Abstract
Objective
Hydroxytyrosol (HT), a polyphenolic component identified in olive oil and leaves, represents one of the most extensively investigated natural phenol due to its diverse beneficial biological properties. Recent studies have shown that HT exhibits protective effects on reproductive function and can improve sperm quality. However, the precise effect and underlying mechanism of HT on oligozoospermia remain unclear. This study aims to investigate the effect and mechanism of HT across varying concentrations on oligozoospermia.
Methods
Oligozoospermia was induced in ICR male mice via intraperitoneal injection of busulfan (BUS, 30 mg/kg). The mice were randomly categorized into five groups as Normal, BUS, HT 10 mg/kg, HT 30 mg/kg and HT 50 mg/kg. The efficacy of HT on oligozoospermia was assessed through sperm concentration and motility using a computer-assisted sperm analysis system. Testicular and epididymal histology was examined via hematoxylin-eosin staining, while cytoskeletal integrity was evaluated by immunofluorescence. Hormone levels, along with reactive oxygen species (ROS) and malondialdehyde (MDA) were examined by ELISA kits. Differentially expressed genes were identified through RNA sequencing, and the expression levels of key genes were validated by qPCR.
Results
The results indicated that HT (50 mg/kg) significantly increased sperm concentration, promoted morphological recovery of testicular tissue, heightened the levels of serum testosterone, follicle-stimulating hormone, luteinizing hormone, as well as decreased the accumulation of ROS and MDA in mice with oligozoospermia. RNA sequencing analysis and RT-qPCR results revealed that HT could restore spermatogenesis and ameliorate oligozoospermia by up-regulating genes related to cilium organization, microtubule-based movement, and spermatid differentiation and development.
Conclusions
HT demonstrates therapeutic efficacy against busulfan-induced oligozoospermia. The mechanism of HT on oligozoospermia may be related to its influence on cilium and microtubule-based movement, along with the differentiation and development of spermatids. HT shows potential as a prospective therapeutic intervention for the treatment of oligozoospermia.
Introduction
Infertility affects about 15% of couples worldwide. 1 Nearly 50% of infertility cases are attributed to male factors. 2 Approximately 30–40% of infertile males still have undetermined causes, a condition known as idiopathic male infertility. 3 oligozoospermia is one of the prevalent causes of idiopathic male infertility, characterized by low sperm count and poor sperm motility. Given the elusive etiology of the disease, empirical medical therapy is commonly employed in clinical practice, yet its efficacy in oligozoospermia remains limited4–6. Natural bioactive substances provide novel avenues for pharmacological interventions in oligozoospermia.
Hydroxytyrosol (3,4-dihydroxyphenylethanol, HT) is a natural polyphenol compound, which is mainly found in the fruits, branches, and leaves of olive plants. Numerous studies have demonstrated that HT possesses a broad spectrum of pharmacological and biological activities, such as anti-aging, anti-proliferation, anti-cancer, anti-inflammatory, antioxidant, and neuroprotection properties7–11. Studies have shown that HT has anti-aging effects and can be used to mitigate age-related diseases. 12 Additionally, HT has been shown to induce anti-proliferative effects and stimulate apoptosis in both human breast cancer and prostate cancer cells.13,14 HT has displayed promising results in ameliorating colitis symptoms 15 and mitigating the inflammatory response in osteoarthritis chondrocytes. 16 HT can protect pancreatic cells from damage and death through its antioxidant effects. 17 HT has garnered increasing attention due to its multiple pharmacological activities. Recently, studies have shown that HT has a protective effect on male sperm. Studies have indicated that HT can effectively improve the quality of sperm from pigs, bulls, and bucks in vitro18–21. HT may help maintain human sperm through its antioxidant activity after centrifugation. 22 In recent years, studies have illustrated the therapeutic effect of HT on testicular damage induced experimentally by diabetes. 23 Studies demonstrated that Hidrox, containing 40–50% HT, can protect testes from cyclophosphamide-induced damage. 24 However, the precise effect and underlying mechanism of HT in vivo on oligozoospermia remain elusive.
Busulfan (BUS) is an extensively utilized chemotherapeutic agent, selectively eliminating endogenous germ cells from the testicular tissue of various animal models. 25 In the present investigation, we have employed an oligozoospermia mouse model induced by BUS administration at a dosage of 30 mg/kg to explore the role and pivotal mechanisms of HT across a spectrum of concentrations. The results showed that HT at a dosage of 50 mg/kg promoted spermatogenesis by regulating genes associated with ciliary microtubule motility (Ccdc65, Dnah2, Zmynd10, Atp1a4, Tssk4) and spermatid differentiation and development (Zfy2, Rnf17, Tssk3, Prm1, Tnp1). Our study provides a theoretical foundation for the therapeutic application of HT, a naturally active compound, in the further development and utilization for treating oligozoospermia.
