Abstract
Introduction
Panax japonicus (PJ) is the Araliaceae Panax family, and it is found in the wild in southwestern China and Japan.1,2 The pharmacological components of PJ mainly include bioactive components such as saponins, organic acids, and polysaccharides, among which saponins are the most abundant. 3 Saponin is a class of triterpenoids or heliosteranes. 4 Chikusetsu Saponin IVa (CHS-IVa) is one of the major saponins in PJ. 5 The parent nucleus of CHS-IVa is a ziasterane-type pentacyclic triterpenoid compound with a molecular weight of 794.97. The chemical formula is C42H66O14, and its chemical structure is shown in Figure 1. Studies have shown that CHS-IVa and its derivatives have various pharmacological activities, including anti-inflammatory, 6 and antioxidant, 7 as well as liver protection. 8 They can also play a role in hypoglycemic effects, 9 regulating metabolism, 10 and anti-virus activity. 11 Studies have shown that CHS-IVa has a positive effect on cardiomyocyte injury in rats, mainly by scavenging the excessive production and accumulation of reactive oxygen species (ROS) caused by high glucose to alleviate the injury. 12 Wang et al found that CHS-IVa can effectively inhibit the production of inflammatory cytokines by human monocytic-leukemia cells (THP-1) macrophages. 6

The chemical structure of CHS-IVa. 10
Intestinal epithelial cells (IPEC) are a component of the intestinal epithelium that can separate the intestinal mucosa from the intestinal lumen environment. 13 They restrict the entry of toxins and pathogens, among other things, into mucosal tissues. 14 Oxidative stress can cause damage to IPEC, leading to tight junction damage and systemic endogenous stress syndrome, and disrupting the self-renewal process of the intestine,15,16 leading to the development of intestinal degenerative diseases such as inflammation and cancer. 17 When cells are stimulated, the intracellular enzyme antioxidant system, including superoxide dismutase (SOD), malondialdehyde (MDA), catalase (CAT), glutathione (GSH), and thioredoxin reductase (TrxR), undergoes changes to maintain good in vivo balance. When cells are stimulated, the intracellular enzyme antioxidant system, including SOD, 18 MDA, CAT, GSH, 19 and TrxR, undergo changes to maintain a fine homeostasis. Many natural products have anti-inflammatory, antioxidant, and other protective effects. Demircigil et al found that the main bioactive components in black cumin essential oil, such as thymoquinone, can exert antioxidant activity by increasing the content of CAT, GSH, and SOD. 20 The study by Ghasemi et al showed that the methanol extract of Teucrium hyrcanicum L. protects mouse embryonic fibroblast cells (NIH/3T3) lines from H2O2-induced oxidative stress by decreasing MDA formation and inhibiting GSH oxidation. 21 Many studies have shown that the saponins from PJ have a protective effect on the gut. Dun et al have shown that Saponins from PJ can inhibit inflammation, regulate intestinal epithelial tight junction damage and down-regulate mitogen-activated protein kinase (MAPK) and nuclear factor kappa-B (NF-κB) signaling pathway phosphorylation in aging rats. 22 PJ total saponins have altered the composition of the intestinal flora in experimental autoimmune encephalomyelitis mice and restored the ratio of Firmicutes to Bacteroides. 23 CHS-IVa is also the main component in PJ total saponins. 24 Therefore, this study took this as a starting point to explore whether CHS-IVa has a protective effect on porcine intestinal epithelial cells (IPEC-J2).
Materials and Methods
Experimental Reagents
CHS-IVa (purity 99.99%) was purchased from Chengdu Must Biotechnology Co., Ltd (Chengdu, China); 30% H2O2 (excellent pure) and absolute ethanol (analytical pure) were purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China); fetal bovine serum and DMEM high-sugar medium were purchased from Gibco (New York, USA); penicillin-streptomycin was purchased from Procell Life Science &Technology Co., Ltd (Wuhan, China); MTT solution was purchased from WellBio (Changsha, China); Formazan was purchased from Conway Century (Jiangsu, China); LDH Cytotoxicity Assay Kit was purchased from Solabel (Shanghai, China); CAT, SOD, GSH, TrxR, and MDA were purchased from Suzhou Keming Biotechnology Co., Ltd (Suzhou, China); Transzol up reagent was purchased from Quanshijin (Beijing, China); Reverse transcription kits were purchased from TaKaRa (Beijing, China); qRT-PCR kit was purchased from Vazyme (Nanjing, China).
