Abstract
Introduction
The genus Linaria Mill. is a member of the Plantaginaceae family and comprises approximately 200 species worldwide.1–4 In phytochemical studies on this genus, it has been determined that plants contain compounds such as alkaloids, phenolic compounds [flavonoids, phenylpropanoids, etc], and terpenoids [monoterpenes (iridoids), diterpenes, triterpenes]. 2 It has been reported that Linaria species are used as a wound healer, tonic, antiscorbutic, diuretic, laxative, and in diabetes, hemorrhoids, and vascular diseases.2,5 In biological activity studies on Linaria species have found that they have antitumor, antiacetylcholinesterase, anti-inflammatory and analgesic, antioxidant, antibacterial, cytotoxic, genotoxic, antidiabetic, and diuretic activities. 2 There are 21 Linaria species in Türkiye and 12 of them are endemic species.1,5,6 In this study, the endemic L. corifolia Desf. plant, which has a limited number of phytochemical and biological activity studies, was studied. Based on the biological activity studies conducted on Linaria species, the in vitro cholinesterase inhibitory activities of aqueous-methanol, ethyl acetate, and n-butanol extracts were investigated. Alzheimer's disease is a multifactorial, complex, and worldwide prevalent neurological disease. Inflammation, oxidative stress, cholinergic pathways, genetic, and environmental factors play a role in causing Alzheimer's.7,8 Senile plaques and amyloid-beta peptide deposition are neuropathological hallmarks of Alzheimer's disease. These symptoms are caused by a decrease in the amount of acetylcholine in the brain, so acetylcholinesterase inhibitors are used in treatment to prevent the hydrolysis of acetylcholine. Cholinesterases are the enzyme family responsible for the hydrolysis of acetylcholine into choline and acetic acid. They consist of 2 types of enzymes: Acetylcholinesterases (AChE) and Butyrylcholinesterases (BChE). AChE are found in peripheral and central tissues, while BChE are found mainly in the liver. The treatment of the disease is symptomatic, and the drugs used have side effects such as gastrointestinal, and hepatotoxicity. Thus, developing natural compounds with fewer side effects is important in treating the disease. In our study, the cholinesterase inhibitory effects of the extracts, and isolated compounds were investigated by in vitro Elmann's method.9–13 When oxidative stress increases, it degrades nucleic acids, proteins, carbohydrates, and fatty acids. This disrupts the integrity of cell membranes, causing apoptosis or amyloid formation. 14 Since oxidative stress and cholinesterase activities affect each other in neurodegenerative diseases such as Alzheimer's disease, 14 the antioxidant effects of plant extracts have been investigated with different methods. We used DPPH, ABTS Radical Scavenging Effects, and CUPRAC Assay, which are commonly used in the evaluation of antioxidant activity, since this assay have not been used before in the evaluation of the activities of the prepared extracts. Since the phenolic compounds found in plants are responsible for the antioxidant effect, we also researched total phenolic, and total flavonoid content of the extracts. We thoroughly investigated the in vitro anticholinesterase activities of various L. corifolia extracts. Also, we identified the chemical composition in the ethyl acetate and n-butanol extracts of the plant for the first.
Materials and Methods
General Experimental Procedures
For the separation and purification of the substances in fractions, chromatographic methods using filler materials such as normal-phase silica gel, Sephadex and reverse-phase silica gel were used. Spectrometer Bruker-Avance Neo 500 MHz (Germany) was used to get nuclear magnetic resonance spectra. Waters Corporation Milford, MA (USA) was used to record Electrospray ionization mass spectrums.
Plant Material
L. corifolia plant was collected from Ankara, Türkiye in June 2020 in the flowering period, plant was identified by Assoc. Prof. Dr. Dilek Ercil and Asst. Prof. Zekiye Ceren Arıtuluk Aydın. Voucher specimens have been previously deposited in Herbarium of the Faculty of Pharmacy, Hacettepe University, Ankara, Türkiye (HUEF 18001).
