biotek, Gen5™) at λ570-630 nm. Briefly, 100 µL cells were distributed into a 96-well plate followed by incubation of the plates for 12 h at 37 °C and 5% CO2. Then 200 µl of each extract (100 µg/mL) or DMSO (0.5% V/V) was added to each well, followed by incubation for additional 48 h under the same conditions. Subsequently, 10 µl of MTT (5 mg/mL prepared in Phosphate Buffered Saline) was added to each well (final MTT conc is 0.5 mg/mL per well), and the plates were incubated as above for 4 h. Formazan crystals was solubilized in 200 µL of 10% acidified SDS solution (containing 0.01N HCl in 1x PBS). After 4 h of incubation, the absorbance was determined at λ570-630 nm using a plate reader (Biotek, Gen5™). To determine the IC50, the cancer cells were treated with a series of dilutions of fruit or leaf extracts (range from 0.10% to 1.00%.) for 48 h, then the cell viability was tested using MTT as described above. All measurements were performed in triplicate and the mean ± SD of the three measurements were expressed as μg/mL.
Investigating Fruit and Leaf Extracts Selectivity Towards Cancer Cells
To test the selectivity of the J procera fruit and leaf extracts against the two cancer cell lines, we used the normal breast cells MCF-10A (MCF-10A, 5 × 104 cells/mL). Cells were disseminated in a 96-well plate followed by incubation for 12 h at 37 °C and 5% CO2. Subsequently, dilutions of the fruit and leaf extracts (100, 50, 25, 12.5, 6.25, or 3.125 µg/mL) were mixed with the cells and the plates were incubated as above for 48 h. The cells viability was monitored using MTT assay. The selectivity index (SI) was calculated using the equation below.27,28
Wound Healing Assay
The cell lines MCF-7 and MDA-MB-231 were used in the wound-healing assay. The cells (5 × 105 cells/mL) were seeded in a 6-well plate and incubated at 37 °C and 5% CO2 for 48 h. A sterile P200 tip was then used to induce wounding in the cells and the medium was changed by a fresh medium containing either fruit or leaf extract or DMSO. The plates were incubated under the same conditions as above. At 0.0 and 48 h after incubation, cells were collected, washed twice using ice-cold 1x PBS, and fixed using ice-cold methanol at 28 °C for 30 min. Afterward cells were stained using 0.5% crystal violet for 30 min, and the extra stain was removed by washing with tap water. The wound areas were photographed at 0 h and 48 h using Optika B-159 (OPTIKA S.r.l.). The wound size was calculated using Image J 1.51 software.
Cell Cycle Analysis
This was carried out by seeding MDA-MB-231 and MCF-7 cells (5 × 105 cells/mL) in a 6-well plate (2 mL/well) followed by incubation overnight at 37 °C and 5% CO2 for adherence. Subsequently, fruit or leaf extracts or DMSO were applied to the cells and the plates were incubated for an additional 48 h. Subsequently, cells were washed twice using cold 1x PBS and collected after trypsinization. The cells were collected by centrifugation for 5 min at 500 x g at 4 °C, suspended in cold 1x PBS, and fixed with absolute ethanol. The fixed cells were kept at −20 °C for 2 h, washed using cold 1x PBS, resuspended in propidium iodide (PI)/RNase (BD Biosciences, BDB550825), and incubated at 28 °C for 30 min in the dark. Using flow cytometry (Accuri C6 Plus flow cytometer, NJ, USA), DNA at each phase was measured within 1 h.
Apoptosis Analysis
To test the extract-induced apoptosis of MDA-MB-231 and MCF-7 cells (5 × 105 cells/mL), if any, Annexin-V and Propidium Iodide DXN kit (eBioscience, 51-66121E, Thermo Fisher Scientific™, USA) was used according to the manufacturer's instruction. Briefly, cells were treated either with fruit or leaf extract or DMSO for 48 h. After trypsinization, the cells were collected and washed twice with ice-cold 1× PBS. Cells were resuspended in 1× binding buffer, 5 µL annexin-V, and 5 µL PI, then incubated at room temperature for 15 min in the dark. The fluorescence was then detected using the Accuri C6 Plus flow cytometer.
