Abstract
Background
Chemotherapy is considered the best approach in the management of malaria pathogenesis symptomized with hyperparasitemia and oxidative stress because of free radicals’ production; however, a crucial step in the production of DNA is the reversible phosphorylation of dTMP to dTDP, which is catalyzed by Plasmodium falciparum thymidylate kinase. Ethnopharmacologically and scientifically, Mangifera indica is used in the treatment of malaria, thereby necessitating antiplasmodial and antioxidant screening of the plant extract, as well as its in silico profiling to unravel the pharmacological and docking potentials of its bioactive compounds against thymidylate kinase.
Methods
The collection of the Mangifera indica stem bark, extract preparation, in vivo antiplasmodial assay, in vitro antioxidant assay, retrieval of Plasmodium falciparum thymidylate kinase, and gas chromatography-mass spectrophotometry (GC-MS) were done using standard protocols. The SwissADME tool was used in the prediction of the physicochemical, drug-likeness, and pharmacokinetic properties, whereas molecular docking analysis was done with the MCULE tool.
Results
The GC-MS profiling revealed mangiferin, d-idopyranoside, lauric acid, fatty acid, isoprene derivatives, etc; these compounds contributed immensely to the plant's antioxidant and antiplasmodial (87.71%) potentials. Also, the compounds (55.7%) successfully docked the active sites of thymidylate kinase; 72.1% and 57.4% revealed high rates of gastrointestinal tract (GIT) absorption and distribution in the interstitial tissues, respectively, whereas 98.4% were not substrates of permeation glycoprotein. However, the % non-inhibition of CYP1A2, CYP2C19, CYP2C9, CYP2D6, and CYP3A4 were 45.9, 98.4, 67.2, 95.1, and 98.4, respectively, while drug-likeness revealed 100% Lipinski's compliance.
Conclusion
The observed antiplasmodial, antioxidant, and pharmacological properties could be due to the presence of the glucosyl xanthone, idopyranoside, fatty acid, and isoprene derivatives.
Background
Malaria is a potentially fatal disease caused by Plasmodium falciparum, abbreviated P. falciparum. 1 In 2019, it was anticipated to be 229 million cases worldwide, with an estimated fatality rate of 409,000 persons, predominantly young children in sub-Saharan Africa. 2 Moreover, reactive oxygen species (ROS) are produced by the parasite's breakdown of hemoglobin in red blood cells (RBCs) in severe malaria cases; as a result, patients exhibit elevated levels of ROS products in urine and lower RBC deformability under shear stress; 3 a clear scenario of oxidative stress. However, research that shows ROS have powerful oxidizing activities and cause damage to biological molecules (proteins, lipids, and DNA) as well as accompanying changes in their structures and functions supports the idea that oxidative stress is a harmful health condition. 4 Traditionally and scientifically, oxidative stress-related disorders are often managed with phytomedicine, and one of the medicinal plants used is Mangifera indica. This plant has already been reported to have antimalarial,5–9 antioxidant and anti-inflammatory10–14 activities; moreover, it contains a bioactive compound (mangiferin) also reported to have antioxidant 11 and antimalarial 12 activities.
Additionally, a study revealed that P. falciparum was resistant to common antimalarial medications, 15 necessitating the development of novel antimalarial medications. 16 Diverse protein targets and pathways have been uncovered through studies on the creation of novel medicines. 16 It is important to note that crucial pathways and targets have been identified for the novel medications that are currently being tested in clinical trials. 17 By screening various chemical libraries against protein targets, it is necessary to investigate new antimalarial targets from novel pathways unrelated to resistance. 17 A crucial step in the creation of cellular DNA is the reversible phosphorylation of deoxythymidine monophosphate (dTMP) to deoxythymidine diphosphate (dTDP), which is catalyzed by one of the protein targets, thymidylate kinase. 17 It differs from other homologs in that it has a broad substrate specificity, which makes it a viable drug target. 17 The pyrimidine biosynthetic pathway, a de novo mechanism that generates pyrimidine is where Plasmodium falciparum thymidine monophosphate kinase (PfTMPK) catalyzes because pyrimidine is required for fast DNA replication, and the parasite is solely dependent on it due to lack of salvage pathway. 17
In this study, lead compounds with antimalarial potentials were predicted after virtual screening of some bioactive compounds against PfTMPK. Antimalarial drug research has recently benefited greatly from the advent of quantitative structure-activity relationship (QSAR) models. 17 As a result, our study utilized in vivo, in vitro, and in silico docking studies to uncover both antiplasmodial and antioxidant potentials of the extracts/compounds as well as the structural insights for PfTMPK inhibition in order to choose antimalarial and antioxidant lead compounds with favourable pharmacological characteristics. 17 Therefore, through the incorporation of in silico techniques, the lead compounds could further be optimized as drug candidates. Hence, the study has revealed the antimalarial and antioxidant mechanism of actions of the lead compounds from Mangifera indica stem bark domesticated in Akpabuyo, Cross River State, Nigeria through experimental and computational studies.