Drug Configuration
900 μL of PBS solution was mixed with 100 μL of a 30% H2O2 solution in a centrifuge tube and filtered through a 0.22-μm sterile needle filter to obtain a 1 × 106 μM H2O2 stock solution. 2 × 105 μg/mL CHS-IVa stock solution was prepared by 0.5 mL of DMSO and 0.1000 g of CHS-IVa powder. Each concentration of reagent in the experiment was obtained by diluting the stock solution according to the corresponding method.
Cell and Cell Culture
IPEC-J2 was purchased from Guangzhou JENNIO Bio-Technology Co., Ltd (Guangzhou, China). The cells were cultured in a thermostatic incubator at 37 °C and 5% CO2, and the complete medium was configured with a ratio of 89% DMEM, 1% penicillin–streptomycin, and 10% fetal calf serum.
Cell Viability Assay
In order to select the optimal H2O2 concentration, growing logarithmic IPEC cells were seeded in 96-well plates. MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) was used to detect the viability of IPEC-J2 cells. According to the experimental group, the corresponding H2O2 dilution (0, 250, 500, 750, and 1000 μM) was added to IPEC cells for 2 hours or 4 hours. Then, discarding the culture medium, 110 μL of MTT solution was added to the wells. After incubation at 37 °C for 2 hours, 150 μL of formazan replaced MTT and was left to stand for 10 minutes. Then we determined the OD of each well at a wavelength of 490 nm and calculated cell viability according to Cell activity = OD490 (treatment-blank)/OD490 (control-blank) × 100%. 25 To select the most suitable CHS-IVa concentration, growing logarithmic IPEC cells were seeded in 96-well plates. CHS-IVa dilution (0, 50, 100, 200, and 400 μg/mL) and complete medium were applied to IPEC cells for 9, 24, and 48 hours. Similarly, CHS-IVa dilution (0, 60, 80, and 100 μg/mL) and complete medium were applied to IPEC cells for 3 and 6 hours. The MTT method was used to detect cell viability.
To investigate the effect of CHS-IVa on cell viability of H2O2-damaged IPEC-J2 cells, logarithmic growth phase cells were seeded in 96-well culture plates. First, 1000 μM H2O2 was added to the air pretreatment for 2 hours. Then 80 and 100 μg/mL of CHS-IVa was respectively incubated for 6 hours. Afterward, the cell activity was tested with the MTT assay.
Cell Scratch Experiment
IPEC-J2 in logarithmic growth phase was seeded in 6-well plates. After placing the 6-well plate lid in the center of each well, the tip of the 10 μL pipette was kept perpendicular to the 6-well plate lid and streaked. After scribing, the medium supernatant was removed after streaking was complete and PBS was added to remove residual cell debris. 26 CHS-IVa solutions at concentrations of 0, 80, 100, and 200 μg/mL diluted with DMEM medium were sequentially added to the wells. Image information was collected after 0, 8, 12, and 24 hours of drug action. Damage rate = scratch area at time t/initial scratch area. 27
Measurement of LDH, CAT, SOD, GSH, TrxR, and MDA
Commercial reagent kits were used to detect the levels of LDH, CAT, SOD, GSH, TrxR and MDA, and follow the steps provided by the manufacturer for the experiment.
qPCR
The total RNA of IPEC-J2 was extracted using TransZol Up reagent. cDNA was extracted using reverse transcription kits. The primer sequences of qPCR gene are listed in the table below (Table 1), and according to the qRT-PCR kit, the reaction reagents were fully mixed with cDNA, 28 and the qPCR procedure was set: 94 °C, 30 s; 94 °C, 5 s, 60 °C, 30 s, 40 cycles; extension at 65 °C.
Oligonucleotide Primers Used for Real-Time PCR.

Statistics and Analysis
The analysis of experiment results in this paper was expressed as “mean ± standard deviation,” and each group of experiments was repeated at least three times. All data were statistically analyzed by GraphPad Prism 8 software. The * sign in the data plot indicates a statistically significant difference between the two groups, P < .05. When the difference is not significant, the P > .05 cell morphology picture was collected using Motic Images Plus 3 software and then Image J was used to calculate the required data.