Extraction and Isolation Process
Dry aerial parts of L. corifolia (899.22 g) were macerated at room temperature with 20% aqueous methanol for a night. The next day, it was extracted at 40 °C, for 6 h, and the extraction process was repeated 4 times. After filtration, the hydroalcoholic filtrate was concentrated at 40 °C under low pressure and dissolved in distilled water. After the lyophilization process, we obtained 75,77 g of extract, yielding 8,43%. Aqueous solution was successively with petroleum ether, CHCl3, EtOAc (ethyl acetate), and n-BuOH. We conducted isolation studies on ethyl acetate and n-butanol extracts.
We applied the n-butanol extract to the column. The column was prepared with Silica gel for pre-fractionation. Elution was started with 10% methanol chloroform. Then, it continued with 17,5% and 50% ratios. We obtained fractions from this chromatography system. We put them on Sephadex Column Chromatography (elution system: 100% methanol). It separated the compounds by their molecular weight. Fractions obtained from Sephadex column chromatography were purified by the reverse phase column chromatography method. In last method, methanol:water (10:90, 20:80, 30:70) solvent systems were used for isolation.
We applied the ethyl acetate extract to Sephadex column chromatography using 100% methanol. The resulting fractions were purified using varying ratios of methanol: water (5:95, 10:90, 15:85, 25:75, 30:70, 40:60, 50:50, 60:40, 70:30) solvent systems.
The Supplemental material has detailed info. It covers general experimental procedures, plant material, extraction and isolation processes, and methods for determining biological activity.
Results and Discussion
Compounds Identification
We isolated six compounds from the L. corifolia plant. We identified their structures using NMR, ESI mass spectroscopy methods, and comparisons with literature data. Compounds are antirrhinoside (63,4 mg) [1], 15 6-ß-Hydroxyantirrhide (8,3 mg) [2], 16 catalpol (24,4 mg) [3],17,18 and aucubin (6,6 mg) [4]19,20 from n-butanol extract, linariin (30,9 mg) [5], 21 and acteoside (verbascoside) (4,3 mg) [6]22,23 from ethyl acetate extract (Figure 1).

Isolated compounds 1-6 from L. corifolia.
The iridoid glycoside antirrhinoside is a chemotaxonomic marker in the Linaria genus. Although antirride is widespread in the Linaria, 6-ß-Hydroxyantirrhide was found in Linaria genistifolia, L. japonica, and L. purpurea. Catalpol (in L. macroura) and aucubin (in L. vulgaris, L. macroura) rarely exist in Linaria. Linariin is a flavonoid glycoside common in Linaria species. Acetoside was identified in L. haelava 2 All compounds are newly isolated from L. corifolia.
Biological Activity Studies
In Vitro Antioxidant Activity
DPPH Radical Scavenging Activity
DPPH (2,2-diphenyl-1-picryl-hydrazyl) radical scavenging effects of extracts were investigated.24,25 High activity was observed in ethyl acetate extract (83,28% at 400 μg/ml concentration) (Figure 2). The IC50 (μg/ml) values for ethyl acetate, n-butanol extract, and ascorbic acid were calculated as 217,75 ± 12,82, 306,58 ± 23,38, and 16,19 ± 2,62 respectively.
