Abstract
Seven known flavonoids were isolated from the ethyl acetate fraction of the 80% ethanol extract of Sanchezia nobilis Hook. f leaves (EASN). The 7 known compounds as hispidulin (4′,5,7-trihydroxy-6-methoxyflavone) (1), hispidulin-7-O-α-galactopyranoside (2), hispidulin-4′-O-β-galactopyranoside (3), hispidulin-7-O-β-galacturonide methyl ester (4), apigenin-7-O-α-glucuronide (5), hispidulin-7-O-α-glucuronide (6), and hispidulin-7-O-β-glucuronide (7), all of which are first records for the Sanchezia genus. The structures of the compounds were confirmed using spectroscopic methods and by comparison with reported data. The antipeptic ulcer and analgesic effects of the extract were also tested in experimental animal models. At a 50 mg/kg/d dose, the ethyl acetate fraction significantly reduced mean ulcer score, gastric volume, and total acidity, and increased gastric pH, while also tending to reduce free acid in comparison with the model group. At a 100 mg/kg/d dose, the ethyl acetate fraction tended to have an analgesic effect. However, a dose of 300 mg/kg/d showed a significant elevation in pain. The extract was also continuously evaluated for subchronic toxicity for 28 days in rats but showed no effect at oral doses of 50 and 250 mg/kg/d. The results of this study have provided more scientific evidence for the chemical composition and use of SNF leaves to treat peptic ulcer disease in Vietnam.
Introduction
According to the classification of Leonard and Smith, the genus Sanchezia contains 59 species. 1 S. nobilis Hook. f (SHF) is distributed mainly in south-western America. One matsutake alcohols (1-octen-3-ol) and 4 matsutake alcohol glycosides (3-O-β-glucopyranosyl-1-octen-3-ol, 3-O-β-glucopyranosyl-(1-6)-β-glucopyranosyl-1-octen-3-ol, 3-O-β-arabinopyranosyl-(1-6)-β-glucopyranosyl-1-octen-3-ol, and 3-O-β-arabinopyranosyl-(1-6)-β-glucopyranosyl-(1-6)-β-glucopyranosyl-1-octen-3-ol) were isolated from the methanol extract of the aerial parts. 2 In 2014, 7 more substances were reported, including 3 flavonoids 3 : apigenin-7-O-β–glucopyranoside, apigenin-7-Ogentiobioside, and apigenin-7-O-β-glucuronopyranoside. Furthermore, a group of Vietnamese authors isolated 4 substances, including 2 flavonoids (quercitrin, hyperoside), from the leaf extract of SHF. 4 Our research group has also isolated 10 substances, including 2 alkaloids (fawcettidine and 13-O-acetyfawcettimine) and 6 flavonoids (apigenin, kaempferol, quercetin, afzelin, hyperoside, and rutin).5,6 This plant has also been studied for its antioxidant, anticancer, antibacterial, antifungal, and anti-inflammatory effects.7-9 In Vietnam, the antioxidant and anti-inflammatory effects of the plant have also been studied. 4 However, people use SHF leaves for the treatment of stomach pain by taking a few fresh leaves, which are then washed and chewed raw with a few grains of salt to relieve pain. 10 In addition to eating the raw leaves, dried leaves can also be used to make a decoction to drink daily instead of water. In this research, we collected the total ethanol extract of SHF leaves and then fractionated this with n-hexane, ethyl acetate, and water. From the ethyl acetate fraction, we isolated compounds and evaluated their subchronic toxicity and antipeptic ulcer effect on the Shay model, as well as their central analgesic effect using the hot plate model and a pain threshold meter.
