Abstract
Background
Methods
Standard protocols were followed in the processing of the plant leaves, extraction, fractionation, isolation, and characterization, evaluation of
Results
The dichloromethane (61.59%) and ethyl acetate (73.15%) fractions had the best therapeutic indices and compared favorably with chloroquine (81.58%) in the curative antiplasmodial assay. The GC-MS analysis revealed 20 already reported antiplasmodial compounds with hexanoic acid 1,1-dimethylethyl ester, octadecanoic acid docosyl ester, and trans-β-ocimene as the lead compounds based on their binding affinities, permeation of the blood-brain-barrier, non-inhibition of metabolizing enzymes, ease of excretion, non-carcinogenicity, as well as non-violation of Lipinski's criteria.
Conclusion
Octadecanoic acid docosyl ester and hexanoic acid 1,1-dimethylethyl ester bonded the tetrahydrofolate-binding sites of PfSHMT, caused inhibition of DNA synthesis, and apoptosis, whereas trans-β-ocimene inhibited PfEMP-1, reversed the attachment of parasitized red blood cells to micro-vascular endothelium as their mechanism of action for parasitemia clearance. Moreover, these lead compounds reported for the first time in the dichloromethane and EtoAc fractions of this plant are responsible for the remarkable antiplasmodial activity observed in this study.
Introduction
Malaria is a potentially fatal disease caused by
Moreover,
Also,
It is worthy of note that the most effective antimalarial medications have been derived from natural sources throughout the history of malaria chemotherapy;
7
conversely, Asanga et al
14
corroborated that the tropical forests in Nigeria are vested with many medicinal plants; so, robust researches and implementation of research findings, the isolation and characterization of bioactive compounds in these plants, as well as the integration of orthodox and traditional medicine could drastically boost health care delivery in Nigeria. One of these medicinal plants with reported ethnopharmacological relevance as well as its natural products with reported pharmacological activities is
In addition, despite the management of malaria with traditional medicine and chemotherapies, the validation of the mode of action of the lead compounds is a serious concern. So, the discovery of therapeutics has been a serious concern on how ligands are docked with their receptors
8
; therefore, the initial phase of drug discovery and development involves lead identification. During this process, chemical compounds are challenged to interact with their protein targets to alter their functions. In order to be effective against some target proteins like PfSHMT and PfEMP-1; lead compounds should ideally exhibit some degree of precision and potency. Hence, this study was geared towards the validation of the antiplasmodial potentials of
Results and Discussion
Results
The Antiplasmodial Activities of the Extract and Fractions of J. insularis Leaves
The leaf extract and its fractions exerted dose-dependent reductions in parasitaemia of the treated mice in the suppressive antiplasmodial assay. From Table 1, the parasitemia reduction was only statistically significant (
Suppressive Antiplasmodial Activities of the Extract and Fractions of J. insularis Leaf (n = 5).