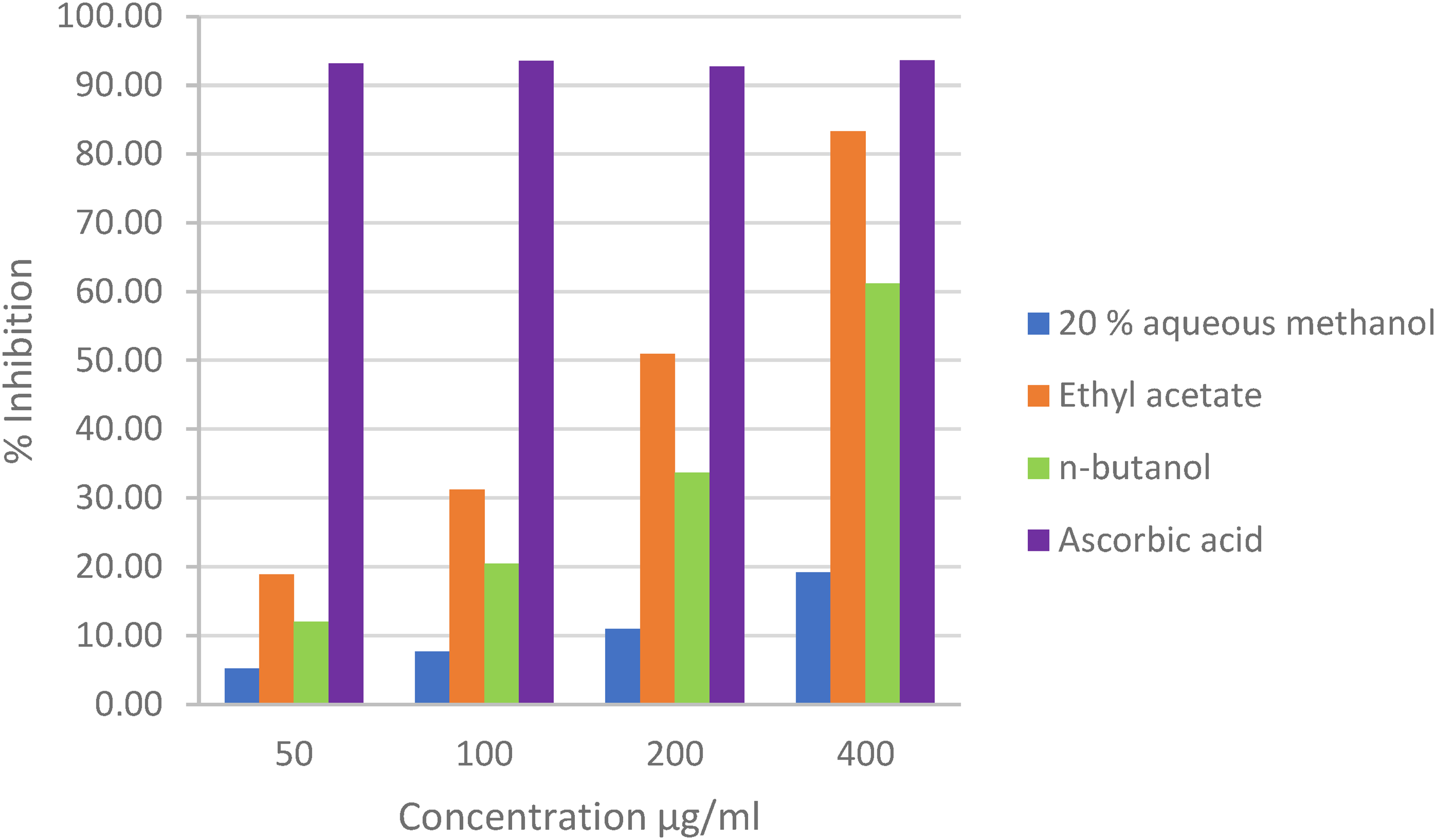
% Inhibition of DPPH radicals.
ABTS Radical Scavenging Activity
ABTS (2,2′-azinobis(3-ethylbenzothiazolin-6-sulphonic acid) radical scavenging activity determinations of extracts were made at concentrations of 100–800 μg/ml.26,27 When the ABTS Radical Scavenging effects of extracts were evaluated, a higher effect was found in ethyl acetate extract than in other extracts (Table 1). Results are given as trolox equivalent antioxidant capacity (TEAC).
TEAC (mg TE /g) Extract Amount of Extracts (TE: Trolox Equivalent) in ABTS Radical Scavenging Activity.

Cupric Ion Reducing Antioxidant Capacity (CUPRAC) Assay
Cupric (II) ıon reduction antioxidant capacity of extracts was determined at concentrations of 50–400 μg/ml.27,28 Similar to the ABTS Radical Scavenging Effect results, the ethyl acetate extract was the most effective (Table 2). Table 3 shows a comparison of all antioxidant activity test results.
TEAC (mg TE /g) Extract Amount of Extracts in CUPRAC Assay.

Antioxidant Activities of Extracts at Different Assay.

Determination of Total Phenolic Content
Total phenolic content of extracts was performed at a concentration of 400 μg/ml.29,30 The highest total phenolic content in ethyl acetate extract was observed (Table 4).
