Abstract
Background/Objective
Cadmium (Cd) is an extremely toxic heavy metal and environmental toxicant, which leads to renal damage by causing oxidative stress. Rhamnazin (RAM) is a novel flavonoid, which possesses multiple pharmacological activities. Therefore, the present study was planned to determine the nephroprotective effects of RAM against Cd-prompted renal toxicity in rats.
Methodology
In this investigation, 24 male albino rats were distributed into four equal groups, i.e., control, CdCl2-intoxicated group (2 mgkg−1), CdCl2 + RAM-treated group (2 mgkg−1 + 20 mgkg−1), and RAM-only treated group (20 mgkg−1). After oral dose administration for 30 days, the alterations in the Nrf2/Keap1 pathway, Cd concentration, antioxidant profile, oxidative stress markers, renal function markers, inflammatory markers, apoptotic markers, and kidney histology were analyzed.
Result
The results of the current study revealed that Cd exposure significantly (P < .05) increased Keap1 expression, while decreasing the expressions of Nrf2 and antioxidant genes and level of total antioxidant status (TAS). Additionally, it decreased the activities of antioxidant enzymes. Whereas, the levels of malondialdehyde (MDA), reactive oxygen species (ROS), and total oxidant status (TOS) were increased. Cd exposure increased Cd concentration in tissues. Additionally, it increased the levels of renal function markers, such as urea, urobilinogen, urinary proteins, creatinine, kidney injury molecule-1 (KIM-1), and neutrophil gelatinase-associated lipocalin (NGAL). On the contrary, Cd exposure reduced albumin and creatinine clearance. Furthermore, it increased the levels of inflammatory markers in the kidneys. Besides, Cd significantly increased the levels of Bax, caspase-3, and caspase-9, whereas lessened Bcl-2 level. Moreover, adverse histoarchitectural changes were observed following the Cd exposure. However, treatment with RAM significantly restored all the damage in the kidneys of the rats.
Conclusion
Our findings highlight the potential of RAM as a valuable therapeutic candidate against heavy metal-induced kidney damage.
Keywords
Introduction
Cadmium (Cd) is listed by the World Health Organization (WHO) as one of the top 10 hazardous substances posing a risk to public health. 1 Anthropogenic activities during the past 120 years have immensely increased Cd concentration in the environment.2,3 Cd is found in air, water, food, and tobacco leaves. Therefore, it enters into the bodies of living organisms via inhalation and ingestion. 4 Mining, smelting as well as industries related to ceramics, electroplating, pigments, and Cd-nickel batteries are major sources of occupational Cd exposure. 5 Additionally, Cd-containing phosphate fertilizersare reincorporating Cd in crops. 6 As microorganisms cannot biodegrade Cd, the heavy metal enters the bodies of living organisms through the food chain, causing health risks. 7 According to the previous literature, people residing in areas with Cd-contaminated crops are reported to retain high Cd levels in their blood and urine. 8 After Cd absorption in the body, it is transported to the tissues by binding with erythrocytes and proteins. 9
kidneys are integral organ for eliminating waste materials of body, managing body fluids, and regulating the electrolyte balance. 10 Heavy metals are potent toxins that disrupt vital biological processes and cause organ damage. 11 Hernández-Cruz et al 12 stated that the kidney is the main target site of heavy metal exposure; therefore, renal tissues are vulnerable to damage even with slight Cd exposure. Furthermore, Cd lacks the ability to be properly metabolized and filtered in the proximal tubules. 13 Therefore, it accumulates in the tissues. Cd has a long half-life ranging from 10 to 35 years in the human body, which further increases its tendency to accumulate in the kidneys and cause renal damage. 14 Cd exposure induces renal toxicity by multiple cellular and pathophysiological events. It elicits damage in biological systems by affecting the antioxidant cell defense and escalating the level of lipid peroxidation. 15 Other toxicity-causing mechanisms include the interaction of Cd with biomolecules as well as its attachment to sulfur, nitrogen, and oxygen ligands, which adversely affect the proteins and enzymes. 16 Besides, it is indicated that Cd reaches the liver and attaches with the metallothionein (MT) to produce Cd-MT conjugates. These conjugates are shifted to the kidneys via blood circulation, 17 filtered by the glomerulus, and again upheld by the epithelial cells of proximal tubules. 18 Within the proximal tubules, Cd-MT conjugates are degraded and Cd ions are released into the kidneys, causing nephrotoxicity. 19 Previous studies demonstrated that Cd exposure induced histopathological damages, such as glomerular necrosis, interstitial fibrosis, tubular degeneration, edema, and hemorrhage in the kidneys of rats.20,21
Flavonoids are secondary metabolites that are extensively used as traditional medicines due to their ROS-scavenging and metal-chelating properties.22,23 Rhamnazin (RAM) is a novel flavonoid, which is naturally found in medicinal plants, such as ginkgo, willows, and sea-buckthorn. 24 It is reported to exert antioxidant, anti-inflammatory, anti-angiogenic, anti-cancerous, anti-bacterial, anti-allergic, anti-viral, and neuroprotective effects.25,26 In a previous study, RAM was shown to reduce the oxidative stress and inflammatory burden in lipopolysaccharide-induced lung injury in rats. 27 This finding indicates that RAM has excellent antioxidant and anti-inflammatory properties. To the best of our knowledge, no studies have reported its effects on renal toxicity to date. Therefore, the current study was conducted to analyze the role of RAM against Cd-prompted renal toxicity by evaluating its role on the Nrf2/Keap1 pathway, biochemical parameters, Cd concentration, renal function markers, inflammatory markers, apoptotic proteins as and histopathological profile of male albino rats.
