Abstract
Objectives
The objective of this research was to investigate the potential anti-inflammatory properties of the Cucurbita pepo fruit. Given the adverse effects associated with long-term use or excessive doses of conventional anti-inflammatory medications, exploring herbal therapies as alternatives has become increasingly important.
Methods
Various preliminary tests and antioxidant assays, both enzymatic and non-enzymatic, were conducted on the C. pepo fruit. Bioactive substances present in the fruit, including alkaloids, carbohydrates, flavonoids, tannins, amino acids, triterpenoids, and saponin glycosides, were identified through early analysis. Different extraction solvents, such as chloroform, methanol, and water, were utilized to extract samples. The methanolic extract was subjected to further testing for its in vitro anti-inflammatory characteristics. This involved evaluating their ability to stabilize RBC membranes, inhibit protein denaturation, and suppress proteinase activity using two concentrations (150 µg/ml and 300 µg/ml) of the extract.
Results
The methanolic extract of the C. pepo fruit exhibited strong free radical scavenging activity in both DPPH and H2O2 assays. Moreover, it demonstrated the dose-dependent stabilization of RBC membranes, resulting in reduced hemolysis, protein denaturation, and proteinase activity. These findings suggest potent anti-inflammatory effects of the methanolic extract.
Conclusion
This research study has found that the C. pepo fruit methanolic extract significantly reduced inflammation. This demonstrates the promise of natural chemicals as less risky substitutes for traditional anti-inflammatory medications. Potentially safe and efficient treatments for inflammatory illnesses could be found in herbal remedies such as the C. pepo fruit. Therefore, this study highlights the significance of exploring herbal remedies as a potential treatment option for inflammation-related diseases.
Keywords
Introduction
Inflammation is indeed a local response of vascularized tissues to various stimuli, both internal and external. It is a protective response of the body that aims to eliminate the causative agent and limit tissue damage.
1
Importantly, inflammation itself is not considered a disease but rather a physiological process. It can occur in response to various factors such as infection, trauma, foreign substances, or autoimmune reactions.
2
Reactive oxygen species (ROS) are inherent byproducts of cellular metabolism that, when present in appropriate quantities, play a crucial role as indispensable signaling molecules.
3
Nevertheless, in cases when levels of ROS become very high, they have the potential to cause harm to cells and tissues. Inflammation is a multifaceted immunological response triggered by noxious stimuli, which entails the secretion of pro-inflammatory biomolecules.
4
The continuous challenge lies in comprehending the complex interrelationship between ROS and these indicators in the development of illnesses. During the process of inflammation, immune cells secrete ROS, which serves as both signaling molecules and mediators of the inflammatory response.
5
Certain pro-inflammatory agents employ ROS as constituents of their signaling pathways. Fabrication of ROS is a key factor in the development of many inflammatory diseases.
6
Specifically, immune cells such as polymorphonuclear neutrophils contribute to endothelial dysfunction and tissue injury by producing excessive ROS at sites of inflammation.
7
The relationship between ROS and inflammation is intricate and varied, necessitating additional investigation to elucidate its intricacies and its therapeutic ramifications.
8
Regardless of the specific trigger, the underlying purpose of inflammation is to protect the body by initiating the immune response and promoting tissue repair. The immune system patrols the body for potentially dangerous invaders and eliminates them.
9
There has been a growing interest in herbal remedies as an alternative treatment strategy to synthetic medications.
10–13
Plants have high concentrations of natural antioxidants, which have been demonstrated to improve health.14,15 Carotenoids in sweet potatoes and spices like rosemary are two examples of phenolic chemicals.
16
Citrus fruits and bell peppers are considered to be highly beneficial sources of vitamin C, also known as ascorbic acid.
17
The effectiveness of these antioxidants is dependent on the quality of their sources and the methods used to extract them. As interest in herbal medicine grows, so does the scientific research investigations on the effectiveness and safety of various herbal remedies. In summary, the interest in herbal remedies as a treatment strategy is driven by a desire for natural approaches, the historical use of herbs in traditional medicine, concerns about safety, individualized treatment options, increasing scientific evidence
The pumpkin, or Cucurbita pepo, belongs to the family Cucurbitaceae. 18 It is a warm-season vegetable with many morphological varieties. It grows in a trailing or climbing vine and has long, slender stems. C. pepo, also known as pumpkin, has been studied for its pharmacological actions. Here are some of the pharmacological activities of C. pepo based on the search results: hepatoprotection, 19 inhibition of benign prostatic hyperplasia, 20 antioxidant, 21 anticancer, 22 antimicrobial, 23 anti-inflammatory, 24 antidiabetic, 25 and antiulcer. 26 This research was conducted to determine whether or not the C. pepo extract has the ability to scavenge free radicals in vitro, as well as its anti-inflammatory effects. The purpose of this study is to determine if the C. pepo extract has therapeutic value as an anti-inflammatory herbal remedy.
Materials and Methods
Collections and Authentication of Plants
Grade A fresh fruits of C. pepo were collected in March 2020 from the herbal garden of Sri Venkateshwara University, Tirupati, Andhra Pradesh, India. Besides, the plant voucher was verified by a group of botany experts at the University and assigned the voucher specimen number (Accession no. CP/2020/001-SNF/2022).
Preparation of C. pepo Crude Extracts
Extraction of plant material from fresh C. pepo fruits involves these steps. It is started by cleaning and peeling the fruit. The fruit puree was then dried in the shade and ground into a fine powder. 48 h of maceration at room temperature with a water-to-material ratio of 1:3 resulted in a viscous, sticky mass. The plant material concentration in the extract was measured and converted into a percentage. The extract's dependability was evaluated, and the concentrated extract was dehydrated and vacuum-sealed in hermetic containers. The extract was suspended in 0.9% normal saline to facilitate utilization. The desiccated pulp material was ground with an electric grinder before being sieved to achieve a finer consistency. The plant material was then extracted using methanol at a concentration of 99.9% via the soxhlation procedure. After vacuum evaporation and condensation, the yield percentage of the distilled alcohol was determined. All extracts were then dehydrated in a desiccator and frozen for additional pharmacological testing. When utilizing chloroform as the extraction solvent, the same procedures were performed. 27
Preliminary Phytochemical Screening
Figure 1 presents the overall studied experiments in the current study. As mentioned, the C. pepo fruit extract was tested in three different solvents for the initial phytochemical analysis: water, methanol, and chloroform. The phytochemical analysis involved diluting 200 mg of extracts in 10 ml of distilled water and filtering the resulting solution. The phytochemical analysis of the C. pepo extracts is presented in Table 1.
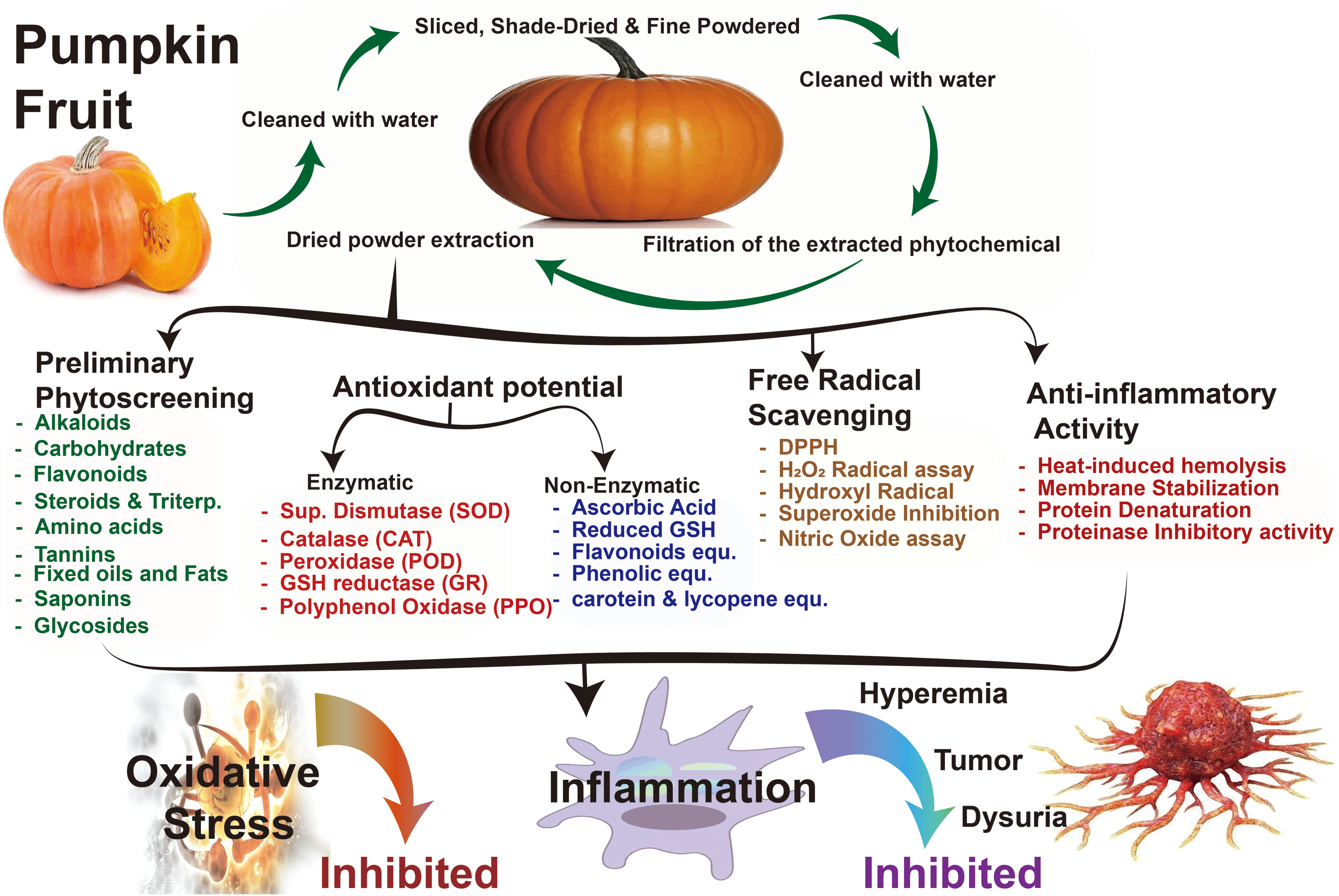
The overall studied experiments flowchart.
Preliminary Phytochemical Screening of C. pepo Extracts.