Results and Discussion
Chemical Results
The structures of the compounds isolated from Sanchezia nobilis leaves (Figure 1) were assigned from NMR spectroscopic and ESI-MS and Q-TOF HRMS data, and by comparison with the respective literature.11-16 The compounds isolated were hispidulin (4′,5,7-trihydroxy-6-methoxyflavone) (1), hispidulin-7-O-α-galactopyranoside (2), hispidulin-4′-O-β-galactopyranoside (3), hispidulin-7-O-β-galacturonide methyl ester (4), apigenin-7-O-α-glucuronide (5), hispidulin-7-O-α-glucuronide (6), and hispidulin-7-O-β-glucuronide (7). All compounds are new records for the Sanchezia genus. Flavonoids have antioxidant, anti-inflammatory, and wound-healing effects.17,18
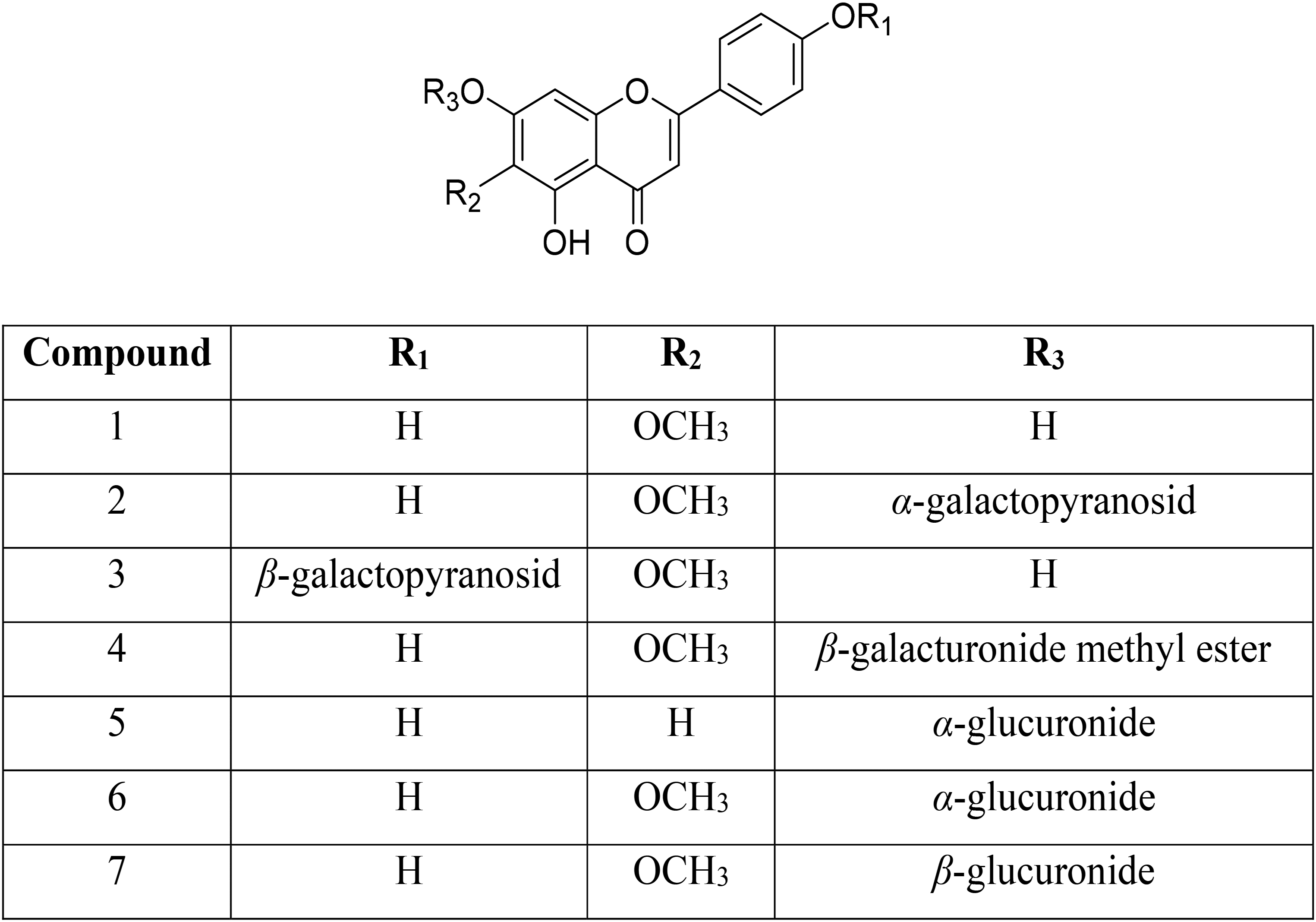
The chemical structures of compounds
Subchronic toxicity
It is evident that there are alkaloids in the plant extract which are potentially toxic. Medicines of medicinal origin are usually used for several days. Thus, for the purpose of safety, we conducted a subchronic toxicity test on this extract. During the experiment, mice in the control group and the 2 groups receiving EASN were active and agile, with bright eyes and dry stools. No significant difference between the 3 groups was observed.
After 14 and 28 days of drinking EASN, the weight of mice increased significantly compared to before the study (P < .05), but there was no difference in the degree of change in body weight between the control and test groups (P > .05).
The red and white blood cell counts, hemoglobin, hematocrit, MCV, and platelets in both group 1 (50 mg/kg/d) and group 2 (250 mg/kg/d) showed no significant differences from the control group both before and after the experiment (P > .05).
The serum biochemical indices after the subchronic administration of EASN to rats showed nonsignificant differences (P > .05) in liver functions between groups (ie, treatment and control groups), either before or after treatment (P > .05).
After 14 and 28 days of administration of EASN, no significant differences were observed in the concentrations of total bilirubin, total albumin, and total cholesterol in the blood of rats in both group 1 (50 mg/kg/d) and group 2 (250 mg/kg/d) when compared with the control group or when comparing the time points before and after the reagent was administered (P > .05). There were also no significant differences in serum creatinine levels (P > .05).