Materials and Methods
Chemicals and Reagents
HT (purity > 98%) was purchased from Macklin (Shanghai, China). BUS and DMSO were purchased from Sigma-Aldrich (Shanghai, China). Hematoxylin and Eosin (HE) were supplied by Beyotime Biotechnology (Shanghai, China). Testosterone (T), luteinizing hormone (LH), follicle-stimulating hormone (FSH), superoxide dismutase (SOD), malondialdehyde (MDA), and reactive oxygen species (ROS) enzyme-linked immunosorbent assay (ELISA) kits were purchased from Herbal Source Biotechnology (Nanjing, China). α-Tubulin antibody was purchased from Cell Signaling Technology. Alexa Fluor 555-labeled Donkey Anti-Rabbit IgG (H + L) and Actin-Tracker Green-488 were purchased from Beyotime Biotechnology (Shanghai, China). Antifade mounting medium for fluorescence (with DAPI) was supplied by Biosharp (Anhui, China). AceQ® Universal SYBR® qPCR Master Mix and HiScript II QRT SuperMix for qPCR were obtained from Vazyme Biotech (Nanjing, China).
Animals and Treatment
Eight-week-old ICR male mice (30 ± 2 g) were obtained from the Experimental Animal Center of Nantong University. Mice were housed under room temperature at 22–24 °C in a 12-h light/dark cycle. Fifty mice were randomly divided into five groups, each consisting of 10 individuals: Normal group, BUS group, BUS + HT low dose group, BUS + HT medium dose group, and BUS + HT high dose group. Each group was treated as follows:
The normal group was fed with normal diet and water.
The BUS group received a single intraperitoneal injection of BUS at 30 mg/kg body weight (BUS were dissolved in 5% DMSO aqueous solution) to induce a mouse model with oligozoospermia. Two weeks later, the mouse received daily intragastric administration of normal saline for four consecutive weeks.
The BUS + HT low dose group received a daily intraperitoneal injection of HT at 10 mg/kg body weight (3 mg HT dissolved in 1.0 mL normal saline) for four consecutive weeks, starting two weeks after the intraperitoneal injection of BUS.
The BUS + HT medium dose group received a daily intraperitoneal injection of HT at 30 mg/kg body weight (9 mg HT dissolved in 1.0 mL normal saline) for four consecutive weeks, starting two weeks after the intraperitoneal injection of BUS.
The BUS + HT high dose group received a daily intraperitoneal injection of HT at 50 mg/kg body weight (15 mg HT dissolved in 1.0 mL normal saline) for four consecutive weeks, starting two weeks after the intraperitoneal injection of BUS.
After the final administration, all mice were weighed, and then euthanized under intraperitoneal injection of pentobarbital sodium, blood samples were collected in gel glass tubes, and centrifuged at 3000 g, 4 °C for 10 min to obtain serum samples. Testes and epididymides were subsequently excised and weighed separately. Additionally, five testes and epididymides from each group were fixed in 4% paraformaldehyde solution, while the remaining samples were snap-frozen in liquid nitrogen and stored at −80 °C, the following experiments were performed immediately.
Analysis of Epididymal Sperm Parameters
The cauda epididymis was cut into fragments and incubated in 2 mL Tyrode's solution at 37 °C for 15 min to allow the spermatozoa to swim out. Aliquots of 20 μL of the solution was sampled for sperm characterization utilizing a computer-assisted sperm analysis (CASA) system, with subsequent collection of digital screenshots depicting the spermatozoa under microscopic examination.
Histological Analysis
Testes and epididymides from each group were fixed overnight in 4% paraformaldehyde, and then dehydrated in 10% and 30% sucrose overnight. Frozen sections were cut into 10 μm thickness, and stained with hematoxylin and eosin (HE). Finally, the sections were observed by a light microscope.
Immunofluorescent Staining
Frozen sections were blocked with 10% donkey serum and 0.1% Triton X-100 in 2% BSA for 60 min at room temperature. Then, sections were incubated with diluted α-Tubulin antibody (1:25, Cell signaling Technology) at 4 °C overnight. Normal rabbit IgG was used as a negative control. The primary antibody was discarded and washed three times (5 min each) with PBST, after which the sections were incubated with the secondary antibody (Alexa Fluor 555-labeled Donkey Anti-Rabbit IgG (H + L), 1:500, Beyotime Biotechnology) for one hour at room temperature in the dark. After removing the secondary antibody and washing three times (5 min each) with PBST, the sections were added with Actin-Tracker Green-488 (1:200, Beyotime Biotechnology) for one hour at room temperature in the dark. Finally, an antifade mounting medium for fluorescence (with DAPI) (Biosharp) sealed all sections. The sections were observed by confocal microscopy (Leica TCSSP8, Leica). Semi-quantitative analysis of fluorescence intensity was performed using image J software.