In the preliminary investigation, researchers looked for antioxidant enzymes and non-enzymatic compounds in C. pepo fruits.
Assay of Enzymic Antioxidants
The antioxidant enzymes superoxide dismutase, catalase, peroxidase, glutathione reductase, and polyphenol oxidases (catechol oxidase and laccase) were measured in Cucurbita pepo fruits. The assay methods that were followed can be found here.
Superoxide Dismutase (SOD)
According to the protocol established by Labidi et al (2021), the SOD activity in the C. pepo fruit was measured. 28
Procedure: The C. pepo fruit (0.5 mg) was crushed with 3.0 ml of potassium phosphate buffer (50 mM, pH 6.4) and centrifuged at 2000 xg for 10 min to extract the enzyme. The assay made use of the supernatant.
Assay: The experimental solution contained a sodium pyrophosphate buffer (1.2 ml), 0.1 ml PMS, 0.3 ml NBT, 0.2 ml enzyme extract, and water (2.8 ml). The reaction was sparked off by adding 0.2 ml of NADH. After incubating the mixture at 30°C for 90 s, 1.0 milliliter of glacial acetic acid was added to put a stop to the reaction. After letting the reaction mixture sit for 10 min and centrifuging it, we added 4.0 ml of n-butanol and shaked it well. A spectrophotometer was calibrated to read at 560 nm to determine the chromogen content in the butanol phase.
Catalase (CAT)
The determination of catalase activity in the fruits of selected plants of C. pepo was conducted using the method described by Labidi (2021). 28
Procedure: The fruit of C. pepo (0.5 mg) was subjected to homogenization in 2.5 ml of phosphate buffer at a temperature of 4 °C. Subsequently, the resulting homogenate was subjected to centrifugation at a force of 2000 xg for a duration of 10 min in order to extract the enzyme. The experimental procedure utilized the supernatant.
Assay: H2O2 in phosphate buffer was pipetted into a quartz cuvette until it reached a volume of 2.9 ml. Rapidly adding the enzyme extract (0.1 ml), it was entirely incorporated. The experiment involved measuring the duration required for the absorbance to decrease by 0.05 units. The H2O2-phosphate buffer was employed for the purpose of comparison. This experiment determined that 0.05 units of decrease in absorbance at 240 nm may be attributed to the presence of one unit of enzyme.
Peroxidase (POD)
The peroxidase activity of the C. pepo extract was assessed using the methodology described by Zhang et al (2021). 29
Procedure: Using 0.1 M phosphate buffer (pH 6.5) and 2.5 ml of the C. pepo fruit, an enzyme extract was generated and used in the experiment by centrifuging the mixture.
Assay: For the experimental procedure, a total volume of 3.0 ml of the pyrogallol solution was mixed with 0.1 ml of the enzyme extract within a cuvette. The spectrophotometer was calibrated to a baseline reading of 0 absorbance units at a wavelength of 430 nm. Subsequently, a volume of 0.5 ml of a 1% H2O2 solution was introduced into the system and agitated. The absorbance fluctuation was monitored at 30-s intervals for a maximum duration of 3 min. The quantification of peroxidase activity is determined by calculating the rate of absorbance change at a wavelength of 430 nm over a period of one minute.
Glutathione Reductase (GR)
The determination of glutathione reductase activity was conducted using the methodology given by Radić et al. 30
Procedure: The enzyme extract was prepared by extracting the juice from crushed C. pepo fruits into a volume of phosphate buffer (2.5 ml). After centrifuging at 5000xg for 10 min to separate the supernatant from the particulates, the experiment was carried out.
Assay: The total volume of the reaction mixture was 3.0 ml, and it contained 0.1 ml of EDTA, 0.1 ml of sodium azide, 0.1 ml of oxidized glutathione, 0.1 ml of the enzyme source, and 3.0 ml of water. After 3 min of incubation, 0.1 ml of NADPH was added to the reaction mixture. For three minutes, readings of absorbance at 340 nm were taken every 15 s. For every set of readings, a control tube with water in it served as a comparison to a tube with oxidized glutathione. Glutathione reductase was measured in units of oxidized NADPH per minute per glutathione reductase unit.
Polyphenol Oxidase (PPO)
The spectrophotometric approach established by Karakus et al (2021) 31 allows for the simultaneous assessment of the activity of polyphenol oxidases, including catechol oxidase and laccase.
Procedure: To homogenise C. pepo fruits, we employed 50 mM Tris HCl, sorbitol, and NaCl in 2.0 ml of a reaction solution. The homogenate was centrifuged at 2000xg for 10 min at 4 °C, and the supernatant was used in the experiment.
Assay: The assay comprised of 0.3 ml of a catechol solution and 2.5 ml of a phosphate buffer. The spectrometer was calibrated to read at 495 nm. After adding the enzyme extract (0.2 ml), the absorbance was measured every 30 s for 5 min. Under the test conditions, the amount of catechol oxidase (or laccase) required to convert 1 mole of dihydrophenol into 1 mole of quinine per minute was considered to be 1 unit of the enzyme.
Catechol oxidase = 0.272 x (ΔA/minute) Laccase = 0.242 x (ΔA/minute)
Determination of non-Enzymic Antioxidants
The levels of non-enzymic antioxidants, including ascorbic acid, reduced glutathione, flavonoids, total phenols, tocopherol, carotenoids, and lycopene, were measured in the fruits of C. pepo. The subsequent section provides an explanation of the methods that were implemented.
Ascorbic Acid
The ascorbic acid content of C. pepo fruits was calculated using the Fadaei (2023) 32 method.
Procedure: Ascorbic acid was extracted by homogenizing C. pepo fruits in 4% trisodium citrate (TSC) and bringing the total amount up to 10 ml. Following centrifugation at a speed of 2000 rpm for a duration of 10 min, the resultant supernatant was subjected to treatment with a small amount of activated charcoal. Subsequently, the mixture was allowed to lie undisturbed for a period of 10 min before being disposed of. In order to eliminate the residual charcoal, further centrifugation was conducted.
Estimation: The assay required 0.5 ml and 1.0 ml aliquots of the supernatant. Ascorbic acid (20-100 g) was removed from the working standard solution (0.2-1.0 ml). All of the test tubes had their volume brought up to 2.0 ml with 4% TSC for the assay. Then, half a millilitre of DNPH reagent and two drops of 10% thiourea solution were added. After incubating the test combination at 37°C for three hours, crystals of osazones were formed. The crystals were dissolved in 2.5 ml of 85% H2SO4 in a cold environment to avoid a drastic rise in the temperature. After H2SO4 was added, only the DNPH reagent and thiourea were added into the control. The absorbance at 540 nm was measured by chilling the tubes in water. Standardized linear regression was used to determine ascorbic acid concentrations using a scientific calculator. The ascorbic acid content was reported in milligrams per milligram of the sample.
Reduced Glutathione (GSH)
To calculate reduced glutathione concentrations, we followed a method suggested by Lu et al (2019). 33
Procedure: Homogenization with 5% TSC and centrifugation at 10000 rpm for 10 min at 4 °C were used to extract the juice from C. pepo fruits. For the assay, the supernatant was collected.
Assay: To perform the assay, 0.1 ml of the supernatant was mixed with 1.0 ml of phosphate buffer and 2.0 ml of DTNB. The mixture turned yellow after 10 min, peaking at 412 nm. A linear regression-programmed computer calculator created a standard curve to measure the reduced GSH concentration. GSH concentrations from 2 to 10 nanomoles were used to construct this curve. The assay yields nanomoles of GSH per gram of tissue, revealing the antioxidant levels in the samples.
Flavonoids
Tripathi et al (2019) provided a method for estimating flavonoids, which was used in this study. 34
Procedure: Flavonoids were isolated from C. pepo fruits using a 9:1 methanol to water ratio. Subsequent extractions used a 1:1 methanol-to-water ratio. After letting the extracts sit out overnight, shaking them thoroughly, combining them, and then clarifying them with n-hexane, the volume was determined. The assay made use of this after it was concentrated.