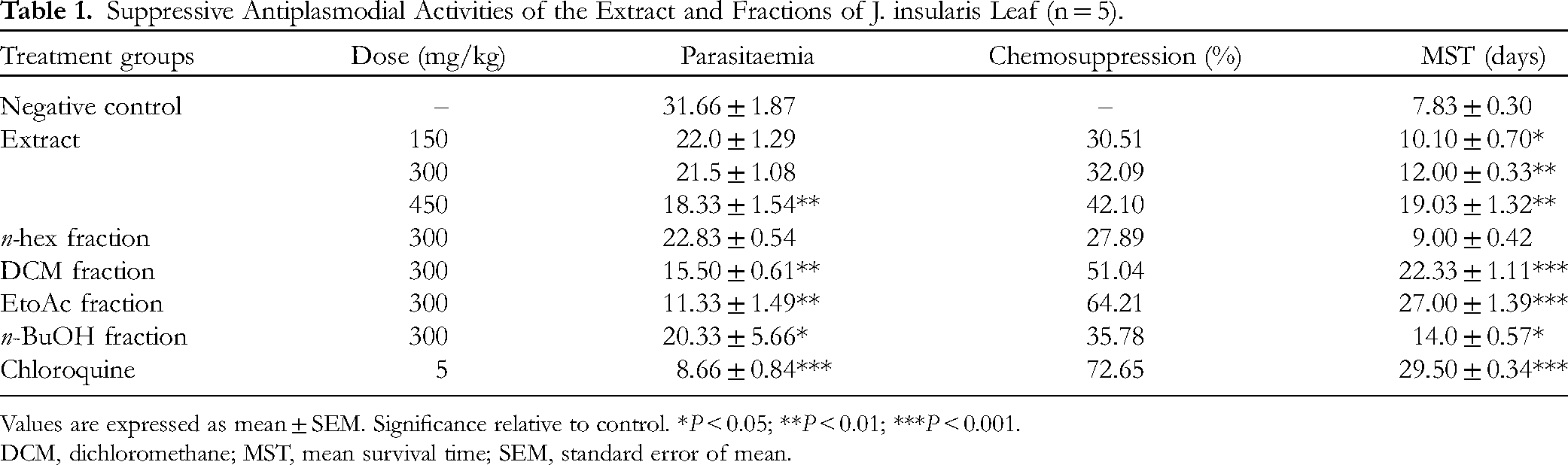
Values are expressed as mean ± SEM. Significance relative to control. *
DCM, dichloromethane; MST, mean survival time; SEM, standard error of mean.
Prophylactic Antiplasmodial Activities of the Extract and Fractions of J. insularis Leaf (n = 5).

Values are expressed as mean ± SEM. Significant relative to control. *
DCM, dichloromethane; MST, mean survival time; SEM, standard error of mean.
Schizonticidal Antiplasmodial Activities of the Extract and Fractions of J. insularis Leaf (n = 5).
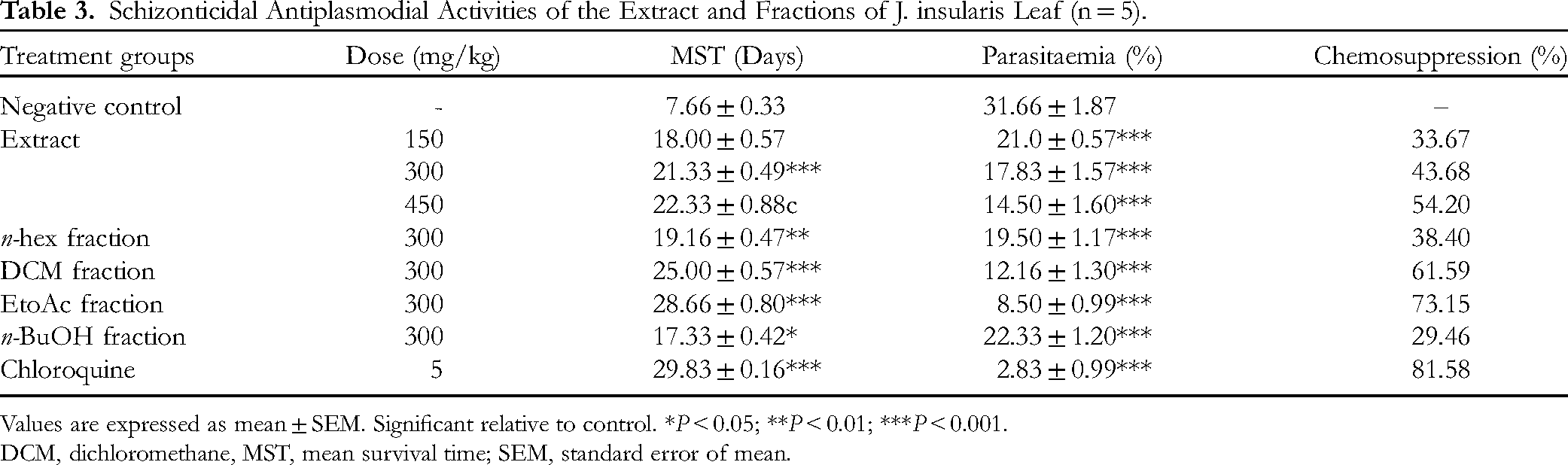
Values are expressed as mean ± SEM. Significant relative to control. *
DCM, dichloromethane, MST, mean survival time; SEM, standard error of mean.