Serum Hormone Levels
Serum levels of T, FSH, and LH were measured by ELISA kits according to the manufacturer's instructions. The sensitivities of the mouse T, LH, and FSH assays were 0.0375 ng/mL, 17.5 pg/mL, and 0.125 U/L, respectively. The coefficients of variation for both intra- and inter-assay variability were below 10% across all conducted assays.
Assessment of Antioxidant Activity
The SOD activity and MDA, ROS concentration were examined according to the manufacturer's instructions. The intra-assay and inter-assay coefficients of variation were < 10% for either of the assays conducted. The sensitivities of the mouse SOD, MDA and ROS assays were 0.25 pg/mL, 0.75 nmol/mL, and 5 IU/mL, respectively.
RNA Sequencing and Data Analysis
Total RNA was extracted from the testes using a Trizol reagent under the manufacturer's instructions. The identification and quantification of total RNA were performed using a NanoDrop spectrophotometer and 2100 Bioanalyzer. The cDNA libraries were constructed utilizing the PCR products. These libraries were subjected to sequencing using the BGISEQ-500/MGISEQ-2000 System (BGI-Shenzhen, China). Fastp was employed for comprehensive quality control and raw data cleanup. The clean data were mapped to the mouse reference genome GRCm38 using STAR software. FeatureCounts software was utilized to enumerate the expression levels of each gene across all samples. Lastly, the differentially expressed genes between different samples were analyzed using the ClusterProfiler package, incorporating Gene Ontology (GO) annotations and Kyoto Encyclopedia of Genes and Genomes (KEGG) annotations.
Quantitative Real-Time PCR
Total RNA was extracted from the testicular tissue, followed by the synthesis of cDNA through the application of a reverse transcription kit. Real-time polymerase chain reaction (PCR) assays were performed with the AceQ® Universal SYBR® qPCR Master Mix (Vazyme Biotech, Nanjing, China), on a CFX Connect Real-Time PCR Detection System (Bio-RAD, USA). The relative mRNA abundance was quantified and normalized to the mean of β-actin mRNA levels. The sequences for the primers are detailed in Table 1.
The Sequences for the Primers are Listed.

Statistical Analysis
Data analysis was conducted utilizing the GraphPad Prism version 8.1 software. The Shapiro–Wilk test for normality was implemented to confirm the Gaussian distribution of the dataset. Inter-group differences within the experimental paradigm were assessed using one-way analysis of variance (ANOVA), and pairwise comparisons were conducted with Student's t-tests. The dataset, obtained from a minimum of three independent replications, was presented as the mean ± standard deviation (SD). Statistical significance was determined at the threshold of P < 0.05.
Results
HT Treatment Reverses the Damage of the Testis and Epididymis Induced by Busulfan
To investigate the impact of HT on oligozoospermia in mouse models, parameters such as testicular dimensions, testicular weight, and epididymal weight were evaluated. Compared with the normal group, mice exposed to BUS exhibited testicular atrophy, characterized by a significant reduction in testicular size and weight (Figure 1A, B). Concurrently, a precipitous decline in sperm concentration was observed in the BUS group, resulting in less than 10% of the concentration found in the normal group (Figure 1D, E). These findings confirm the successful establishment of an oligozoospermia mouse model. Nonetheless, a concentration-dependent augmentation in testicular dimensions was observed after HT administration at dosages of 10 mg/kg, 30 mg/kg, and 50 mg/kg, with the 50 mg/kg HT dosage resulting in marked testicular enlargement (Figure 1A). A pronounced elevation in both testicular and epididymal weight was observed following the 50 mg/kg HT treatment, achieving statistical significance (P = 0.074, 0.0349) compared with the BUS group (Figure 1B, C). In addition, the sperm concentration within the cauda epididymis across various groups is discernible through CASA assay, as depicted in Figure 1E. The results unveiled that the number of sperm in the epididymis was substantially (P = 0.0002) reduced after BUS treatment. The administration of 50 mg/kg HT greatly (P = 0.0210) ameliorated the busulfan-induced reduction in epididymal sperm count in mice (Figure 1D, E). These results indicate that HT treatment has the capability to reverse the testicular and epididymal damage caused by busulfan.

