Abstract
Phytolacca americana L. is traditionally used in Korea, Japan, and China as a diuretic, antibacterial, antiviral, anticancer, and anti-inflammatory agent, and also in the treatment of hepatitis B, psoriasis, edema, and rheumatism. In this study, we evaluated the subchronic toxicity of an aqueous extract of P. americana (PAAE) in male and female F344 rats. The rats were orally administered PAAE (0, 500, 1000, and 2000 mg/kg body weight) once daily for 13 weeks. Mortality rate, body weight, food consumption, and organ weights were measured and assessed. Additionally, ophthalmological, hematological, and histopathological parameters were evaluated. Urinalysis and necropsy were also performed. The clinical chemistry values for potassium in the treated female groups (500, 1000, and 2000 mg/kg/ body weight/day) were higher than those in the control. Further, the relative weights of the kidneys in the treated female groups (1000 and 2000 mg/kg/ body weight/day) were higher than those in the control. However, these changes were not consistent in either sex, and no abnormalities were found in the corresponding pathological findings. Thus the results showed no adverse effects in all the parameters assessed. The findings show that after 13 weeks of treatment, the “no-observed-adverse-effect level” of PAAE is 2000 mg/kg body weight in both male and female F344 rats under the experimental conditions applied. Although treatment-related adverse effects were not seen, potassium-level changes in the blood should be examined to establish the safety profile of PAAE after long-term treatment.
Herbal medicines have been traditionally used in many countries for a very long time and are still widely used in Korea, Japan, and China. Currently, herbal medicines are also popular as alternative medicines, even in western countries. However, they are often formulated for human use without being subjected to rigorous scientific and toxicity testing. Consequently, safety issues concerning herbal products have increased and the adverse effects of herbal medicines are continuously reported. 1,2 Therefore, it is important to evaluate their safety through toxicity studies.
Phytolacca americana L. (P. esculentum, P. radix, P. acinosa; American pokeweed), family Phytolaccaceae, is a popular medicinal herb in Korea, Japan, and China. The chemical constituents of P. americana mainly consist of triterpenoid saponins 3 ; phytolaccosides A, B, C, D, and E 4 ; jaligonic acid; esculenic acid; phytolaccagenin 5 ; flavone; phenolic acid; sterol; and polysaccharides. 6 They have been used for their diuretic, antibacterial, antiviral, anti-inflammatory, and anticancer effects, and also in the treatment of hepatitis B, psoriasis, 7 edema, and rheumatism. 8 An increasing number of studies have focused on the bio-activities of P. americana, including its diuretic, antibacterial, antiviral, anti-inflammatory, anti-tumor, and anti-mammary gland hyperplasia effects. 7 Aside from its biological activities, toxicological information has been reported, including in vitro erythrocyte agglutination and leukocyte mitosis. 9,10 Moreover, in Turkey, the fruit of P. americana was found to reduce body weight and led to swelling of the hock joints. 11 An understanding of the general toxicity of a certain compound is an essential step in supporting its safe use. Therefore, the objective of this study was to evaluate the toxicity of an oral P. americana aqueous extract (PAAE) in male and female F344 rats under a high research practice standard system. This study was performed in compliance with the Good Laboratory Practice (GLP) of the Organization for Economic Cooperation and Development 12 and the Ministry of Food and Drug Safety (MFDS, Korea, 2009). Toxicity evaluations herein included mortality, clinical signs, body weight, food consumption, ophthalmologic examination, urinalysis, hematology, clinical chemistry, gross findings at necropsy, organ weight, and histopathological examination.
Results and Discussion
Characterization of the PAAE
Constituents of herbal drugs can be influenced and modified by a variety of factors, including differences among plant species and the conditions of strain, growth, harvest, and storage. Therefore, before the standardized extracts were prepared, P. americana was authenticated by 5 independent experts, and the standard crude drug was selected. The certification tests were performed at Catholic University of Daegu (Daegu, Republic of Korea) according to the general testing method of the Korean Pharmacopeia 13 to prevent any external contamination. The authenticated plant was proved to be safe from typical contaminations such as heavy metals [As (<3.0 mg/kg), Cd (<1.0 mg/kg), Hg (<0.2 mg/kg), and Pb (<5.0 mg/kg)] and pesticides [aldrin (<0.01 mg/kg), endrin (<0.01 mg/kg), dieldrin (<0.01 mg/kg), total dichlorodiphenyltrichloroethane (<0.2 mg/kg), and total hexachlorocyclohexane (<0.1 mg/kg)], and aflatoxin B1 (<0.01 ppm).