Materials and Methods
Collection and Identification of Plant Material
On April 8, 2023, the stem bark of Mangifera indica was obtained from the botanical farm, Arthur Jarvis University, in Akpabuyo, Cross River State, Nigeria. Mr Godwin Eteng, a plant taxonomist from the University of Calabar in Nigeria, performed the identification, and voucher number UCH432G was deposited.
Extraction of Mangifera indica stem bark
The stem bark of Mangifera indica plant (763 g) were washed with running tap water, chopped with machete, and dried under the sun. Pulverization with electric blender was done to generate coarse particles (509.34 g); maceration in 10 L of 90% ethanol for 72 h yielded 8.60% of stem bark extract (43.8 g) after the processes of filtration and concentration in a rotary evaporator (at 40 oC). 18
Evaluation of in vitro antioxidant activities
The methods described by Ruch et al, 19 Carter, 20 Re et al, 21 Klein and Perry, 22 Yen and Chen, 23 and Liyang-Pathiranan and Shahidi 24 were used to measure the hydrogen peroxide scavenging properties, ferrous ion chelating properties, total antioxidant capacity, vitamin C concentrations, ferric reducing antioxidant power, and 2,2-diphenyl-1-picrylhydrazyl (DPPH)-radical scavenging properties, respectively.
Acquisition, Handling of Mice, and Ethical Approval
The Animal House, located in the Faculty of Science, Arthur Jarvis University, Cross River State, Nigeria, provided thirty (30) Albino Wistar mice of (13-27 g) that were used in the study. The NIH guidelines for handling and caring for experimental animals (NIH publication No. 8023; revised 1978) were judiciously implemented. The mice were given the freedom to a standard pellet diet, water, and kept at ambient temperature. The approval (approval number: AJUFNAS/0136) for this study's experimental protocol was given by the Animal Ethics Committee of our faculty, and compliance was judiciously monitored.
Determination of Acute Toxicity
Lethal dose (LD50) and effective doses (ED50) were geometrically calculated by using the method reported by Lorke, 25 which involved phases I and II. The rats were observed for 24 h while the number of deaths was recorded in order to establish the median dosage of the plant as well as the one that might induce toxicity in a short term in mice. After administering the herbal formulation (50-2000 mg/kg) through the peritoneum, toxicity symptoms such as gasping, slowed breathing, palpitations, and death could be seen within 24 h.
Parasite and Inoculum Preparation
The National Institute of Medical Research (NIMR), Nigeria, provided the donor mice infected with chloroquine-sensitive Plasmodium berghei (NK-65) strain used for the study. An inoculum of Plasmodium berghei-infected erythrocytes was prepared according to the method.5,6
Drug Formulation and Administration to Mice
Exactly 100 ml of distilled water was used in the dissolution of tablets of artemisinin combination therapy (ACT) (artemeter 80 mg/lumenfantrine 480 mg) (560 mg), Fidson Healthcare Plc, Nigeria, and administered at a dosage (8 mg/kg body weight of mice), as well as 20 ml of distilled water, which enhanced the dissolution of tablets of artesunate (100 mg) and administered at a dosage (5 mg/kg body weight of mice), both used as a positive control in the study. 5
In vivo antiplasmodial determination
A method reported by some researchers5,26 was used for the evaluation of in vivo chemotherapeutic antimalarial activity of the plant. Following the completion of the administration on the fourth day, the euthanasia procedure of CO2 asphyxiation using CO2 chamber with a concentration of 30–70% was applicable; unconsciousness and death of mice were observed within 30–60 s and 1-2 min, respectively,27,28 and the dead animals were judiciously buried. However, an anesthetic agent was not used in this study because the animals were not sacrificed. The parasite density (parasites/µL), mean survival time (MST) of each treatment group over a period of 30 days (D0-D29), as well as the percentage growth inhibition of the parasites, were calculated as follows:
Examination Procedure
Each slide was prepared by measuring blood (2 μL) for the thin film as well as 6 μL for the thick film according to a report by Owusu-Agyei et al, 29 whereas their parasite densities and % growth inhibition of Plasmodium berghei were calculated and expressed as reported. 30
Gas Chromatography Mass Spectrometry (GC-MS)
Ten grams (10 g) of Mangifera indica's stem bark extract was weighed, dissolved in dimethyl sulfoxide (DMSO), and subjected to GC-MS analysis according to the method described by some researchers. 6 The GC-MS data of the extract was recorded on an Agilent 7890A gas chromatograph connected with an Agilent MS model 5975C MSD detector (Agilent Technologies, USA). An HP5-MS column with 5% phenyl-methylpolysiloxane, 30 m × 0.25 mm × 0.25 µm, was used with a helium gas flow under a pressure of 10 psi. The injector temperature was set at 280 °C. The oven temperature started from 150 °C for 3 min and increased to 300 °C at 10 °C/minute and held for 5 min at 300 °C. The mass spectrometer was operated using the electron ionization mode at 70 eV. The identification of compounds was achieved based on their retention indices, area sum %, and by comparison of their mass spectral fragmentation patterns with the National Institute of Standards and Technology (NIST) database/ChemStation data system.