Effect of HT on testicular and epididymal weight and sperm parameters. (A) Changes in testicular size in 8-week-old mice treated with BUS, BUS + HT 10 mg/kg, BUS + HT 30 mg/kg, and BUS + HT 50 mg/kg. (B) Testicular weight. (C) Epididymal weight. (D) Sperm concentration. (E) Sperm morphology in the cauda epididymis. *P < 0.05, **P < 0.01, and ***P < 0.001, n ≥ 5.
HT Promotes the Recovery of Testicular Spermatogenesis and Increases the Number of Epididymal Sperm
To examine the histological architecture of the testis and epididymis, Hematoxylin and eosin (HE) staining was performed. The findings revealed that the spermatogenic tubules in the normal group were structurally intact with orderly arranged spermatogenic cells. Previous studies have manifested that exposure to BUS can lead to substantial impairment of the seminiferous tubules within the testis and a concomitant decrease in sperm count within the epididymis. 26 Compared with the normal group, the BUS-exposed group exhibited significant structural impairment of the seminiferous tubules, characterized by a marked (P < 0.0001) reduction in the numbers of spermatogenic cells and mature spermatozoa within the tubular lumen (Figure 2C), a disrupted arrangement of the spermatogenic cells, and the presence of severe vacuolization (Figure 2A). Moreover, seminiferous tubular diameters were observed to diminish following BUS exposure, with a subsequent partial recovery upon HT administration (Figure 2D). Testicular tissue from mice received 50 mg/kg HT displayed evident restoration of tubular structure and a normalization of spermatogenic cell count. (Figure 2A, C, D). Examination of the epididymal tissue revealed a substantial presence of sperm in the caput, corpus, and cauda epididymis in the normal group. However, in the BUS group, sperm within any segment of the epididymis was barely visible. In addition, a significant increase in sperm count was observed in the cauda epididymis after HT treatment compared with the BUS group (Figure 2B). These results demonstrate that HT treatment possesses the capacity to alleviate the structural damage in the testicular and epididymal regions and mitigate the decreased sperm count in the epididymis caused by busulfan.

He staining of representative structures in testicular and epididymal tissues. (A) Testicular tissue morphology in normal, BUS, and HT 50 mg/kg groups. The top row is 20 × , the bottom row is 40 × . (B) Histology of the caput epididymis, corpus epididymis, and cauda epididymis in each group. Scale bar = 25 μm. (C) Spermatogenic cells count in normal, BUS, and HT 50 mg/kg groups. (D) Tubular diameters in normal, BUS, and HT 50 mg/kg groups. *P < 0.05, **P < 0.01, and ***P < 0.001, n ≥ 3.
HT Restores the Cytoskeleton Damaged by Busulfan
To visualize the cytoskeleton, which encompasses microfilaments and microtubules, immunofluorescence staining was implemented. In the normal group, both microfilaments (F-actin) and microtubules (α-tubulin) displayed a uniform and orderly arrangement. However, in the BUS group, there was a noticeable disarray of microfilaments and a concomitant reduction in α-tubulin expression when compared to the normal group. The administration of HT effectively ameliorated the BUS-induced disruption of microfilaments and led to an up-regulation of α-tubulin expression (Figure 3A). An analysis of fluorescence intensity related to microfilaments and microtubules was conducted, both of which decreased in the BUS group but significantly increased (P < 0.0001, P = 0.0022) after HT treatment (Figure 3B, C). These findings manifest that HT could improve the cytoskeleton integrity disrupted by busulfan and facilitate cytoskeleton regeneration.

Visualization of the cytoskeleton. (A) Immunofluorescence staining of the testicular cytoskeleton in each group. Green: F-actin; Red: α-tubulin; Blue: DAPI; Scale bar = 25 μm. (B) F-actin intensity in each group. (C) α-tubulin intensity in each group. *P < 0.05, **P < 0.01, and ***P < 0.001, n ≥ 3.
HT Mitigates the Reduction of Serum Hormone Levels Caused by Busulfan
In order to understand the reproductive endocrine function, the levels of T, FSH, and LH in serum were detected by ELISA kits. Measurement of serum hormone levels illustrated that BUS administration significantly (P < 0.0001) diminished the levels of T, FSH, and LH in the serum compared with the normal group. HT treatment observably (P = 0.0021, 0.0037, 0.0002) increased the levels of T, FSH, and LH after BUS administration (Figure 4). These results suggest that HT treatment is competent to modulate hormone levels and mitigate the decrease in hormone levels induced by busulfan.

