Abstract
Objectives
To investigate the safety profile of subchronic oral administration of hydroethanol leaf extract of Newbouldia laevis (NL) in rats.
Methods
NL was administered orally (20, 100, and 500 mg/kg) to groups of rats for 90 days. Distilled water served as control. Some rats were spared for an extra 30 days with no extract administration (reversibility study). Blood, urine and selected vital organs were collected for analyses.
Results
In male rats, NL (20, 500 mg/kg) irreversibly increased (P˂.05) bodyweight and feeding pattern with opposite trend of results in females. NL (20 mg/kg) elicited irreversible decrease (P˂0.05) in RBC in male rats; a reversible decrease in RBC and Hb, and an irreversible increase (500 mg/kg) in WBC and neutrophils in females. In male rats, NL irreversibly decreased (P˂.01) HDL-c and increased uric acid; in female rats, it elicited a reversible increase (P˂.05, 500 mg/kg) in AST. NL (500 mg/kg) irreversibly decreased (P˂.05) sperm motility and count, and produced an irreversible increase (P˂.01) in testosterone levels in male rats (100 mg/kg). It reversibly decreased (20-500 mg/kg) luteinizing hormone levels (both sexes).
Conclusions
NL is fairly safe at lower doses. However, it could cause anemia at lower doses, infertility (both sexes), liver and kidney injuries at high doses.
Introduction
Plant medicines and related products have, in our recent history, witnessed widespread popularity all over the world. 1 In Africa and other developing nations, cultural preference, affordability, accessibility, and availability are the integral reasons for their use. 2 But for the convincing beliefs that herbal medicines are natural and safer, the upsurge in their patronage in the advanced and developed countries would not have been possible. 3 Cultural and spiritual use, food and nutrition and medicines for treating a wide range of diseases in man and animals highlight the diverse purposes of plants. People generally believe that traditional medicine cures do not cause any harm and accordingly they are consumed without any regard to quantity or dose. 4 However, scientific advancements have triggered the quest to ascertain if truly green medicines are actually devoid of harmful effects as claimed. 5 Plants have many chemical constituents, some of which may be potentially and intrinsically injurious to the body. 6 Wrong identification of plants, incorrect preparation, irrational use, overdosing, herb–drug interactions, etc, can also result in toxicity.7,8 The majority of the scientific studies conducted on medicinal plants focused largely on their therapeutic benefits at the expense of their toxicity profiles.9,10–12 Several toxicity studies (acute and subacute) of Newbouldia laevis extract have been documented.13–16 Meanwhile, there has been no documented subchronic study on any form of the extract of N. laevis.
N. laevis (P. Beauv.) Seem. is a deep greenish plant from the Bignoniaceae family with a widespread presence in the tropics. In Nigeria, the plant is used for different purposes, including medicinal, cultural and ornamental values. Its native name in Yoruba is “Akoko,” “Aduruku” in Hausa and “Ogilisi” in Igbo; in folk medicine, the plant (leaf, root, and stem) is used for different medicinal purposes. 17 In Nigeria, N. laevis is used traditionally for rheumatic swellings, syphilis, piles, headache, convulsion, epilepsy, and manic disorders.18–20 The root and leaves have been reported in the management of fever, headache, and CNS disorders. 19
An extensive literature search revealed no detailed information on subchronic (90 days) toxicological profile of the plant. Accordingly, this study investigated 90 days of oral treatment of hydroethanol (50% aqueous ethanol) leaf extract of N. laevis in rats and the possibility for reversal of effects sequel to the stoppage of treatment.
Materials and Methods
Plant Material
Leaves of N. laevis were obtained from Ikenne Town, Ogun State, Nigeria and identification was done by Prof J.D. Olowokudejo of the Department of Botany, Faculty of Science, University of Lagos, Nigeria. A voucher specimen (LUH8017) was kept in the institutional herbarium.