Assessment of the Physicochemical and Pharmacokinetic Properties of the Bioactive Compounds
The SMILES of the different compounds were retrieved from the PubChem web platform (http) and were subsequently converted into mole files using MarvinSketch software (version 15.11.30).6,31 The drug-likeness and absorption, distribution, metabolism, and excretion (ADME) properties of the bioactive compounds were determined using previously reported methods. 30 Potential drug-like lead compounds against the target protein were found using the SWISSADME program. Moreover, information on all the parameters, including Lipinski's rule of five, Egan, Muegge, Veber, and Ghose parameters, was reported. 30
Plasmodium Falciparum Thymidylate Kinase Protein Properties and Molecular Docking of the Bioactive Compounds
The Research Collaboratory for Structural Bioinformatics (RCSC) protein database was used to retrieve the three-dimensional structure of the thymidylate kinase protein. The following characteristics of the recovered protein were noted: Thymidylate kinase is the name of the protein (source: sc-PDB; PDB ID: 2WWG; Uniprot name: Q8I4S1_PLAF7; Uniprot Accession ID: Q8I4S1; Uniprot taxonomic ID: 36329; organism: Plasmodium falciparum). The protein's default binding sites for binding centers X, Y, and Z were 8.9677, −12.0499, and −5.6692, respectively. Using the MCULE online tool, the ligands were docked blindly against the bioactive molecules. 30 The thymidylate kinase protein's posture and the binding affinities (scores) of the various compounds were predicted by the MCULE docking tool. The binding interactions came after the blind docking; their 3-D format, the amino acid residues, and the binding poses were retrieved for all the interactions.32,33
Statistical Analysis
GraphPad Prism version 9.0 (GraphPad Software, Incorporated, San Diego, CA, USA) was used to analyze the statistics and expressed as mean ± SEM (standard error of mean). The significant differences within and between the different groups were analyzed using one-way ANOVA (analysis of variance); then, their means were compared through the use of Turkey's post hoc test. The differences at a confidence level of 95% were considered to be statistically significant.
Results
The GC-MS analysis of the ethanol extract of Mangifera indica stem bark. The GC-MS result (Table 1) revealed a total of thirty-two (32) bioactive compounds; they included but were not limited to mangiferin silyl ester; 9,12-Octadecadienoic acid, methyl ester; Z-8-Methyl-9- tetradecenoic acid ester; 7,11-Hexadecadienal acetate; oleic acid, methyl ester; 1,6-undecadiene; 1-(p-toluidino)-1-deoxy-beta-d-idopyranose, silyl ester; and 3,7-dimethyl-1,6-Octadiene ester, etc.
GC-MS Analysis of Mangifera indica Stem Bark.

In Vivo Antiplasmodial (Suppressive Test Model)
Moreover, the chemo-suppressive efficacy of the drugs, extract, and formulation are presented in Table 2. The decreasing order of % growth inhibition of the parasites were as follows: stem bark + artesunate (88.69%) > artesunate (88.04%) > stem bark (87.71%) > ACT (84.60%) > negative control.
In Vivo Antiplasmodial (Suppressive Test Model) (n = 6).

a*significant decrease; p < 0.05 versus control; a** implies significant increases p < 0.05 versus control.
The Interpretation of the Pharmacokinetic Properties of the Bioactive Compounds
According to Table 3, out of the thirty-two (32) compounds screened, 72.1% showed a high rate of absorption in the GIT with 57.4% showing abilities to permeate the blood-brain barrier (BBB). Also, the predictor of toxicity and renal clearance, P- gp showed that the majority 98.4% of the screened bioactive compounds were not substrates of permeation glycoprotein (P-gp). Further analysis of the predictors of drug metabolism for possible inhibition of the different mono-oxygenases revealed non-inhibition of CYP1A2, CYP2C19, CYP2C9, CYP2D6, and CYP3A4 as 45.9%, 98.4%, 67.2%, 95.1%, and 98.4% respectively. Similarly, the screened compounds followed the rules of Lipinski (Pfizer) 100%, Ghose (Amgen) 29.5%, Egan (Pharmacia) 70.5%, Veber 50.8%, Muegge 13.1% and had high bioavailability score as 0.55 (88.5%) and 0.85 (11.5%).
Pharmacokinetic Properties of the Bioactive Compounds.

Physicochemical Properties and Drug-Likeness of the various Bioactive Compounds
The result (Table 4) highlighted the physicochemical and drug-likeness of the bioactive compounds. All (100%) of the screened compounds obeyed Lipinski's rule of five with the highest of only one violation for the compounds whereas the molecular weights of the lead compounds were less than 500 Daltons.
Physicochemical Properties and Drug-Likeness of the Bioactive Compounds.