Results
Effect of RAM Against Cd Accumulation in Renal Tissues
Assessment of toxin accumulation in tissues serves as an indicator of toxicity. In the current study, high Cd levels were observed in the Cd-treated group of rats as compared to the control. However, Cd level was significantly decreased (P < .05) in the co-treatment (Cd + RAM) group when compared to the rats of the Cd-induced group. Nonetheless, no significant difference was observed between the RAM-only treated group and the control group (Table 1).
Effect of RAM on Accumulation of Cd in Renal Tissues.

Data is shown as mean ± SEM (6 rats/group). Results with various superscripts in columns are dissimilar from each other (P < .05).
Effect of Cd and RAM on Nrf2/Keap1 Signaling Pathway
Role of RAM against Cd-induced toxicity was determined on Nrf2/Keap1 signaling pathway by analyzing the expressions of Nrf2, Keap1, and antioxidant genes, including catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione reductase (GSR), glutathione-S-transferase (GST) and heme oxygenase-1 (HO-1). Cd exposure significantly reduced (P < .05) the expressions of Nrf2 and antioxidant genes, while increasing Keap1 expression as compared to the control group. On the other hand, co-treatment of RAM and Cd significantly restored (P < .05) the expressions of the aforementioned genes as compared with the Cd-induced group. Besides, no significant differences were found between RAM-only treated group and the control group (Figure 1).
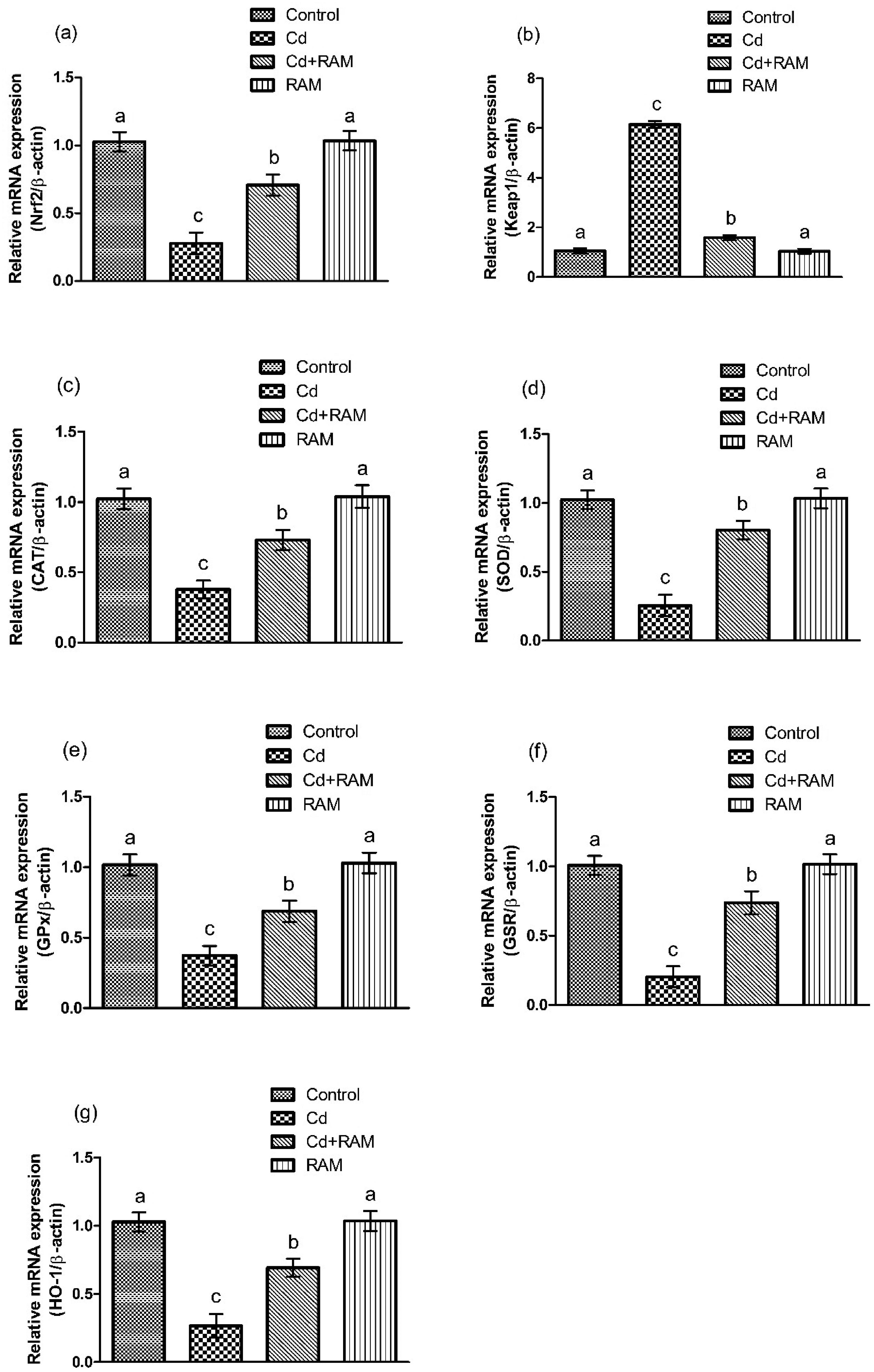
Expressions of (a) Nrf2, (b) Keap1, (c) CAT, (d) SOD, (e) GPx, (f) GSR, and (g) HO-1. Bars show mean ± SEM values (n = 6). Different superscripts show significant differences among various groups (P < .05).