The Effect of the Extract/Fractions of J. insularis Leaves on the Rectal Temperatures of Infected Mice
The administration of the extract and fractions of
Effect of the Extract and Fractions of J. insularis Leaves (n = 5) on Rectal Temperature of Mice Infected with P. Berghei During Established Infection.

Values are expressed as mean ± SEM.
DCM, dichloromethane; SEM, standard error of mean.
GC-MS Analysis
GC-MS analysis (Table 5) of DCM fraction revealed some pharmacologically active compounds such as glyceraldehyde; hexanoic acid; 1,1-dimethylethyl ester; hexanoic acid, butyl ester; hexanoic acid, 2,4-dimethyl-, methyl ester; E-2-tetradecen-1-ol, oxirane, tetradecyl-; trans-β-ocimene; α-pinene among others. Also, the GC-MS analysis (Table 6) of EtoAc fraction indicated that it contains unsaturated fatty acids such as hexanoic acid; pentanoic acid, 3-methyl-; hexanoic acid, 1,1-dimethylethyl ester; hexadec-9-enoic acid; 7-tert-butyldimethylsilyloxy-, methyl ester; heneicosanoic acid, methyl ester; octa-2,4,6-triene; 1,3,6-heptatriene, 5-methyl-, (E)-; phytol, acetate; octadecanoic acid, 2-hydroxy-1,3-propanediyl ester; octadecanoic acid, docosyl ester; and others.
GCMS Analysis of DCM Fraction of J. insularis Leaf.

DCM, dichloromethane.
GCMS Analysis of EtoAc Fraction of

Molecular Docking Analysis
The result (Table 7, Figures 1 and 2) of the docking analysis revealed the interactions of 1BJ4 with selected ligands (A1, A2, A3, A4, A5, and A6) and standard drug (CQ). Ligand A3 binding energy (−6.1 kcal/mol) was lower than that of CQ (−4.8 kcal/mol); however, that of ligands A1 (−4.7 kcal/mol) and A5 (−4.3 kcal/mol) were higher but compared favorably with the standard drug. Further analysis on the binding pocket of the protein revealed that ligand A3 formed an alkyl, pi-alkyl, and 3 hydrogen bonds with the amino acid residues at the active site of the protein. Also, the functional groups in ligand A1 were able to form 2 hydrogen bonds via Thr163 and Phe125 as well as pi-sigma bond with Ala162 at the active site of IBJ4. Consequently, ligand A5 generated both alkyl and pi-alkyl interactions with Ala162, Leu149, and Phe125 as the amino acid residues at the active site of IBJ4 at various bond distances whereas CQ interaction with IBJ4 revealed that Thr163 formed a conventional hydrogen bond while both Leu149 and Ala162 formed alkyl and pi-alkyl interactions with the functional groups in chloroquine, respectively.

2D and 3D visualization of protein-ligand interaction of 1BJ4 with CQ, A1, A2, and A3.