Effect of HT on serum hormone levels. (A) Testosterone (T) concentration. (B)Follicle-stimulating hormone (FSH) concentration. (C) Luteinizing hormone (LH) concentration. *P < 0.05, **P < 0.01, and ***P < 0.001, n = 5.
HT Alleviates Oxidative Stress Induced by Busulfan
Oxidative stress is an important factor in male infertility, high levels of ROS can cause male infertility by lipid peroxidation. 27 As the terminal product of lipid peroxidation, MDA is also a marker to evaluate the level of oxidative stress. 28 Meanwhile, it has been reported that HT has antioxidant effects. 22 Therefore, the levels of ROS and MDA in testes were detected with ELISA kits. The results exhibited substantially (P < 0.0001, P = 0.0079) elevated concentrations of both ROS and MDA in the BUS group compared with the normal group. Notably, ROS and MDA concentrations were decreased significantly (P < 0.0001, P = 0.0020) following HT administration (Figure 5). These findings indicate that HT could ameliorate oligozoospermia by alleviating oxidative stress.
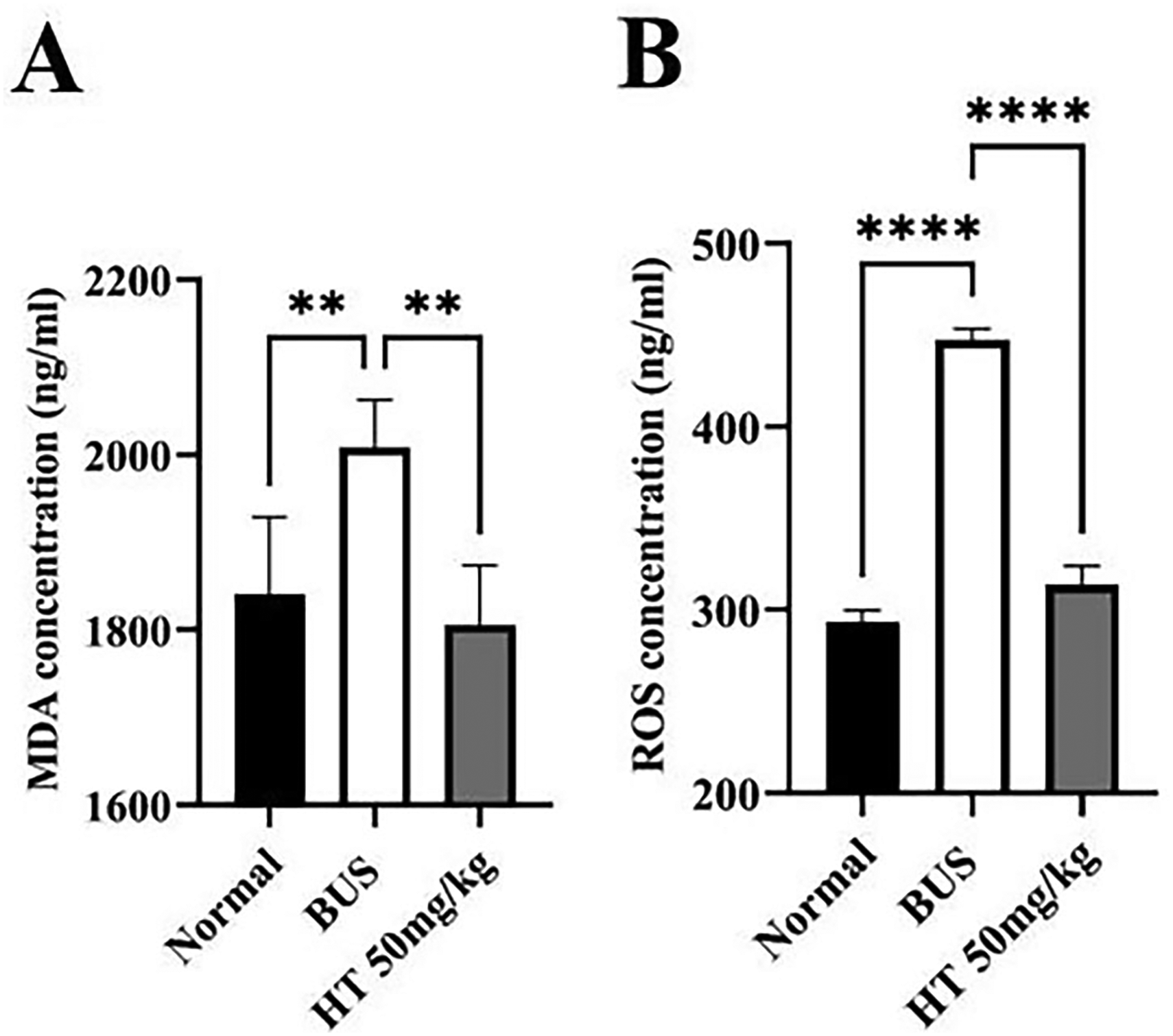
Effect of HT on ROS and MDA levels in testes. (A) MDA concentration, (B) ROS concentration. *P < 0.05, **P < 0.01, and ***P < 0.001, n = 5.