Extraction
Leaves of N. laevis were dried under a shade until a constant weight was obtained. The air-dried leaves were ground into powder and soaked in hydroethanol (50% aqueous ethanol; 1.5 L) for 48 hours, after which it was decanted and filtered. The residue was resoaked in hydroethanol (×2) to ensure complete extraction. Under reduced pressure at a temperature of 40 °C, the cumulative filtrate was evaporated to dryness. A solid extract was obtained (12.75% yield), and carefully dissolved as required in sterile water to get the needed concentrations (mg/mL).
Experimental Animals
Albino mice (15-25 g) and Wistar rats (100-150 g) of either sex were procured from the Laboratory Animal Centre of the College of Medicine, University of Lagos (CMUL), Lagos, Nigeria. The animals were housed in a clean and conducive environment with 24-hour access to feed (Livestock Feeds PLC, Lagos, Nigeria) and water. An adaptation period of 2 weeks was allowed before the studies began. Ethical approval was obtained from the Health Research Ethics Committee (HREC) of CMUL (CMUL/HREC/08/18/415).
Sample size determination was based on our previous experimental design in similar toxicological assessment studies.9,11
Acute Toxicity Test
For 12 hours, groups of mice with 5 animals each were denied access to feed after which N. laevis (≤2 g/kg) was given orally. Another set of mice (5 groups) were treated intraperitoneally with different extract doses (50-800 mg/kg). The two groups designated as vehicles received distilled water (10 mL/kg; p.o. and i.p.). Behavioral responses and toxicity tendencies were carefully observed for 2 hours after treatment. Deaths in each group within 24 hours were noted and the remaining animals were kept under close observation for a further 2 weeks for any signs of delayed toxicity. Using the Behrens-Karber method, the lethal dose (LD50) was estimated.21,22
Subchronic Toxicity Test
The OECD-408 guideline was followed in this case. Eighty Wistar rats were selected into 4 groups of 20 rats each (10:10; male:female) without bias and were daily given distilled water (control) and N. laevis at 20, 100, and 500 mg/kg orally for 90 days. Doses selection (×1/5, ×1, and ×5 respectively) was in accordance with the procedures of Afolabi et al 9 and Akindele et al. 11 Bodyweight changes of rats in each group were recorded on a weekly basis for 90 days. Changes in feeding and drinking habits were noted daily. On the eve of the 90th day, urine samples were collected for urinalysis. 23 Twenty-four hours post 90th day treatment; some animals in each group were anaesthetized. Through the retro-orbital plexus vein of the rat eye, blood samples were collected for different analyses (blood, blood chemistry, and hormones). The rats were sacrificed and ethylenediaminetetraacetate (EDTA), lithium heparin, and sterile (plain) sample bottles were respectively used for the collection of blood samples for hematological, biochemical, and hormonal analyses. Blood specimens in sterile sample bottles were allowed to coagulate for 1 hour after which centrifugation (Uniscope Laboratory Centrifuge; Model SM902B, Surgi-field Medicals, England, UK) was done at 3500 r/min for 10 min. According to Cheesbrough. 24 Ogli et al 25 and Kale et al, 26 sperm was extracted from male rats and its movement, population (count) and abnormality (%) were evaluated.
Internal Organs
Upon sacrifice, rats were laparatomised and vital organs were picked out and weighed (regimented to 100 g bodyweight of corresponding rats). Fixed (in 10% neutral buffered formalin) internal organs were examined histopathologically. The same methods were employed for the reversibility study.
Hematological Analysis
Blood specimens in EDTA bottles were probed for Full blood count using an automated hematology analyzer. 11
Biochemical Analysis
Blood specimens in lithium heparin bottles were studied for cardiovascular, liver, and kidney function tests using an automated analyzer (Randox Laboratories Limited, Crumlin, County Antrim, BT294QY, UK).