Comet Assay
Comet assay was carried out as described by Dhawan et al. 29 with slight modifications. Cells were treated and collected as indicated above and stained with trypan blue (15250061, Gibco™). Afterwards, the cells were mixed with 0.5% low-melting-point agarose/PBS, pH 7.4 at 37 °C and pipetted onto a frosted glass slide precoated with a layer of 1% normal melting point agarose (NMA)/H2O. The slides were kept for 8 min at 4 °C then immersed in lysis solution (2.5 M NaCl, 100 mM EDTA, and 10 mM Tris-HCl) for 2 h in the dark at 4 °C. The slides were maintained in alkaline buffer (10 N NaOH and 200 mM EDTA, PH >13) for 20 min prior to electrophoresis (24 V and 300 mA) for 30 min for DNA unwinding. Slides were then washed 3 times (5 min each) using a neutralization buffer (0.4 M Tris-HCl, pH 7.5), dehydrated by absolute ethanol for 20 min, stained with ethidium bromide (20 μg/mL), and dipped in dH2O for removal of excess stain. The images were obtained using an Olympus BX43 connected to Olympus Digital Camera.
Detection of Apoptosis by Fluorescence Microscope
MDA-MB-231 and MCF-7 cancerous cells were disseminated on a coverslip in 6-well plate and kept at 28 °C for 12 h. Cells were treated with fruit extract for 48 h then fixed with 4% paraformaldehyde for at 28 ˚C for 15 min. The nuclear damage was then tested via staining the cells using PI for 45 min at 28 ˚C in the dark. PBS was used to wash cells twice and the cells were mounted on a glass slide. Cells were photographed using Olympus BX43 connected to Olympus PEN Lite E-PL3 camera at 10× magnification.
RT-PCR Analysis
This was carried out to determine the expression of the Aromatase enzyme-encoding gene. MDA-MB-231 and MCF-7 cells were treated with either fruit or leaf extract as well as with DMSO for 12 h. TRIzol™ Reagent (Invitrogen, Cat. No. 15596018) was used for RNA extraction from the treated cells. Amounts (4 µg) RNA were reverse transcribed to cDNA using RevertAid First Strand cDNA Synthesis kit according to manufacturer's instructions. The expression of Aromatase and GAPDH was monitored using SensiFAST™ SYBR® High-ROX Kit and gene-specific primers in StepOne™ Real-Time PCR System (Applied Biosystems™). The expression level was firstly normalized to the level of GAPDH then calculated using the 2−(ΔΔCt) method where ΔCt = (Ct-CtGAPDH); ΔCtDMSO = (CtDMSO-CtGAPDH); ΔΔCt = (ΔCt- ΔCtDMSO). Ct is the cycle threshold. The sequences of the primers are shown in Table 1.
Primers Used in the Current Study.

Western Blot Analysis
MDA-MB-231 and MCF-7 cells (2.5 × 105 cells/mL) were distributed and processed as described above. Cells were then treated with the fruit or leaf extracts and incubated for 48 h. Subsequently, cells were lysed using RIPA buffer containing 1× protease and phosphatase inhibitor cocktail (Thermo Scientific™). The lysates were centrifuged for 5 min at 4 °C and the supernatants-containing proteins were collected. The proteins were then fractionated on SDS-PAGE (100 V for 45 min) and blotted to a 0.45 µm nitrocellulose membrane followed by incubation with a primary antibody against p27Kip1, PI3K, Akt, pAKT S473, mTOR, matrix metallopeptidase 9 (MMP9) or FASN overnight at 4 °C on a rocker (Supplemental Information: Table S1). Then an appropriate HRP-conjugated secondary antibody was used. GAPDH was used as a loading control. WesternBright® ECL (K-12045-D20) was used to develop the membrane whereas ChemiDoc MP Imaging System (Bio-Rad) was used for the detection of the developed signals.
Statistical Analysis
All results are presented as the mean ± standard deviation (SD). Statistically significant differences among the responses of the tested cell lines were calculated using Student's t test or one-way ANOVA. A P-value <.05 was considered as the threshold for statistical significance. All calculations were performed using GraphPad Prism software (7.0).
Results
Phytochemical Analysis and Total Free Radical Scavenging Activity
Our analysis revealed significant differences in phenolic content between the J procera fruits and leaves; (82.59 ± 7.5 µg/g fruits vs 65.14 ± 3.9 µg/g leaves; P < .05). In contrast, the leaves had five folds higher flavonoid content than fruits (15.22 ± 4.9 µg/g leaves vs2.83 ± 1.1 µg/g fruits; P < .05). The DPPH assay indicated a significantly higher reactive oxygen species (ROS) quenching activity of J procera fruits (19.0 ± 0.2 μg/mL) than leaves extract (41.8 ± 1 μg/mL P < .05). Consistent results were also obtained by ABTS assay with a significant higher ROS scavenging power in the J procera fruits (27.19 ± 1.21 μg/mL) than leaves (65.05 ± 2.51 μg/mL; P < .05). The data is presented in Table 2.