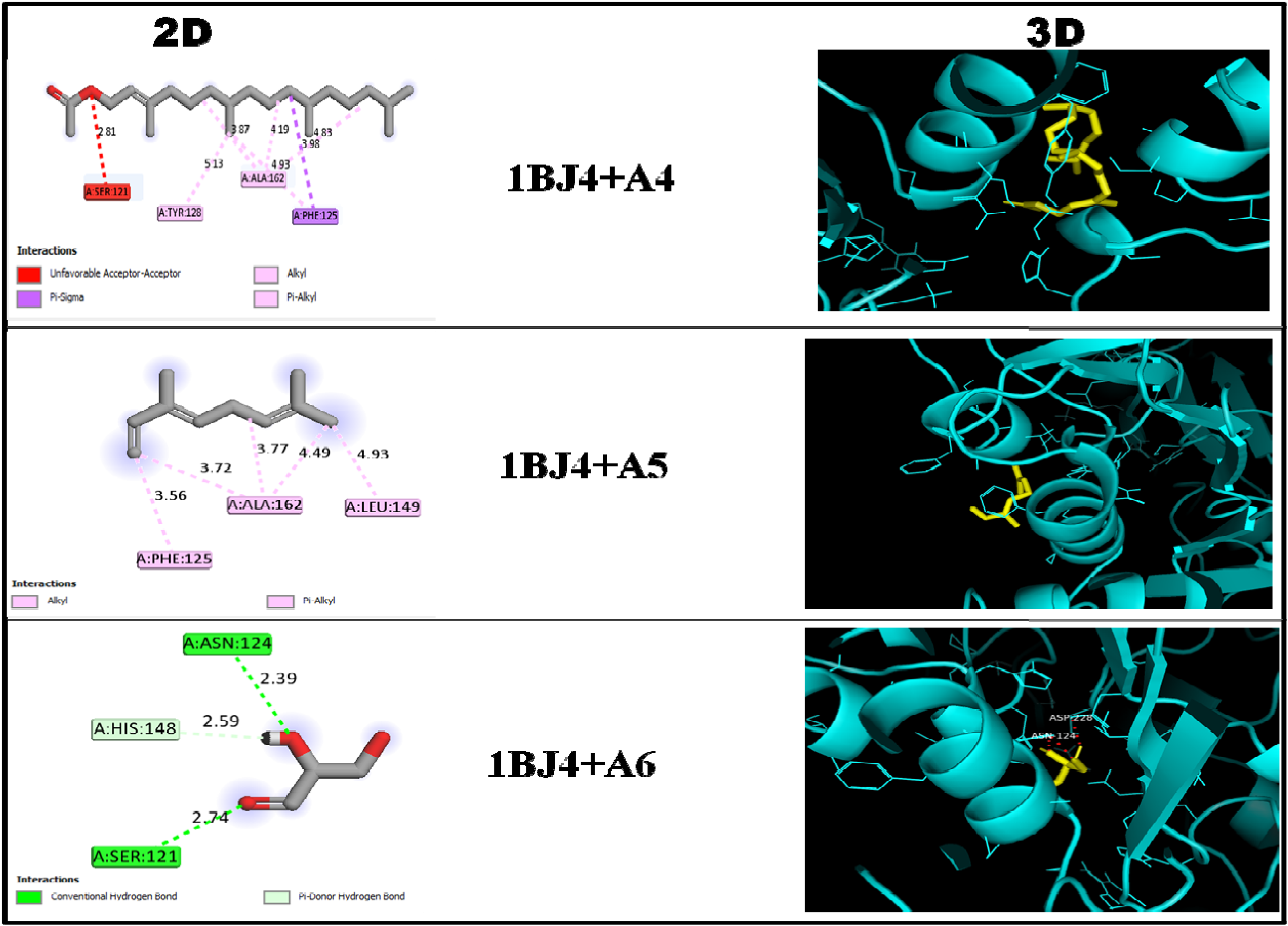
2D and 3D visualization of protein-ligand interaction of 1BJ4 with A4, A5, and A6.