Semen Analysis
Sperm was removed from rat epididymis and sperm movement (motility), population (count) and % abnormality (morphology) were determined.24–26
Urine Chemistry
Urine samples collected from individual rats for 12 hours (8:00 p.m. to 8:00 a.m.) on the 89th/90th and 119th/120th days were used for urine chemistry analysis using standard urine strips (Uricheck, Ref. U003b, Veda Lab, Alencon, France). 27
Hormone Analysis
Luteinizing hormone (LH), estrogen, testosterone, follicle-stimulating hormone (FSH) and progesterone were analyzed from the plasma.28,29
Histopathological Assessment
Tissues were fixed in 10% neutral buffered formalin, dehydrated in graded alcohol, embedded in paraffin, cut into 4 to 5 μm thick sections and stained with hematoxylin-eosin for photomicroscopic assessment using a Model N-400ME photomicroscope (CEL-TECH Diagnostics, Hamburg, Germany).11,30
Statistical Evaluation
GraphPad Prism 6 was used for statistical analysis. Results are presented as mean ± SEM Comparison of data was done using One-way ANOVA alongside Dunnett's post hoc test for multiple comparisons. Results with P-value of greater than .05 were regarded as significant (P < .05).
Results
Acute Toxicity
There was no death when N. laevis was given up to 2 g/kg. However, inactivity, slow movement and quietness were noticed within 2 hours after extract treatment. Progression to severity or signs of delayed toxicity in the animals was not observed following 14 days of close monitoring. The LD50 (i.p.) was 390 mg/kg.
Effect on Bodyweight, Food and Water Intakes
In male rats, N. laevis produced significant increase in bodyweight (P ˂ .05, 500 mg/kg) and food intake (P ˂ .01, 20 mg/kg) compared to control. The effect of the extract on bodyweight was sustained but was reversed in respect of food intake after 30 days of no extract administration. In female rats, N. laevis (20-500 mg/kg) produced a significant irreversible decrease in bodyweight (P ˂ .001, .01) and a reversible decrease in food intake (P ˂ .001) relative control (Table 1).
Effect of hydroethanol leaf extract of Newbouldia laevis on bodyweight alterations, food and water consumptions in male and female rats (main and reversibility studies).

Values indicated as mean ± S.E.M (n = 10).
P ˂ .05.
P ˂ .01.
P ˂ .001.
P ˂ .0001 versus distilled water (one-way ANOVA alongside Dunnett's multiple comparison test).
Effect on Weight of Vital Organs
In male rats, with the exception of the pancreas and spleen, N. laevis significantly increased (P ˂ .05-.0001) the weight of the organs across all doses against the control. Upon cessation of the administration of the extract, the increase in the weight of the kidney was sustained at 500 mg/kg, while at 20 mg/kg, the effect of the extract on most of the organs was reversed (Supplemental Table S1). In female rats, N. laevis (20-100 mg/kg) increased (P ˂ .05-.001) the weight of most of the organs. However, the heart elicited a postadministration reduction (P ˂ .05) at 500 mg/kg compared to the control (Supplemental Table S2).
Effect on Blood Indices
In male rats, N. laevis produced an insignificant effect (P > .05) on most of the parameters relative to the control. However, there was a significant decrease in RBC (P ˂ .05) at 20 mg/kg of the extract compared to control. Postadministration increase (P < .01-.0001) in WBC, its differentials, RBC and platelets were elicited. At 100 to 500 mg/kg, PCV (P < .001) and Hb (P < .05, .01) were significantly decreased in the 30 days of the reversibility study (Table 2). In female rats, the extract (500 mg/kg) produced an irreversible increase (P < .01, .001) in WBC, neutrophils, MCV, MCH, and MCHC against the control. At 20 mg/kg, Hb and RBC were reversibly decreased (P < .05, .01) compared to control. In the postadministration study, RBC, Hb, and PCV were all significantly increased (P < .01, .001, 20 mg/kg) compared to distilled water (Table 3).