The two active ingredients of the herbal extract, phytolaccoside E (1.67 mg/g of aqueous extract, tR = 32.31 minutes) and phytolaccoside B (0.75 mg/g of aqueous extract, tR = 35.15 minutes), were identified by comparison with authentic samples using HPLC. The content of phytolaccoside E, the marker compound of P. americana, was 1.67 mg/g. PAAE was stable at 5 °C for 6 months. 14
General Observations
No treatment-related mortality was observed in any of the study groups during the 13-week repeated-dose toxicity study. PAAE induced salivation in 8 males and 9 females at 2000 mg/kg body weight (bw)/day. However, no histopathological correlations were observed. Changes in body weight during the treatment period are shown in Figure 1. No treatment-related changes in body weight were observed in the treated groups. In addition, the results show no clear differences in food consumption between the control and treated groups (Figure 2).
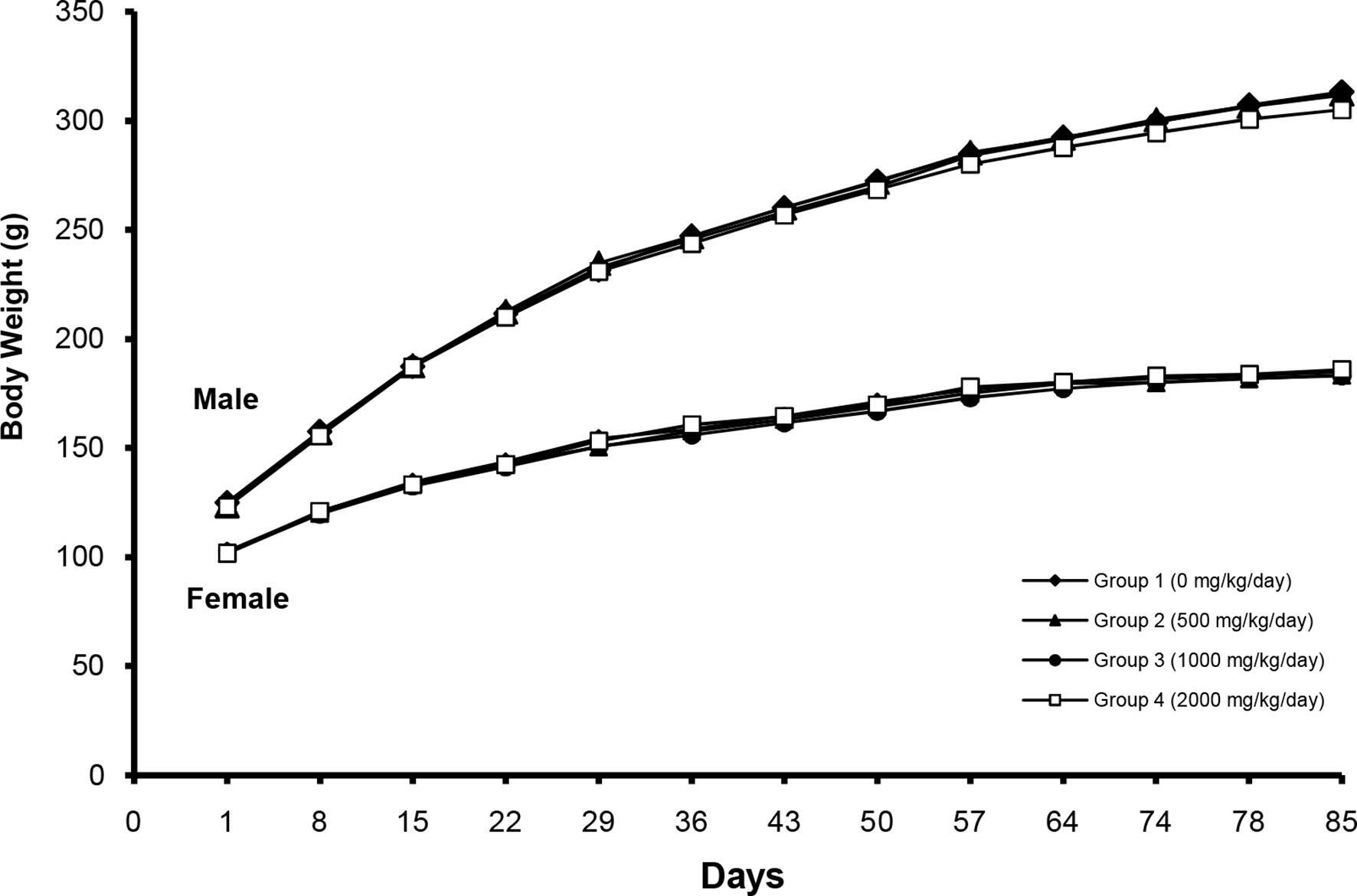
Changes in body weight after treatment with Phytolacca americana aqueous extract at 500, 1000, and 2000 mg/kg bw/day.