After 28 days of oral administration of EASN at doses of 50 and 250 mg/kg/d, no gross pathological changes were observed in the hearts, lungs, hepatosplenomegaly, pancreas, kidneys, or digestive systems of the experimental rats (in both the control and 2 test groups),
Effect of protecting ulcers according to Shay's method
From the dose of the total extract (150 mg/kg/d) in Shay's gastric ulcer model was found to be effective and the ethyl acetate fraction was approximately 35% of the total extract. So we used a 50 mg/kg/d dose of EASN for white rats to evaluate the antiulcer effect using Shay's model. Doses of 100 and 300 mg/kg/d were used to evaluate central analgesia, while doses of 50 and 250 mg/kg/d were administered to evaluate toxicity.
Ratio of ulcers in each group. Ulcers were not observed in the control mice (0/10 rats) whereas the incidence of gastric ulcers after pyloric ligation was 100% (10/10 rats) in the model group and the group using EASN. In the ranitidine group, the rate of rats with post-pyloric ulcers was 90% (9/10 rats). There was no difference in the percentage of rats with ulcers between the groups taking the drug in comparison to the model group (P > .05).
Negative group. The rate of grade II ulcers was 27.5% and of grade I lesions 72.5%. For the group taking ranitidine, the extent of ulcer lesions improved when compared to the model group, with a reduction in the rate of grade II lesions (6.8%). Moreover, the rate of grade I lesions was 93.2%. The group taking EASN at a 50 mg/kg dose also showed an improvement in ulcer severity in comparison to the model group (Table 1). Doses of ranitidine (50 mg/kg) and EASN (50 mg/kg) reduced the mean ulcer score and ulcer index in comparison to the model group (P < .01) (Table 2).
Effect on Mean Ulcer Score, Ulcer Index.

*P < .05; **P < .01; ***P < .001 (Kruskal-Wallis H test, χ2 = 25,527, df = 5).
Effect on Gastric Juice Volume, Free Acidity, Total Acidity, and Gastric pH.

*P < .05; **P < .01; ***P < .001 (Student t test).
Furthermore, ranitidine with a 50 mg/kg dose significantly reduced gastric volume and free acidity while increasing pH when compared with the model group (P < .001 and P < .05, respectively). After a 50 mg/kg/day dose, the EASN proved its capacity to reduce gastric volume and total acidity while also increasing pH in comparison to the model group (P < .001 and P < .05, respectively). Simultaneously, it also tended to reduce the free acidity when compared to the model group (P > .05) (Table 3) (Figure 2).
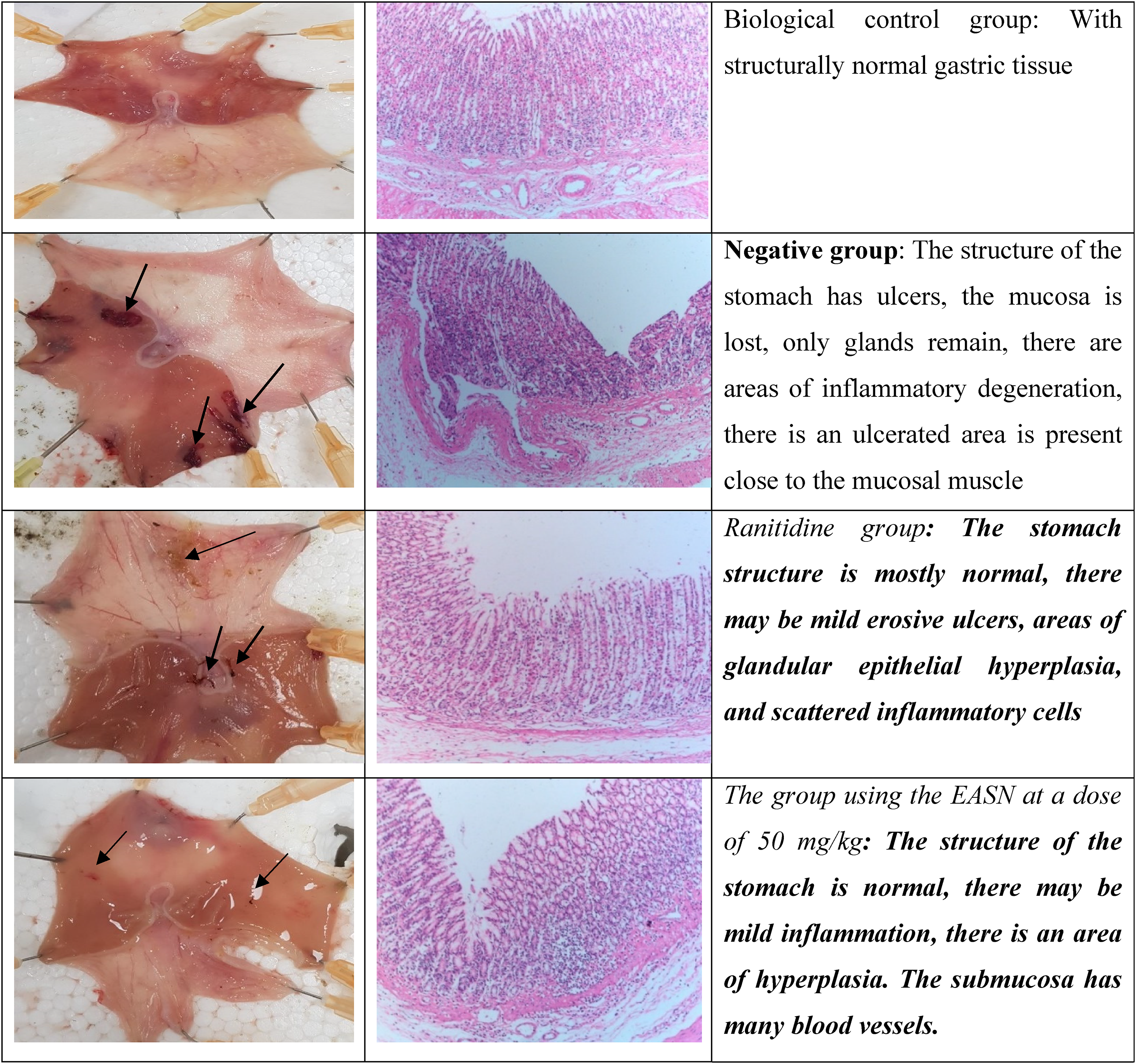
Macroscopic and microscopic (40 times magnification) images of rats’ stomach.
Effect on Reaction Time to Temperature of White Mice.

Differences from group 1 (control group): *P < .05; **P < .01; ***P < .001.
Analgesic effect determination using a hot plate