Effect of Newbouldia laevis on Blood Indices in Male Rats.

Values indicated as mean ± SEM (n = 10).
P ˂ .05.
P ˂ .01.
P ˂ .001.
P ˂ .0001 versus distilled water (one-way ANOVA alongside Dunnett's multiple comparison test). Rev: Reversibility study.
Effect of Newbouldia laevis on Blood Indices in Female Rats.

Values indicated as mean ± SEM (n = 10).
P ˂ .05.
P ˂ .01.
P ˂ .001.
P ˂ .0001 versus distilled water (one-way ANOVA alongside Dunnett's multiple comparison test). Rev: Reversibility study.
Effect on Blood Chemistry
In male rats, N. laevis (100 mg/kg) reversibly increased (P ˂.05) the albumin level compared to control. There was an irreversible decrease (P ˂ .01) in albumin level at 500 mg/kg against the control. The extract produced an irreversible decrease (P ˂ .01, .001, 20-500 mg/kg) in HDL-c level, and sustained increase (P ˂ .05, .01, 20-500 mg/kg) in uric acid level compared to distilled water (Supplemental Table S3). In female rats, blood parameters were largely not affected (P > .05) relative to the control. However, the extract at 500 mg/kg reversibly increased (P ˂ .05) the AST level in relation to control. Meanwhile, the extract decreased (P ˂ .01, 20 mg/kg) the ALP level compared to control. There was a delayed increase (P ˂ .05, 100 mg/kg) in ALP level compared to distilled water (Supplemental Table S4).
Effect on Sperm Parameters
N. laevis at 500 mg/kg produced significant reductions (P ˂ .05, .01) in sperm motility and count which were not reversed in 30 days posttreatment study relative to control (Table 4).
Effect of Newbouldia laevis on Sperm Parameters in Male Rats.

Rev: Reversibility study.
Values indicated as mean ± SEM (n = 8).
P ˂ .05.
P ˂ .01.
P ˂ .001 versus distilled water (one-way ANOVA alongside Dunnett's multiple comparison test).
Effect on Urine Chemistry
In male rats, no major effect (P > .05) was produced on urine indices by N. laevis against the control. Upon the cessation of the extract administration, there was a significant increase (P ˂ .05) in pH at 100 mg/kg of the extract relative to the control (Supplemental Table S5). In female animals, N. laevis produced an irreversible increase (P ˂ .01, 20-500 mg/kg) in pH and a reversible decrease (P ˂ .05, 100 mg/kg) in specific gravity compared to distilled water (Supplemental Table S6).
Effect on Hormones
In the male rats, N. laevis decreased (P ˂ .05-.001, 20-500 mg/kg) the FSH and LH levels, and irreversibly increased (P ˂ .01, 100 mg/kg) the testosterone level relative to control. In the 30-days postadministration study, the decrease in FSH level (P ˂ .01) was sustained while LH level increase was reversed (Table 5). In female rats, N. laevis (20-500 mg/kg) produced a decrease (P ˂ .01, .05) in LH level compared to control. There was a delayed increase (P ˂ .05, .01, 100-500 mg/kg) in FSH and LH levels against the control (Table 6).
Effect of Newbouldia laevis on Hormones in Male Rats.

FSH, follicle-stimulating hormone; LH, luteinizing hormone; Rev: Reversibility study.
Values indicated as mean ± SEM (n = 8).
P ˂ .05.
P ˂ .01 versus distilled water (one-way ANOVA alongside Dunnett's multiple comparison test).
Effect of Newbouldia laevis on Hormones in Female Rats.

FSH, follicle-stimulating hormone; LH, luteinizing hormone; Rev: Reversibility study.
Values indicated as mean ± SEM (n = 8).
P ˂ .05.
P ˂ .01 versus distilled water (one-way ANOVA alongside Dunnett's multiple comparison test).