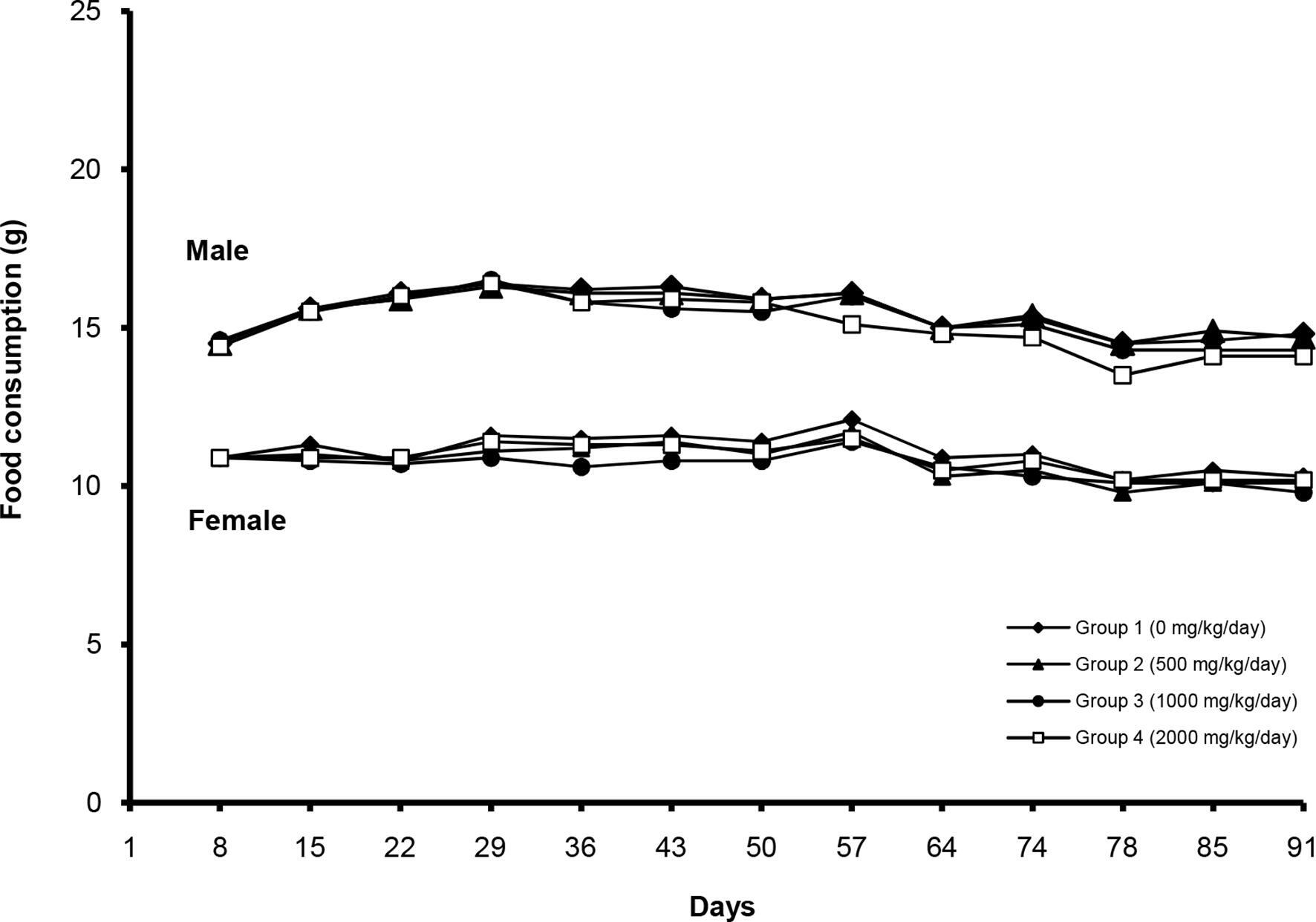
Changes in food consumption after treatment with Phytolacca americana aqueous extract at 500, 1000, and 2000 mg/kg bw/day.