Results
Different Concentrations of H2O2 Reduced the Viability of IPEC-J2 Cells
Due to its strong oxidizing properties, H2O2 is often used to establish an oxidative damage model. 29 The viability of IPEC-J2 cells was detected after 2 hours of different concentrations of H2O2. Cell viability was significantly reduced after 2 hours treatment with 750 μM and 1000 μM H2O2 compared to the control group (P < .05) (Figure 2a). After 4 hours of treatment, the cell activity of the H2O2 group at a concentration of 1000 μM decreased significantly (P < .05) (Figure 2b). 1000 μM H2O2 can reduce cell viability at 2 hours, which was chosen for subsequent experiments.

Cell viability of IPEC-J2 cells at different times treated with different concentrations of H2O2. (a) 2 hours; (b) 4 hours. Data are presented as mean ± SD. *P < .05 was statistically significant compared to the control group.
Different Concentrations of CHS-IVa Regulated the Viability of IPEC-J2 Cells
Firstly, the viability of IPEC-J2 cells treated with different concentrations of CHS-IVa for 9, 24, and 48 hours was detected. When 50, 100, and 200 μg/mL CHS-IVa were treated for 9 hours, the viability of IPEC-J2 cells was increased significantly (P < .05) (Figure 3a). At 24 hours of treatment, IPEC-J2 cell viability still showed a significant upward trend in the 50 and 100 μg/mL groups (P < .05), but there was no significant change in cell viability (P > .05) at 