Total Phenolic and Flavonoids in the Fruit and Leaf Extracts. The Concentration that is Required to Quench 50% of the ABTS or DPPH radicals (IC50).

*Significant difference between fruit and leaf extracts.
Anticancer Activity of J procera Extracts
The anticancer activities of the J procera fruits and leaves extract were tested against two types of breast cancer cells: ER-positive breast cancer cells (MCF-7) and triple-negative breast cancer cells (MDA-MB-231) and normal epithelial breast cells (MCF-10A), (Table 2 and Figure 1). Interestingly, both fruit and leaf extracts showed anticancer activities against the tested two breast cancer cells with significant differences between the two extracts; MCF-7 (15 ± 1.5 µg/mL vs 25 ± 0.9 µg/mL; P = .009), and MDA-MB-231 (22 ± 0.3 µg/mL vs 42 ± 2.8 µg/mL; P = .001), respectively. Also, the fruit extract showed significantly lower IC50 than the leaves extract on both MCF-7 cell line (15 ± 1.5 µg/mL vs69 ± 1.1 µg/mL; P = .001) and MDA-MB-231 cells (22 ± 0.3 µg/mL vs 69 ± 1.1 µg/mL; P = .007) when compared with the normal epithelial breast cells (MCF-10A). Further, there was a significant difference between IC50 of leaf extract on MCF-7 cell line (25 ± 0.9 µg/mL vs 72 ± 1.7 µg/mL; P = .003) and MDA-MB-231 cells (22 ± 0.3 µg/mL vs 72 ± 1.7 µg/mL; P = .021) when compared with the normal breast cells (MCF-10A).
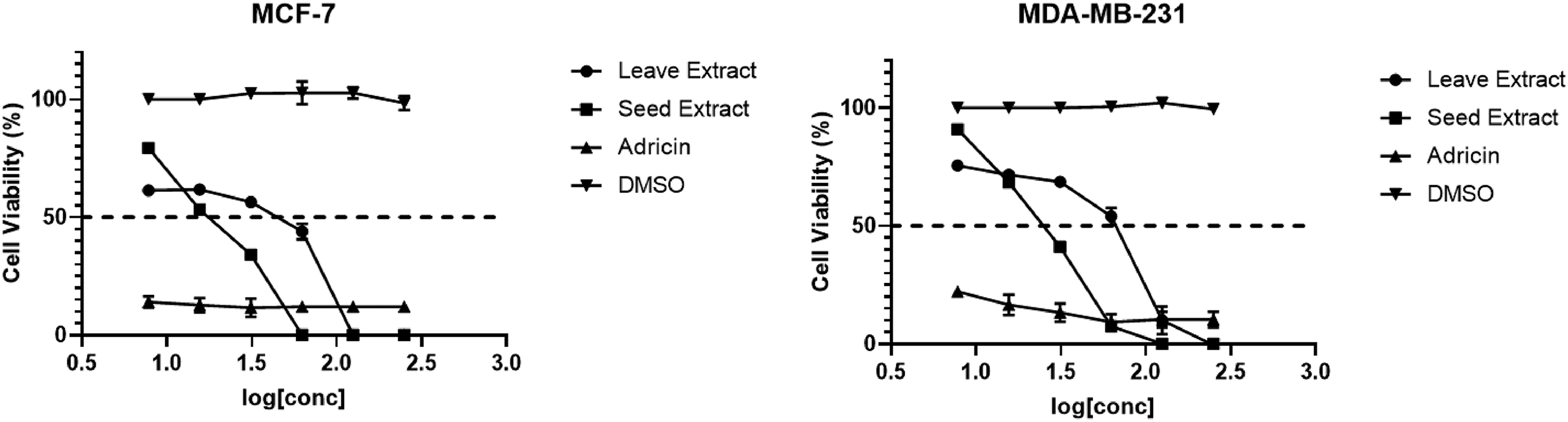
The cytotoxic activity of Juniperus procera fruit and leaf extracts against MCF-7 and MDA-MB-231.
It is worth mentioning that, both the fruit and leaf extracts showed higher selectivity towards the tested cancerous cells than normal ones (SI > 2). The results of the initial screening analysis using MTT assay indicated higher selectivity of the fruit extract than leaf extract. Because of the higher anticancer activity and selectivity of the J procera fruit extract over those of leaf extract against MCF-7 and MDA-MB-231, only the fruit extract was used during the downstream investigation.