Effect of Cd and RAM on Antioxidant Profile
Cd exposure significantly (P < .05) reduced the activities of antioxidant enzymes and the level of glutathione (GSH) in Cd-induced rats as compared to the control group. On the other hand, RAM treatment to the rats significantly (P < .05) increased the activities of antioxidant enzymes and GSH level in the co-treatment group as compared with Cd-induced group. Moreover, no significant differences were observed between the RAM-only treated group and the control group (Table 2).
Effect of Cd and RAM on Antioxidant Profile.

Data is shown as mean ± SEM (6 rats/group). Results with various superscripts in columns are dissimilar from each other (P < .05).
Effect of Cd and RAM on MDA, ROS, TOS, and TAS
Cd administration significantly increased (P < .05) the levels of MDA, ROS, and TOS, while TAS level was decreased in Cd-induced rats compared with control. Whereas, RAM significantly reduced (P < .05) the levels of MDA, ROS, and TOS, while increasing TAS level in the co-administered (Cd + RAM) rats as compared with the Cd-administered rats. Nevertheless, no significant differences were found between the RAM-only treated group and the control group (Table 3).
Effect of Cd and RAM on Oxidative Stress.

Data is shown as mean ± SEM (6 rats/group). Results with various superscripts in columns are dissimilar from each other (P < .05).
Effect of Cd and RAM on Renal Function Markers
Cd exposure significantly (P < .05) increased the levels of urea, urobilinogen, creatinine, urinary proteins, KIM-1, and NGAL, while significantly (P < .05) decreasing the levels of albumin and creatinine clearance in Cd-induced rats as compared with the control group. On the other hand, RAM significantly reversed (P < .05) all of the imbalance in the levels of renal function markers in the co-administered (Cd + RAM) group as compared to the Cd-induced group. Nonetheless, no significant differences were observed between RAM-only treated group and the control group (Tables 4 and 5).
Effect of Cd and RAM on Renal Marker Profile.

Data is shown as mean ± SEM (6 rats/group). Results with various superscripts in columns are dissimilar from each other (P < .05).
Effect of Cd and RAM on KIM-1 and NGAL.

Data is shown as mean ± SEM (6 rats/group). Results with various superscripts in columns are dissimilar from each other (P < .05).
Effect of Cd and RAM on Inflammatory Markers
Cd administration significantly increased (P < .05) the levels of inflammatory markers, such as nuclear factor-kappa B (NF-κB), tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), interleukin-6 IL-6, and cyclooxygenase-2 (COX-2) in the rats of Cd-induced group compared to the control group. On the other hand, RAM significantly (P < .05) reduced the levels of these inflammatory markers in the co-administered (Cd + RAM) rats compared to the Cd-induced rats. Furthermore, no significant differences were found between RAM-only treated group and the control group (Table 6).
Effect of Cd and RAM on Inflammatory Profile.

Data is shown as mean ± SEM (6 rats/group). Results with various superscripts in columns are dissimilar from each other (P < .05).
Effect of Cd and RAM on Apoptosis
Cd exposure significantly increased (P < .05) the levels of Bax, caspase-3, and caspase-9, whereas decreased Bcl-2 level in Cd-induced rats compared to the rats of control group (Table 7). However, RAM treatment significantly (P < .05) restored the imbalance in the levels of these apoptotic markers to the normal in the co-administered group (Cd + RAM) compared with Cd-induced group. Nevertheless, no significant differences were observed between RAM-only treated group and the control group.
Effect of Cd and RAM on Apoptotic Profile.

Data is shown as mean ± SEM (6 rats/group). Results with various superscripts in columns are dissimilar from each other (P < .05).