24 hours in CHS-IVa at 200 μg/mL (Figure 3b). By 48 hours, the cell viability of the CHS-IVa group at a concentration of 200 μg/mL decreased significantly (P < .05), and the cell viability was no longer significantly increased in the 100 μg/mL group (Figure 3c). It shows that 100 μg/mL CHS-IVa has no toxic effect on cells within 24 hours. Then, the viability of different concentrations of CHS-IVa on IPEC-J2 cells was detected for 3 and 6 hours (Figure 3d and e), and it was found that the cell viability was significantly increased after 60, 80, and 100 μg/mL CHS-IVa acted on IPEC-J2 for 3 and 6 hours (P < .05). In order to better exert the pharmacological activity of CHS-IVa, we chose 80 and 100 μg/mL CHS-IVa to act on IPEC-J2 cells for 6 hours in subsequent experiments.
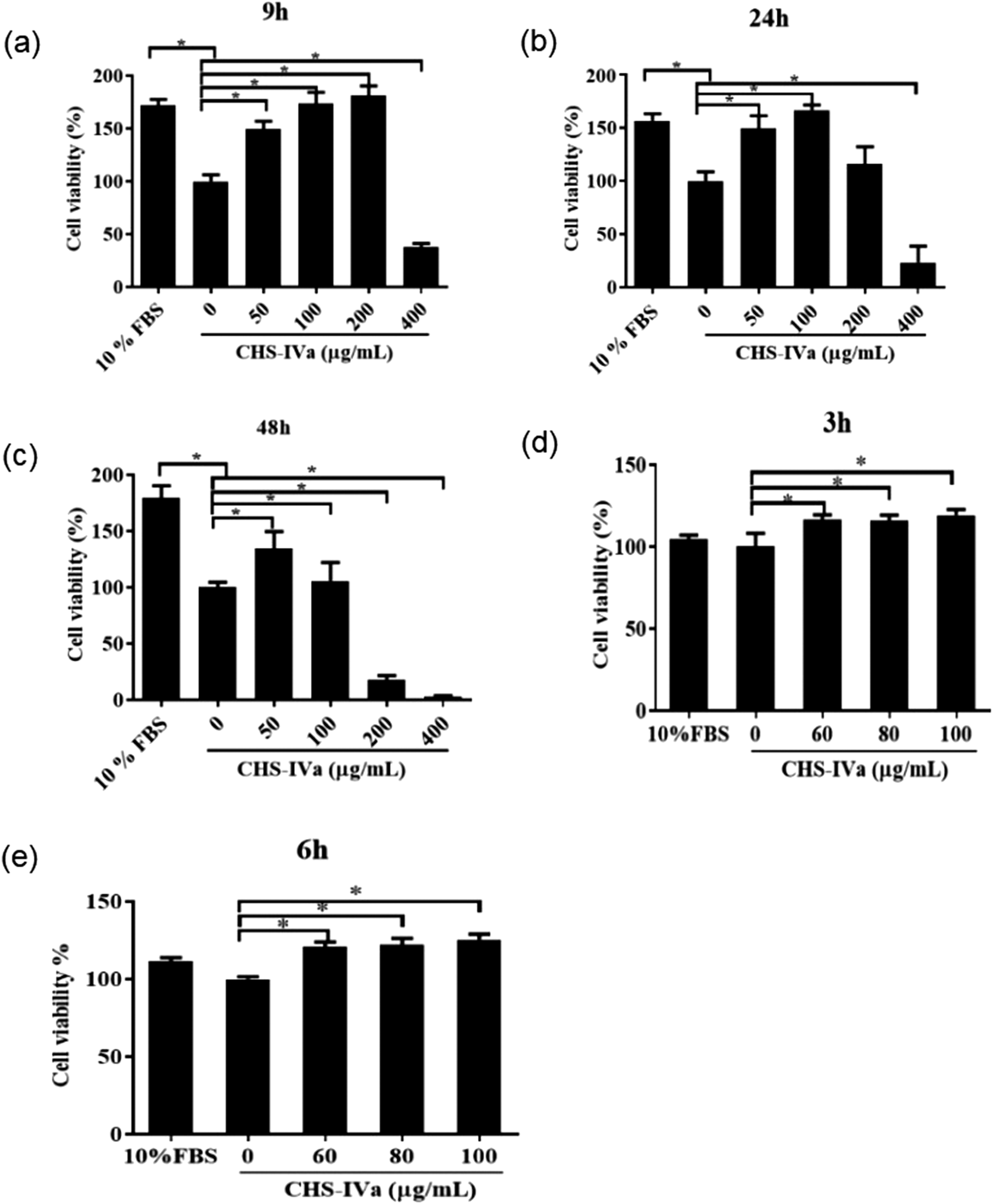
Cell viability of IPEC-J2 cells at different times treated with different concentrations of CHS-IVa. (a) 9 hours; (b) 24 hours; (c) 48 hours; (d) 3 hours; (e) 6 hours. Data are presented as mean ± SD. *P < .05 was statistically significant compared to the control group.
Different Concentrations of CHS-IVa Repaired the Scratch Damage of IPEC-J2 Cells
After CHS-IVa treatment of 80 and 100 μg/mL for 0 to 24 hours, the damage rate at the cell scribing gradually decreased (Figure 4a and b). The restoration effect of CHS-IVa at 100 μg/mL was obvious. The repair effect of CHS-IVa at 200 μg/mL on cells was slight after 8 and 12 hours, but the repair rate of cells significantly decreased after 24 hours of treatment.