Assay: A portion of the extract was removed using a pipette and then concentrated by evaporation. Withdrawals of standard catechin ranged from 0.2 to 1.0 ml, with the latter being diluted to 1.0 ml with distilled water. Each test tube contained an aliquot of vanillin reagent (4.0 ml), which was heated in a water bath and boiled for 15 min before being cooled. The solution's optical density was measured to be 340 nm. Flavonoid concentrations were determined after the standard curve was generated using a scientific calculator programmed for linear regression. Flavonoids per milligram of tissue were used as the unit of measurement.
Total Phenols
The quantification of the overall phenolic content of the plant material was conducted utilizing the methodology established by Mandal et al (2021). 35
Procedure: Extraction of phenols: Homogenizing C. pepo fruit in 10 times their volume of 80% ethanol yielded the final homogenate. Twenty minutes were spent centrifuging the homogenate at 10,000 xg. A second extraction was made from the residue using 80% ethanol. The supernatants were collected into a pool and then evaporated. The leftovers were dissolved in an exact amount of clean water.
Assay: A solution containing phenols is prepared, and its volume is brought to a total of 3.0 ml by adding distilled water. The solution is then treated with 0.5 ml of the Folin-Ciocalteau reagent and 2.0 ml of 20% sodium carbonate solution. The final blend turns into a blue liquid. After heating the blue solution for one minute, it is chilled. Using a spectrophotometer, we determined the absorbance of the chilled solution at a wavelength of 650 nm. A reagent blank (a solution without the sample) was used as a reference. Known concentrations of a catechol solution (ranging from 0.2 to 1.0 ml or 2.0 to 10 g) are entered into an electronic calculator with a linear regression mode. This generates a standard curve relating the concentration of catechol to its absorbance at 650 nm. Using the generated standard curve, the absorbance of the sample is compared to the curve to determine its corresponding concentration of phenols. The total phenol content of the sample is expressed in milligrams of phenols per gram of tissue.
Tocopherol
Kahrıman (2021) described a spectrophotometric method for estimating the tocopherol content in plant samples. 36
Procedure: The C. pepo fruit is homogenized in a small volume of 0.1N H2SO4.
Volume adjustment: The volume of the homogenate is increased to 50 ml by slowly adding 0.1N H2SO4 without shaking. The mixture is left to stand for one night. On the next day, the contents of the flask are shaken well. Whatman No.1 filter paper is used to filter the mixture and remove any solids or debris that may have made it into the final product.
Estimation: Three centrifuge tubes were prepared, each containing 1.5 ml of the plant extract (test), reference solution, and water (blank). In each tube, a volume of 1.5 ml of ethanol and xylene is introduced and subsequently homogenized. The centrifugation process is employed to achieve the separation of the xylene layer from the ethanol and protein layers within the tubes. The xylene layer is meticulously isolated from the surrounding layers and subsequently transferred to a fresh tube. The 2,2'-dipyridyl reagent (1.0 ml) is introduced to the xylene layer (1.0 ml). The reaction mixture is transferred into cuvettes specifically designed for spectrophotometric analysis. The absorbance of the sample, standard, and blank was then measured using a spectrophotometer, with a reference wavelength of 460 nm. Commencing with an empty cuvette, a precise volume of 0.33 ml of FeCl3 solution is introduced, followed by a meticulous agitation of the contents. The absorbance of the test and standard cuvettes was measured against the blank at a wavelength of 520 nm precisely 15 min after the start of the experiment. The concentration of tocopherol was determined by using the obtained absorbance values and expressed in micrograms of tocopherol per milligram of material.
Total Carotenoids and Lycopene
Thitipramote et al (2023) provided a method for estimating total carotenoids and lycopene. 37
Procedure: In order to prepare the extract, C. pepo fruits were homogenised with 5 mL of 0.05% BHT in acetone, 5 mL of 95% ethanol, and 10 mL of hexane. Clear liquid was used in the experiment after centrifuging the mixture.
Assay: Supernatant dilution: The supernatant (presumably obtained after centrifugation) was diluted with 3.0 ml of water. The diluted supernatant was placed on ice and allowed to cool for 5 min. The cooled supernatant was vigorously shaken to ensure thorough mixing. After incubating the tubes at room temperature for 5 min, phase separation occurs. This results in the formation of two distinct layers. The absorbance readings were taken at 450 nm for carotenoid estimation and 503 nm for lycopene estimation. It is important to note that these specific wavelengths are commonly used for measuring the absorbance of carotenoids and lycopene, respectively. After recording the absorbance readings, the top hexane layer, which likely contained the targeted carotenoids and lycopene, was carefully transferred to a new tube. This layer was separated from the bottom aqueous layer.
Determination of Free Radical Scavenging Activity
DPPH Radical Scavenging Assay
The capacity of the fruit extracts to effectively neutralise the DPPH radical was quantified. 38
Procedure: One millilitre of methanol was added to twenty microliters of the C. pepo fruit extract and fifty microliters of DPPH solution. The reaction mixture was analysed after it was kept at room temperature and had been vigorously shaken for 30 min. Decolorization of the purple colour was spotted at around 518 nm. As a negative control, we used methanol, and as a positive control, we used DPPH dissolved in methanol.
H2O2 Scavenging Assay
The approach employed by Kanupriya et al (2019) was used to assess the scavenging ability of the fruit extracts against H2O2. 39
Procedure: The method called for mixing 0.6 millilitres of hydrogen peroxide solution with 10 millilitres of C. pepo fruit extract. The volume was adjusted to 3.0 ml with phosphate buffer. The reaction mixture's absorbance was tracked at 230 nm. The H2O2 was left out of the blank solution, which was phosphate buffer.
Hydroxyl Radical Scavenging Assay
Quantification of H2O2-generated hydroxyl radicals was achieved by spectrophotometrically measuring the formation of TBARS (thiobarbituric acid reactive compounds) as described by Kavitha (2018). 40
Procedure: Into the reaction mixture was added 0.1 millilitres each of 2-deoxyribose, FeCl3, EDTA, hydrogen peroxide, ascorbic acid, and a buffer. One millilitre of the plant extract was utilised to make up the difference, which was twenty microliters. The reaction mixture was incubated at 37 degrees Celsius for an hour, and then 0.5 millilitres were added to a solution containing 1 millilitre of trichloroacetic acid. After adding 1.0 ml of a 1% aqueous solution of TBA, the mixture was incubated at 90 °C for 15 min to promote colour development. After the sample had cooled to the appropriate temperature, the absorbance was measured at 532 nm using a blank solution as a control. To determine the relative degree of TBARS generation in the extract-treated groups, we set the positive control (H2O2) at 100%.
Superoxide Radical Inhibition Assay
Superoxide formation was inhibited in-vitro using a method similar to that developed by Jancy et al (2020) using an aqueous, methanolic, and chloroform extract of C. pepo fruits. 41
Procedure: In a tube, a volume of 0.02 ml of plant extract (or dimethyl sulfoxide in the control tube), 0.2 ml of EDTA, 0.1 ml of nitro blue tetrazolium, and 0.05 ml of riboflavin were amalgamated. To achieve a final volume of 3.0 ml, a phosphate buffer was incorporated into the mixture. The tubes were vigorously agitated to achieve comprehensive homogenization of the constituents. The measurement of the mixture's absorbance was conducted at a wavelength of 560 nm with a spectrophotometer. The tubes were subjected to consistent and even irradiation from a fluorescent bulb for a period of 30 min. Following a period of 30 min of exposure to light, the absorbance of the combination was subsequently determined at a wavelength of 560 nm using a spectrophotometer. The determination of the percentage of inhibition involves the comparison of the alteration in the optical density (absorbance) prior to and after illumination. The precise computation will be contingent upon the particular formula or equation employed in the experimental framework for ascertaining the magnitude of superoxide generation and suppression.