Total Phenolic Content of Extracts.

Determination of Total Flavonoid Content
Total flavonoid content of extracts was investigated at a concentration of 200 μg/ml. 31 The n-butanol extract had more flavonoids than other extracts. The total flavonoid contents of the extracts are given in Table 5.
Total Flavonoid Content of Extracts.

In another study, Fe +3/ ferricyanide reduction, DPPH radical, and superoxide scavenging activity were measured for ethanol, ethyl acetate, and dichloromethane extracts of L. corifolia aerial parts. The ethanol extract showed higher effects than the other extracts. When the results of total phenolic content and total flavonoid determination were examined, it was found that the ethanol extract had higher phenolic compound and flavonoid content than other extracts. 32 In our study, we found that the ethyl acetate extract (at 400 μg/ml) had 83,28% DPPH radical inhibition. In ABTS Radical Scavenging Activity, and CUPRAC Assay TEAC values respectively;114,66 mg, 202,50 mg TE/g extract. It was more active than other extracts in all antioxidant tests. This was linked to its high phenolic compound content. According to the DPPH Radical Scavenging Activity results, it is seen that ethyl acetate extract (83,28% inhibition) at a concentration of 400 μg/ml has a similar effect to ascorbic acid (93,61% inhibition). After ethyl acetate extract, n-butanol extract had 61,19% DPPH radical inhibition at 400 μg/ml. In ABTS Radical Scavenging Activity, and CUPRAC Assay its TEAC values respectively; 69,68 mg, 99,71 mg TE/g extract. It was moderately effective. The 20% aqueous methanol extract had 19,16% DPPH radical inhibition at 400 μg/ml. In ABTS Radical Scavenging Activity, and CUPRAC Assay, its TEAC values respectively; 45,57 mg, 42,21 mg TE/g extract. It was low effective. It has been determined that ethyl acetate and n-butanol extracts have more power to reduce cupric (II) ions. But, they have less power to scavenge ABTS radicals.
In Vitro Anticholinesterase Activity
Extracts and isolated compounds of L. corifolia were investigated for their inhibition effects of acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) enzymes (Figures 3, 4, 5, and 6).9,12,33,34 Physostigmine salicylate was used as a standard compound. It had IC50 1,17 ± 0,18 for AChE and 9,13 ± 0,06 μg/ml for BChE. % Inhibition AChE and BChE values of the standard compound are given in Table 6.

% Inhibition AChE of extracts.
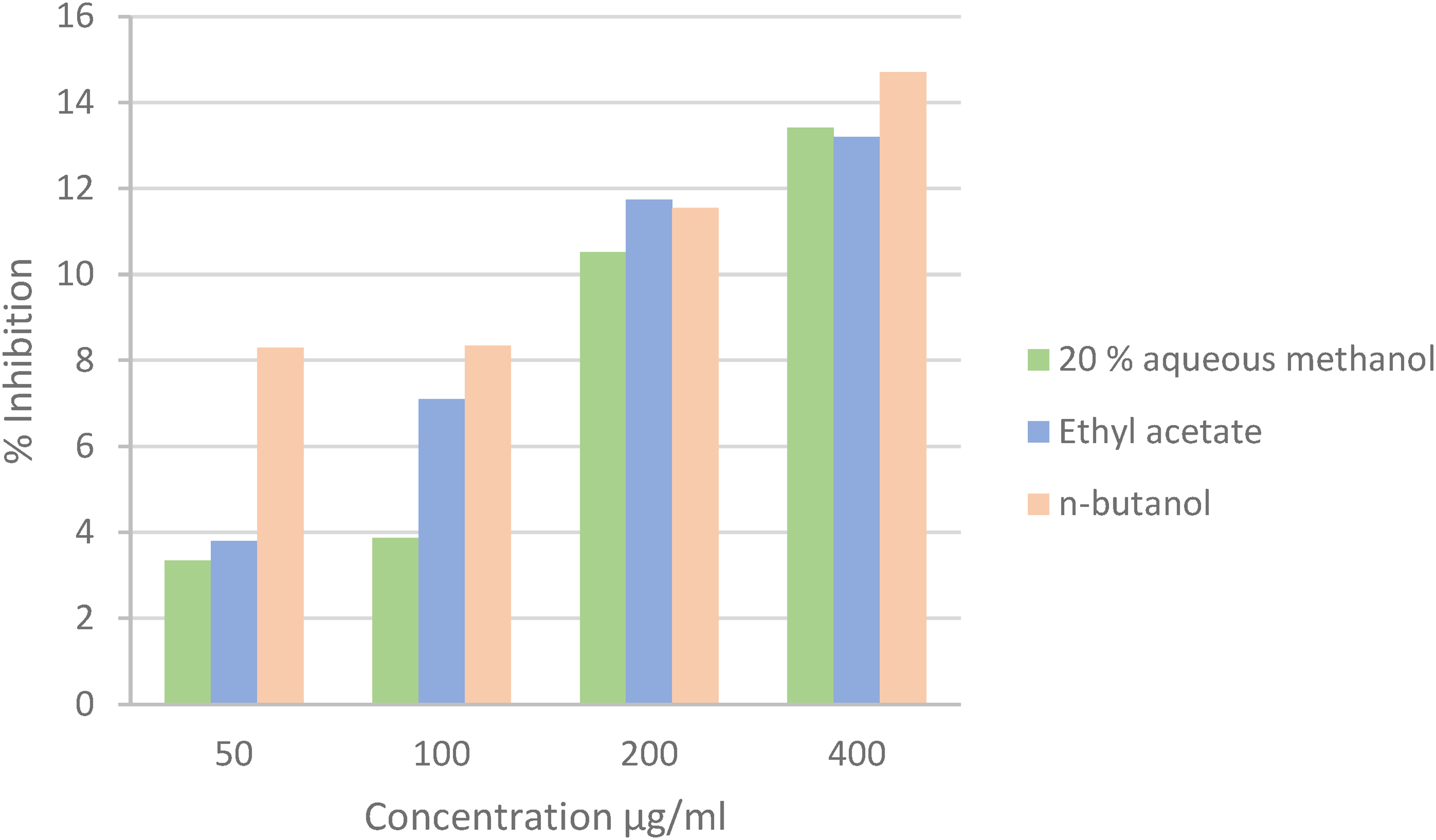
% Inhibition BChE of extracts.

% Inhibition AChE of isolated compounds.

% Inhibition BChE of isolated compounds (The inhibition value of 6-ß-hydroxyantrirrhide compound at 100 μg/ml could not be calculated.).