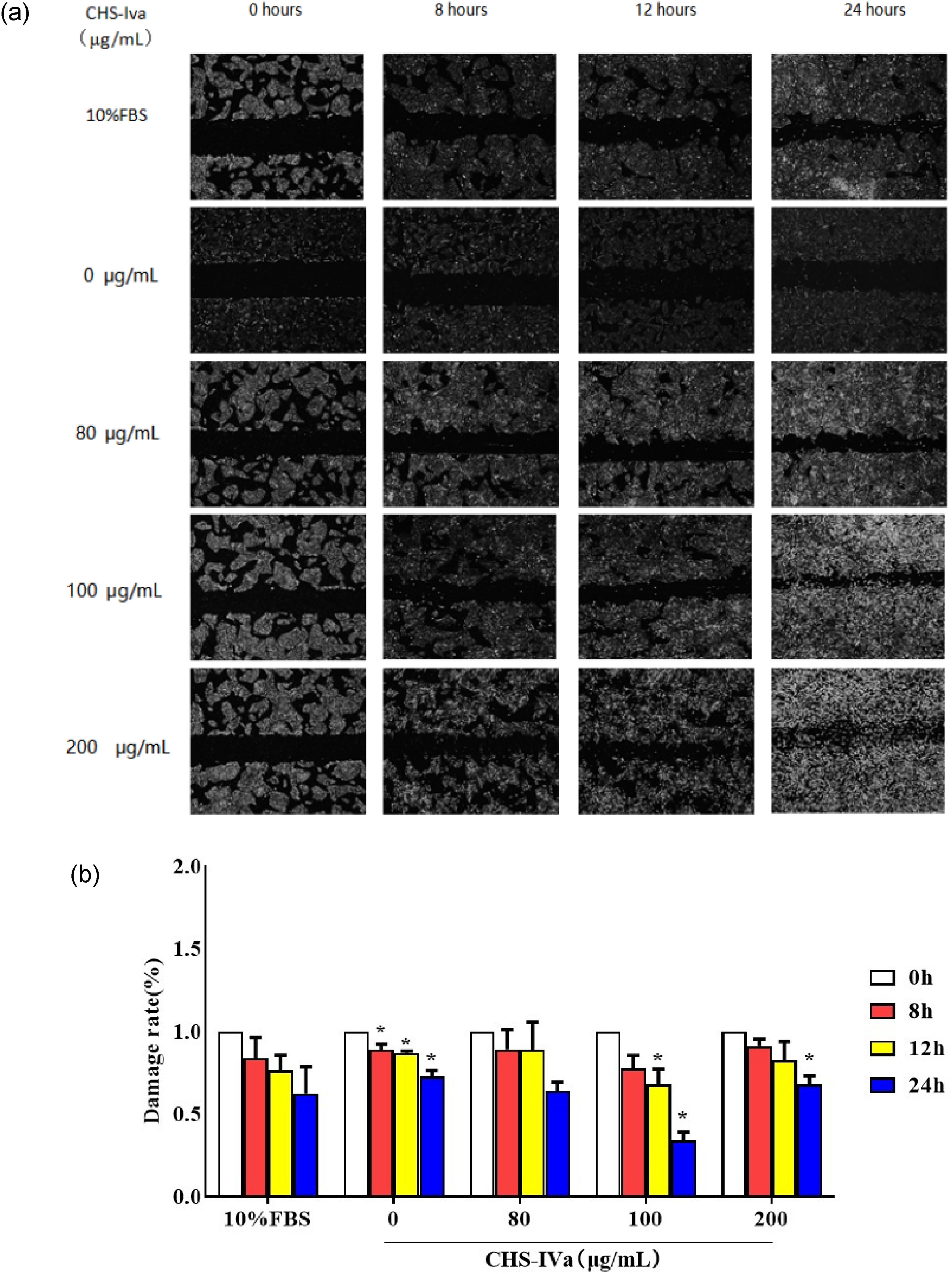
Effects of different concentrations of CHS-IVa on the repair of IPEC-J2 cells after injured. (a) Results of scratch test; (b) Damage rate (%). The scales are 100 μm. Data are presented as mean ± SD. *P < .05 was statistically significant compared to the control group.
CHS-IVa Repaired the Damage of IPEC-J2 Cells after H2O2 Treatment
Compared with the group treated with 1000 μM H2O2 for 2 hours, the activity of IPEC-J2 cells significantly increased after 6 hours in the 80 and 100 μg/mL CHS-IVa groups (P < .05) (Figure 5a). The results showed that 80 and 100 μg/mL had a certain protective effect on cells damaged by H2O2. LDH exists inside the cell and is released outside the cell when it is damaged or dies. 30 We detected changes in LDH viability in the culture supernatant of IPEC-J2 cells injured by 1000 μM H2O2 under different concentrations of CHS-IVa. LDH activity was significantly decreased in the 80 and 100 μg/mL CHS-IVa treatment groups compared with the 1000 μM H2O2 single treated group (Figure 5b).
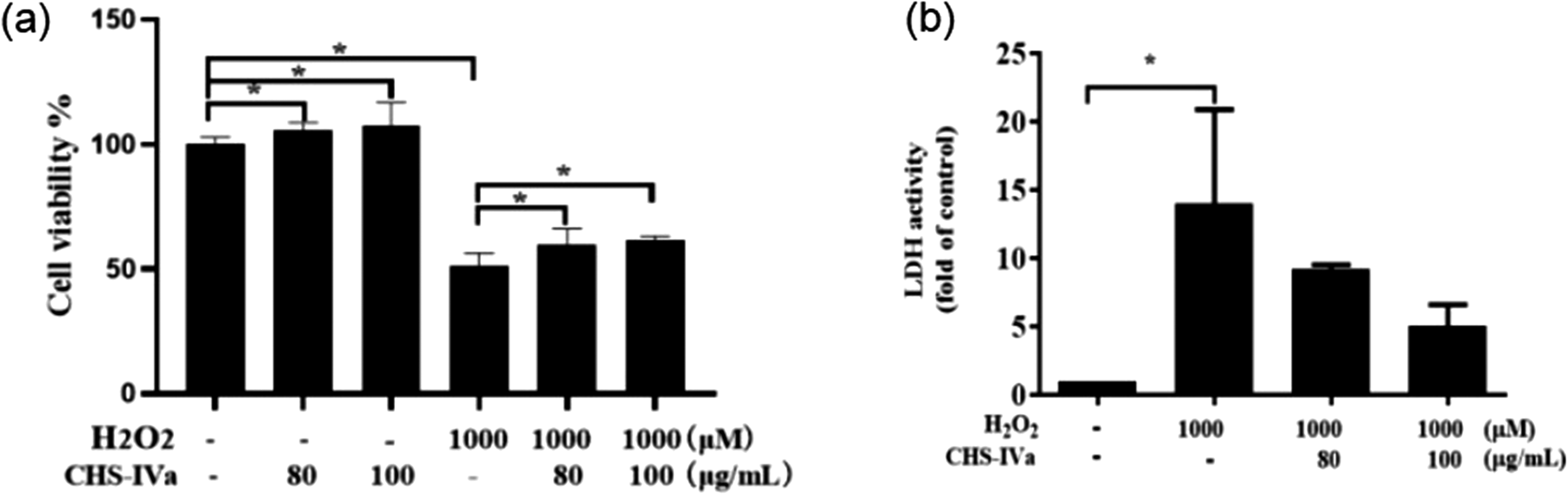
Effects of different concentrations of CHS-IVa on cell viability (a) and LDH activity (b) of IPEC-J2 cells treated with H2O2. Data are presented as mean ± SD. *P < .05 was statistically significant compared to the control group.