Docking Output of Ligands Against Thymidylate Kinase
The docking of the thirty-two (32) bioactive compounds (Figure 1A) revealed that 75.7% compounds successfully docked thymidylate kinase (2WWG) (Figure 1B) with scores ranging from −5.5 to −9.3 as presented in Table 5 and Figures 2–5. Docking simulation for 2WWG against 3,7-dimethyl-(S)-1,6-octadiene ester revealed the amino acid residues as Tyr98, Asn65, Phe42, Pro43, Ile150, Glu149, Leu157, Tyr151, Asp15, Try95/98, Ser101/106. However, 2-Glucopyranosyl-1,3,6,7-tetrahydroxy-xanthen-9-one, silyl ester (mangiferin silyl ester) docking with 2WWG revealed the amino acid residues at its active sites as Tyr98, Tyr41, Leu57, Phe42, Ile150, Tyr151, Asp15, Glu152, Ser17, Glu149, Glu12, Ser101, Gly102, and Ala99. Also, shikimic acid, methyl ester revealed the amino acid residues at the site of interaction with 2WWG as Pro43, Ile150, Tyr151, Asp15, Glu12, Ser101, Tyr98, Asn65, Phe42, and Arg97. Moreover, Ile150, Glu149, Tyr151, Lys19, Asp15, Glu152, Leu157, Arg16, Ser17, Phe11, and Asp96 influenced the bonding of z-8-methyl-9-tetradecenoic acid ester with thymidylate kinase. Similarly, the amino acid residues, including Glu49, Ile150, Asp15, Pro43, Phe42, Tyr98, Asn65, Glu12, Ser106, Tyr95, and Ser101 interacted in the bonding of 7,11- hexadecadienal acetate to 2WWG active sites. 1-(p-toluidino)-1-deoxy-beta-d-idopyranose, silyl ester docking simulation with 2WWG showed that the ligand interacted through the following amino acid residues: Leu157/150, Glu149, Asn65, Phe42, Pro43, Asp15, Tyr151, Tyr98, Ser101/106, Asp15, Gly102, Ala99, Arg97, Tyr98, Glu12, Gly102, Tyr105/109, Glu12, Lys19, and Glu149. In addition, dodecanoic acid, methyl ester bonded with 2WWG through the interactions of Leu157/150, Glu149, Asn65, Phe42, Pro43, Asp15, Tyr151, Tyr98, Ser101/106, Asp15, Gly102, Ala99, Arg97, Tyr98, Glu12, Gly102, Tyr105/109, Glu12, Lys19, and Glu149 amino acids.




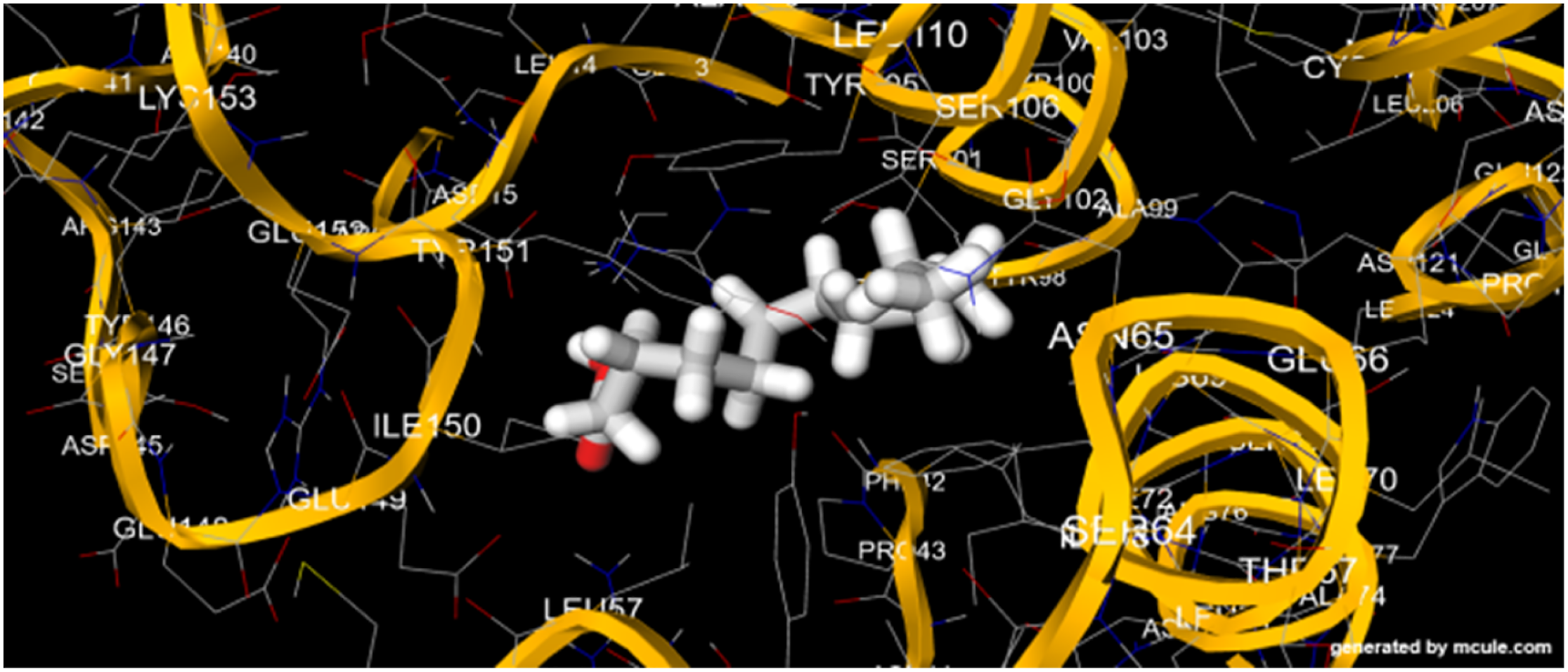
Interaction Between Dodecanoic Acid, Methyl Ester and 2WWG.