Clinical Pathology
Farnes et al 10 reported the in vitro toxicity of P. americana on blood, including erythrocyte agglutination and leukocyte mitosis. However, the toxicity of a compound can vary based on the conditions it is exposed to and the material preparation method. 15
In the present study, most hematology values were not significantly altered by PAAE treatment, except for the mean corpuscular volume (MCV), mean cell hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), and platelet count (PLT) in certain groups. However, these values remained within the normal range; the changes in PLT in male rats were not dose proportional. 16,17 Furthermore, no abnormal ophthalmological findings were observed during the study (data not shown).
The clinical chemistry analyses showed that blood urea nitrogen (BUN) and total protein (TP) in male rats, and albumin (ALB), creatine kinase (CK), Ca, and K in female rats were altered by PAAE treatment to a statistically significant extent. However, the altered values remained within the normal range, with the exception of the K value in female rats. 16 Daily PAAE treatment (500, 1000, and 2000 mg/kg bw/day) increased the potassium level 1.10-, 1.13-, and 1.13-fold, respectively, in the female groups (Tables 1 and 2). These results are different from the control mean values and out of the normal range for potassium levels in rats. 16 Since these changes were treatment-related, the long-term effects of PAAE on potassium level must be studied.
Hematological Values Obtained After Treating F344 Rats With Phytolacca americana Aqueous Extract at 500, 1000, and 2000 mg/kg bw/day.
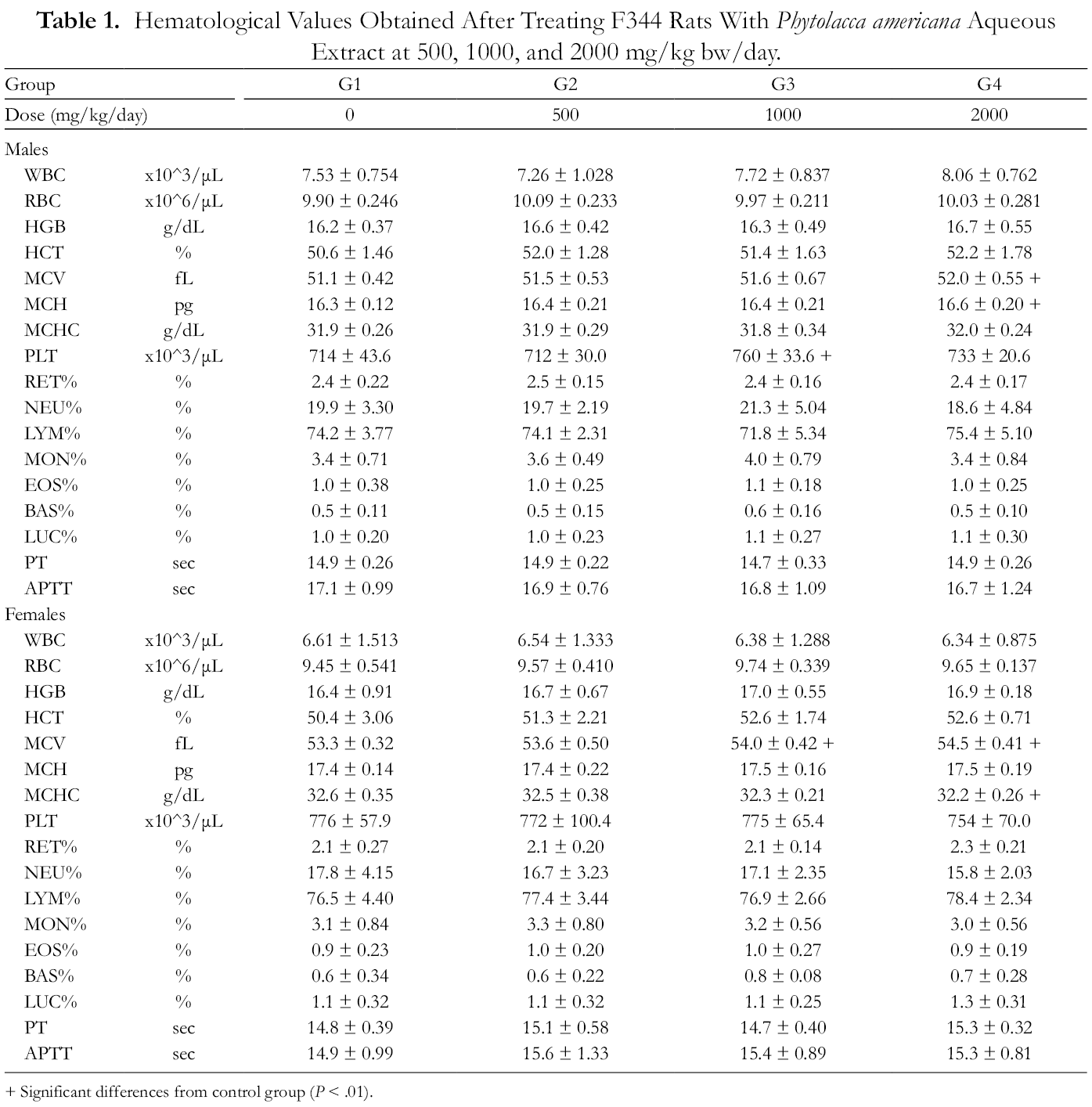
+ Significant differences from control group (P < .01).