Regulation of CHS-IVa on the Changes of MDA Content and Antioxidant Enzyme Activity in IPEC-J2 Cells Treated With H2O2
Next, we examined the effect of different concentrations of CHS-IVa on the activity changes of antioxidant enzymes (including CAT, SOD, GSH, TrxR) in the culture supernatant of IPEC-J2 cells after the treatment of 1000 μM H2O2. Compared with the control group, the activities of GSH, SOD, CAT, and TrxR in the cultured supernatant were significantly reduced after 1000 μM H2O2 treatment for 2 hours (P < .05) (Figure 6a–6d). After adding 80 and 100 μg/mL CHS-IVa for 6 hours, the activities of GSH, SOD, CAT, and TrxR in the supernatant all showed an upward trend, and the activities of TrxR, GSH, and CAT increased significantly (P < .05). Next, changes in MDA content were detected. Compared with the control group, the content of MDA in the culture supernatant of the 1000 μM H2O2 group increased (Figure 6e). However, compared with the H2O2 single treatment group, the MDA content in the culture supernatant of cells treated with 80 and 100 μg/mL CHS-IVa showed a decreasing trend.

Effect of CHS-IVa on CAT (a), SOD (b), GSH (c), TrxR (d) activity, and MDA (e) content in the culture supernatant of IPEC-J2 cells treated with H2O2. Data are presented as mean ± SD. *P < .05 was statistically significant compared to the control group.
CHS-IVa Reduced H2O2-Induced Up-Regulation of Gene Expression of Inflammatory-Related Factors in IPEC-J2 Cells
Next, we investigated the effect of different concentrations of CHS-IVa on inflammatory-related factors in IPEC-J2 cells. Compared with the control group, the relative expression of IL-6 and IL-1α mRNA in H2O2 showed an upward trend, and the increase of IL-6 was significant (P < .05) (Figure 7a and b). After the addition of 80 and 100 μg/mL CHS-IVa, the relative expression of IL-6 mRNA showed a downward trend with a significant difference (P < .05), while the relative expression of IL-1α showed a downward trend only after the addition of 80 μg/mL CHS-IVa. After treatment with 1000 μM H2O2, the relative expression of TNF-α mRNA in the cells showed an upward trend (Figure 7c). After treatment with CHS-IVa at 80 and 100 μg/mL, the relative expression of TNF-α showed a downward trend. Similarly, compared to the control group, the relative expression of P65 and NF-κB mRNA in the cells of the H2O2 single treatment group showed an upward trend (Figure 7d and e). After adding CHS-IVa at concentrations of 80 and 100 μg/mL, the relative expression of P65 and NF-κB mRNA in the cells showed a downward trend, and 100 μg/mL CHS-IVa downregulated the expression of P65 and NF-κB mRNA more obviously.