Cell Cycle and Apoptosis
To evaluate the impact of the J procera fruit extract on the cell cycle progression, flow cytometry analysis for the PI-stained single cells was used. For the MCF-7 cancer cell line, the fruit extract successfully arrested ER-positive breast cancer cells (MCF-7) at S-phase with an arrest of 19.4% compared to 14.1% in the DMSO-treated cells. In the triple-negative breast cancer cells (MDA-MB-231), the fruit extract arrested the cell cycle at G1 phase with an arrest of 70% compared to 52.3% in the control cells (Figure 2) suggesting a differential response of the two cancer cell lines to the fruit extract. The application of the fruit extract induced 13.7% late apoptosis in the MCF-7 cells compared to 0.7% in the control cells. The corresponding values of the extract-induced apoptosis in the MDA-MB-231 cells approached 15.1% compared to 0.3% in the control cells (Figure 2).

Flow cytometric analysis showing the effects of Juniperus procera fruit extract treatments on cell cycle activity and apoptosis in breast cancer cells (MDA- MB-231 and MCF-7).
Apoptosis Induction was via DNA Damage
To test the ability of the J procera fruit extract in inducing DNA damage and apoptosis in the MCF-7 and MDA-MB-231 breast cancer cell lines, Comet assay was adopted. Relative to the DMSO-treated cells, the application of the fruit extract induced DNA damage in both cell types and the affected cells proceed to the apoptosis pathway (Figure 2). The fruit extract-induced DNA damage was further confirmed by immunofluorescence technique which revealed clear nuclear fragmentation and apoptosis in the fruit extract-treated MCF-7 and MDA-MB-231 cells relative to DMSO-treated cells, (Figure 3).

Comet and immunofluorescence assays: top panel: comet assay indicating the comet and the tail formed after application of the Juniperus procera fruit extract to MCF-7 cells and MDA-MB-231cells. Lower panel: Immunofluorescence staining against nuclear DNA showing nuclear fragmentation in treated cell lines compared to dimethyl sulfoxide (DMSO). (A) Untreated MCF-7 cells. (B) Untreated MDA-MB-231cells. (C) Fruit extract-treated MCF-7 cells. (D) Fruit extract-treated MDA-MB-231 cells. The pictures were taken by Olympus BX43 connected with Olympus PEN Lite E-PL3 camera at 10× magnification.
Effects of J procera Fruit Extract on Wound Healing in Cancer Cells
Figure 4 revealed that J procera fruit extract induced significant inhibitions of migration and healing process in both breast cancer cells (MDA-MB-231 and MCF-7) compared to the control cells.