Analysis of Differentially Expressed Genes in Testis Between Oligozoospermia and HT Groups
To delve into the underlying mechanisms of HT in the treatment of oligozoospermia, RNA sequencing was performed. Considering that the effect of HT on oligozoospermia may play a role in the early spermatogenic cycle, differentially expressed genes were examined between the testes of oligozoospermia and HT groups after two weeks of HT administration. A total of 3402 genes were extremely up-regulated, while 31 genes were down-regulated in the HT-treated group when compared with the BUS group (Figure 6A-B).

Analysis of transcriptome sequencing. (A) The statistical analysis of differentially expressed genes. (B) Volcano map of differentially expressed genes between BUS and HT groups. (C) GO enrichment analysis of up-regulated genes in HT treatment. The X-axis shows the number of genes enriched in GO, and the Y-axis shows the GO classification.
To elucidate the biological functions of these differentially expressed genes, Gene Ontology (GO) classification analysis was carried out based on RNA sequencing data. A comparative analysis was undertaken on differentially expressed genes that exhibited significant up-regulation following HT in contrast to the busulfan (BUS) group. The preeminent GO terms enriched in biological process, cellular component, and molecular function were depicted in Figure 6C. Notably, among the GO terms, “cilium movement”, “microtubule-based movement”, “cilium organization”, “spermatid differentiation” and “spermatid development” are closely associated with the restorative impact of HT on spermatogenesis (Figure 6C). These findings indicate that HT significantly modulates germ cell development.
Validation of the Expression Levels of Differentially Expressed Genes After HT Treatment
Based on RNA sequencing data, HT treatment significantly up-regulated the expression of genes pivotal to cilium movement and microtubule-based movement, such as Ccdc65, Dnah2, Zmynd10, Atp1a4, and Tssk4. The relative mRNA expression levels of these genes were validated by qPCR, consistent with the RNA sequencing results. Concurrently, in comparison to the BUS group, there was a marked increase in the expression of genes linked to spermatid differentiation and development, including Zfy2, Rnf17, Tssk3, Prm1 and Tnp1, within the HT group (Figure 7).

Verification of differentially expressed genes by qPCR. *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001, n ≥ 3.
Discussion
Male infertility affects millions of men worldwide and has emerged as a prominent global health concern. 29 Oligozoospermia is mainly divided into obstructive oligozoospermia and non-obstructive oligozoospermia. Non-obstructive oligozoospermia is a leading cause of idiopathic male infertility due to its intricate pathogenesis. 30 Busulfan, an alkylating agent used in cancer chemotherapy, presents a variety of side effects in cancer patients. Damage to the reproductive system is one of the major negative effects. 31 Busulfan treatment can cause severe testicular damage resulting in oligozoospermia, and it serves as a suitable model for studying non-obstructive oligozoospermia in mice. 25 Therefore, it is necessary to find effective therapeutic drugs against the adverse effects of busulfan on the reproductive system. In our study, hydroxytyrosol was found to be competent to alleviate the damage of spermatogenesis in busulfan-induced oligozoospermia (Figure 1).
Hydroxytyrosol, a polyphenol derived from olive oil, is a naturally active substance with antioxidant properties in vitro. 8 Virgin olive oil is the main dietary source of hydroxytyrosol. Because of its bioavailability, non-toxicity, and ease of formulation, hydroxytyrosol is considered an excellent food supplement by the nutraceutical and food industries. 7 In recent years, hydroxytyrosol has garnered increasing attention due to its various pharmacological activities, including antioxidant, anti-inflammatory, cardiac, and neural protective effects. 12 Phenolic compounds are absorbed in the intestine in a dose-dependent manner. 9 Therefore, after 4 weeks of intragastric administration of HT at 10 mg/kg, 30 mg/kg, and 50 mg/kg, the testicular volume and weight, epididymal weight, and sperm concentration of mice were found to increase in a dose-dependent manner. The drug effect was most pronounced when HT was administered at a concentration of 50 mg/kg (Figure 1).