Docking Scores and Corresponding Amino Acid Residues.

Antioxidant Activities of Mangifera indica Stem Bark
The result of the DPPH-radical scavenging activity of the extract of the plant (Figure 6A) as compared with ascorbic acid (vitamin C) showed that the extract exhibited lower DPPH-radical scavenging activity at varying concentrations. Also, the result (Figure 6B) showed that the ferric reducing antioxidant power (FRAP) of the stem bark as compared with ascorbic acid showed that both exhibited similar FRAP with very little variation. Moreover, the result (Figure 6C) of the ferrous ion chelating activity of the plant's stem bark as compared with ascorbic acid showed that the positive control had better ferrous ion chelating activity than the extract at all concentrations; nevertheless, the increase in ferrous ion chelating activity of the extract was concentration dependent. Furthermore, the total antioxidant capacity (TAC) as shown in Figure 6D showed that ascorbic acid had better TAC than the extract. In addition, the result (Figure 7) of hydrogen peroxide (H2O2) scavenging activity of the plant showed a better activity of the control over the extract and the increase in H2O2 scavenging activity was concentration dependent.

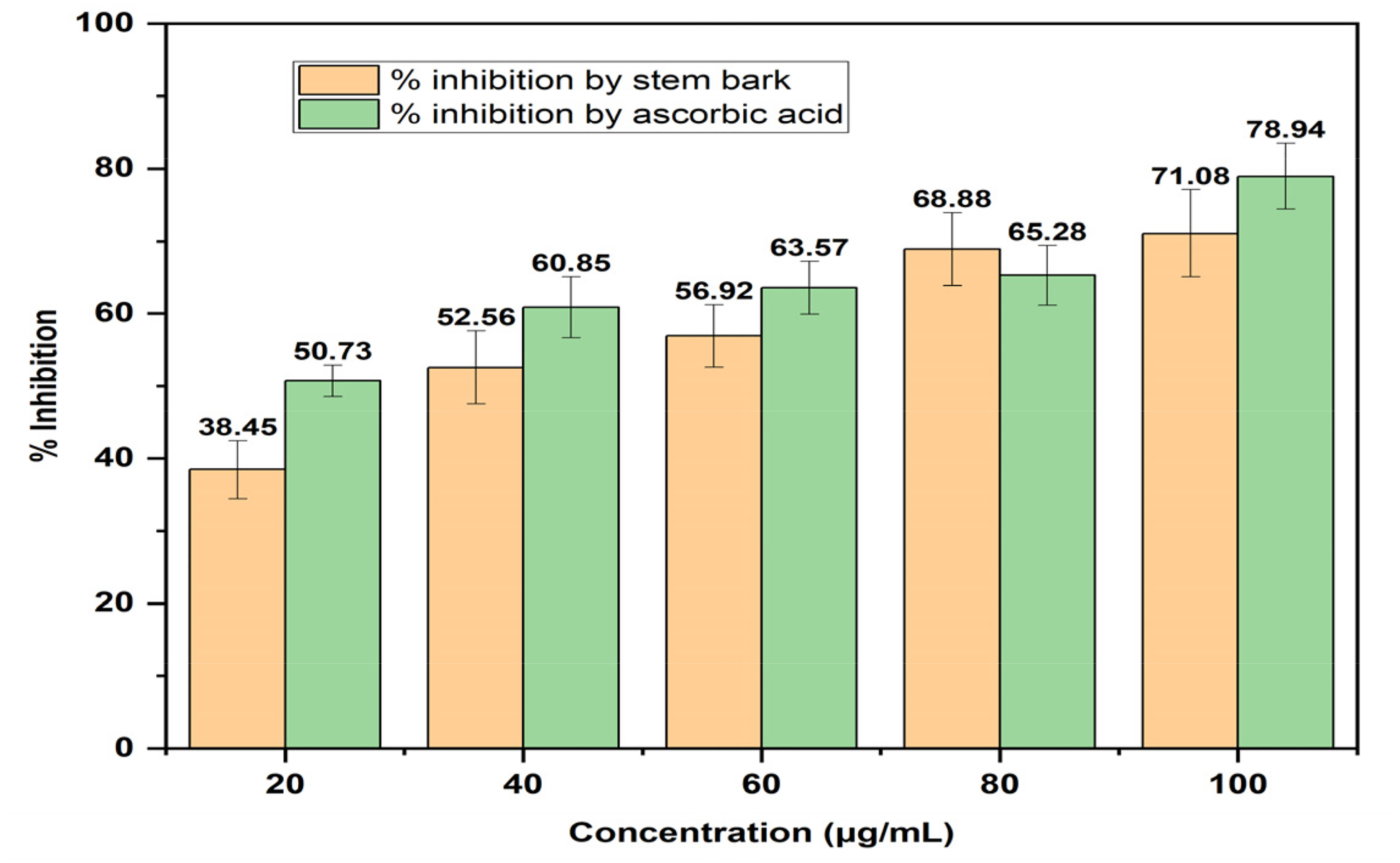
H2O2 Scavenging Activity of Ethanol Extract of Mangifera Indica Stem Bark.