Effect of Cd and RAM on Kidney Histopathology
Cd exposure caused structural injuries in the renal tissues, including degeneration of proximal tubules, glomerular atrophy, obstruction of capillaries, and Bowman's capsule damage in the Cd-treated rats compared to the control rats. On the other hand, RAM supplementation significantly recovered the aforementioned morphological changes and improved the structure of the glomerulus, proximal tubules, and Bowman's capsule as well as decreased the blockage in capillaries in the co-administered rats compared with Cd-exposed rats. However, no significant differences were observed between RAM-only treated rats and control rats (Figure 2).
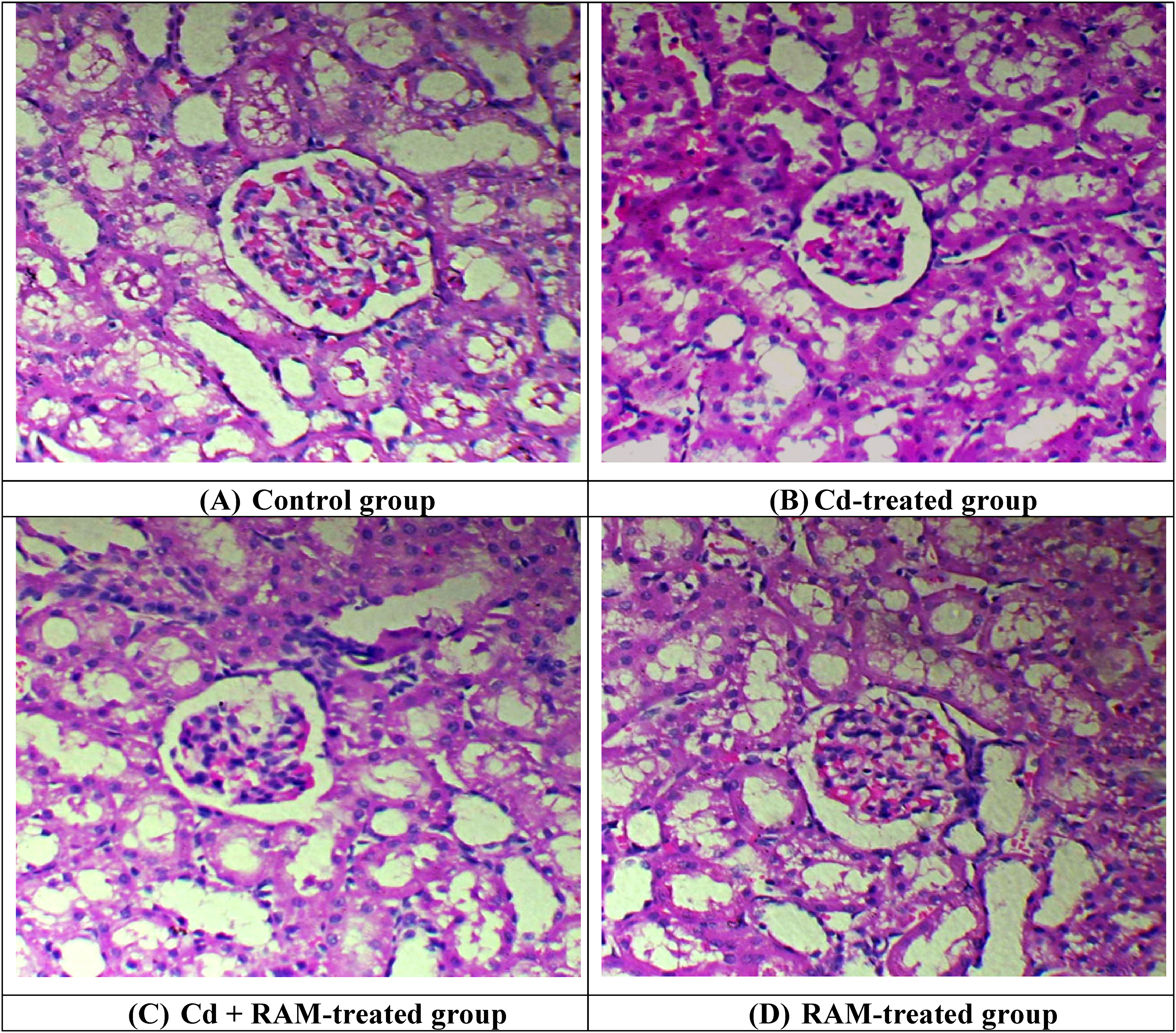
Effect of Cd and RAM on histopathology of kidney tissues. (a) Renal tissues of control group displayed healthy and normal structural integrity having a proper arrangement of tubules in kidneys. (b) Cd exposure resulted in damage in morphology with congested central and portal veins, extra-dilated sinusoids, and disorganized cellular state. (c) RAM supplementation led to the reduction in congestion of blood vessels and dilation of sinusoids in co-treated group (CD + RAM) (d) RAM treatment group showed normal morphology similar to the control group. Cd: cadmium, RAM: rhamnazin. (A) Control group. (B) Cd-treated group. (C) Cd + RAM-treated group. (D) RAM-treated group.