Results of Clinical Chemistry Analysis After Treating F344 Rats With Phytolacca americana Aqueous Extract at 500, 1000, and 2000 mg/kg Bw/day.
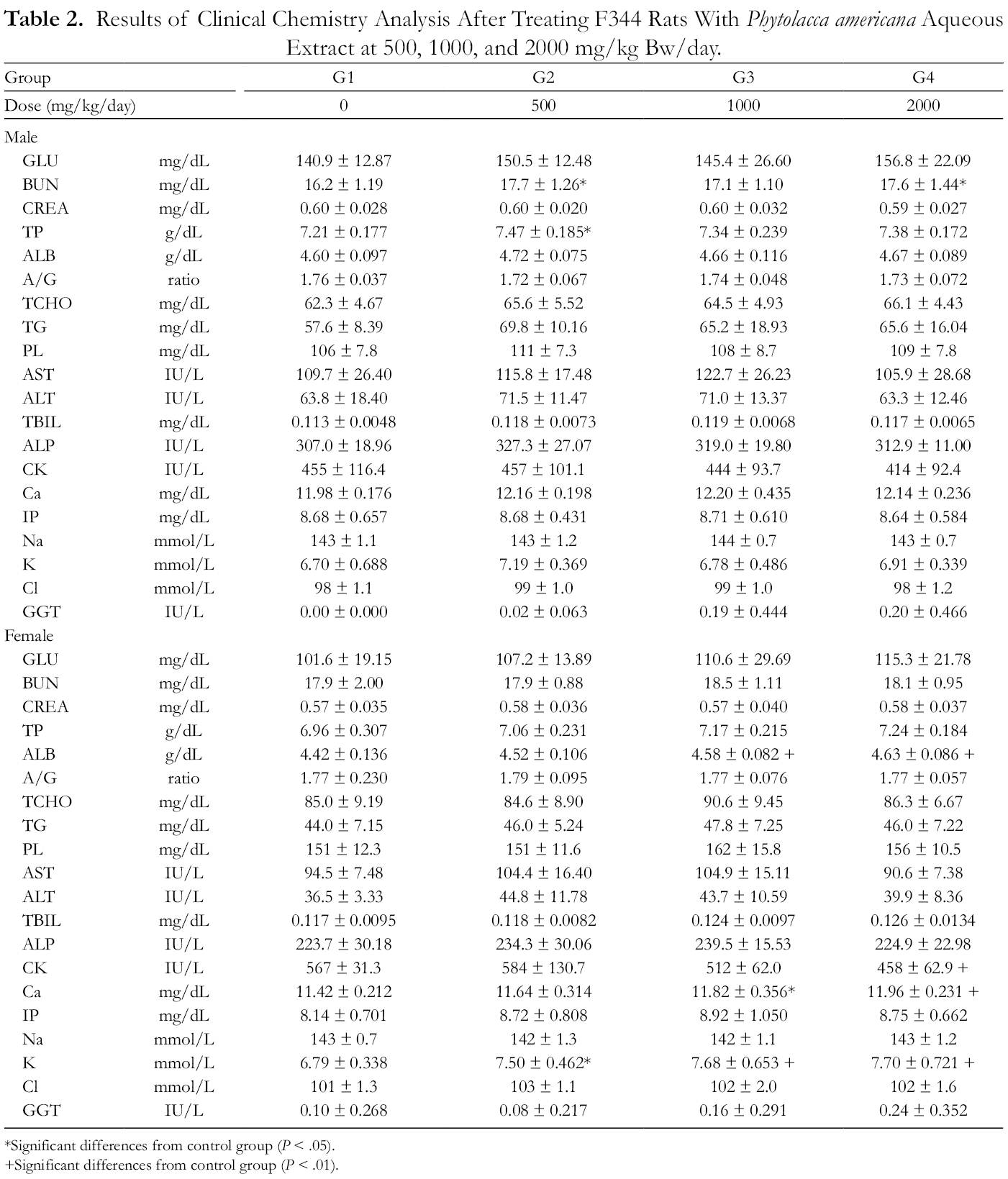
*Significant differences from control group (P < .05).