Spermatogenesis is a complex physiological process involving the development of spermatogonia into mature sperm, including spermatogonia proliferation, spermatocyte meiosis, and sperm cell morphogenesis. 32 Reduced or loss of spermatid function can disrupt spermatogenesis and lead to male infertility or sterility. 33 We found that busulfan damaged seminiferous tubules and disrupted the spermatogenesis process, and HT could promote spermatogenesis and increase mature sperm (Figure 2). In addition, the cytoskeletal network of the testis plays an important role in spermatogenesis. As the major cytoskeletal components, microfilaments and microtubules are able to maintain and promote spermatogenesis.34,35 Actin is the major component of microfilaments, so we visualized the expression and distribution of the cytoskeleton by actin tracker and α-tubulin. In this study, we found that HT could promote the regeneration of busulfan-damaged cytoskeleton (Figure 3). Meanwhile, testosterone is crucial for the regulation of spermatogenesis.36,37 Our study revealed that T, FSH, and LH were in a low hormonal disorder state in oligozoospermia, and HT administration could improve these hormone levels (Figure 4).
Previous studies have shown that busulfan treatment stimulates oxidative stress and leads to oligozoospermia. 38 Oxidative stress is caused by high levels of ROS, which is involved in the pathological process of oligozoospermia. Excessive production of ROS can lead to spermatogenesis damage and infertility through lipid peroxidation. 39 In this study, HT was able to reduce ROS levels and reduce MDA concentrations in oligozoospermia (Figure 5).
We further explored the potential molecular mechanisms for the recovery of spermatogenesis in HT treatment of oligozoospermia by transcriptome sequencing. The GO enrichment analysis manifested that spermatid differentiation and development, cilium and microtubule-based movement were strongly associated with the effect of HT treatment on spermatogenesis recovery (Figure 6). Ccdc65 is required for flagellar structure in human sperm and its mutation causes primary ciliary dyskinesia.40,41 Dnah2 mutations can lead to human and mouse sperm flagella morphological abnormalities resulting in male infertility. 42 Zmynd10 plays an important role in dynein assembly and is mutated in response to motile cilia defects.43,44 Atp1a4 is essential for the maintenance of sperm flagella morphology and male fertility.45,46 Tssk4 is closely related to protein phosphorylation in spermatogenesis and takes effect on maintaining the structural integrity of sperm flagella. 47 According to transcriptome sequencing, HT treatment can up-regulate these genes related to cilium and microtubule-based movement to improve oligozoospermia. In addition, transition proteins (Tnps) and protamine (Prms) are important nuclear proteins involved in chromatin remodeling during spermatogenesis. 48 The roles of Prm1 and Tnp1 in spermatogenesis are remarkable. 49 Deletion of Zfy2 leads to meiotic arrest and spermatogenesis disorders. 50 Rnf17 is an essential component of the germ cell nucleus and affects round spermatid development. 51 The loss of Tssk3 leads to the disorder of spermatogenic stages and is essential for a variety of the infertility-related protein phosphorylation. 52 Transcriptome sequencing analysis revealed that HT treatment could increase the expression of these genes associated with the differentiation and development of sperm cells, thereby ameliorating spermatogenesis.
The beneficial impact of HT on spermatogenesis was further validated by qPCR, elucidating its involvement in the cilium and microtubule-based movement and the differentiation and development of spermatids (Figure 7). It has been reported that HT can maintain sperm quality in vitro through its antioxidant mechanisms. 22 We hypothesize that HT may reestablish the regulatory control of these genes in vivo by mitigating the heightened ROS environment within the testes caused by busulfan exposure (Figure 8). Nevertheless, the study is not without its limitations, particularly in identifying the specific molecular targets that HT exerts its effects to augment spermatid development. Consequently, there is a necessity for additional research and in-depth analysis to elucidate the specific targets.