Discussion
Cd intoxication induces toxicity primarily by escalating lipid peroxidation and causing oxidative stress. 28 However, flavonoid supplementation plays a protective role in the mitigation of oxidative stress generated after toxin exposure. 29 Due to increasing environmental pollution, the demand for antioxidant-enriched phytochemicals is increasing globally due to their potential health benefits. 30 RAM is a bioactive flavonoid, which reportedly holds the capacity to reduce oxidative stress owing to its free radical-scavenging efficacy. 26 Therefore, the current investigation was aimed to assess the mitigative effect of RAM against Cd-induced renal toxicity by analyzing the alterations in the Nrf2/Keap1 pathway, Cd concentration, antioxidant profile, oxidative stress, renal function profile, inflammatory markers, apoptotic proteins, and kidney histology.
In the current study, Cd level was observed to be significantly (P < .05) increased after the administration of Cd. The observed significant increase in Cd levels reflects the uptake and accumulation of this toxic metal in the renal tissues. The deposition of Cd in the body contributes to various pathophysiological disorders, notably kidney dysfunction. 31 It is noticed that the use of phytochemicals as chelating agents has become increasingly important due to the continuous increase of Cd in the environment as well as in living organisms. Chelating agents have the tendency to counter heavy metal intoxication by binding with the metal ions, thereby shielding the target sites from the toxic ions by eliminating them from the body. 32 In the current study, RAM treatment decreased the accumulation of Cd from the Cd-exposed renal tissues. It is well-documented that hydroxyl ions (OH−) have the capability to act as metal chelators by conjugating with the Cd2+ ions. 33 Xiao et al 34 reported the occurrence of three hydroxyl groups in the structure of RAM. It was inferred that these functional groups interact with Cd ions, sequester them from renal tissues, and facilitate their excretion via urine, making RAM an effective chelating agent for heavy metals.
In the current research, Cd exposure increased Keap1 expression, while reducing the expressions of Nrf2 and antioxidant genes (SOD, CAT, GPx, GSR, GST, and HO-1) in the renal tissues. Nrf2 is regarded as the transcription factor and multi-organ protector, which plays a significant role in the cellular homeostasis by transcription of antioxidant genes and reduction of oxidative stress. 35 Under normal homeostatic conditions in the cells, Nrf2 is bound to Keap1 in the cytoplasm. Keap1 regulates Nrf2 levels by promoting its ubiquitination and subsequent degradation through the ubiquitin-proteasome pathway. 36 However, during excessive oxidative stress, the expression of Keap1 is increased, while bringing down the expressions of Nrf2 and antioxidant genes. 37 According to Aranda-Rivera et al, 38 disturbance in Nrf2 signaling pathway was observed as a potential mechanism behind renal toxicity. Therefore, it was deduced that Cd exposure escalated the Keap1 expression, while decreasing the expression of Nrf2, which in turn, reduced the expressions of antioxidant genes and caused nephrotoxicity. However, RAM treatment upregulated the expressions of Nrf2 and antioxidant genes, while decreasing the Keap1 expression in the rats. It was inferred that the antioxidant role of RAM is due to its capacity to mitigate oxidative stress by enhancing Nrf2 expression, promoting its nuclear translocation, and upregulating the expressions of antioxidant genes, thereby improving the cellular antioxidant defense. The above-mentioned phenomena reveal the defensive role of RAM against Cd toxicity by mediating the Nrf2/Keap1 signaling pathway.
Cd exposure significantly reduced the activities of antioxidant enzymes, including SOD, CAT, GPx, GSR, GST, and HO-1 as well as the level of GSH in the rats. It is well understood that cells have their own enzymatic and non-enzymatic antioxidant defense system, which assists them in countering the adverse effects of free radicals. 39 Cd is reported to cause toxicity by generating ROS, including HO− and O2−. 40 These hydroxyl radicals penetrate the cell membrane and react with DNA in the nucleus, ultimately leading to tissue and organ toxicity. 41 Antioxidant (enzymatic and non-enzymatic) defense system shields the organisms from the adverse effects of free radicals. 42 CAT is an antioxidant enzyme that cleaves the extremely toxic, H2O2, into H2O and O2. 43 SOD is a chain-breaking antioxidant, which is engaged in the conversion of superoxide ions into H2O2 and O2. 44 GSR acts as a reducer to convert oxidized glutathione (GSSG) into GSH. 45 GSH is a non-enzymatic antioxidant that plays a crucial role in maintaining cell integrity and metabolic activities. 46 Moreover, it provides the cellular defense against Cd toxicity. 47 GST is an essential cofactor that conjugates GSH with xenobiotics to ensure their elimination from the body. 48 HO-1 is another vital enzyme that is a crucial member of the catabolic pathway of heme metabolism. Additionally, it shields the cells from oxidative stress. 49 However, Cd exposure retards the activities of antioxidant enzymes due to the direct interaction of Cd with SH groups at enzyme active sites, proteins, and nucleic acids. 50 Therefore, it is demonstrated that inhibition of free radical-scavenging due to reduction in activities of cellular antioxidant enzymes and ROS elevation leads to oxidative stress and causes renal toxicity.51,52
In the current research, Cd administration elevated the levels of MDA, ROS, and TOS, whereas TAS level was reduced. The increase in lipid peroxidation level was evident by the increased level of MDA. Several Cd-induced toxic effects in the body are the result of increased lipid peroxidation, which is attributed to the reduction in activities of antioxidant enzymes as well as the elevation of ROS. 53 TOS is an indicator of the overall oxidation status, 54 whereas TAS shows the total antioxidant status of the body. 55 Decrease in the activities of antioxidant enzymes was also reflected by the reduction in TAS. While the oxidative stress was evident by the increase in TOS. Our findings are in line with the previous investigations, in which Cd intoxication reduced the activities of antioxidant enzymes, while increasing the level of ROS.56,57 A previous study reported that the therapeutic efficacy of polyphenols is majorly attributed to the multiple hydroxyl moieties in their configuration. 58 Therefore, it was deduced that RAM, owing to its polyphenolic nature, potentially increased the activities of antioxidant enzymes and the level of TAS, while reducing the level of lipid peroxidation, ROS, and TOS in the kidney tissues.