Molecular Docking Analysis of Selected Compounds from the Fraction of

Keys: CQ = Chloroquine; A1= Hexanoic acid, 1,1-dimethylethyl ester; A2= Octa-2,4,6-triene; A3= Octadecanoic acid, docosyl ester; A4= Phytol, acetate; A5= Trans-β-Ocimene; A6= Glyceraldehyde
According to the results (Table 7, Figures 3 and 4) of the docking analysis, the interactions of 3C64 with chloroquine (CQ) and selected ligands (A1, A2, A3, A4, A5, and A6) occurred. The binding energy of CQ (−5.4 kcal/mol) was lower than those of the selected ligands; however, the binding energies of ligands A5 (−4.8 kcal/mol), A4 (−4.6 kcal/mol), A3 (−4.5 kcal/mol), and A2 (−4.5 kcal/mol) compared favorably with that of CQ. Further analysis of these ligands’ interactions at the active site of 3C64 revealed that CQ formed some carbon-hydrogen bonds via Asp21 and Lys24; Tyr55 and Ile20 revealed alkyl and pi-alkyl interactions with the functional groups in CQ, whereas Trp25 formed a pi-pi stacked interaction with the aromatic ring in CQ. Also, ligand A5 through Trp25 formed a pi-sigma bond with the protein whereas the presence of Lys24 at the protein's active site enhanced the formation of alkyl and pi-alkyl interactions with the functional groups in CQ. The interaction of 3C64 with ligands A2, A3, and A4 via their various amino acid residues at the protein's active site revealed majorly alkyl and pi-alkyl interactions with these ligands.

2D and 3D visualization of protein-ligand interaction of 3C64 with CQ, A1, A2, and A3.

2D and 3D visualization of protein-ligand interaction of 3C64 with A4, A5, and A6.
ADMET Studies on Selected Ligands and Chloroquine
The absorption, distribution, metabolism, excretion, and toxicity (ADMET) results (Table 8) revealed that the Caco-2 permeability values for ligands A1, A2, A4, A5, and A6 were −4.162, −4.726, −4.568, −4.557 and −5.123, respectively. More so, the gastrointestinal tract (GI) absorption indices for ligands A1 and A3 were high whereas ligands A1 and A2 showed maximal plasma protein binding (PPB) in percentages as 87.559% and 10.061%. Also, ligands A1, A3, and A5 revealed they could cross the blood-brain-barrier (BBB); however, of all the ligands, it was only A3 and A4 that showed they could inhibit CYP2C9. Moreover, ligands A1, A2, A4, and A5 revealed excellent clearance levels as 11.519, 9.583, 5.081, and 7.761, respectively. Although ligands A1 and A2 revealed they could induce minimal carcinogenicity, ligands A4, A5, and A6 are non-carcinogens. In addition, all the ligands with exception of ligands A3 obeyed Lipinski rule of five with at most one violation.
ADMET Properties of Selected Bioactive Compound.

GI, gastrointestinal tract; BBB, blood-brain-barrier; PPB, plasma protein binding; ADMET, absorption, distribution, metabolism, excretion, and toxicity.
Discussion
Malaria pathogenesis normally reveals hyperparasitaemia, hyperthermia, and hyperpyrexia
20
; however, these symptoms if not averted with chemotherapies and medicinal plants could trigger deleterious complications with possibilities of mortality; so,
Also, fever is one of the cardinal symptoms of malaria; however,
Moreover, molecular docking as a computational tool is designed to accentuate the best binding orientation that compounds interact with their target proteins; it reveals the biological efficacy as well as the binding affinities of compounds, thereby, enhancing the prediction of their potential therapeutic effect.