% Inhibition AChE and BChE of Physostigmine Salicylate (*:It has not Been Tested at Relevant Concentrations.).

In cholinesterase inhibitor effect studies conducted on Linaria species, L. reflexa ethyl acetate extract (IC50: 185,6 ± 1,2 µg/mL), a high effect was determined in the isolated linariin, isolinariin A and B compounds, and a lower effect was observed in the linariin (IC50: 0,30 ± 0,05 μM). 35 In another study, L. scariosa butanol extract was found to have high acetylcholinesterase inhibitory effect (IC50: 75,47 ± 3,5 µg/mL). It was thought that butanol extract may have high activity because it contains the compound pectolinarin. It was concluded that the extracts may be active due to their flavonoid content.35,36
Various studies have shown that catalpol does not have an AChE inhibitory effect, and in animal models, it has been shown that it can increase memory and learning by decreasing the M receptor (brain muscarinic acetylcholine receptor), increasing BDNF receptor density, and reducing cholinergic degeneration. In the study examining the effect of catalpol on female mice with neurodegenerative damage, catalpol was administered at concentrations of 5, 15, and 45 mg/kg for 3 months. It has been observed that catalpol strongly increases BDNF expression, memory, and learning ability. 37 It is thought that the catalpol compound, which has antioxidant, anti-inflammatory, and anti-apoptotic effects in in vitro and in vivo studies, may be effective in Alzheimer's disease with its neuroprotective effect. 38 In the study catalpol, aucubin, acteoside, and other compounds from Verbascum mucranatum Lam. AChE and BChE inhibitory effects were tested, The acteoside compound had a medium AChE inhibitory effect. But, the catalpol, and aucubin were not effective. 39
When our experimental results were evaluated; an increase was viewed in the acetylcholinesterase inhibitor activity of n-butanol extract concentration-dependent. Iridoid compounds isolated from n-butanol extract, such as the compound catalpol, which has been found effective in various studies, may be the compounds responsible for the activity of n-butanol extract. However, ethyl acetate extract was determined more active than other extracts at 200 and 400 μg/ml. It has been reported that flavonoids and iridoids may be effective for neurodegenerative diseases such as Alzheimer's due to their neuroprotective and healing effects.2,36,40 In our study, it is thought that the acetylcholinesterase inhibitory effect of ethyl acetate extract (at 200, and 400 µg/mL) may be due to the phenolic compounds it contains and other flavonoids found together with the isolated linariin, acteoside compound.