Nitric Oxide Radical Inhibition Assay
The ability of the fruit extracts to quell nitric oxide radicals produced in vitro was evaluated using the protocol established by Abd Ghafar et al (2018). 42
Procedure: The reaction was initiated by mixing 0.5 ml of each fruit extract with 2.0 ml of sodium nitroprusside and 0.5 ml of PBS. The assay mixture was properly mixed and incubated at 25 °C for 150 min. A final 0.5 mL of the Griess reagent was added to the test mixture after waiting for 30 min. The untreated tube served as a comparison and contained no fruit extract. Pink chromogen absorbance was measured at 546 nm and compared to a blank reagent.
In Vitro Anti-Inflammatory Activity
Heat-Induced Hemolysis
The heat-induced hemolysis assay was carried out using the method proposed by Johnson et al (2022). 43
A total of 10 ml of fresh whole human blood was taken and centrifuged at 3000 rpm for 10 min. Centrifugation separates the RBCs from other components of the blood. After centrifugation, the supernatant (containing plasma and other cellular components) was carefully removed, leaving behind the RBC pellet. The RBC pellet was washed three times with an equivalent volume of isotonic saline. This washing process helps in removing any residual plasma or contaminants from the RBCs. The volume of the RBC pellet obtained from the centrifugation was measured or estimated to determine the blood volume. Based on the calculated blood volume, a 10% v/v RBC suspension was prepared. This means that the RBCs are suspended in isotonic saline at a concentration of 10% of the total volume.
Procedure: The reaction mixture was prepared by combining the C. pepo fruit extract at concentrations of 150 µg/ml and 300 µg/ml (in water, methanol, and chloroform) with the red blood cell suspension at a concentration of 10%. The exact volumes of each component in the reaction mixture were not specified. A control tube was prepared by filling it with saline instead of the C. pepo fruit extract. Whereas this control was for testing the effects of the fruit extract on hemolysis. One of the reaction tubes probably had the gold standard drug, aspirin, put to it. The reaction tubes, including the control tube, containing the reaction mixture, were placed in a water bath and heated to 56 degrees Celsius for 30 min. When the incubation period was over, the tubes were cooled to room temperature by running water. Each reaction tube, including the control tube, had its supernatant centrifuged at 2500 rpm for 5 min. Red blood cells that were lysed were recovered by centrifugation. A spectrophotometer was used to determine the absorbance of the supernatant at 560 nm. The absorbance at this wavelength can indicate the degree of hemolysis. By comparing the absorbance of the reaction tubes containing the C. pepo fruit extract with the control tube, the percentage of hemolysis prevented by the fruit extract could be determined. The exact calculation method has not been provided.
Membrane Stabilization Test
Damage to macromolecules and membrane lipid peroxidation produce tissue harm caused by lysosomal enzymes generated during inflammation. Stabilising the lysosomal membrane is crucial for controlling inflammation by preventing the escape of lysosomal contents. Similarities exist between the membranes of lysosomes and erythrocytes. Since the extract was found to stabilize erythrocyte membranes, this likely also implies that it can stabilize lysosomal membranes. 44
Under in vitro conditions, the anti-inflammatory activities of aqueous, methanolic, and chloroform extracts of fruits of C. pepo was measured by their ability to prevent the lysis of human red blood cell (HRBC) membranes in response to hypotonicity. With a few tweaks, the Gupta (2019) approach was used. 45
Procedure: A mixture was prepared by combining an extract with concentrations of 150 µg/ml and 300 µg/ml, phosphate buffer (1.0 ml), hypo saline (2.0 ml), and a suspension of human red blood cells (HRBC) (0.5 ml). Following a 30-min incubation period at 37 °C, the reaction mixture was subjected to centrifugation at 3000 rpm for a duration of 20 min. Spectrophotometric analysis was conducted to measure the content of haemoglobin in the supernatant at a wavelength of 560 nm. Aspirin was used as the gold standard medication. The control group was given everything except the test samples for use. HRBC stabilization and protection as percentages were determined.
Protein Denaturation Assay
Inflammatory and arthritic disorders are linked to the denaturing of tissue proteins. Denaturing tissue proteins may lead to auto-antigen formation in several arthritic conditions. 46 An effective anti-inflammatory medication may be compounds that prevent protein denaturation. Extracts of the C. pepo fruit were therefore evaluated for their ability to prevent protein denaturation in three different solvents: water, methanol, and chloroform. The protein denaturation experiment was performed using a modified version of the protocol described by Yesmin et al (2020). 47
Procedure: The test solution, which included the aqueous extract of fruits of C. pepo extract/aspirin, was mixed with 100 l of egg albumin solution after incubation at 37 degrees Celsius for 15 min. The denaturing temperature of the reaction mixture was kept at 70 °C in a water bath. After the tubes had cooled, the turbidity at 660 nm was measured using a spectrophotometer (Genesys, USA). There was no test solution in the control tube. Denaturation inhibition percentages were determined.
Proteinase Inhibitory Activity
Proteinases are found to be involved in a number of biological processes such as inflammation and tissue injury. Serine proteinases from neutrophils, one of the inflammatory cells, are implicated in inflammatory disorders like pulmonary emphysema. 48 Thus, the proteinase inhibitory potential of the aqueous, methanolic, and chloroform extract of fruits of C. pepo was tested following the method of Sanmuga Priya et al (2018). 49
Procedure: The assay mixture contained 1.0 ml of the test sample and 1.0 ml of the Tris-HCl buffer containing 0.06 mg of trypsin. The components were mixed well and then incubated for 5 min at 37 °C. After that, 1 mL of 1.0 % W/V of casein was added and then the mixture was incubated again for 20 min. Perchloric acid with a concentration of 2.0 mL 70% was used to terminate the reaction. The absorbance of the clear supernatant was measured at 210 nm after centrifuging the cloudy suspension. Buffer was used to fill up the void. Aspirin was used as a baseline medication for this study. Inhibition of proteinase activity was calculated as a percentage.
Statistical Analysis
Mean and standard deviations were used to describe the data. Student's t-test was applied to the paired data to find the level of statistical significance. A p-value of less than 0.05 was taken as an indicative of statistical significance. The data for biogenic amines were presented as mean standard errors of the means. To test for statistically significant differences between the groups, a one-way analysis of variance (ANOVA) was used. Statistical significance was assumed for P values of less than 0.05 in both the test and Dunnet's test. All of the calculations were done in Graph Pad 6.0.
Results
Preliminary Qualitative Phytochemical Screening of the Extract
An initial qualitative phytochemical examination of the C. pepo fruit aqueous extract was performed. Table 2 displays the tabulated data. Extracts of C. pepo were found to contain alkaloids, carbohydrates, flavonoids, proteins, amino acids, steroids, fats and oils, and saponins in the fruit juice; alkaloids, carbohydrates, flavonoids, triterpenoids, saponins, and glycosides in the chloroform extract, and alkaloids, carbohydrates, flavonoids, proteins, amino acids, and saponins in the methanolic extract.
Preliminary Qualitative Phytochemical Screening of the Fruits of C. pepo.