31
The lowest binding affinities observed for octadecanoic acid docosyl ester and hexanoic acid, 1,1-dimethylethyl ester interaction with PfSHMT as well as trans-β-ocimene, phytol acetate, and octadecanoic acid docosyl ester interaction with PfEMP-1 that were better than chloroquine proved that these compounds’ antiplasmodial activities were through the inhibition of PfSHMT that led to the mitigation of DNA synthesis in the parasites as well as the inhibition of PfEMP-1, a receptor that mediates the attachment of pRBCs to micro-vascular endothelium, thereby, resulting in the parasitemia clearance. It is worthy of note that some researchers
8
posited that lead compounds had lower docking scores; therefore, the lower score function during docking simulations is an advantage for the prediction of both strength and type of signals produced during the structure-based design of drugs. Conversely, the lower binding affinity in these compounds could be attributed to the presence of carbonyl group (C = O) that participated in the hydrogen bonding, Vander Waal forces, and electrostatic interactions, thus, facilitating the compounds interaction with these proteins. Consequently, the interaction of trans-β-ocimene (a monoterpenoid) with PfEMP-1 revealed a pi-sigma bond; however, Li et al
32
reported that pi-sigma interaction contributed to specificity and stability of receptor-ligand interaction as well as influenced their binding affinities during docking simulations. Therefore, hexanoic acid 1,1-dimethylethyl ester and octadecanoic acid docosyl ester with the least binding energies had the best interaction and antiplasmodial activities against PfSHMT, implying that they competed with THF and
The evaluation of the ADMET properties of the ligands as part of
Conclusion
The EtoAc and DCM fractions of
Materials and Methods
Collection and Identification of Plant Material
The leaves of
Extraction
The leaves were washed with distilled water and shade dried at temperature (26 OC) for 2 weeks. The dried leaves (1728 g) were cut into smaller pieces, pulverized to powder, and divided into two parts. One part (497 g) was macerated in 4 L of ethanol (80%) for 72 h, while the remaining part (1231 g) was successively macerated for 72 h in 7.3 L of n-hexane (n-hex), 6.8 L of DCM, 4.1 L of ethyl acetate (EtoAc), and 3.2 L of butanol (BuOH), respectively. The filtrate of each extract and fraction was concentrated and evaporated to dryness
Microorganisms (Parasites)
Chloroquine-sensitive strain of
Parasite Inoculation
Each mouse used in the experiment was inoculated intraperitoneally with 0.2 mL of infected blood containing about 1 × 107 
Experimental Animals
Swiss Albino Wistar mice (18-25 g) of both sexes used in the study were obtained from the University of Uyo's animal house. They were kept in standard plastic cages in a well-ventilated room and acclimatized for 10 days before the experiments. The mice were fed on standard pelleted diet and water
Drug Administration
The extract, fractions, chloroquine, and pyrimethamine used in the study were obtained from a commercial pharmacy in Uyo, Nigeria, and administered orally with the aid of a stainless metallic feeding cannula.
Determination of Median Lethal Dose (LD50)
The determination of median lethal dose (LD50) of the extract was carried out in mice using oral (p.o) route by modified method of Lorke. 39 The mice in groups of three each were administered different doses of the extract (100-5000 mg/kg). They were observed for the manifestation of physical signs of toxicity such as writhing, decreased motor activity, decreased body/ limb tone, decreased mobility, and death. The mortality in each group within 24 h was recorded. The LD50 value was calculated as geometrical means of the maximum dose producing 0% (a) and the minimum dose producing 100% mortality (b); therefore, LD50 =√ab and was estimated to be = 5000 mg/kg body weight of mice.
Antiplasmodial Assay
The Evaluation of Suppressive Antiplasmodial Activities of the Drug, Extract, and Fractions of J. insularis Leaves (4-day test)
This test was used to evaluate the chemosuppressive activity of the extract, fractions, and chloroquine against early 
The Evaluation of Prophylactic Antiplasmodial Activities of the Drug, Extract, and Fractions of J. insularis Leaves
This was evaluated using the method.42,43 The mice were randomly divided into nine groups of five mice per group. Groups (1-3) were given extract (150, 300, and 450 mg/kg) body weight of mice, respectively; groups (4-7) were given
The Evaluation of the Curative Antiplasmodial Activities of the Drug, Extract, and Fractions of J. insularis Leaves
This test was used to evaluate the schizonticidal activity of the extract, fractions, and chloroquine in established