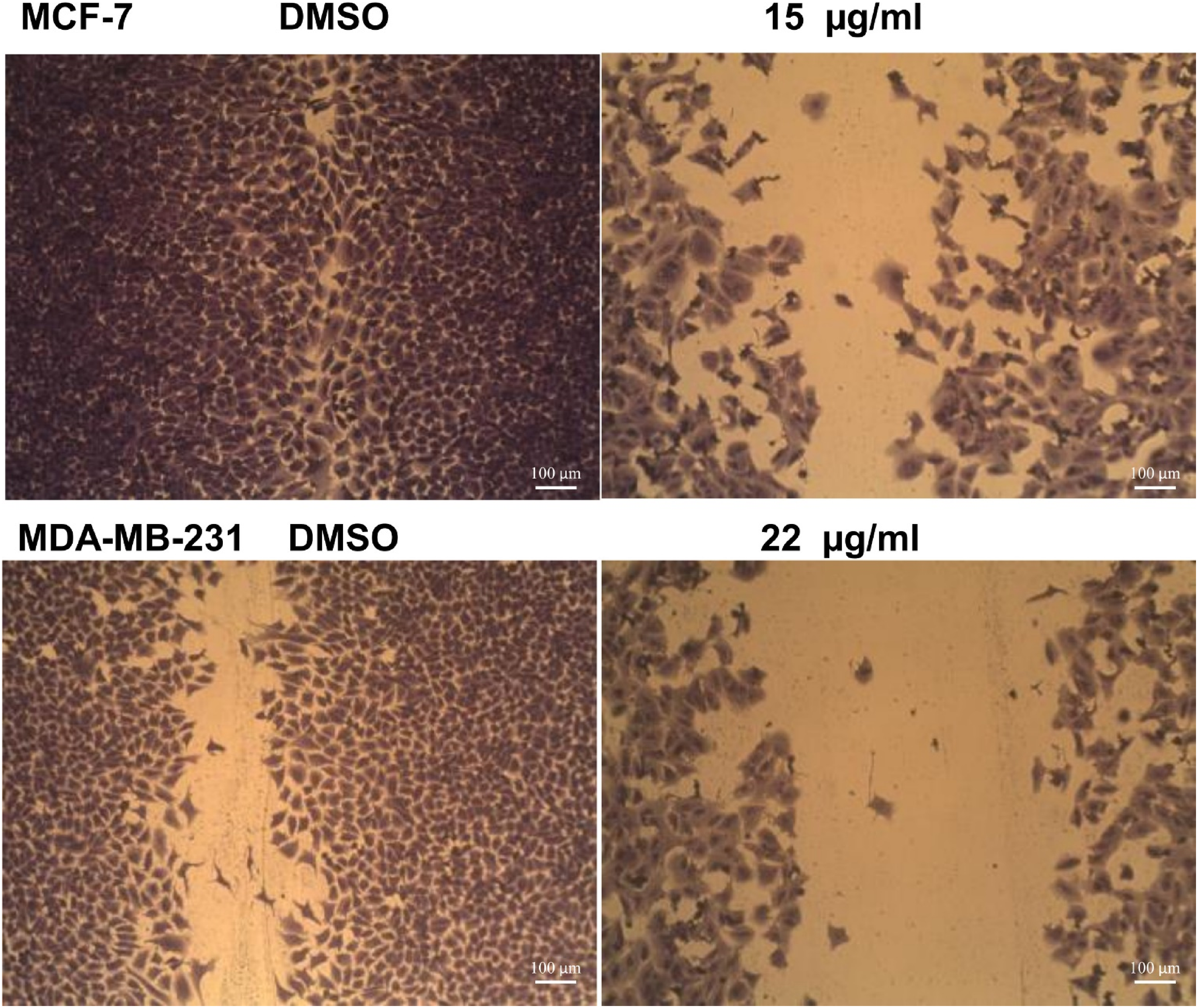
Effects of Juniperus procera fruit extract on wound healing in breast cancer cells (MDA- MB-231 and MCF-7).
Molecular Mechanisms of the Anticancer Action of J procera Fruit Extract
To gain insights into the potential mechanism of the J procera fruit extract-induced cell death, we investigated the level of critical cancer cell survival and metastasis-related signaling proteins such as PI3K/Akt/mTOR pathway. Interestingly, fruit extract significantly downregulated the expression of crucial proteins involved in cell cycle and apoptosis such as PI3K and total and the phosphorylated Akt. These responses were also associated with downregulation of the mTOR (Figure 5). Our results also revealed significant downregulation of the level of MMP9, and fatty acid synthase (FASN) proteins in response to the application of the Juniperus fruit extract. Further, the application of the Juniperus fruit extract downregulated the level of p27 in MCF-7, however, the extract induced the expression of p27 in the MDA-MB-231(Figure 6).

Effects of Juniperus procera fruit extract on cell cycle and apoptosis’ related proteins in breast MDA-MB-231 and MCF-7.
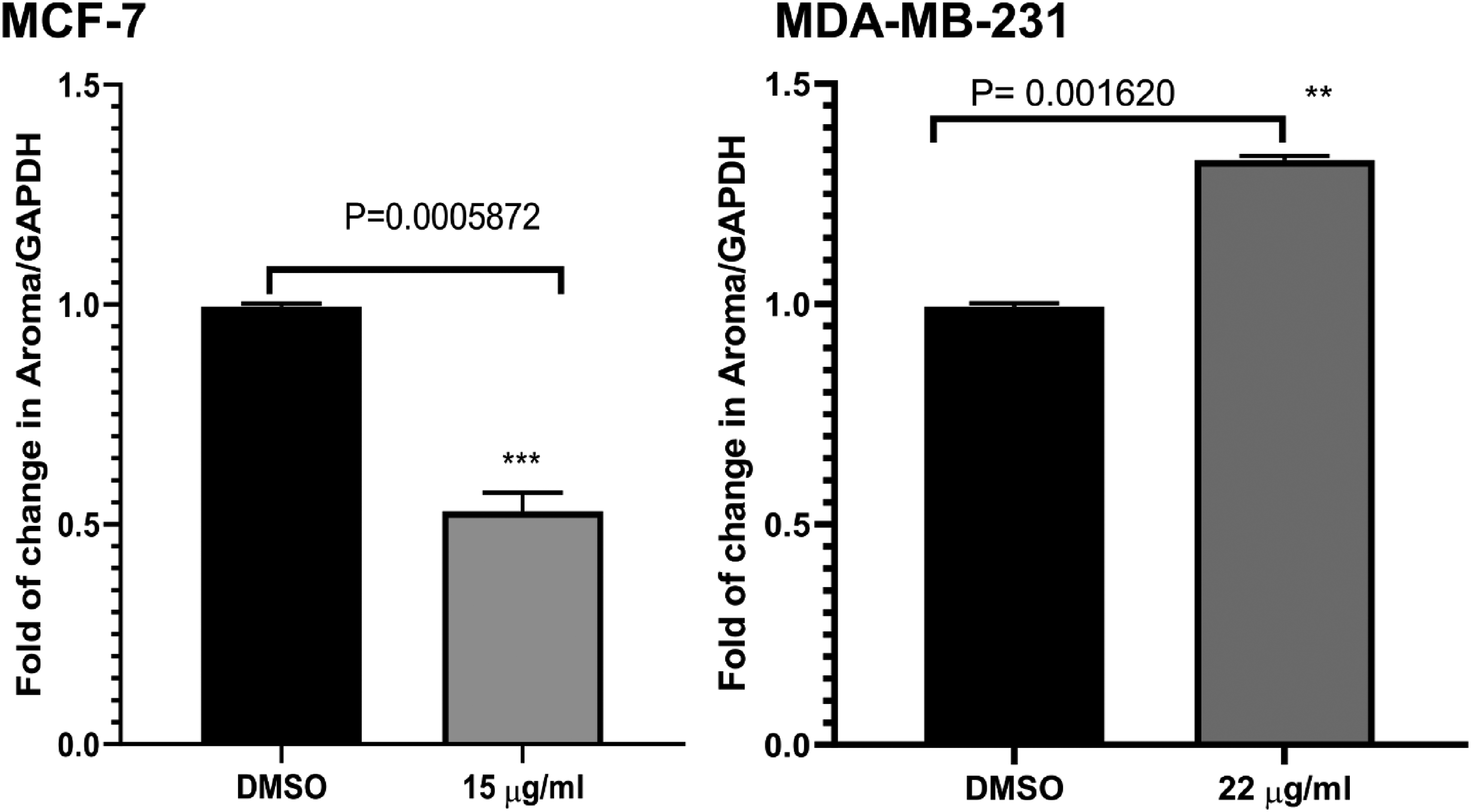
Effects of J Juniperus procera fruit extract on the aromatase expression MDA-MB-231 and MCF-7.
We also tested the impact of the fruit extract on the aromatase-encoding gene. Our results revealed differential effects of Juniperus fruit extract on gene expression of the aromatase gene in two breast cancer cells MDA-MB-231 and MCF-7. The fruit extract significantly downregulated the aromatase-encoding gene in MCF-7 cells (P = .0006). However, the expression of this gene was substantially upregulated in the MDA-MB-231 cells (P = .0016), (Figure 6).
Discussion
For decades, trials to find effective treatments for cancers are going on, however, all the scientific efforts to find a cure for cancer reached a consensus that there will be no success for monotherapy as a comprehensive cancer treatment with a bed need for more effective integrative or combination therapies.7,17 Due to their effectiveness in preventing or reversing the development of cancer and having a lower risk of side effects than those associated with synthetic anticancer compounds, plant-derived anticancer agents have been applied in chemotherapy as possible sources of anticancer compounds. 8 In this study, the anticancer activities of the extracts of the Saudi J procera fruit and leaves and their possible molecular mechanisms in controlling growth of human cancerous cells were evaluated.