Codeine significantly prolonged the response time to temperature in treatment mice when compared to the control group (P < .001). At a 100 mg/kg/d dose, the EASN tended to prolong rats’ response time to temperature in comparison to the control group (P > .05). At a 300 mg/kg/d dose, the EASN markedly prolonged the response time to temperature in rats (P < .01) (Table 4).
Effect of Pain Force on Pain Threshold Meter.

Differences from group 1 (control group): *P < .05; **P < .01; ***P < .001.
Analgesic Effect Determination Using a Pain Threshold Meter (Dynamic Plantar Aesthesiometer)
Codeine had the effect of significantly increasing the force causing pain reflex on the pain threshold meter readings of treatment mice when compared with those of the biological control group (P < .001). At a 100 mg/kg/d dose, the EASN tended to prolong the pain response time of treatment rats when compared with biological controls; however, this difference was not statistically significant (P > .05). At a 300 mg/kg/d dose, the EASN significantly prolonged the pain response time of treatment rats compared based on their pain threshold meter readings in comparison to those of the biological controls (P < .05) (Table 5).
Effect of Pain Response Time on Pain Threshold Meter.

Differences from group 1 (control group): *P < .05; **P < .01; ***P < .001.
Based on the pain threshold meter readings of treatment rats, codeine significantly prolonged pain response time in comparison to the control group (P < .01). At a 100 mg/kg/d dose, the EASN tended to prolong the pain response time of treatment rats when compared with biological controls albeit statistically insignificant (P > .05). At a 300 mg/kg/d dose, the EASN significantly prolonged the pain response time of treatment rats compared based on their pain threshold meter readings in comparison to those of the biological controls (P < .05).
Studies on the acute toxicity of Sanchezia nobilis Hook.f showed that the n-hexane and ethyl acetate segment of leaves had LC50 values of 19.95 and 12.88 µg/mL, respectively, 8 while the dichloromethane extract of SHF root had significant toxicity with an IC50 of 2.52 g/mL. 9 In the preliminary study, we evaluated the acute toxicity of SHF leaf extracts in rats. Although it resulted in determining the maximum possible dose for rats, this study did not determine the LD50 dose. In the ethyl acetate segment, there were alkaloids 9 and also some isolated alkaloids. 5 This is a group of highly toxic/biologically active substances that can be toxic when used in large doses or long-term. Moreover, medicinal herbs are often used for a long period of time. Therefore, the study was conducted to evaluate the subchronic toxicity of the EASN. From the results obtained, the study can confirm that the use of SHF leaves is quite safe.