Cd exposure significantly increased the levels of urea, urobilinogen, creatinine, urinary proteins, KIM-1, and NGAL, while decreasing the levels of albumin and creatinine clearance. Estimating the fluctuation in renal function markers levels reveals the homeostatic state of kidneys. 59 Heavy metal exposure disturbs the normal state of kidneys by increasing the level of serum urea and creatinine levels. 60 Previous investigations reported that Cd possesses the capability to alter the levels of kidney function markers.61,62 It suppresses the assimilation of amino acids into proteins, which ultimately increases the level of urea in renal tissues. 50 Additionally, it was stated that the increase in the level of albumin probably takes place due to the impairment of proximal tubules of kidneys due to Cd exposure, which averts the reabsorption of glomerular-filtered albumin. 63 Whereas, poor creatinine clearance due to Cd toxicity could be the possible reason behind the decrease in glomerular filtration rate. 64
KIM-1 and NGAL are among the most promising markers of acute kidney injury. 65 Increase in the levels of both glycoproteins helps in the early diagnosis of nephrotoxicity. 66 KIM-1 is a transmembrane protein having a mucin and immunoglobulin domain, which is only detected during renal damage. 67 It is present in the proximal-tubular cells of kidneys, whose apical membrane is released in the lumen. It cleaves and decomposes into soluble fragments, which can be easily discharged into urine. Therefore, it specifically acts as a urinary biomarker. 68 NGAL is the second emerging renal biomarker belonging to the lipocalin superfamily. This glycoprotein is produced by epithelial and neutrophil cells, released after glomerular filtration, and completely reabsorbed in normal tubular tissues. 69 Nevertheless, NGAL is not reabsorbed in the damaged tubules due to renal injury; therefore, its elevated level is expressed in the urine. 70 However, RAM supplementation brought down the levels of KIM-1 and NGAL in the rats. Summing up, RAM treatment significantly reduced the levels of urea, urobilinogen, creatinine, urinary proteins, KIM-1, and NGAL, while increasing the levels of albumin and creatinine clearance, which revealed the mitigative effect of RAM against renal injuries.
Cd administration remarkably increased the levels of inflammatory biomarkers (NF-kΒ, TNF-α, IL-1β, IL-6 and COX-2) in the renal tissues of rats. Oxidative stress is considered to trigger various inflammatory events in tissues. 71 NF-κB activation is an initial but crucial step for the stimulation of various pro-inflammatory mediators that cause inflammatory burden in tissues. 72 Activated NF-κB is shifted into the nucleus to instigate the generation and liberation of inflammatory cytokines, namely TNF-α, IL-1β, and IL-6. 73 Previous investigations have revealed that the high level of TNF-α contributes to renal dysfunction by vasoconstriction and generation of cytotoxicity, which activates the inflammatory cycle, ultimately causing kidney damage. 74 IL-6 is another inflammatory biomarker that primarily activates inflammatory responses. 75 COX-2 is an inducible enzyme and inflammatory marker that contributes to inflammatory stress but is expressed only after receiving inflammatory stimuli from other molecules. 76 It is demonstrated that Cd activates NF-κB and stimulate a series of inflammatory processes, including the subsequent increase in the levels of IL-1ß, IL-6, and TNF-α. 28 High levels of interleukins and TNF-α further contribute to the inflammatory stress and toxicity in the renal tissues. 74 However, RAM supplementation significantly (P < .05) reduced the levels of these inflammatory markers. Nrf2 activation plays a key role in inhibiting NF-κB activation, which ultimately decreases the production of inflammatory cytokines. 36 In light of the above-mentioned finding, it was deduced that RAM treatment activated Nrf2, which not only increased the expressions of antioxidant genes but also reduced the levels of inflammatory markers.