J procera fruit and leaf extracts had high phenolics and flavonoids content. However, seed extracts had higher phenolics than leaves whereas the reverse was true for flavonoids (Table 2). In addition, the fruit extract was more effective in quenching free radicals than the leaf extract (Table 2). Phenolic compounds are known for their potent antioxidant activities. Therefore, the higher phenolic content in the J procera could explain the superior free radical quenching activity by fruit compared to leaf extract. This view has been supported by the reported direct correlation between phenolic content and elevated antioxidant activity of plant extracts.1,8,23 Several studies have reported the potential role of flavonoids in delaying or reversing carcinogenic progression in a wide range of cancer types.30–32 In fact, numerous epidemiological studies have linked the lower risk of several types of cancer to the consumption of flavonoid-rich fruits, vegetables, and whole grains.32,33
Our cytotoxicity analysis showed that J procera fruit extracts exert more cytotoxic effects than leave extracts against MCF-7 and MDA-MB-231 and both extracts showed interesting selectivity for cancer cells rather than the healthy cells (Table 3). These results are consistent with those of, 34 who reported the absence of the cytotoxic effects of J procera extracts against normal cells. 34 A successful anticancer treatment should also incapacitate cancer cells while causing minimum damage to normal cells, implying minimal side effects. 35 Both fruit and leave extracts of J procera exhibited substantial anticancer properties, as they inhibited the proliferation of the tested cancer cell lines, thus it could be a source for pharmacologically active products for chemotherapeutic treatment. Along with their anticancer activities, J procera fruits and leaves extracts contain a variety of antioxidants including phenolics and flavonoids and thus have the ability to scavenge ROS and blocking lipid peroxidation which have been implicated in the induction of human diseases. 13
The Selectivity of the Anticancer Activity of the Fruit and Leaf Extracts of Juniperus procera Towards Cancer Cells. Shown are the Mean Values of IC50 (µg/mL) ± Standard Deviation of Three Independent Experiments.