When the % Inhibition values of pure compounds were compared, linariin was found to be more effective. Among the iridoid compounds, the catalpol compound was more effective than other iridoids at 25 and 100 μg/ml (33,87 ± 1,94, 26,49 ± 8,62%, respectively). A higher butyrylcholinesterase inhibitory effect was observed in dichloromethane extract of L. scariosa (IC50: 96,02 ± 0,69 µg/mL). Fatty acids such as α-linolenic and palmitic acid have been held responsible for the high butyrylcholinesterase inhibitory effect of the extract. 36 Butyrylcholinesterase inhibitory activities of the extracts and isolated compounds are generally low compared to the acetylcholinesterase inhibitory activity. Since the extracts contain mostly polar compounds, it can be said that butyrylcholinesterase activities are low. The cholinesterase inhibitory effects of the extracts and pure compounds were found to be lower compared to physostigmine salicylate.
Detailed information is also available in the supplemental material.
Limitations of Study
Although the ethyl acetate extract contained a large number of substances in our isolation studies, structure determinations could not be carried out because the amounts of the substances obtained were very small. Again, the n-butanol extract contained flavonoids. But, they could not be obtained in pure form despite the using different chromatographic methods. The amounts of aucubin and acteoside compounds were too low. So, we could not evaluate their cholinesterase inhibitory effects. The in vivo effects of antirrhinoside and 6-ß-Hydroxyantirrhide compounds, whose cholinesterase inhibitory effects have not been studied before, can be investigated. The in vivo effects of these compounds can be evaluated together with the in vitro effect results.
Conclusions
In our study, we report for the first time different antioxidant, anticholinesterase activities of L. corifolia extracts. We also report isolation of pure metabolites of ethyl acetat, and n-butanol extracts. Six compounds were isolated from the L. corifolia plant and their structures were identified through NMR, ESI mass spectroscopy methods, and comparisons with literature data. Antirrhinoside, 6-ß-Hydroxyantirrhide, catalpol, aucubin, linariin, and acteoside (verbascoside) compounds are new compounds for the L. corifolia. It has been determined that L. corifolia contains iridoid glycosides, flavonoids, and phenylpropanoids, similar to other Linaria species. As a result of antioxidant activity studies, ethyl acetate extract was found to be more effective extract than other extract due to its high phenolic content. Compared to physostigmine salicylate, all extracts, and pure compounds have lower acetylcholinesterase and butyrylcholinesterase inhibitory activity. However, higher antiacetylcholinesterase activity was observed at concentrations of 200 and 400 μg/ml, depending on the active components of the ethyl acetate extract. Among pure compounds, linariin appears to be more active than other compounds.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241272734 - Supplemental material for Phytochemical Content, In Vitro Antioxidant, and Cholinesterase Inhibitory Activities Determination of Endemic Linaria corifolia Desf
Supplemental material, sj-docx-1-npx-10.1177_1934578X241272734 for Phytochemical Content, In Vitro Antioxidant, and Cholinesterase Inhibitory Activities Determination of Endemic Linaria corifolia Desf by Melike Utlu and Dilek Ercil in Natural Product Communications
Footnotes
Acknowledgments
We thank Prof. Dr. Hakan Göker (Ankara University, Pharmacy Faculty, Department of Pharmaceutical Chemistry) for performing our NMR analyses.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Hacettepe University Scientific Research Projects Coordination Unit (Project No: THD-2021-19442).
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