Shay's model of pyloric ligation caused excessive acid secretion, mucosal resorption, mucosal barrier disruption, ulceration, and gastric congestion in white rats. The research results show that with a 50 mg/kg/d dose (equivalent to the dose of medicinal herbs used in humans), significant ulcer reduction was observed in comparison to the model group. In particular, ulcer score, ulcer index, gastric juice volume, free acidity, and total acidity were significantly lower when compared to the model and ranitidine groups. In both models, central analgesia resulted in pain relief—especially with a 300 mg/kg/d dose, which provided good results with statistical significance (P < .05). Notably, this is close to the effect of reducing pain through codeine phosphate administration at a 20 mg/kg dose. Gastric ulcers often have symptoms of pain, heartburn, and burning in the epigastrium due to excess gastric acid. Endoscopic findings showed mucosal surface congestion, gastrointestinal mucosal detachment, and mild to severe ulceration. The study results also showed that the EASN was effective for all gastric ulcer symptoms. Based on this result, scientific evidence has been added to people's experience of using it.
In this segment, 7 flavonoids were isolated. Notably, 6 flavonoids were also previously isolated. Flavonoids are known for their antioxidant, antibacterial, anti-inflammatory, and antiviral effects.17-19 This may suggest that the effect of this fraction comes from flavonoids.
Conclusions
In conclusion, the findings of the present study highlight the nontoxic nature and promising antipeptic ulcer and analgesic effects of the ethyl acetate of Sanchezia nobilis Hook.f (SHF). Seven flavonoids, hispidulin (4′,5,7-trihydroxy-6-methoxyflavone) (1), hispidulin-7-O-α-galactopyranoside (2), hispidulin-4′-O-β-galactopyranoside (3), hispidulin-7-O-β-galacturonide methyl ester (4), apigenin-7-O-α-glucuronide (5), hispidulin-7-O-α-glucuronide (6), and hispidulin-7-O-β-glucuronide (7) were separated from the ethyl acetate of Sanchezia nobilis Hook.f. The present study represents the first time that the 7 known compounds were isolated from the Sanchezia genus. Since flavonoids have strong antioxidant and anti-inflammatory effects, this group of substances may be responsible for the main effect of the ethyl acetate fraction of SHF leaves. Our research results provide additional scientific evidence supporting the use of SHF leaves to treat peptic ulcer disease in Vietnam.
Materials and Methods
Plant Material
The leaves of Sanchezia nobilis Hook.f (SHF) was accumulated in Nam Dinh province during January 2018 and authenticated by the School of Medicine and Pharmacy, Vietnam National University, Hanoi, Vietnam (No: 190DV18 SMP-VNU). A voucher specimen has been deposited in the aforementioned institution.
General Procedures
Nuclear magnetic resonance (1H, 13C, HMBC, HSQC, and COSY NMR) experiments were conducted using both a Bruker AM500 FT-NMR and a Bruker AvanceNEO 600 NMR spectrometers for 1H (500 and 600 MHz) and 13C (125 and 150 MHz) in DMSO-d6 or MeOH-d4 as solvents, using tetramethylsilane as an internal standard. Electrospray ionization-mass spectrometry (ESI-MS) spectra were recorded on an LC/MS-8045 Shimadzu system and an Agilent 1100 LC/MSD in both negative and positive modes. An Agilent 1290 LC coupled to a quadrupole-time of flight (Q-TOF) 6530 UHD accurate mass spectrometer (MS) was used to tentatively identify the metabolites in the extracts. Normal-phase and reversed-phase chromatography were performed on silica gel (Merck, 63-200 µm) and YMC RP-18 resins (30-50 µm, Fuji Silysia Chemical). Precoated silica gel 60 F254 plates (Merck) and RP-18 F254S plates (Merck) were carried out thin-layer chromatography. The spots were detected by spraying with a 2% FeCl3-ethanol solution. Preparative high-performance liquid chromatography was operated by using a Shimadzu Preparative High-performance liquid chromatography (LC-20AP pump, SPD-M20A PDA detector, HS C18 column 25 cm × 21.2 mm, 10 μm).
Experimental Animals
This study used adult Wistar rats of both sexes weighing 180 to 250 g, as well as Swiss albino mice weighing 20 ± 2 g. All animals were provided by the Central Institute of Hygiene and Epidemiology. All animals were kept in appropriate cages at standard controlled laboratory conditions (25 °C, 12-h light/12-h dark cycle). Mice/rats were provided with food and water ad libitum. Experiments were performed at the Institute of the Department of Pharmacology - Hanoi Medical University following Vietnamese ethics laws regarding the care and use of laboratory animals. This study was approved by the Scientific Board Committee of Hanoi Medical University (ref number: IRB00003121).