+Significant differences from control group (P < .01).
Gross Findings and Organ Weight Measurement
The relative weights of the heart and kidneys in the male rats and kidneys in the female rats significantly increased in a dose dependent manner compared with the control group (Tables 3 and 4). However, considering the normal relative organ weight (0.28, 0.32, and 0.68-0.75 for heart and kidneys, respectively), the increased relative weights were within the normal ranges. 16,18 In addition, no treatment-related gross findings were observed during necropsy in any of the rats.
Absolute Organ Weights in F344 Rats After Treatment With Phytolacca Americana Aqueous Extract at 500, 1000, and 2000 mg/kg bw/day.

*Significant differences from control group (P < .05).
Relative Organ Weights in F344 Rats After Treatment With Phytolacca americana Aqueous Extract at 500, 1000, and 2000 mg/kg Bw/day.

*Significant differences from control group (P < .05).
+Significant differences from control group (P < .01).
Histopathological Examination
Histopathological examination showed no treatment-related changes in any of the treated animals. However, inflammatory cell foci, mineralization, and tubular dilatation or regeneration were observed in the kidneys. Additionally, inflammatory cell foci, fibrosis, hepatodiaphragmatic nodules, and vacuolated areas were observed in the livers of the animals. Minimal changes were observed in the heart, spleen, thymus, lung, adrenal gland, and mesenteric lymph nodes. The abovementioned histopathological findings were randomly distributed between the control and treated groups, and were considered spontaneous or incidental. 19 Thus, no consistent treatment-related histopathological lesions were found in the rats of either sex. Furthermore, no PAAE-related macroscopic changes were observed in the liver or kidney of any of the rats (Table 5).
Histopathological Examination of F344 Rats After Treatment With Phytolacca Americana Aqueous Extract at 500, 1000, and 2000 mg/kg Bw/day.

In conclusion, the results of our 13-week repeated-dose toxicity study in F344 rats revealed no treatment-related changes in mortality, clinical signs, body weight, food consumption, clinical chemistry, gross findings at necropsy, or organ weight. Additionally, the findings from the ophthalmic examination, urinalysis, hematology study, and histopathological investigation showed no PAAE-related changes. Therefore, the “no-observed-adverse-effect-level” of PAAE may be considered to be 2000 mg/kg bw/day in male and female F344 rats under the present experimental conditions. However, further research is required to assess the activities of the compounds in PAAE, as well as the chronic toxicity of the extract.
Materials and Methods
Preparation of PAAE
Freeze-dried PAAE was prepared from the roots of P. americana and used for the study. The plant was collected from Korea by Prof. Byung-Sun Min (College of Pharmacy, Catholic University of Daegu, Republic of Korea). Phytolacca americana was authenticated by 5 independent experts. Botanical identification and high-performance liquid chromatography (HPLC) analyses were performed by Prof. Byung-Sun Min, and a voucher specimen (CUD-1289-1) was deposited at the herbarium of the College of Pharmacy of the Catholic University of Daegu. The authenticated plant was proved to be free of typical contaminations such as pesticides (total dichlorodiphenyltrichloroethane, total hexachlorocyclohexane, aldrin, endrin, and dieldrin), heavy metals (As, Cd, Hg, and Pb), and aflatoxin B1.
Air-dried and powdered roots of P. americana (40.1 kg) were boiled for 2 hours in 500 L of hot distilled water according to the standard hot water extraction method of the Korean Pharmacopeia. After filtration through a cheesecloth, the filtrates were collected and concentrated by lyophilization, yielding an aqueous extract of 9.74 kg [24.3% (w/w) of dried P. americana]. The active components, phytolaccoside E and phytolaccoside B, were measured using an established HPLC method (gradient solvent system of H2O:acetonitrile (30→90%, 0-60 minutes); flow rate 1 mL/min; UV detection at 210 nm; an Eclipse XDB-C-18, 5 µm, 4.6 × 150 mm column).