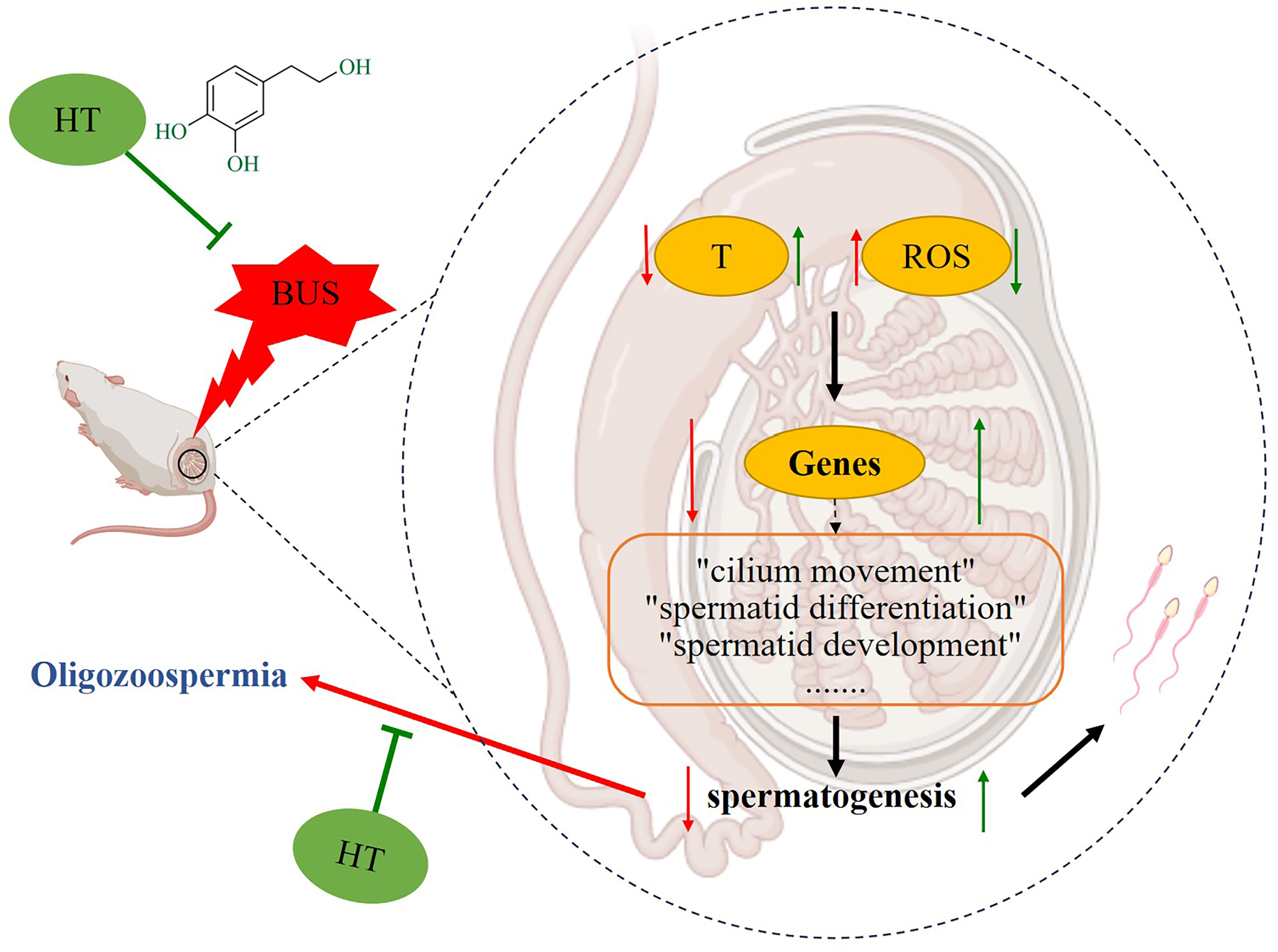
The possible mechanism of HT on oligozoospermia mice.
Conclusion
To sum up, our research represents a pioneering effort to establish the therapeutic efficacy of HT in the context of busulfan-induced oligozoospermia. HT possesses the potential to mitigate busulfan-induced testicular damage, remodel testicular cytoskeleton, and rehabilitate spermatogenic parameters. Our findings suggest that HT may facilitate the resumption of regulatory control over genes associated with cilium and microtubule-based movement, as well as spermatid differentiation and development, by augmenting testosterone levels in the testes and mitigating testicular oxidative stress, thereby restoring spermatogenesis. Looking ahead, we aim to delve into the direct targets of HT and elucidate its specific signaling pathways, thereby laying a robust experimental foundation for utilizing HT in the treatment of male infertility.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study is approved by the Animal Protection Ethics Committee of Nantong University (permission no. IACUC20230511-1002).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (81901528 to X. W.); the Jiangsu Health Innovation Team Program; the Nantong University Clinical Medicine Specialized Research Fund Project (2023JY008 to X. W.); the Nantong Basic Research Program Project (JC12022009 to Y. M.); the Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX24_3562 to T. Y.), and the Large Instruments Open Foundation of Nantong University (KFJN2429 to C. W.).
Statement of Human and Animal Rights
All procedures involving animals were conducted in accordance with the Animal Protection Ethics Committee of Nantong University's (permission no. IACUC20230511-1002) approved protocols.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