Extraction and Isolation
The dried leaves of Sanchezia nobilis Hook.f (SHF) (3.8 kg) were extracted with 80% EtOH (3 times × 3 days) (10L × 3 times) at normal temperature. After removal of the solvent under reduced pressure, a residue extract (316 g) was afforded, successfully suspended in H2O, and extracted with n-hexane and ethyl acetate, respectively. After concentration in vacuo, the ethyl acetate fraction (119 g) was separated by normal-phase column chromatography (CH2Cl2:MeOH:H2O, 2:2:1) to obtain 5 fractions (E1 through E5). Fraction E2 (2.3 g) was purified by normal-phase flash chromatography using a silica column (23 g) and gradient system of dichloromethane:CH3OH with a flow of 20 mL/min to give compound
Spectral Data
Hispidulin (4′,5,7-trihydroxy-6-methoxyflavone) (1)
A yellow powder. LC-ESI MS m/z 301.0 [M + H]+ (calcd for C16H13O6+ 301.1), m/z 298.8 [M − H]− (calcd for C16H11O6−299.1). 1H NMR (600 MHz, DMSO-d6) δH (ppm): 3.75 (3H, s, OCH3), 6.59 (1H, s, H-3), 6.92 (2H, d, J = 8.8 Hz, H-3′,5′), 6,77 (1H, s, H-8), 7.92 (2H, d, J = 8.8 Hz, H-2′,6′), 13,07 (1H, brs, 5-OH). 13C NMR (150 MHz, DMSO-d6) δC (ppm): 60.0 (OCH3), 1313 (C-6), 94,2 (C-8), 104.1 (C-10), 116.0 (C-3′,5′), 121.2 (C-1′), 128.5 (C-2′,6′), 102.4 (C-3), 152.4 (C-9), 157.2 (C-7), 152.4 (C-5), 161.2 (C-4′),163.8 (C-2), 182.1 (C-4).
Hispidulin-7-O-α-galactopyranoside (2)
A yellow powder. LC-ESI MS m/z 460.9 [M − H]− (calcd for C22H21O11− 461.1), [M + Na]+ 485.0 (calcd for C22H23O11Na+ 485.1). 1H NMR (600 MHz, DMSO-d6) δH (ppm): 3.21 (1H, t, J = 8.9 Hz, H4″), 3.45–3.33 (3H, m, H-5″,3″,2″), 3.46 (1H, m, H-6″b), 3.75 (1H, d, J = 10.6 Hz, H-6″a), 3.76 (3H, s, OCH3), 5.10 (1H, d, J = 4.7 Hz, H-1″), 6.81 (1H, s, H-8), 6.90 (1H, d, J = 8.8 Hz, H-3′,5′), 7.00 (1H, s, H-3), 7.92 (2H, d, J = 8.4 Hz, H-2′,6′), 13.00 (1H, brs, 5-OH). 13C NMR (125 MHz, DMSO-d6) δC (ppm): 60.3 (C-6″), 60.6 (OCH3), 69.6 (C-4″), 73.2 (C-2″), 76.8 (C-3″), 77.3 (C-5″), 1326 (C-6), 100.2 (C-1″), 94,3 (C-8), 105.7 (C-10), 116.3 (C-3′,5′), 120.1 (C-1′), 128.6 (C-2′,6′), 102.3 (C-3), 152.2 (C-9), 152.5 (C-5), 156.4 (C-7), 162.7 (C-4′),164.5 (C-2), 182.3 (C-4).
Hispidulin-4′-O-β-galactopyranoside (3)
A yellow powder. Q-TOF HRMS m/z 463.1232 [M + H]+ (calcd for C22H23O11 463.1235), m/z 461.1088 [M − H]− (calcd for C22H21O11− 461.1089), m/z [M + Cl]− 497.0851 (calcd for C22H22O11Cl− 497.0856). 1H NMR (600 MHz, DMSO-d6) δH (ppm): 3.19 (1H, t, J = 9.0 Hz, H-4″), 3.38–3.27 (3H, m, H-5″,2″,3″), 3.49 (1H, m, H-6″b), 3.65 (3H, s, OCH3), 3.70 (1H, dd, J = 2.4 Hz, 11.7 Hz, H-6″a), 4.98 (1H, d, J = 7.5 Hz, H-1″), 6.05 (1H, s, H-8), 6.54 (1H, s, H-3), 7.15 (2H, J = 8.9 Hz, H-3′,5′), 7.90 (2H, d, J = 8.9 Hz, H-2′,6′), 12.78 (1H, brs, 5-OH). 13C NMR (125 MHz, DMSO-d6) δC (ppm): 58.9 (OCH3), 60.7 (C-6″), 69.7 (C-4″), 73.3 (C-2″), 76.6 (C-5″), 77.2 (C-3″), 96.2 (C-8), 99.5 (C-10), 100.0 (C-1″), 133.8 (C-6), 116.6 (C-3′,5′), 125.0 (C-1′),127.5 (C-2′,6′), 1028 (C-3), 153.4 (C-9), 151.6 (C-5), 159.6 (C-4′),160.8 (C-2), 1562 (C-7), 179.8 (C-4).