Preliminary Qualitative Phytochemical Screening of the Fruits of C. pepo.

Key fact: ‘+’ indicates the presence of compounds; ‘-’ indicates the absence of compounds.
Enzymic Antioxidant Activities in Fruits of C. pepo
The enzymatic antioxidant activity of the Cucurbita pepo fruit extract is presented in Table 3. The results showed that superoxide dismutase and catalase activities were both increased in the C. pepo fruit extract.
Enzymic Antioxidant Activities in Fruits of C. pepo.

Enzymic Antioxidant Activities in Fruits of C. pepo.

Values are Mean ± SD of triplicates; @ 1Unit = Quantity of enzyme required to reduce NBT oxidation by half; # 1Unit = The amount of enzyme needed to cause a 0.05 unit/min drop in the absorbance at 240 nm; $ 1 Unit = Change in absorbance at 430 nm/minute; and 1 Unit = µmoles of NADPH oxidized/minute; * 1 Unit = Catechol oxidase/laccase activity expressed as the rate at which 1 unit of dihydro phenol is converted into quinine per minute.
Table 4 displays the amounts of non-enzymatic antioxidants present in C. pepo fruits, including ascorbate (282.60), reduced glutathione (653.41), flavonoids (169.76), total phenols (273.52), tocopherol (8.54), carotenoids (791.11), lycopene (51.40), and chlorophyll.
Non-Enzymic Antioxidant Activities in Fruits of C. pepo.

Non-Enzymic Antioxidant Activities in Fruits of C. pepo.

Values are Mean ± SD of triplicates
The radical scavenging activity of the aqueous extract of fruits of C. pepo was probed against a series of free radicals like DPPH, H2O2, hydroxyl, superoxide, and nitric oxide radicals.
DPPH, H2O2, and Hydroxyl Radical Scavenging Activity
Antioxidant in vitro assays, such as DPPH, H2O2, and hydroxyl radical scavenging activity, play a significant role in assessing antioxidant capacity. Identifying and characterizing the potential health benefits of various natural products, pharmaceutical compounds, and dietary substances requires the evaluation of their antioxidant activity in vitro. The antioxidant property of the of the C. pepo fruit extract was determined using DPPH, H2O2, and hydroxyl radical scavenging assays (Tables 5 and 6). The DPPH scavenging and H2O2 scavenging activities are maximum in the methanolic extract of C. pepo fruits.
DPPH and H2O2 Scavenging Activities of Fruits of C. pepo.