Administration of Cd raised the levels of apoptotic proteins, Bax, caspase-9, and caspase-3, while reducing the level of anti-apoptotic protein, Bcl-2. Apoptotic damage is regarded as a causative factor underlying renal toxicity. 77 Bax and Bcl-2 are associated with Bcl-2 family which regulates the mitochondria-dependent apoptotic pathway. 78 Bax attachment to the membrane of mitochondria leads to the eviction of apoptotic proteins, second mitochondria-derived activator of caspase (SMAC) and Omi, in the cytosol which mediate the liberation of cytochrome C. Both of these apoptotic proteins trigger the apoptosis initiator (procaspase-9) which is evidenced to activate caspase-9 and caspase-3, thereby cleaving several protein substrates and causing apoptosis. 79 Another study indicated that Cd exposure increases membrane permeability of mitochondria, causing the discharge of cytochrome C that stimulates caspases and culminates in apoptotic damage. 80 However, RAM treatment reversed the levels of these apoptotic proteins in the rats. Nrf2 possesses the tendency to maintain the integrity of mitochondria during oxidative injury, 81 which inhibits the liberation of cytochrome C and ultimately suppresses apoptosis. Additionally, increase in the level of Bcl-2 and reduction in Bax and caspases following RAM supplementation led to the mitigation of Cd-induced apoptosis, thereby revealing the anti-apoptotic potential of RAM.
The histopathological assessment revealed that Cd exposure caused histological damage, including degeneration of proximal tubules, glomerular atrophy, obstruction of capillaries, and Bowman's capsule damage in the renal tissues (Figure 1). Cd exposure has tendency to reduce the activities of antioxidant enzymes 82 and increase the levels of free radicals and lipid peroxidation. Both these phenomena cause oxidative stress and disrupt the biomolecules (DNA, lipids, and proteins), which consequently leads to renal toxicity. 83 Prozialeck et al 67 stated that Cd-induced degeneration in renal tissues is linked to the increased level of KIM-1. However, RAM administration remarkably decreased the Cd-prompted histological damage in the kidneys of rats. Therefore, it is inferred that RAM has the tendency to show alleviative potential in restoring the structure of renal tissues due to its antioxidant efficacy. However, the limitation of the present study is that a single dose of RAM was used, therefore, a dose dependent study of RAM is recommended in future. Besides, the present investigation emphasizes the need for clinical trials to determine its safety, bioavailability, and therapeutic efficacy in humans, especially in populations with high Cd exposure.
Conclusion
Collectively, it was revealed that Cd exposure increased Cd concentration in tissues and adversely affected Nrf2/Keap1 pathway, biochemical profile, renal function markers, inflammatory markers, apoptotic proteins, and histopathological state of renal tissues. Nonetheless, RAM treatment reversed all renal damages caused by Cd. Summing up, the current investigation unveiled the nephroprotective efficacy of RAM due to its effects on Nrf2/Keap1 signaling pathway, depicting its strong antioxidant potential. Due to its antioxidant potential, RAM mitigated oxidative stress as well as prevented the consequent instigation of inflammatory burden, apoptotic damage, and renal toxicity. Moreover, the decrease in the Cd level in tissues suggested that RAM could act as a potent metal-chelating agent that may prevent heavy metal toxicity.
Materials and Methods
Chemicals and Reagents
Cd and RAM were bought from the Sigma-Aldrich (Germany). The remaining reagents used in the experiments were of analytical grade and also purchased from Sigma-Aldrich (Germany).
Animals
In the current trial, 24 male albino rats (200 ± 20 g) of approximately 7 weeks were categorized into four groups (six rats/group). Rats were housed in Animal Facility of University of Agriculture, Faisalabad (UAF) at 23 ± 2 °C and a 12 h light/dark cycle was maintained. Moreover, tap water and animal food were provided to animals. Before starting the experiment, rats were subjected to laboratory conditions for 7 days to acclimatize them to the laboratory environment. The experiment was conducted in accordance with the ARRIVE guidelines to ensure rigorous and ethical study design and animal welfare standards.
Experimental Design
Rats were treated with Cd and RAM intraperitoneally for 30 days. Rats were randomly assigned to each treatment group to minimize selection bias, and the individual conducting the experiments was blinded to the group allocations to ensure objective data collection. Group 1, ie, Control, was orally administered with normal saline. Cd was provided to the rats in the form of CdCl2. Group 2 was orally treated with 2 mgkg−1 of CdCl2 dissolved in saline. Group 3 was orally co-treated with 2 mgkg−1 of CdCl2 and 20 mgkg−1 of RAM. Both compounds were dissolved in saline. Group 4 was orally provided with 20 mgkg−1 of RAM dissolved in saline. The dose of Cd (2 mgkg−1) was selected based on our former investigation. 28 While the dose of RAM (20 mgkg−1) was chosen according to the study of Wu et al 27 Urine samples were collected from fasted rats in metabolic cages a day before decapitation. Samples were preserved in containers at −80 °C for urinary markers analysis. After the 30-day trial period, rats were anesthetized by diethyl ether and decapitated. Blood was stored in test tubes and serum was obtained from the blood, which was preserved at −80 °C to analyze biochemical markers. Kidneys were removed and fixed in the 10% formalin solution for histomorphological assessment.