Histopathological Assessments
The leaf extract of N. laevis at subtherapeutic and therapeutic doses did not alter the histological architecture of the rats’ brain (both sexes) samples examined after 90 days of repeated administration. Neurons appeared generally normal and showed no visible lesions. However, few foci of neuronal degeneration and necrosis, and moderate congestion of cerebral blood vessels were observed at the supratherapeutic dose of the extract. In the reversibility study, these effects were reversed as neurons appeared normal and no visible lesions were seen (Supplemental Figure S1).
For the rat liver, hepatocytes were generally normal; there was no visible sign of lesion and a few foci of mild thinning of hepatic plates were observed at lower doses. At higher doses, hepatic plates were closely parked; there were scanty foci of monohepatocellular necrosis and a mild aggregate of mononuclear inflammatory cells in the portal tracts in both sexes of the rat liver. In the postadministration study, all these effects were reversed. The liver cells appeared generally normal (Figure 1).

Male and female liver histology of rats treated with DW (10 mL/kg: A, AO) and NL (20 mg/kg: B, BO); (100 mg/kg: C, CO); (500 mg/kg: D, DO) (×100). AO, BO, CO, and DO: Implies no extract treatment (reversibility) study.
The rat kidneys at lower doses appeared normal, the glomerular and tubular structures were normal and there was no sign of apoptosis in the kidneys. However, there was a sign of congestion, cellular degeneration and apoptosis, and glomerular and tubular atrophy at the supratherapeutic dose. In the reversibility study, the kidney appeared normal; there was no sign of congestion or apoptosis (Figure 2).
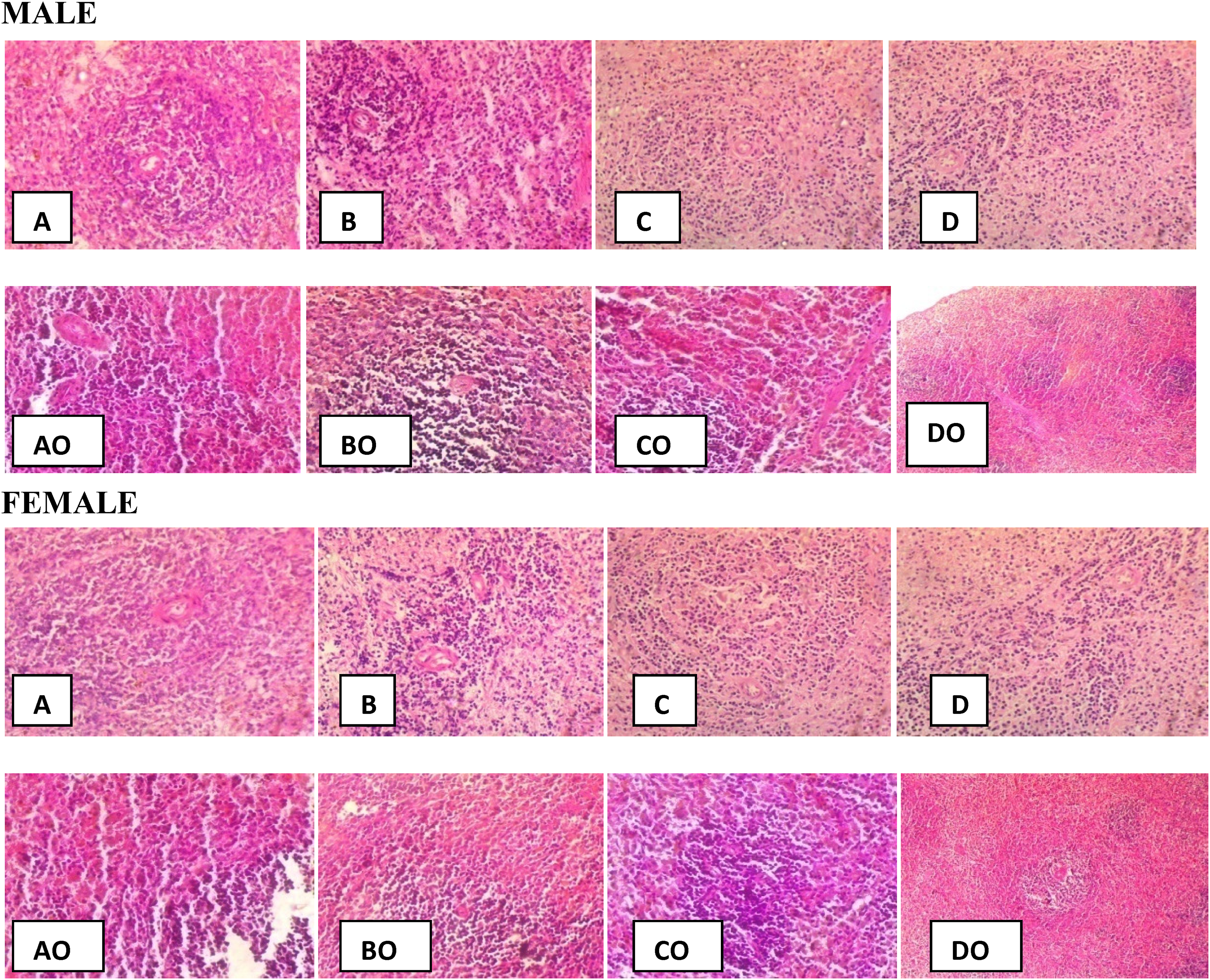
Male and female kidney histology of rats treated with DW (10 mL/kg: A, AO) and NL (20 mg/kg: B, BO); (100 mg/kg: C, CO); (500 mg/kg: D, DO) (×100). AO, BO, CO, and DO: Implies no extract treatment (reversibility) study.
The testes did not show any visible lesions on interstitial cells at all doses. There was mild congestion of blood vessels at therapeutic and supratherapeutic doses. Evidence of loss of spermatogenic cells was observed at higher doses. In the postadministration study, there was no visible lesion on the testicular cells (Supplemental Figure S2).