DPPH and H2O2 Scavenging Activities of Fruits of C. pepo.

Values are mean ± SD of triplicates.
Hydroxyl Radical Scavenging Activity of Fruits of C. pepo.

Values are Mean ± SD of triplicates.
The value in the H2O2-treated group was set at 100%, and the percentage values for the other groups were determined relative to this number.
Antioxidant assays in vitro for superoxide and nitric oxide inhibiting activity are crucial for determining the potential health benefits of various compounds and substances. Involved in oxidative stress and inflammatory processes, these assays provide crucial insights into the ability of these compounds to mitigate the detrimental effects of ROS and RNS. Antioxidant activity must be evaluated in vitro in order to identify and characterize substances with potential therapeutic applications, especially in the context of conditions involving oxidative damage and inflammation. Keeping in mind the toxicity of superoxide and nitric oxide, the inhibitory activity of chloroform, methanol, and aqueous extracts of fruits of C. pepo on the generation of superoxide anion and nitric oxide was studied. The results are presented in Table 7.
Superoxide and Nitric Oxide Inhibitory Activities of C. pepo.

Superoxide and Nitric Oxide Inhibitory Activities of C. pepo.

Values are mean ± SD of triplicates.
The scavenging effect of the methanolic extract of the C. pepo fruit was found to be the most significant in both the DPPH and H2O2 assays. Consequently, the methanolic extract was chosen for subsequent examination. The objective of this study was to evaluate the effectiveness of the methanolic extract of C. pepo fruits (MECP) in maintaining the structural integrity of the HRBC membrane. This was achieved by investigating its ability to suppress hemolysis generated by heat and hypotonicity, as well as its potential to prevent protein denaturation and proteinase activity. Two distinct dosage levels of the extract were employed, specifically the recommended dosage of 150 µg/ml and a higher dosage of 300 µg/ml. Aspirin was employed as the reference drug for comparative analysis.
Heat-Induced Hemolysis
The inhibition of heat-induced hemolysis by the MECP was checked and the results are displayed in Table 8. Heat-induced hemolysis was inhibited by MECP, and the effect was dose-dependent. The maximum percentage of inhibition was seen for MECP at 300 µg/ml (80.44), which was very comparable to that observed for aspirin (89.28).
Effect of the MECP on Heat-Induced Hemolysis.

Effect of the MECP on Heat-Induced Hemolysis.

Values are mean ± SD of triplicates.
The potential of the extract to inhibit hemolysis by stabilizing the HRBC membrane was determined. The results obtained are presented in Table 9. HRBC membrane stabilization was inhibited by MECP, and the effect was dose-dependent. The maximum percentage of inhibition was seen for MECP at 300 µg/ml (67.55%), which was comparable to that observed for aspirin (86.23%).
Effect of the MECP on HRBC Membrane Stabilization.

Effect of the MECP on HRBC Membrane Stabilization.

Values are mean ± SD of triplicates.
Protein denaturation has been implicated in inflammatory diseases. Thus, the anti-denaturation assay of protein has been used as a convenient tool to check the extent of inhibition rendered by the MECP. It is inferred from the result (Table 10) that the extract inhibited albumin denaturation in a concentration-dependent manner. At 150 µg/ml, about 60.10% inhibition was observed, which further enhanced to 75.89% on increasing the concentration to 300 µg/ml. The result was comparable with that of the standard drug aspirin, which exhibited 84.73% inhibition.
Effect of the MECP on albumin denaturation.

Effect of the MECP on albumin denaturation.

Proteinases are enzymes that catalyze the hydrolysis of the peptide bonds in protein, thereby bringing structural and functional changes in them (Vyas et al, 2013). With this view, the proteinase inhibitory activity of the methanolic extract of fruits of Cucurbita pepo was screened in vitro at two doses 150 µg/ml and 300 µg/ml, respectively. It can be inferred from the result (Table 11) that the extract inhibited proteinases in a dose-dependent manner. On increasing the concentration from 150 µg/ml to 300 µg/ml, the inhibition increased correspondingly from 55.06% to 76.51%.
Effect of the MECP on HRBC Proteinase Inhibition.

Effect of the MECP on HRBC Proteinase Inhibition.