Assessment of Cd Level
Accumulated Cd level in renal tissues was evaluated by the spectrophotometric method using a chromogenic reagent, o-diazoaminophenylarsonic acid azobenzene. 84
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
The renal tissue samples were homogenized and total mRNA was isolated from the homogenized mixtures using QIAzol Lysis Reagent (Qiagen, Cat: 79306, Germany). RNA purity was assessed using 260/280 ratio with the NanoDrop 2000c (Labtech International, UK). Complementary DNA (cDNA) was then synthesized from the total mRNA using Prime Script TM RT Reagent Kit (Takara, Japan). qRT-PCR assessments were conducted to assess the variations in mRNA expressions using SYBR premix Ex Taq 2 kit (Takara). Relative gene expressions were determined using 2−ΔΔCT method. β-actin was kept as standard. 85 Primer sequences are shown in Table 8. 86
Primer Sequence for qRT-PCR.

Biochemical Analysis
CAT activity was determined by following the protocol of Chance and Maehly. 87 SOD activity was estimated using the protocol given by Spitz and Oberley. 88 GSH content was estimated by the methodology presented by Griffith. 89 GPx activity was measured by a protocol explained by Lawrence and Burk. 90 GSR activity was assessed by the procedure described by Carlberg and Mannervick. 91 GST activity was determined via the method of Habig et al 92 The HO-1 activity was determined by evaluating bilirubin formation according to the technique described by Magee et al 93 MDA level was measured to take an estimate of lipid peroxidation, using a method stated by Hochstein and Utley. 94 The level of ROS was gauged according to the technique of Hayashi et al 95 Levels of TAS and TOS were measured by Rel Assay Diagnosing kit using the procedures stated by Erel 54 and Erel, 55 respectively.
Assessment of Renal Function Markers
Assessment of kidney function was performed by evaluating the levels of renal function markers. The levels of urea (Cat No. ab83362; Abcam, USA), urinary protein (Cat No. 9040; Chondrex, USA), albumin (Cat No. ab108789; Abcam, USA), urobilinogen (Cat No. EBT-BC089; Biozol, Germany), creatinine (Cat No. ab65340; Abcam, USA) and creatinine clearance (Cat No. ab65340; Abcam, USA) were evaluated by standard diagnostic kits. The levels of urinary KIM-1 and serum NGAL were evaluated using ELISA kits (Cusabio, Huston, USA). Moreover, the company's guide given with the kits was followed.
Assessment of Inflammatory Markers
The level of inflammatory markers, NF-κB (CSB-E13148r), IL-1β (CSB-E08055r), IL-6 (CSB-E04640r), TNF-α (CSB-E11987r) COX-2 (CSB-E13399r) in renal tissues of rats were assessed by the commercially available ELISA kits (Cusabio, Huston, USA) and the manufacturer's guidelines were followed.
Analysis of Apoptotic Markers
The assessment of Bcl-2 (Cat No. EKU02675-96 T; Biomatik Corporation, Canada), Bax (Cat No. EKU02681-96 T; Biomatik Corporation, Canada), caspase-9 (E-EL-R0163; Elabscience, USA) and caspase-3 (Cat No. EKF57872-96 T; Biomatik Corporation, Canada) level was carried out using ELISA kits.
Histopathological Assessment
Histopathology was performed to analyze the role of RAM against Cd-exposed renal tissues of rats. Tissues were fixed in the 10% formaldehyde. After that, tissues were dehydrated in the increasing grades of alcohol (80%-100%), washed with xylene, and embedded in paraffin wax blocks for microtomy. Thin sections (4 μm) were sliced and stained by hematoxylin/eosin for slide preparation. In the end, slides were carefully observed under a light microscope at 400x and microphotographs were captured.
Statistical Analysis
Data was shown as mean ± SEM. The One-way analysis of variance (ANOVA) was used to estimate the statistical variations. Later one, Tukey's test was applied and the GraphPad Prism 5 was used. P < .05 was considered as the level of significance.
Footnotes
Acknowledgment
The authors extend their appreciation to Taif University, Saudi Arabia, for Supporting this work through project number (TU-DSPP-2024-219).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The current experiment was performed in strict compliance with the guidelines of the Ethical Board of University of Agriculture, Faisalabad, with approval number DGS/1841-44.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Taif University, Saudi Arabia, Project No. (TU-DSPP-2024-219).
Statement of Human and Animal Rights
All the experimental procedures were conducted as per the Institutional Guidelines for the Care and Use of Animals of University of Agriculture, Faisalabad, Pakistan.
Statement of Informed Consent
There are no human subjects in this article. Therefore, informed consent is not applicable.