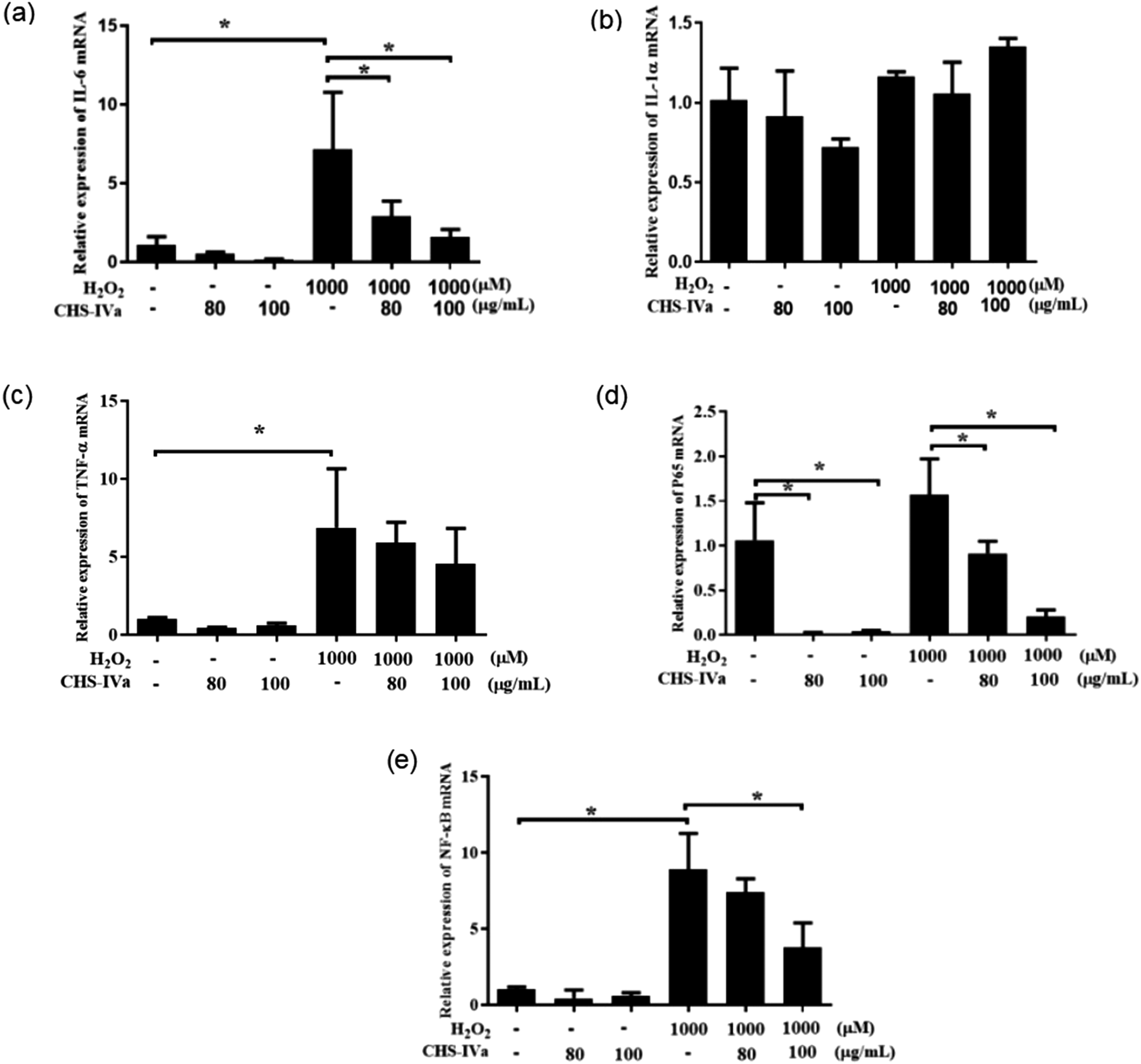
Effect of CHS-IVa on the relative expression of IL-6 (a), IL-1α (b), TNF-α (c), NF-κB (e), and P65 (d) dmRNA in IPEC-J2 cells treated with H2O2. Data are presented as mean ± SD. *P < .05 was statistically significant compared to the control group.