Freeze-dried PAAE powder was suspended in the vehicle (distilled water; Dai Han Pharm. Co., Ltd., Seoul, Korea) and used at concentrations of 50, 100, and 200 mg/mL. The appropriate quantity of the test drug was measured and mixed with distilled water to prepare a 200 mg/mL mixture, which was then diluted to prepare the other concentrations. Fresh mixtures were prepared weekly and stored in a container at approximately 5 °C for use.
Animals and Maintenance
Six week-old male and female specific-pathogen-free F344 rats (n = 48 each) were obtained from Orient Bio Inc. (Seongnam-si, Republic of Korea) for the study. The animals were acclimated to their new surroundings for 9 days, after which only the healthy animals were used in the study. Eighty rats were randomly assigned to 4 groups (1 control and 3 treatment groups) using the Path/Tox system (version 4.2.2; Xybion Medical Systems Corporation, Lawrenceville, NJ, USA). Each group consisted of 10 rats of each sex. The body weight ranges, prior to the start of dosing, were 115.4-134.3 g for the males and 93.5-113.2 g for the females. The animals were housed in polycarbonate cages with bedding (Laboratory animal Aspen bedding, Abedd Baltic Ltd., Jelgava, Latvia) throughout the study period. Sterilized tap water and pelleted food for experimental animals (PMI Nutrition International, USA) were given to the rats. The rats were kept in a room maintained under the following conditions: 12/12 hours light/dark cycle; temperature, 23 ± 3 °C; relative humidity, 50 ± 10%; air ventilation, 10–20 times/hour; and light intensity, 150–300 lux.
The protocol for the study was approved by the Institutional Animal Care and Use Committee of Korea Institute of Toxicology (Daejeon, Korea, IACUC No. 091125-G09136). All procedures were performed in compliance with Testing Guidelines for Safety Evaluation of Drugs (notification no. 2005-60, issued by the Korea Food and Drug Administration on October 21, 2005).
Treatment and Toxicity Assessment
Oral administration was chosen because it is the clinically-intended administration route for PAAE in humans and has been used in a previous nonclinical study. A dosing volume of 10 mL/kg was used based on calculations using the Path/Tox system. A previous 2-week study by the Korea Institute of Toxicology revealed that PAAE doses of 500, 1000, and 2000 mg/kg bw/day were well tolerated. 20 Therefore, the same doses were selected for this 13-week repeated-dose study. All the measurements and examinations were performed using the Path/Tox system. The experimental animals were checked for their general condition and behavior once daily throughout the acclimation period. However, examination and recording of clinical signs were done twice daily (before and after dosing) during the treatment period, and the day before the necropsy was performed.
The animals were weighed on the day of arrival, prior to randomization, before the first dose was administered and once weekly thereafter, and on the day of necropsy. Cage food consumption was recorded once during the acclimation period and once weekly during the treatment period. Individual food consumption was calculated and expressed as g/rat/day. External eye examinations were performed on all animals during the acclimation period; however, both external and fundus examination of the animals in the control and high-dose (2000 mg/kg/day) groups were performed using a binocular indirect ophthalmoscope (IO-H; Neitz Instruments Co., Ltd., Tokyo, Japan) at week 13 before the necropsy was performed. Prior to examination via binocular indirect ophthalmoscopy, a mydriatic compound (Midrin-P; Santen Pharmaceutical Co., Ltd., Osaka, Japan) was instilled into each eye.
Urine samples were collected overnight (for approximately 17 hours) from animals housed in metabolism cages during the last week of treatment. Each animal was housed in an individual metabolism cage. Food was withdrawn overnight before urine was collected; however, water was available ad libitum. Urinalysis was performed using a urine automatic analyzer (CliniTek-500; Bayer, Pittsburgh, PA, USA) and urine stick (Multistix 10 SG; Siemens, Munich, Germany). The following parameters were evaluated: urine volume, specific gravity, color, pH, ketone body, glucose, protein, occult blood, nitrite, bilirubin, and urobilinogen. Urine volume was measured by reading the scales on the urine collection tube. Additionally, a microscopic examination for urine cast, epithelial cells, white blood cells, and red blood cells was performed.