Discussion
Dangerous adverse reactions or organ toxicities from herbal remedies have been reported. 31 Detailed toxicological studies are imperative to reveal hazards of some commonly used plants which will help the regulatory bodies and policy makers to properly advise the public on their use. It will also help with their standardization and drug development. Accordingly, an extensive toxicological investigation of hydroethanol leaf extract of N. laevis was conducted. An acute toxicity test is a standard in-vivo method to evaluate drug safety. Adeyemi et al 23 and Akindele et al 10 reported that therapeutic agents with oral LD50 values greater than 2 g/kg are considered relatively safe. The LD50 (oral) value of N. laevis was estimated to be >2 g/kg in mice; therefore, N. laevis can be said to be safe. The LD50 (i.p.) was 390 mg/kg.
N. laevis produced a significant irreversible increase in bodyweight at the supratherapeutic dose and a reversible increase in feeding pattern at the subtherapeutic dose in male rats. In female rats, the extract at all doses irreversibly reduced the bodyweight and reversibly decreased the feeding pattern. On vital organs, the extract (20-500 mg/kg) irreversibly increased the weight of all the soft structures (organs) in male rats except the pancreas and spleen which were not affected. In female rats, N. laevis (20-500 mg/kg) increased the weight of all the soft structures; an effect which was not reversed except for the heart which showed a reversible reduction in its weight at the supratherapeutic dose. Reduction in food and water intakes may be suggestive of CNS effect based on the suppression of appetite and growth rate.12,32,33 Reduction in bodyweight gain and internal organ weights are indices of toxicity after exposure to toxic substances.11,34,35 Based on the above findings, N. laevis can be said to be safe in male rats as the extract did not reduce the bodyweight gain, vital organs weight and food intake. However, in female rats, the extract reduced the bodyweight gain, weight of the heart and feeding pattern; suggesting toxicity.