Hispidulin-7-O-β-galacturonide methyl ester (4)
Yellow powder. LC-ESI MS m/z 488.9 [M − H]− (calcd for C23H22O11− 489.1), m/z 491.0 [M + H]+ (calcd for C23H23O11+ 491.1). 1H NMR (500 MHz, DMSO-d6) δH (ppm): 3.42–3.37 (3H, m, H-2″,3″,4″), 3.66 (3H, s, 6″-OCH3), 3.75 (3H, s, 3-OCH3), 4.20 (1H, d, J = 9.6 Hz, H-5″), 5.35 (1H, d, J = 7.2 Hz, H-1″), 6.86 (1H, s, H-3), 6.95 (2H, d, J = 8.8 Hz, H-3′,5′), 7.07 (1H, s, H-8), 7.95 (2H, d, J = 8.8 Hz, H-2′,6′), 13.0 (1H, s, 5-OH). 13C NMR (125 MHz, DMSO-d6) δC (ppm): 52.0 (6″-OCH3), 60.4 (3-OCH3), 71.3 (C-4″), 72.8 (C-2″), 75.3 (C-5″), 75.7 (C-3″), 93.9 (C-8), 99.4 (C-1″),102.8 (C-3), 105.9 (C-10), 116.1 (C-3′,5′),121.1 (C-1′), 128.6 (C-2′,6′), 132.5 (C-6), 152.2 (C-9), 152.7 (C-5), 156.0 (C-7), 161.4 (C-4′),164.4 (C-2), 169.2 (C-6″), 182.4 (C-4).
Apigenin-7-O-α-glucuronide (5)
Yellow powder. LC-ESI MS m/z 446.8 [M + H]+ (calcd for C21H19O11+ 447.1), m/z 444.9 [M − H]− (calcd for C21H17O11− 445.1). 1H NMR (500 MHz, DMSO-d6) δH (ppm): 3.30–3.25 (3H, m, H-3″, 2″,4″), 3.68 (1H, d, J = 7.2 Hz, H-5″), 5.09 (1H, d, J = 4.9 Hz, H-1″), 6.41 (1H, s, H-8), 6.81 (2H, s, H-6, H-3), 6.91 (2H, d, J = 7.7 Hz, H-3′,5′),7.90 (2H, d, J = 7.7 Hz, H −2′, H-6′), 12.97 (1H, brs, 5-OH). 13C NMR (125 MHz, DMSO-d6) δC (ppm): 182.4 (C-4), 173.4 (C-6″), 164.7 (C-2), 163.4 (C-7), 162.1 (C-4′), 157.5 (C-5), 157.4 (C-9), 128.9 (C-2′,6′), 121.2 (C-1′), 116.5 (C-3′,5′), 105.8 (C-10), 103.4 (C-6), 100.1 (C-1″), 99.9 (C-8), 95.1 (C-3), 76.8 (C-3″), 74.6 (C-5″), 73.4 (C-2″), 72.4 (C-4″).
Hispidulin-7-O-α-glucuronide (6)
Yellow powder. LC-ESI MS m/z 476.9 [M + H]+ (calcd for C22H21O11+ 477.1), [M − H]− 474.9 (calcd for C22H19O12− 475.1). 1H NMR (500 MHz, DMSO-d6) δH (ppm): 3.33–3.29 (3H, m, H-2″,3″,4″), 3.74 (1H, m, H-5″), 3.76 (3H, s, OCH3), 5.15 (1H, d, J = 5.8 Hz, H-1″), 6.73 (1H, s, H-3), 6.85 (2H, d, J = 8.2 Hz, H-3′,5′), 6.91 (1H, s, H-8), 7.82 (2H, d, J = 8.2 Hz, H-2′,6′), 12.97 (1H, s, 5-OH). 13C NMR (125 MHz, DMSO-d6) δC (ppm): 60.3 (OCH3), 76.6 (C-3″), 74.3 (C-5″), 73.0 (C-2″), 72.0 (C-4″), 1325 (C-6), 99.6 (C-1″), 94.1 (C-8), 105.7 (C-10), 116.0 (C-3′,5′), 120.6 (C-1′), 128.4 (C-2′, 6′), 1024 (C-3), 152.2 (C-9), 152.5 (C-5), 156.4 (C-7), 161.8 (C-4′),164.3 (C-2), 173.1 (C-6″), 182.2 (C-4).
Hispidulin-7-O-β-glucuronide (7)
Yellow powder. LC-ESI MS m/z 474.9 [M − H]− (calcd for C22H19O12− 475.1). 1H NMR (600 MHz, MeOH-d4) δH (ppm): 3.69–3.61 (3H, m, H-4″,3″,2″), 3.96 (3H, s, OCH3), 4.01 (1H, d, J = 9.1 Hz, H-5″), 5.22 (1H, d, J = 7.4 Hz, H-1″), 7,02 (1H, s, H-8), 6.96 (2H, d, J = 8.8 Hz, H-3′,5′), 6.68 (1H, s, H-3), 7.92 (2H, d, J = 8.8 Hz, H-2′,6′). 13C NMR (125 MHz, DMSO-d6) δC (ppm): 60.3 (OCH3), 71.9 (C-4″), 73.0 (C-2″), 73.9 (C-5″), 76.7 (C-3″), 94.1 (C-8), 99.7 (C-1″), 102.4 (C-3), 105.6 (C-10), 115.9 (C-3′,5′), 120.6 (C-1′),128.4 (C-2′,6′), 132.4 (C-6), 152.1 (C-9), 152.4 (C-5), 156.5 (C-7), 161.7 (C-4′), 164.3 (C-2), 172.1 (C-6″), 182.3 (C-4).