Significant difference between IC50 of fruit extract on MCF-7 cell line (P = .001) and MDA-MB-231 cells (P = .007) when compared with the normal breast cells (MCF-10A).
Significant difference between IC50 of leaf extract on MCF-7 cell line (P = .003) and MDA-MB-231 cells (P = .021) when compared with the normal breast cells (MCF-10A).
Significant difference between IC50 of Adricin on MCF-7 cell line (P = .043) when compared with the normal breast cells (MCF-10A).
*Significant difference between IC50 of fruit and leaf extracts on MCF-7 cell line (P = .009).
**Significant difference between IC50 of fruit and leaf extracts on MDA-MB-231 cell line (P = .001).
Flow cytometry analyses were performed to evaluate the fruit extract's effects on the progression of the cell cycle. Our results revealed that J procera fruit extract arrested breast cell line MCF-7 at the S phase, while the triple-negative cell line (MDA-MB-231) was arrested at the G1 phase, (Figure 2), suggesting differential behavior of the fruit extracts on the two cell lines. Defects in the apoptotic mechanism and cell cycle checkpoints cause oncogenic resistance. 36 This form of resistance contributes to the development of cancerous phenotypes. In reality, cancer is characterized by the avoidance of apoptosis and the loss of cell cycle control. 37 The cell cycle analysis revealed that fruit extract induced a progressive accumulation in the G1/M phase and a reduction in the number of cells in the S phase in the MDA-MB-231cell line. In the MCF-7 cell line, the fruit extract showed a significant reduction in cell entered in G0/G1 phase. These results indicate that fruit extract exerts its effects on the MDA-MB-231 cells by arresting cell cycle and apoptosis pathway. However, in MCF-7 cells it acts as apoptotic inducer.
In the current study, damaged DNA induces cell cycle and apoptosis was detected by Comet assay (Figure 2). There was a significant DNA damage induced after treatment of the tested breast cancer cell lines with either leave or fruit extract in comparison to the DMSO-treated cell lines (P < .05). The extracts-induced DNA damage was further confirmed by immunofluorescence which revealed significant induction of nuclear fragmentation and apoptosis in both MCF-7 and MDA-MB-231 cell lines in response to J procera fruit extract (Figure 3).
To gain insights into the possible mechanisms underlying the anticancer activity of J procera fruit extract, we examined changes in PI3K/AKT/mTOR signaling proteins. Interestingly, our results revealed that the fruit extract downregulated the expression of PI3K, reducing total levels and phosphorylating AKT, thus downregulating the levels of mTOR (Figure 5). Therefore, these results highlight the potential of J procera fruit extract to contribute significantly to breast cancer treatment by enhancing the extrinsic apoptotic-signaling pathway. This conclusion is supported by a number of evidence (1) The constitutive activation of the PI3K/AKT/mTOR pathway induces tumor cell proliferation and survival in a variety of human cancers, including breast cancer, (2) the PI3K/AKT/mTOR signaling pathway contributes significantly to cancer cell survival and metastasis and thus blocking this route can induce apoptosis and/or cell-cycle arrest,38,39 and (3) AKT inhibitors contribute to progression-free survival in triple-negative breast cancer patients along with activation of the PI3K/AKT pathway. Altogether, the inhibitory effects of J procera fruit extract on PI3K/AKT/mTOR signaling pathway can contribute significantly to confronting the urgent need for inhibitors that can target the critical signaling pathways for TNBC treatments. 17
PI3K signaling pathway diverges at many points and has a variety of functional consequences. The range of functional responses may represent the effect of spectrum, level, and extent of activity of components of the PI3K pathway. Yet, it has been suggested that various nodes in the signaling pathway play a dominant role in certain outcomes. 18 AKT-mediated activation of mammalian target of rapamycin (mTOR) is critical for induction of cell proliferation. 18 mTOR regulates translation in response to nutrients/growth factors via phosphorylating components of protein synthesis mechanisms such as ribosomal protein S6 kinase (p70S6 K) and 4E binding protein (4EBP), leads to the release of the translation initiation factor eIF4E. It's in vitro transforming, and antiapoptotic activity have also been reported. 40
Several molecular signaling pathways including extracellular signal-regulated kinase (ERK), mitogen-activated protein kinase and PI3K regulate MMP9 expression pathways known to be altered in Breast Cancer. 41 Our results revealed that J procera fruit extract downregulated (MMP9) levels in protein lysates, which confirms inhibition of this signaling pathway. In addition, the fruit extract-induced downregulation of mTOR was associated with significant reduction in the levels of FASN which catalyzes the production of fatty acids necessary for cell membrane integrity, metabolism, and protein posttranslational modifications. FASN also has a significant role in tumor growth and survival. 42