Gas Chromatography-mass Spectrometry Analysis of the Fractions
GC-MS data of the fractions (DCM and ethyl acetate) (10 mg each) being the best based on their antiplasmodial therapeutic indices were recorded on an Agilent 7890 A gas chromatograph connected with an Agilent MS model 5975C MSD detector (Agilent Technologies, USA). A HP5-MS column 5% phenyl-methylpolysiloxane, 30 m × 0.25 mm × 0.25 µm was used with a helium gas flow under a pressure of 10 psi. The injector temperature was set at 280 °C. The oven temperature started from 150 °C for 3 min (min), increased to 300 °C at 10 °C/min, and held for 5 min at 300 °C. The mass spectrometer was operated using the electron ionization mode at 70 eV. The fractions were either directly injected in
The Assessment of the Physicochemical and Pharmacokinetics Properties of the Bioactive Compounds from J. insularis Leaves
Prediction of Target and ADMET Properties of the Ligands
This analysis was carried out using two online tools: swissADME (http://www.swissadme.ch/) and ADMET lab 2.0 (https://admetmesh.scbdd.com/) 8 ; the ADMET properties of the ligands employed in this study were predicted. The different ADMET properties of the ligands and the chloroquine were predicted by using their different canonical strings or Simplified Molecular-Input Line-Entry System strings that were retrieved from the PubChem web platform (https://pubchem.ncbi.nlm.nih.gov/pccompound) in their 3D conformation. All the relevant parameters including Lipinski's rule of five and Ghose parameters were recorded. Using the SWISSADME Target Prediction tool, the targets of the different ligands were determined. 44
Target Proteins Properties and Molecular Docking of the Bioactive Compounds
In this study, the three-dimensional structures of the target proteins: recombinant serine hydroxylmethyltransferase and MC179 portion of the cysteine-rich inter-domain region of PfEmp-1 with PDB ID, 1BJ4 and 3C64, respectively, were obtained from protein data bank (PDB). The ligands and chloroquine were gotten from Pubchem (https://pubchem.ncbi.nlm.nih.gov), whereas the target proteins were retrieved and downloaded in PDB format from the Research Collaboratory for Structural Bioinformatics (RCSB) (www.RCSBPDB.org) and docked blindly. 8 The downloaded proteins were prepared using Biovia Discovery Studio version 21.1.020298 8 ; the protein preparation steps involved the removal of water molecules and native ligand groups present, as well as the addition of polar hydrogen atoms. After the proteins preparation, molecular docking analysis was carried out using PyRx. 45 The results of this analysis were evaluated based on the compound with highest negative binding energy. Also, the 2D visualization of the docking experiment was carried out by using the Biovia discovery studio as it provided an understanding of the specific amino acid residues and their types of interaction between the ligands and the target proteins. Moreover, the PyMol software 46 was employed in providing the 3D visualization of the ligand-receptor interactions, and these visualizations provided better understanding of the docking processes.
Statistical Analysis
Data collected were analyzed using one-way analysis of variance followed by Turkey's multiple comparison post-test (Graphpad prism software Inc. La Jolla, CA, USA). Values were expressed as mean ± standard error of mean (SEM) and significance relative to control were considered at
Footnotes
Acknowledgements
The authors are grateful to Mr Nsikan Malachy and other staff Animal House of Pharmacology and Toxicology Department, University of Uyo for providing technical assistance as well as the editor and reviewers for the review processes.
Authors’ Contributions
Conceptualization and methodology: Enyiekere VJ, Okokon JE, and Asanga EE. Software and statistical analysis: Ekeleme CM and Asanga EE. Resources: Asanga EE, Enyiekere VJ, and Okokon JE. Writing, review, and editing: Asanga EE, Enyiekere VJ, Okokon JE, Anagboso MO, and Ise UP. Supervision: Okokon JE.
All the authors read the manuscript and the authors’ list and consented to the publication.
Availability of Data and Materials
Datasets generated in this study are available from the corresponding author on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The NIH guidelines for the care, handling, and use of experimental animals (NIH Publication, 1996) and compliance with ARRIVE guidelines were judiciously followed. Moreover, the Animal Ethics Committee of the University of Uyo, Akwa Ibom State, Nigeria (approval number: UUY/0420) approved the experimental protocol.
Informed Consent
All the authors contributed their quota to the research, they have thoroughly read, and have approved the manuscript to be published.