Subchronic Toxicity
Subchronic (28-day) oral toxicity studies of the EASN were conducted with white rats.20,21 A repeat-dose oral toxicity study was conducted according to OECD Guideline 407. 20 The control group was administered water at o dose of 10 mL/kg body weight. Rats in groups 2 and 3 were administered the EASN doses of 50 and 250 mg/kg body weight, respectively. The extract was administered at the same time each day every day for 28 days and observed at least twice a day for morbidity and mortality. All rats were assessed weekly for body weight. Experimental animal blood samples were collected for hematology and biochemistry analysis. At the termination of the experiment, rats were operated on to observe all organs’ macroscopic structures. Histology samples (liver, kidney) were collected from 30% of rats to assess microscopic morphology.
Antiulcer Essay
Effect of treating stomach ulcers using a pyloric ligation model
The method developed by Shay et al,
22
with slight modifications, was used to perform pyloric ligation on the rats. A total of 40 rats were randomly divided into 4 groups of 10 rats each. The control group was group 1, which was administered with the solvent used to mix the medicine (vehicle) (p.o). The negative group was group 2, which was orally administered with the solvent used to mix the medicine (vehicle) (p.o). The positive group was control 3, which was orally administered 50 mg/kg ranitidine at 1 - Ratio of rats with a gastric ulcer in each group. - Average number of ulcers: Assesses the extent of damage by counting the number of ulcers and scoring according to the method23,24: +0 points: no ulcers; +1 point: superficial ulcer; +2 points: deep ulcers; +3 points: mucosal perforation ulceration. - Ulcer index (Ui): Calculated according to the formula - Macroscopic image of the mouse stomach. - Microscopic images (40× magnification) of the stomach of 30% of white rats in each group.
where Un is the average number of ulcers for one mouse, Us is the average ulcer score, and Up is the percentage of rats with ulcers.
Analgesic Assay Using a hot plate 25
White mice were randomly divided into 4 groups of 10 each:
- Group 1 (control): Mice were administered water at 20 mL/kg/d. - Group 2 (positive control): Mice were administered codeine phosphate at 20 mg/kg. - Group 3: Mice were administered the EASN at 100 mg/kg/d. - Group 4: Mice were administered the EASN at 300 mg/kg/d.
Mice from all groups were given distilled water/positive control/reagent once per day in the morning, with a volume of 20 mL/kg/d for 5 consecutive days. Rats’ reaction times to temperature were measured before taking the reagent and 1 h after the last dose of the reagent. Mice were placed on a hot plate and were always maintained at 56 °C by the thermostatic system. The time from the mouse placed on the hot plate to it licking its hidden legs or jumping was considered reaction time. We eliminated mice that reacted too quickly (before 8 s) or slowly (after 30 s). Comparison of reaction times to thermal stimulation before and after reagent ingestion (expressed as a percentage).
Analgesic Assay Using a Pain Threshold meter26,27
White mice were randomly divided into 4 groups with 10 mice each. Mice in the groups were given distilled water/positive control/reagent once a day in the morning, with a volume of 20 mL/kg/d for 5 consecutive days. We measured the pain response time and pain force before taking the reagent and 1 h after taking the last reagent. The increase in pain threshold and time to painful stimuli before and after reagent ingestion for drug-treated groups was compared to that of the control group (expressed as a percentage).
Statistics
Statistical analysis was performed using the software SigmaPlot 12.0 (SYSTAT Software Inc). Data are presented as the mean ± SD. Research data before and after drinking distilled water/positive control/reagent for the same batch were statistically processed using the paired t test. Comparisons between biological controls and positive/reagent oral controls were conducted using Student t test. Differences were considered statistically significant when P < .05.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221126306 - Supplemental material for Flavonoids and the Subchronic Toxicity, Antipeptic Ulcer, and Analgesic Effects of the Ethyl Acetate Soluble Fraction of the Ethanol Extract from Sanchezia nobilis Hook. f Leaves
Supplemental material, sj-docx-1-npx-10.1177_1934578X221126306 for Flavonoids and the Subchronic Toxicity, Antipeptic Ulcer, and Analgesic Effects of the Ethyl Acetate Soluble Fraction of the Ethanol Extract from Sanchezia nobilis Hook. f Leaves by Thi Xuan Bui, Minh Ngoc Tran, Thanh Ha Tran and Thi Thu Hien Dang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