Discussion
Severe malaria pathogenesis induces hyperparasitemia with an attendant oxidative stress due to free radicals (superoxide anions, hydrogen peroxide) generated through the parasites’ metabolic pathways. Consequently, the effect of hyperparasitemia is devastating; therefore, chemotherapies could mitigate the parasite-induced and oxidative stress-induced mortality through different mechanisms of action including parasite chemosuppression and schizont suicidal attacks.5,26 From the study, the chemosuppressive potential of the Mangifera indica stem bark showed that all the treatment groups produced significant reductions in their parasite densities as compared with the group of negative controls; hence, the extract and drugs greatly suppressed the expression of P. berghei-induced infection in the mice as evidenced in their remarkable increases in % growth inhibition of parasites and the significant increases in their mean survival time (MST). Therefore, the chemosuppressive activity of the extract against the parasite could be attributed to the predominant mangiferin silyl ester; 1-(p-toluidino)-1-deoxy-.beta.-d-idopyranose, silyl ester; Z-8-methyl-9-tetradecenoic acid ester; 3,7-dimethyl-1,6-octadiene ester; and 7,11-hexadecadienal acetate compounds present in the plant as they could have acted through the inhibition of the activities of thymidylate synthesis; thereby, leading to the mitigation of protein synthesis in the parasites and subsequent death. Conversely, the prolonged MST for the parasitized mice was due to better parasitemia clearance by the bioactive compounds; hence, better chemosuppressive activities. This position was earlier corroborated by some researchers 18 on the significant increases in MST of mice in a similar study.
According to some researchers, Mangifera indica is ethnopharmacologically used in the treatment of malaria. The plant's bioactive compounds’ mechanism of action could be through the inhibition of PfTMPK, 33 which in turn affects the parasite's metabolic pathways and processes. The docking of PfTMPK with bioactive substances in this study revealed the three structural properties (-NH fragment, -OH group, and significant numbers of oxygen atoms) that led to an inhibitory effect and appropriate binding at the active sites of thymidylate kinase. 12 Further docking simulation revealed the best five lead compounds in decreasing order based on their docking scores as follows: mangiferin silyl ester (a C-glycoside polyhydroxylated xanthone ester); 1-(p-toluidino)-1-deoxy-.beta.- d-idopyranose, silyl ester (an N-arylated-β-d-idopyranoside); Z-8-methyl-9-tetradecenoic acid ester (a branched-chain monounsaturated fatty acid ester); 3,7-dimethyl-1,6-octadiene ester (a branched-chain isoprene derivative); and 7,11-hexadecadienal acetate (a polyunsaturated aldehyde-derived flavor compound) that compared favorably with chloroquine (a 4-aminoquinoline antimalarial agent). Moreover, hydrogen bonds were formed with the Asp residues in each compound whereas pi-pi-stacked interactions with their Tyr and Phe residues ensued. The inhibitory effect of the protonated forms of mangiferin ester, specifically the C6-O- species, appearing at pH 7.5, paired with two docked positions of either a xanthone or glucose moiety, positioned inside the thymidylate kinase-binding pocket, was discovered by additional simulations using the silyl derivatives of mangiferin and 1-(p-toluidino)-1-deoxy-.beta.-d-idopyranose. 12 The presence of O-atoms enhanced the formation of more hydrogen bonds with important residues like Arg. In addition, the OH groups in the lauric acid, methyl ester; 1-(p-toluidino)-1-deoxy-.beta.-d-idopyranose, silyl ester; 3,7-dimethyl-1,6-octadiene ester; and mangiferin silyl ester facilitated electrostatic and hydrophobic interactions with the various amino acid residues in the thymidylate kinase binding pocket; hence, the reason for their lower binding energies and docking scores. This observation was similar to the report by Ejeh et al 34 and Asanga et al 30 that lead compounds had lower docking energy scores; therefore, the lower score function during docking simulations is an advantage for the prediction of both strength and type of signals produced during the structure-based design of drugs. Therefore, mangiferin silyl ester; 1-(p-toluidino)-1-deoxy-.beta.-d-idopyranose, silyl ester; Z-8-methyl-9-tetradecenoic acid ester; 3,7-dimethyl-1,6-octadiene ester; and 7,11-hexadecadienal acetate inhibited thymidylate synthase and blocked the production of DNA through the prevention of 2’-deoxyuridine-5’-monophosphate (dUMP) conversion to 2’-deoxythymidine-5’-monophosphate (dTMP); thereby, blocking parasites’ DNA syntheses and cell death occurred.