The challenges of controlling metastasis and cancer invasion are the most awesome hurdles to therapeutic success, making regulation of metastasis and invasion an important therapeutic goal. The biological process of metastasis requires disruption of the extracellular matrix to encapsulate mesenchymal collagen and endothelial basement membrane. mTOR controls the expression of metastatic proteins like MMP9 as well as the lipogenesis pathway. 42 Our analysis indicated that J procera fruit extract reduced the quantity of MMP9 in the protein lysate, indicating the inhibition of the wound healing processes. The J procera fruit extract also increased the cell cycle inhibitor p27 in MCF-7 cell lysate while decreasing it in MDA-MB-231 lysate. Unlike p53 and other tumor suppressor pathways, the PI3K pathway is triggered in cancer. Therefore, it represents an ideal target for therapy because of the ease of suppression of activation events relative to replacing the loss of tumor suppressor function. 40
Aromatase, an enzyme that drives synthesis of estrogen, is important in the development of ER-positive breast cancer. Many tissues, including the breast, express aromatase, which gives them the ability to synthesize estrogen locally. 43 Aromatase expression is under the control of tissue-specific promoters that are regulated by different transcription factors. 44 The present study demonstrated that J procera fruit extract substantially decreased aromatase activity in MCF-7 but increased it in MDA-MB-231 (Figure 6). These fruit extract-induced inhibition reveals the aromatase inhibitor activity of the fruit extract in estrogen-positive breast cancer cell line MCF-7. However, the fruit extract-elicited activation of aromatase in the triple-negative cell line (MDA- MB-231) may attributed to the expression of high levels of EGFR, which is associated with a high likelihood of loss of susceptibility to hormone therapy and/or metastasis. 41 A strong association between aromatase (Cyp19) gene expression and both cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2) expression in breast cancer samples has been reported. 43 In the breast, the main source of aromatase is breast stromal-adipose-fibroblasts called breast adipose fibroblasts (BAFs). In breast cancer tissue, BAFs provide structural support for tumors, and malignant epithelial cells secrete factors such as prostaglandin E2 and cytokines that act on BAFs, preventing BAF differentiation, so resulting in an increase in both aromatase expression and increases local estrogen synthesis. 45 Moreover, Aromatase is the rate-limiting enzyme in estrogen biosynthesis.44,46 Targeting these partially tissue-selective promoters in breast cancer offers a potential way to specifically inhibit aromatase activity in breast tissue without the usual side effects of aromatase inhibitors in other organs, such as bone. 44
Conclusion
In conclusion, the Saudi J procera extracts exhibited anticancer activities via three essential processes, cell cycle arrest, apoptosis induction, and cell migration prevention. The mechanism of action includes DNA damage, downregulating the expression of PI3K/Akt/mTOR pathway proteins, and reducing aromatase expression levels. The anticancer activities of J procera fruit extract were more evident than those of its leaves.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241232778 - Supplemental material for Evaluation of Saudi Juniperus procera Extracts Cytotoxicity and Regulatory Mechanisms of Tumorigenesis Against Two Breast Cancer Cell Lines
Supplemental material, sj-docx-1-npx-10.1177_1934578X241232778 for Evaluation of Saudi Juniperus procera Extracts Cytotoxicity and Regulatory Mechanisms of Tumorigenesis Against Two Breast Cancer Cell Lines by Ateeq A. Al-Zahrani, Farag Ibraheem, Mohamed El-Hefny and Fardous El-Senduny in Natural Product Communications
Supplemental Material
sj-pptx-2-npx-10.1177_1934578X241232778 - Supplemental material for Evaluation of Saudi Juniperus procera Extracts Cytotoxicity and Regulatory Mechanisms of Tumorigenesis Against Two Breast Cancer Cell Lines
Supplemental material, sj-pptx-2-npx-10.1177_1934578X241232778 for Evaluation of Saudi Juniperus procera Extracts Cytotoxicity and Regulatory Mechanisms of Tumorigenesis Against Two Breast Cancer Cell Lines by Ateeq A. Al-Zahrani, Farag Ibraheem, Mohamed El-Hefny and Fardous El-Senduny in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by the Deanship of Scientific Research at Umm Al-Qura University (Grant Code. 18-SCI-1-01- 0028).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.