In respect of hematological parameters, N. laevis (20 mg/kg) produced an irreversible decrease in RBC in male rats; a reversible decrease in RBC, Hb and an irreversible increase in WBC and neutrophils in female rats. The extract (500 mg/kg) also produced an irreversible increase in WBC, neutrophils, MCV and MCHC in female rats. Evaluating hematological parameters in animal studies has been reported to have high predictive value for humans.35,36 Akindele et al 11 reported anemia sequel to significant reductions in RBC, PCV, and Hb upon chronic usage of an extract. Therefore, the decrease in RBC and Hb by N. laevis in both sexes portends the likelihood of N. laevis inducing anemia at the subtherapeutic dose. Adebayo et al 37 reported that MCHC, MCH, and MCV are related to RBC, increase or decrease in these parameters would affect RBC. Therefore, the increase in MCHC and MCV in female rats may help to reduce anemia. An increase in white blood cells and lymphocytes could be linked to the immune-stimulatory effects of the plant extract. 38 The increase in WBC, neutrophils and lymphocytes in female animals is an indication of the immune-boosting effect of N. laevis and this could be due to the stimulatory effect of N. laevis on bone marrow.
On blood chemistry, the extract did not produce any significant effect on most of the parameters in both sexes. However, in male rats, the extract (100 mg/kg) increased the albumin level. Meanwhile, at 500 mg/kg, the extract decreased albumin, HDL-c and glucose levels. In female rats, N. laevis (500 mg/kg) reversibly increased the AST level; decreased the ALP (20 mg/kg) and increased the glucose level. The uric acid level was increased in male rats at all doses. AST and ALT are major enzymes assayed in liver function tests and increase in the levels of these enzymes above normal are clear indices of liver toxicity.22,39,40 Increase in serum concentration of liver biomarkers (ALT, AST, and ALP) is a reflection of liver toxicity.41,42 The increase in AST level by the extract at the supratherapeutic dose suggests possible risk with respect to the liver, while the decrease in ALP at the subtherapeutic dose suggests possible protective tendencies of the extract on the liver. A low level of HDL-c indicates cardiovascular toxicity while an increased serum level of HDL-c shows cardio-protective properties. 43 Accordingly, the decrease in HDL-c by N. laevis (500 mg/kg) suggests cardiovascular toxicity in male animals. According to Ma et al, 44 acidic urine can contribute to uric acid stone formation in the kidneys, ureters, or bladder. Creatinine, urea, and uric acid determinations are critical markers of kidney function. 45 Based on these findings, the significant increase in uric acid level may be unhealthy for the bladder and kidneys, hence their damage.
With respect to sperm parameters, N. laevis at the supratherapeutic dose irreversibly reduced sperm motility and count. Poisoned bucks demonstrated decreased sperm production, impaired sperm morphology and testicular degeneration with restoration of spermatogenesis upon discontinuation of the poison.11,46 Accordingly, N. laevis at the supratherapeutic dose could impact male fertility negatively.
For urinalysis, in male rats, N. laevis did not produce any major alterations in urine parameters. Upon the cessation of the extract administration, N. laevis increased the pH at the therapeutic dose. In female animals, N. laevis irreversibly increased the pH and reversibly decreased the specific gravity. The decrease in specific gravity suggests reduced excretion of solute occasioned by increased water consumption. The increase in the pH of the urine is an indication of metabolic alkalosis.
On hormonal assay, in male rats, N. laevis (20-500 mg/kg) decreased the levels of FSH and LH; and significantly increased the testosterone level (100 mg/kg) which was not reversed. In female rats, N. laevis (20-500 mg/kg) decreased the level of LH. There was a delayed (post 90 days) increase (100-500 mg/kg) in FSH and LH levels. Significant increases in the serum testosterone and LH levels by plant extracts may be helpful in the management of erectile dysfunction and infertility. 47 Decrease in serum testosterone level suggests a possible decrease in libido and reproduction. 48 Significant decrease in FSH and LH levels by plant extracts may induce reproductive anomalies. 49 Based on these submissions, the increase in testosterone level at the therapeutic dose suggests possible enhancement of male fertility, while the decrease in LH at the supratherapeutic dose may impair fertility in female rats.