The Lipinski's rule of five, the most commonly used methodology in the early stages of drug development and optimization, ensures that lead compounds behave like drugs. 30 The result showed 100% compliance with Lipinski's rule, thereby suggesting that all the lead compounds were potential drugs with acceptable physicochemical and pharmacological properties. The lead compounds being part of 72.1% of compounds with a high rate of absorption suggested high absorption in GIT and better bioavailability in the systemic circulation for the compounds. Indices of drug distribution (abilities to cross the blood-brain barrier and the likeness of acting as substrates of permeation glycoprotein) were also evaluated to ascertain the likelihood of the compounds to penetrate the brain as well as their efficacy and toxicity with respect to acting as substrates or inhibitors of permeation glycoprotein. More so, the lead compounds being part of the 57.4% with abilities to permeate the blood-brain barrier (BBB) unraveled faster distribution of the compounds in the biological system; therefore, the compounds’ penetration of the brain may be through lipid-mediated free diffusion or receptor-mediated transport. In addition, drug metabolism is mediated by mono-oxygenases to forestall toxicity and enhance biotransformation for possible therapeutic responses; therefore, the non-inhibition of the mono-oxygenases by the lead compounds suggested better biotransformation, faster clearance, and elimination of their metabolites, as well as lower toxicity of the compounds; this result corroborated that earlier reported. 30 Similarly, the compounds’ high bioavailability score suggested high GIT absorption and distribution in the interstitial tissues with remarkable pharmacokinetic properties; hence, the lead compounds are drug-like molecules. This observation was consistent with earlier reports by some researchers on the drug-likeness of some molecules.34,35
The most effective antimalarial medications have been derived from natural sources throughout the history of malaria chemotherapy; 17 conversely, some researchers 18 corroborated that the tropical forests in Nigeria are vested with many medicinal plants; so, robust research and implementation of research findings, the isolation and characterization of bioactive compounds in these plants, as well as the integration of orthodox and traditional medicine could drastically boost health care delivery in Nigeria. In addition, these studies revealed some of the natural products with some reported pharmacological activities. Some researchers 12 reported the antimalarial activities of mangiferin, whereas others 5 reported that fatty acid esters in Mangifera indica are responsible for the plant's antimalarial activity through the inhibition of fatty acid biosynthesis in the parasite's apicoplast. More so, Okokon et al 36 reported the antimalarial activities of polyunsaturated fatty acid esters. Therefore, Kumaratilake et al 37 and Shaikh et al 38 stated that the degree of unsaturation in polyunsaturated fatty acids is crucial for their antiplasmodial effect against parasites. Conversely, Qidwai and Khan 39 posited that the inhibition of fatty acid synthase II is the drug target for fatty acids in Plasmodium falciparum. Therefore, the derivatives of mangiferin; 1-(p-toluidino)-1-deoxy-.beta. -d-idopyranose; Z-8-methyl-9-tetradecenoic acid; 3,7-dimethyl-1,6-octadiene; and 7,11-hexadecadienal docking with PfTMPK suggested their abilities in the inhibition of the replication, transcription, and translation of fatty acid synthase II, the major enzyme in lipid biosynthesis in the parasite's apicoplast, as the likely antiplasmodial mechanism of action of these lead compounds.