Values are Mean ± SD of triplicates
The current study offers a thorough analysis of the C. pepo fruit extract's phytochemical screening, antioxidant properties, and anti-inflammatory activities. Based on the results of a preliminary phytochemical investigation, it was observed that the C. pepo extract exhibited the presence of various chemical constituents in the fruit extract, including alkaloids, carbohydrates, flavonoids, proteins, amino acids, steroids, fats and oils, and saponins. Similarly, the chloroform extract contained alkaloids, carbohydrates, flavonoids, triterpenoids, saponins, and glycosides. Furthermore, the methanolic extract displayed the presence of alkaloids, carbohydrates, flavonoids, proteins, amino acids, and saponins. Through a comprehensive analysis of the chemical constituents present in various solvent extracts, including proteins, alkaloids, and flavonoids, the study lays the groundwork for a fundamental comprehension of the biochemical profile of the extract. Furthermore, the incorporation of data pertaining to enzymatic antioxidants (catalase and superoxide dismutase) enriched the mechanistic comprehension of the antioxidant capability of the studied extracts. Where, the plants synthesize enzymes, namely superoxide dismutase, glutathione peroxidase, glutathione reductase, ascorbate oxidase, and glucose 6-phosphate dehydrogenase, with the purpose of mitigating the harmful effects of ROS. They have a crucial role in removing reactive oxygen species from plants. 50 Plant viability is dependent on the superoxide dismutase activity, which is increased in response to oxidative stress. 51 To counteract the negative effects of high H2O2 levels on the tissue metabolism, an increase in the catalase activity develops as an adaptive characteristic. 53 52,The peroxidase activity of the C. pepo fruit extract was found to be quite high. Boosting the NADP+/NADPH ratio requires a high level of glutathione reductase activity. Several plant species have been tested for the presence of enzymatic antioxidants, and their results are presented here. The study conducted by Jangid et al (2023) revealed that the application of the methanolic extract of Prosopis cineraria leaves to oxidant-exposed goat liver slices resulted in an enhancement of the antioxidant state and provided protection against oxidative damage. 54 Catalase, peroxidase, glutathione S-transferase, superoxide dismutase, and polyphenol oxidase are examples of enzymatic antioxidants. Vigneshwar et al (2016) conducted a study and found that the activity of these enzymes is notably greater in the leaves of Bacopa monnieri compared to the stolon or the roots. 55 Therefore, the presence of enzymic antioxidants in the extract of C. pepo fruits has beneficial therapeutic benefits, and regular consumption may aid in the fight against diseases brought on by oxidative stress. The highest concentrations of ascorbate, reduced glutathione, tocopherol, carotenoids, and chlorophyll were found in the C. pepo fruit.
The ability of plants to scavenge free radicals depends, in large part, on the concentration of antioxidants in their tissues. Researchers have discovered that polyphenols and flavonoids help plants deal with a wide range of stressors. There are numerous phenolic chemicals in plants that are used in important ways. 56 Total phenols, flavonoids, and flavanols were extracted in high concentrations from the seeds of Foeniculum vulgare and Trachyspermum ammi using both water and methanol. 57
The comparisons with analogous research conducted on other plant species showed a significant illustration of the high antioxidant potential of the C. pepo extract compared with various botanical sources. Indeed, this study has established assays for assessing the antioxidant potential, including the DPPH and H2O2 scavenging assays, thereby furnishing quantitative data to substantiate its assertions. For example, the antioxidant efficacy of the C. pepo fruit extract was further validated by comparisons with research conducted on other plant extracts, including Murraya exotica L. and Kyllinga nemoralis. In contrast, DPPH, a stable nitrogen-centered free radical, is commonly used to measure a compound's ability to either scavenge free radicals or donate hydrogen. 58 H2O2 is freely diffusible, and hence, readily permeates the membrane and damages proteins, nucleic acids, carbohydrates, and lipids. It can give rise to singlet oxygen by reacting with superoxide anion or with HOCl or chloramines. In terms of oxidizing power, OH• is among the most destructive. It can be produced by the Fenton reaction or by radioactively transforming water. 59 Lipids and DNA are oxidized by hydroxyl radicals. It forms the 8-hydroxy-2'-deoxyguanosine (8-OHdG) radical by reacting with the guanine ring structure of DNA, which can change DNA base pairs and break DNA strands, leading to mutations, cell growth arrest, or apoptosis. 60 The results of DPPH and H2O2 scavenging assays revealed that the aqueous extract of fruits of the Cucurbita pepo extract of the leaves exhibited maximum scavenging ability. The hydroxyl radical scavenging assay showed comparable results, with H2O2 exposure inducing the most damage to 2'-deoxyribose, and the aqueous extract of C. pepo fruits considerably mitigating this effect. The extract inhibited TBARS formation to a greater extent. Hence, the result of the present study revealed that the aqueous extract of fruits of Cucurbita pepo showed strong antioxidant potential. According to Ang et al (2022), the methanolic extract of Kyllinga nemoralis had high quantities of total phenols, flavonoids, flavonols, and tannins and effectively scavenged hydroxyl, DPPH, and hydrogen peroxide radicals. 61 The water fraction of Murraya exotica L. leaves showed the highest capacity to scavenge hydroxyl radicals compared to the other tested crude extracts. 62 In light of the above studies, the observations of the present study clearly revealed the antioxidant potential of the methanolic extract of fruits of C. pepo. In its entirety, the discourse offers a strong conceptual justification for the study's conclusions through the integration of various lines of evidence, the comparison of results to the pre-existing literature, and the clarification of underlying mechanisms of action. For example, the study on superoxide anion (O2ˉ•), which is of critical importance among the free radicals, as O2ˉ• is the principal species produced in the cells, and H2O2, hydroxyl radical (OH•) and peroxynitrite (OONOˉ) 63 are obtained as products from O2ˉ• in the downstream reaction cascade. Besides, it could induce cellular damage and inflammation, free radicals such as ROS and RNS operate at the molecular level, and as inflammatory mediators and signalling molecules, ROS are produced naturally by cells like plasma membrane nanoparticles (PMNs) and other physiological activities. 64 These free radicals, on a molecular level, increase the levels of adhesion molecules and proinflammatory cytokines, which leads to inflammation. 65 They trigger pathways like as NF-κB, which is essential for regulating inflammation, producing cytokines like IL-1β and TNF-α, and adhesion molecules like ICAM-1 and VCAM-1. 66 Conditions such as cancer, sepsis, and autoimmunity can develop as a result of chronic inflammation, which in turn increases the generation of free radicals. 67 In order to effectively manage inflammatory illnesses, it is crucial to find biomaterials and anti-inflammatory drugs that can protect the tissues from the ROS production.
In both assays, the maximum percent of inhibition was observed in the methanolic extract, closely followed by aqueous and chloroform extracts. Shahane et al (2023) found that the methanolic extract of Calendula officinalis leaves displayed stronger nitric oxide and superoxide radical inhibitory properties compared to aqueous and chloroform extracts, which was consistent with the current findings. 68 These results indicated that the extraction of most of the enzymatic and non-enzymatic antioxidants is into methanol, thus attributing to its high antioxidant potential. Having ascertained this, the ability of the extracts to protect the major cellular macromolecules against oxidative damage was studied in vitro.
Lysosomal proteases and bactericidal enzymes are produced by active neutrophils, setting off a chain reaction that degrades biomolecules, denaturates proteins, and peroxidizes membrane lipids. The inflammatory response is controlled by the stability of the lysosomal membrane. 69 Inflammation results in the production of proteases, which can cause auto-antigens and, in turn, auto-immune diseases. Valid indicators of anti-inflammatory efficacy in vitro include HRBC membrane stability, protection against protein denaturation, and proteinase activity. 70 These results showed that the methanolic extract inhibited heat-induced hemolysis in a dose-dependent manner. In the experiment, the higher dose of MECP had an inhibitory effect on hemolysis that was similar to the standard drug aspirin. This suggests that the tested extract, at the higher dose, demonstrated anti-hemolytic properties comparable to those of aspirin. Thus, the inhibiting activity of the extract increased with increasing concentration. The methanolic extract of Erythrina indica effectively inhibited the lysis of the HRBC membrane and rendered membrane protection dose-dependently. 71 The hemolysis-inhibiting ability of methanolic, ethanolic, and chloroform extracts of Erythrina variegate bark was compared. The ethanol extract was shown to be the most effective, followed by methanolic and chloroform extracts. 72 The above studies support our observation that the methanolic extract of fruits of C. pepo exhibits heat-induced lysis of erythrocytes and renders membrane protection, as a mark of its anti-inflammatory potential.