All the animals were fasted overnight before either the necropsy or blood sampling was done. The rats were anesthetized with isoflurane and blood samples were collected from the vena cava during necropsy for hematological and clinical chemistry analyses. The samples for hematological analysis were collected into tubes containing EDTA-2K. The following were analyzed using an ADVIA 120 hematology system (Bayer): white blood cell count (WBC), red blood cell count (RBC), hematocrit (HCT), hemoglobin concentration, MCV, MCH, MCHC, PLT, lymphocyte (LYM), monocyte (MON), neutrophil (NEU), basophil (BAS), eosinophil (EOS), and large unstained cell (LUC), and reticulocyte (RET) count. In addition, blood samples treated with 3.2% sodium citrate were analyzed for prothrombin time and activated partial thromboplastin time using ACL 9000 (Instrumentation Laboratory, Milano, Italy).
Blood samples for clinical chemistry analysis were collected into tubes without anticoagulant simultaneously with those for hematological analysis, placed at room temperature for at least 90 minutes, and centrifuged at 3,000 rpm for 10 minutes at room temperature to obtain serum. The following parameters were measured using an automatic analyzer (TBA 120FR NEO; Toshiba Corp., Tokyo, Japan): alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), BUN, creatinine (CREA), glucose (GLU), total cholesterol (TCHO), TP, albumin/globulin ratio (A/G), ALB, CK, triglycerides (TG), total bilirubin (TB), gamma glutamyl transferase (GGT), phospholipids (PL), calcium, chloride, inorganic sodium, phosphorus, and potassium.
After blood sampling, the animals were sacrificed by exsanguination through the vena cava and aorta under isoflurane anesthesia. Complete necropsy was performed on all the experimental animals. The absolute weights of the brain, pituitary gland, liver, kidneys, spleen, heart, lungs, thymus, salivary gland, thyroid gland, testes, epididymis, prostate, seminal vesicle, uterus, and ovaries were determined and the relative organ weights (% of terminal body weight) were calculated.
Following detailed external and internal examinations, tissue samples (skin, mammary gland, testes, epididymis, prostate, seminal vesicle, urinary bladder, ovaries, uterus, vagina, spleen, pancreas, stomach, duodenum, jejunum, ileum, cecum, colon, rectum, kidneys, heart, lungs, adrenal gland, liver, salivary gland, mesenteric lymph node, mandibular lymph node, thyroid gland, aorta, thymus, trachea, tongue, esophagus, sciatic nerve, skeletal muscle, sternum/marrow, femur/joint/marrow, thoracic spinal cord, harderian gland, brain, pituitary gland, and eyes/optic nerve) were taken from each animal and preserved in 10% neutral buffered formalin. The eyes and optic nerves were fixed in Davidson’s fixative, whereas the testes were fixed in Bouin’s fixative for approximately 48 hours and transferred into 70% ethanol. Preserved tissues from the animals in the control and high-dose groups, as well as the kidneys, liver, heart, spleen, thymus, lung, adrenal glands, and mesenteric lymph nodes of all animals in the medium- and low-dose groups, were embedded in paraffin wax. The tissues were sectioned, stained with hematoxylin and eosin, and examined microscopically.
Statistical Analysis
Data were statistically analyzed by multiple comparison methods. When Bartlett’s test showed no significant deviations from variance homogeneity, analysis of variance (ANOVA) was used to determine if any of the group means differed at the P < .05 level. Additionally, Dunnett’s test was used to determine differences in data between the control and treatment groups when the data were found to be significant from the ANOVA test. Furthermore, when significant deviations from variance homogeneity were observed from the Bartlett’s test, a nonparametric comparison test, Kruskal-Wallis (H) test, was conducted to determine if any of the group means differed at the P < .05 level. When a significant difference was observed from the Kruskal-Wallis (H) test, the Dunn’s Rank Sum test was conducted to quantify the specific pairs of group data that were significantly different from the mean. Fisher’s exact test was conducted to compare pairs of data (including prevalence and percentage). The level of probability was taken as 1% or 5%. Statistical analyses were performed by comparing the data from the different treatment groups with those of the control group using Path/Tox.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Korea Food and Drug Administration (09162KFDA540), the Ministry of Science and ICT (NRF-2016M3A9C4953144 and NRF-2020R1F1A1054226), research fund of Chungnam National University and the Korea Institute of Toxicology (grant no. KK‑2001).