It is worthy of note that free radicals as initiators of oxidative stress have been reported to cause mutations or cell death through their interaction with macromolecules like deoxyribonucleic acid (DNA), proteins, and lipids; 40 however, oxidative stress is therefore thought to contribute to the development and pathogenesis of malaria; nevertheless, endogenous antioxidants shield the organism from the negative effects of these free radicals. 40 Without mincing words, some pathological conditions could interfere with these endogenous antioxidants’ protective actions, necessitating the prescription of exogenous antioxidants. 40 Furthermore, some compounds’ antioxidant activities have been linked to pathways, including the binding of transition metal ion catalysts, peroxide decomposition, stoppage of hydrogen absorption, reductive capacity, and radical scavenging.41,42 In this study, hydrogen ion donation was responsible for the concentration-dependent suppression of DPPH radical by the compounds in Mangifera indica extract; 43 this result was consistent with the findings of Itoh et al. 13 However, the observed radical scavenging activities could result from the derivative of glucosyl xanthone (mangiferin), whose formulations are reportedly used as food supplements for oxidative stress-related disorders. 14 Also, the ferric reducing antioxidant power (Fe3+ to Fe2+) of the plant showed that the extract had concentration-dependent reducing ability due to the presence of antioxidant compounds like mangiferin, lauric acid, fatty acid, and isoprene derivatives. The oxidized form of the metal ion is stabilized by metal-chelating chemicals, which lower the concentration of transition metals in lipid peroxidation by reducing their redox potentials.44,45 The high chelating power of the extract demonstrated its concentration-dependent chelating activities and antioxidant potentials, supporting earlier findings by Andreu et al 46 that the ability of mangiferin to bind iron served as the main mechanism for preventing lipid peroxidation caused by Fe2+ citrate in the rat liver mitochondria. Additionally, the extract had a high total antioxidant capacity that varied with concentration, comparable to that reported by Vyas et al 47 and Halliwell et al. 48 But Sun et al 49 and Dar et al 11 reported that mangiferin has a complete antioxidant capability. Furthermore, the concentration-dependent moderate H2O2 scavenging effect of the extract as compared with ascorbic acid revealed the ease of H2O2 scavenging to avert its hydroxyl radical generation and toxicity to biological membranes.33,50
Conclusion
The derivatives of mangiferin; 1-(p-toluidino)-1-deoxy-.beta.-d-idopyranose; Z-8-methyl-9-tetradecenoic acid; 3,7-dimethyl-1,6-octadiene; and 7,11-hexadecadienal characterized from Mangifera indica stem bark are the new antimalarial and antioxidant compounds targeting PfTMPK as evidenced through experimental and computational studies; therefore, these lead compounds are candidates for lead optimization and further rational drug design processes.
Strength and Limitations
The strength of this study included:
The provision of data on the antiplasmodial and antioxidant activities of Mangifera indica stem bark. The GC-MS profiling revealed mangiferin, d-idopyranoside, lauric acid, fatty acid, isoprene derivatives, etc; these compounds contributed immensely to the plant's antioxidant and antiplasmodial (87.71%) potentials. The derivatives of mangiferin; 1-(p-toluidino)-1-deoxy-.beta.-d-idopyranose; Z-8-methyl-9-tetradecenoic acid; 3,7-dimethyl-1,6-octadiene; and 7,11-hexadecadienal characterized from Mangifera indica stem bark are the new antimalarial and antioxidant compounds targeting PfTMPK
The limitations included:
In silico nature: Although molecular docking is a powerful tool, its predictions need to be validated through experimental studies such as in vitro and in vivo assays. Limited financial resources: Limited financial resources restricted the access to advanced instrumentation and technologies that could have enhanced the depth and breadth of the research. Non-validation of the docking with co-crystalized ligands due to computational cost
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251332125 - Supplemental material for Glucosyl Xanthone, d-Idopyranoside, Fatty Acid and Isoprene Derivatives as New Anti-Malarial Compounds Targeting Plasmodium Falciparum Thymidine Monophosphate Kinase (PfTMPK)
Supplemental material, sj-docx-1-npx-10.1177_1934578X251332125 for Glucosyl Xanthone, d-Idopyranoside, Fatty Acid and Isoprene Derivatives as New Anti-Malarial Compounds Targeting Plasmodium Falciparum Thymidine Monophosphate Kinase (PfTMPK) by Edet Asanga, Henshaw Okoroiwu, Uwem Edet, Usenobong Ufot, Akaninyene Joseph, Jude Okokon, Promise Nelson, Olorunfemi Eseyin, Ani Nkang, Mfon Akpan, Nikita Umoaffia, Casmir Isienyi, Goodnews Samuel, Samuel Essang, Olamide Kolawole, Tonia Edet, Praise Iwara and Deborah Ani in Natural Product Communications
Footnotes
Abbreviations
Acknowledgements
The authors are grateful to University of Uyo management for providing enabling environment and Mrs Ekaette Umoh of Pharmaceutical Chemistry Department, University of Uyo for technical assistance.
Declaration of AI-Assisted Technologies
The authors used Quillbot software to edit and review the content of this manuscript.
Statement of Human and Animal Rights
The NIH protocol as well as the ARRIVE guidelines for handling and care for laboratory animals were judiciously followed; moreover, euthanasia procedure of CO2 asphyxiation using CO2 chamber with concentration 30–70% was applicable.
Clinical Trial Number
Not applicable
Ethical Considerations
The NIH protocols for laboratory animals were judiciously followed and the experimental protocol was authorized by the Faculty of Science, Arthur Jarvis University's Animal Ethics Committee (approval number: AJUFNAS/0136), and compliance was judiciously monitored.
Informed Consent
This was not applicable as human subjects were not part of this study
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The author(s) hereby declare that the research was carried out in the absence of any financial and non-financial, intellectual, or commercial relationships that could lead to a conflict of interest.
Data Availability
Data will be made available by the corresponding author on request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