Discussion
As the main organ of nutrient digestion and absorption in the body, the intestine is also the largest immune defense organ in the animal's body, and is essential for maintaining the health and growth of livestock and poultry. 31 Intestinal injury caused by a variety of factors can lead to intestinal mucosal damage and dysfunction.3233-34 Oxidative stress is a key factor in gut barrier disruption in intestinal diseases. 35 Many natural herbs have a protective effect on the intestines. Post-treatment with ginsenoside Rb1 has been shown to improve the mesenteric venous albumin leakage induced by lipopolysaccharide, thereby protecting the intestine. 36 There are also studies indicating that Ginsenoside RD can prevent experimental colitis by reducing the accumulation of leukocytes and downregulating multiple pro-inflammatory cytokines. 37 As a natural active ingredient in Panax japonicus saponin, CHS-IVa has various medicinal effects such as anti-injury and anti-apoptosis. 38 The results showed that 50 to 200 μg/mL CHS-IVa could promote the growth of IPEC-J2 cells within 9 hours, while 400 μg/mL of CHS-IVa reduced cell activity. At 48 hours, 200 μg/mL of CHS-IVa showed toxic effects on cells, reducing cell viability. Then, the cell viability of cells treated with different concentrations of CHS-IVa was subsequently detected for 3 and 6 hours, and it was found that the cell viability also increased. In Anmin Shao's study, concentrations of CHS-IVa at 40 and 50 μM significantly reduced the viability of primary neurons within 6 hours, while CHS-IVa at 10, 20, and 30 μM had no significant effect on cell viability. 39 It was shown that high concentrations of CHS-IVa can inhibit cell activity. We also showed that CHS-IVa has a repairing effect on intestinal epithelial cell injury through scratch experiments. Studies have shown that CHS-IVa can promote cell repair by inhibiting the migration of ovarian cancer cells. 40
Many studies have shown that CHS-IVa has antioxidant effects. 12 Oxidative stress refers to the phenomenon that interferes with the oxidative balance of the intracellular redox system due to the excessive production of ROS in the cell under the influence of external harmful factors. 41 There are many natural Chinese herbs that have antioxidant effects.42,43 They play an important role in antioxidants by changing the activities of SOD, CAT, GSH, and other antioxidant enzymes to scavenge reactive oxygen radicals produced in the process of cellular oxidative metabolism. 44 The results of this study showed that the decrease in SOD activity, TrxR activity, CAT activity, and GSH activity induced by H2O2 could be alleviated after CHS-IVa treatment at 80 and 100 μg/mL, which indicating that intracellular reactive oxygen radicals were controlled under CHS-IVa treatment. Wang's findings also show that CHS-IVa at 10, 20, and 40 μM increased SOD and GSH activity in pancreatic β-cell. 45 At the same time, our study also showed a decrease in LDH levels in the cell supernatant. Studies have shown that CHS-IVa can reduce LDH release.45,46 In conclusion, we know that CHS-IVa has an antioxidant effect on IPEC that can relieve or inhibit intestinal damage caused by oxidative stress and maintain the normal life activities of the body. It is speculated that CHS-IVa can be used for further research on potential antioxidants.
The body's oxidative stress response pathway is closely related to inflammation and cytokine secretion. As pro-inflammatory factors, IL-1α and IL-6 are involved in the inflammatory and immune response of cells.47,48 Studies have shown that inhibition of the NF-κB pathway can exert anti-inflammatory effects. 49 Yuan's research showed that CHS-IVa can reduce the expression of NF-κB and IL-1β in mouse epididymal adipose tissue and macrophages induced by high fat. 50 Meanwhile, it was reported that CHS-IVa could suppress the levels of IL-1β, IL-6, and IL-10 in mouse macrophages by lipopolysaccharide. 51 Our study found that CHS-IVa attenuated the H2O2-induced increase in the relative expression of IL-6, P65, and NF-κB in IPEC. It shows that CHS-IVa has a certain protective effect on intestinal inflammation. NF-κB is the key to inflammation and oxidative stress. In this study, we found that CHS-IVa can alleviate the upregulation of NF-kB caused by hydrogen peroxide, but we did not conduct in-depth experiments on regulating molecular mechanisms related to oxidative stress and inflammation, which requires further research in the future.
Conclusions
The concentration of 80 and 100 μg/mL CHS-IVa can enhance the viability of IPEC-J2 cells, promote the repair of cell scribing damage, and alleviate the decline of cell activity caused by H2O2. It can also slow down the decrease of SOD, TrxR, CAT, and GSH activity, as well as the increase of expression of inflammatory cytokines IL-6, P65, and NF-κB caused by H2O2. These findings provide fundamental research for CHS-IVa as an effective anti-injury drug.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Hunan Province, China (2022JJ30022, 2022JJ30293) and the Hunan Province Technology Breakthrough Project of 2021 for the open competition mechanism to select the best candidates (2021NK1030).
Statement of Human and Animal Rights
Not applicable.
Statement of Informed Consent
Not applicable.