In respect of histopathology, the brain, liver, kidney, and testes samples showed normal histological architectures at the subtherapeutic and therapeutic doses for both sexes. However, the cytoarchitectural appearances of these organs were distorted at the supratherapeutic dose which is an indication of toxicity.
We had previously reported the GC-MS fingerprint of the hydroethanol leaf extract of N. laevis revealing 44 peaks; octadecanoic acid ethyl ester; (E)-9-octadecenoic acid ethyl ester; (E)-1,3-dosenoic acid; oleic acid; and phytol were found to be most abundant. 50 We also reported the presence of saponins, steroids, glycosides, flavonoids, phenols, alkaloids, and tannins in the extract; total antioxidant capacity, total phenolics, and total flavonoids contents of the extract were reported to respectively be 42.65 ± 0.16 mg/100 g ascorbic acid equivalent, 44.49 ± 0.15 mg/100 g gallic acid equivalent, and 23.61 ± 0.18 mg/100 g quercetin equivalent. 16 Flavonoids have been reported to exhibit significant antioxidant and antiinflammatory activities which portends great medicinal importance owing to the association of oxidative stress and inflammation in the pathophysiology of numerous diseases. 51 Saleem et al52,53 previously reported that phenolic and polyphenolic compounds hold great importance among phytochemicals and constitute a significant amount of natural phytoproducts in foods.
In terms of limitation of the study, the reversibility study could only accommodate 30 days of cessation of administration of the extract to the different groups although the main study, with the administration of the extract at various doses, was for 90 days. Also, specific renal function and cardiac parameters could not be assayed and reported due to unforeseen challenges.
Conclusion
The findings in this study suggest that the hydroethanol leaf extract of N. laevis is relatively nontoxic and safe when administered at lower doses. However, the subchronic administration of subtherapeutic dose of N. laevis may cause anemia in both sexes; while the supratherapeutic dose of N. laevis extract has the tendency to cause infertility (in both sexes), liver (female) and kidney (male) injuries. These outcomes call for caution in relation to excessive and long-term intake of extract of the plant for ethnomedicinal purposes and mechanistic laboratory studies.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241257080 - Supplemental material for Ninety-Day Toxicological Assessment of Preparation of the Medicinal Plant Newbouldia laevis (P. Beauv.) Seem. (Bignoniaceae) in Rats
Supplemental material, sj-docx-1-npx-10.1177_1934578X241257080 for Ninety-Day Toxicological Assessment of Preparation of the Medicinal Plant Newbouldia laevis (P. Beauv.) Seem. (Bignoniaceae) in Rats by Akanji A. Murtala, Abidemi J. Akindele and Ibrahim A. Oreagba in Natural Product Communications
Footnotes
Acknowledgments
The authors appreciate Mr Luqmon Osipitan, Mr Busayo Kasumu (Department of Pharmacology, Olabisi Onabanjo University, Nigeria), Mrs Adeniyi Abimbola Taiwo, and Mr Olanrewaju Adegoke (Hematology Department, Olabisi Onabanjo University Teaching Hospital, Nigeria) for their technical assistance.
Author Contributions
Akanji A. Murtala: investigation, formal analysis, writing—original draft, writing—review and editing, and funding acquisition; Abidemi J. Akindele: conceptualization, methodology, writing—review and editing, project administration, and supervision; and Ibrahim A. Oreagba: writing—review and editing, and supervision.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This was obtained from the Health Research Ethics Committee (HREC) of the College of Medicine, University of Lagos, Nigeria (CMUL/HREC/08/18/415).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