The anti-inflammatory activities were comprehensively examined in the study through an analysis of multiple mechanisms, such as protein denaturation inhibition and proteinase activity support and membrane stabilisation. By consulting research on Enicostemma axillare and Aster lanceolatus, two additional plant extracts that possess comparable anti-inflammatory properties, the discourse provides a more comprehensive framework for comprehending the potential therapeutic advantages of the C. pepo fruit extract. During inflammation, neutrophils empty their lysosomal contents, releasing proteases and bactericidal enzymes that damage tissue. 73 Anti-inflammatory drugs (NSAIDs) keep the lysosomal membrane intact, which stops the contents from leaking out. Therefore, drugs that stabilize membranes may be useful anti-inflammatories. 74 Since the HRBC membrane is analogous to the lysosomal membrane, the membrane stabilizing ability of the methanolic extract of fruits of C. pepo can be extrapolated to that of the lysosomal membrane. It is inferred from the result that the extract rendered considerable protection to the membrane by inhibiting hypotonicity-induced hemolysis in a concentration-dependent manner. The MECP extract showed 39.9% protection at 150 µg/ml, increasing to 67.55% at double concentration, indicating a dose-dependent response. It is a known fact that the intracellular calcium level will affect the shape and volume of the cell. The methanolic extract of fruits of C. pepo might have stabilized the RBC membrane by adjusting the calcium level inside the cell, thereby increasing the surface area to volume ratio of the cell. The membrane-protecting property of the extract could also be attributed to the interaction of the plant components with the cell membrane proteins.
A comparative investigation was undertaken to assess the anti-inflammatory potential of various extracts (petroleum ether, chloroform, methanol, and aqueous) derived from the leaves of Aerva lanata. The results revealed a noteworthy capacity of the methanolic extract to stabilize the RBC membrane, surpassing the effectiveness of the standard pharmaceutical diclofenac sodium. 75 The RBC membrane stabilization effect was observed in a dose-dependent way when the ethanolic extract of the aerial components (leaf, stem, and flowers) of Pergularia daemai was administered. This effect was shown to be comparable to that of the conventional medicine diclofenac sodium. 76 The hydroalcoholic extracts of the stem bark of three Pinus varieties namely, Pinus roxburghii, Pinus wallichiana, and Pinus gerardiana, have been shown to possess significant anti-inflammatory activity by inhibiting hemolysis and protein denaturation. 77 In light of these reports, it can be inferred from our findings that MECP renders a strong membrane stabilizing potential, which can be extrapolated as a strong anti-inflammatory effect.
Protein denaturation can provoke delayed-type hypersensitivity as effectively as native proteins. 78 Salicylic acid, phenylbutazone, and flufenamic acid are examples of anti-inflammatory drugs that have been shown to increase inflammation by inducing thermally induced protein denaturation. 79 Protein denaturation was suppressed by the extract, and the degree of inhibition was shown to be concentration-dependent. At 150 µg/ml, about 60.10% inhibition was observed, which further enhanced to 75.89% on increasing the concentration to 300 µg/ml. The result was comparable with that of the standard drug aspirin, which exhibited 84.73% inhibition. Chandra et al (2012) examined the anti-denaturing activity of the aqueous extract of the coffee beans Coffea Arabica. 80 An isolated compound from the methanolic extract of the Butea monosperma bark significantly inhibited protein denaturation at a lower dose of 1 µg/ml, whereas the activity decreased on increasing the concentration. 81 Inhibition of protein denaturation by a methanolic extract of the Piper betle leaf was observed to be concentration dependent. 82 Thus, with the support of the above reports, the anti-denaturation property of the methanolic extract of fruits of C. pepo has been confirmed by our result.
Proteinases are enzymes that catalyze the hydrolysis of the peptide bonds in protein, thereby bringing structural and functional changes in them. 83 Serine proteinases that are reported to be abundantly present in the lysosomal granules, promote the release of pro-inflammatory cytokines during inflammatory response and cause tissue damage. 84 Our findings are correlated with another study, in which Leelaprakash and Dass (2011) found the methanolic extract of Enicostemma axillare to exhibit anti-inflammatory activity by inhibiting the proteinase trypsin in a significant manner. 85 The methanolic extract of the leaves of Aster lanceolatus was found to exhibit a significant anti-proteinase activity in vitro. 86 The findings of the present study showed that the methanolic extract of fruits of C. pepo stabilized the HRBC membrane, and inhibited the heat-induced hemolysis of the HRBC membrane, protein denaturation, and proteinase activity in vitro. These properties of the extract confirmed its strong anti-inflammatory activity.
On the other hand, the clinical validation of the anti-inflammatory activities was declined in the study on the C. pepo fruit extract, while its phytochemical composition and antioxidant capabilities were thoroughly investigated. Therefore, clinical study will add to the statistical robustness and reliability of the results that might be compromised due to the in vitro used study assays taking into account aspects such as bioavailability and metabolism. Furthermore, concerns regarding the extract's effectiveness and safety profiles persist due to the lack of clinical research that compare it to standard anti-inflammatory drugs. Thus, a complete comprehension is impeded because the study does not investigate underlying biological pathways or address possible confounding factors. Additionally, it is possible to miss species-specific differences in phytochemical profiles when drawing broad conclusions about C. pepo. Thus, to improve our understanding of C. pepo's anti-inflammatory potential, future studies should focus on clinical validation, larger samples, comparative trials, mechanistic inquiry, and confounding factor evaluation.
The limitations of the current study encompass several elements about how the in vitro findings impact the in vivo systems’ reliability for clinical applications. This covers issues related to the complexity of biological systems and the microenvironments within their cells, tissues, and organs. For example, concerns about specificity, sensitivity, and relevance to in vivo circumstances such as the overall antioxidant defense systems could be raised in enzymatic and non-enzymatic assays. Furthermore, assay variability factors such as sample preparation and the used RBCs’ incubation conditions (temperature, pH, and osmolarity) that significantly contribute to the inconsistencies in the results of heat-induced hemolysis assays needs to be optimized. Meanwhile, the proteinase inhibitory activity protocol standardization, substrate concentrations, and reaction conditions are essential for minimizing its variability and improving result consistency. Thus, by understanding these limitations and employing appropriate experimental strategies, researchers can enhance the accuracy and translatability of the current study in vitro assays for advancing our understanding of the biological processes and facilitating the development of the target therapies.
Conclusions
Inflammation is a defensive immunological response to injury that causes pain, redness, heat, and swelling. Limited inflammatory response is beneficial, but uncontrolled inflammatory response causes chronic problems like allergies, cancer, autoimmune diseases, metabolic syndrome, diabetes, obesity-related diseases, and cardiovascular and neurological disorders. Pilot investigations showed that C. pepo fruits had higher levels of both enzymatic and non-enzymatic antioxidants; their chloroform, methanol, and water extracts showing antioxidant potential were prepared for further study. Findings denote, that these extracts have free radical quenching activities. Methanolic extracts were more active than water and chloroform extracts. DPPH radical scavenging experiments were performed at various extract concentrations ranging from 25 to 1000 μg/ml. The finding denotes that the higher dose of the methanolic extract has significant anti-inflammatory properties. Several assays were used to assess the extract's in vitro anti-inflammatory efficacy through measuring its capacity to stabilize RBC membranes, prevent protein denaturation, and inhibit proteinase activity. The extract dose-dependently stabilized HRBC membranes, and decreased hemolysis, protein denaturation, and proteinase activity. This study shows that the methanolic extract of the C. pepo fruit has anti-inflammatory properties. Thus, the plant can serve as a potential candidate for the development of safer drugs to treat inflammation-related disorders.
Footnotes
Acknowledgments
The authors would like to thank Dr K. Madhav Chetty, College of Botany, Sri Venkateshwara University, India, for his assistance in verifying the plant voucher specimen assigned number.
Ethical Approval
Ethical approval is not applicable to this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the “Belt and Road” joint project fund between Zhejiang University, China, and National Research Centre, Egypt (Project No: SQ2023YFE0103360). Also, it was funded by the Deputyship for Research Innovation, Ministry of Education in Saudi Arabia for funding this research work through the project number (GrantA421).
Statement of Informed Consent
There are no human subjects in this article and informed consent and/trial registration is not applicable.
Statement of Human and Animal Rights
There are no human or animal subjects in this article that need right consent.
