Abstract
Background
Inflammations, oxidative stress, and pains underlie the pathogenesis of non-communicable diseases (NCDs) like respiratory disorders and cancer; however, they are scientifically treated with Curcuma longa. Therefore, the pharmacological screening (antioxidant, anti-inflammatory, and analgesic) of the plant's rhizome, isolation and characterization of its compounds, as well as their in silico profiling with cyclooxygenases, phosphodiesterase, superoxide dismutase, glutathione peroxidase, and catalase could yield lead compounds with better benefit-to-risk ratio against oxidative stress and inflammation.
Methods
The collection of the plant's rhizomes, extract preparation, anti-inflammatory, analgesic, and antioxidant studies, retrieval of target proteins, GC-MS, ADME, and docking analyses were done using standard protocols.
Results
Curcuma longa's percentage edema inhibition and analgesic potentials were better than aspirin in both acute and sub-acute inflammations as well as chemical and thermal-induced pains; moreover, its antioxidant activities were better than vitamin C. Moreover, twenty compounds with anti-inflammatory, analgesic, and antioxidant activities were unraveled from the GC-MS analysis. Ar-tumerone, curcumin, γ-sitosterol, α-tocopherol, isocurcumenol, adamantane, and curdione were the lead compounds based on their binding affinities (for PDE-4B, COX-1, COX-2, SOD, CAT, GPx), lower molecular weights, non-permeation of blood brain barrier, bioavailability profiles, non-inhibition of metabolizing enzymes, faster renal clearance, and non-violation of Lipinski's rules.
Conclusion
Adamantane, α-tocopherol, and γ-sitosterol reported for the first time in the plant's rhizome could mitigate oxidative stress and inflammation as mediators of apoptosis, cytotoxic impaired autophagy, DNA damages, and mitochondrial dysfunction that contributes to the progression of non-communicable diseases (NCDs).
Background
Non-communicable diseases (NCDs) including cardiovascular diseases, cancer, metabolic diseases are becoming a global epidemic and poses severe threat to humanity. Oxidative stress aggravates respiratory and cardiovascular disorders due to the generation of free radicals normally produced during metabolic processes; thereby, promoting the development of NCDs. However, human living cells have an excellent detoxifying enzyme system (superoxide dismutase, catalase, and glutathione peroxidase) vested with the mandate of neutralizing these oxidant species in order to avert pathologies. Moreover, inflammation is the tissue's immunologic reaction to injury and is characterized by the mobilization of antibodies and leucocytes as well as swellings and fluid accumulation. Oxidative stress and inflammation can trigger a range of secondary injuries such as apoptosis, cytotoxic impaired autophagy, DNA damages, and mitochondrial dysfunction that contributes to the progression of NCDs. Conversely, pain is a distressing emotional and sensory experience connected to existing or possible tissue damage. 1 On the contrary, chronic inflammations contribute to cancer and other human diseases;2,3 therefore, the human body requires adequate regulation of inflammatory responses by anti-inflammatory mediators; for example, interleukin-10. The chemical mediators act through the modulation of pro-inflammatory enzymes and cytokines such as interleukin-6 (IL-6), IL-1β, tumor necrotic factor alpha (TNF-α), neutrophils, monocytes, and macrophages; 4 therefore, both pains and inflammations entail a variety of biochemical pathways, including the activation of enzymes, release of inflammatory mediators, absorption of fluid, cell migration, damage to tissues, and repairs. 5
In addition, the inhibition of pro-inflammatory enzymes such as cyclo-oxygenases (COX) in the inflammatory cascades6,7 is observed as the mechanism of action of some pharmacotherapies. The other includes, the inhibition of prostaglandins production being the end-product in the COX pathways of immunologic responses. 8 COX-1 as an isoform of COX found in the platelets and smooth muscle cells, it aids the modification of homeostatic functions whereas COX-2, present in the parenchyma cells of many tissues, is induced as an early response to pro-inflammatory mediators and stimuli such as cytokines 9 and produces prostaglandins that mediate pains and inflammations.10,11 Consequently, diclofenac, a NSAID is a selective COX-1 and COX-2 inhibitor that acts through the inhibition of DMH-induced colon tumorigenesis by suppressing MIP-1α, VEGF, and MCP-1. 10
Moreover, phosphodiesterase-4B (PDE-4B) found in the immune, brain, and epithelial cells manifests as an intracellular non-receptor enzyme for the regulation of inflammation. 12 The inhibition of PDE-4B results in cyclic adenosine monophosphate (cAMP) surge and its resultant modulation of proteins that prevent the recruitment of immune cells and pro-inflammatory mediators release. PDE-4B has been suggested as a promising therapeutic target for the curative use of several pulmonary and serious neurological conditions. 12 Numerous PDE-4 inhibitors have been developed and synthesized, with the administration of roflumilast approved for the therapeutic management of inflammatory airway illnesses. 12 Unfortunately, this class of drugs has undesirable effects despite their efficacy; however, substantial efforts should be geared to mitigate these outcomes and improve the benefit-to-risk ratio; hence, the necessity for new chemotherapies.
Furthermore, the profiling of medicinal plants with acclaimed antioxidant, analgesic, and anti-inflammatory properties may enhance the discovery of novel substances with improved efficacy as well as safety. 13 Turmeric (Curcuma longa) is a member of Zingiberaceae family. It has served as a culinary spice with religious significance 14 and is a rich source of polyphenolic curcuminoids, 15 volatile oil (Ar-turmerone, curcumin), and essential oil (sesquiterpenes, curcumin). 16 It has been reported to possess anti-inflammatory,17,18 antioxidant and anti-inflammatory, 19 anti-cardiovascular 20 activities. Moreover, curcumin on its part, possesses a variety of pharmacological activities, including anti-inflammatory and cancer chemopreventive,21,22 antioxidant,23,24 and analgesic 21 activities.
Generally, the bioassay, isolation, and characterization of bioactive compounds, as well as the validation of the mechanism of action of the lead compounds from medicinal plants are tasking and tedious but very rewarding in the quest of discovering new chemotherapies. So, as part of drug discovery processes, researchers profile many ligands to unravel the lead compounds that may further be subjected to optimization. Therefore, the objectives of this study were to evaluate the antioxidant, analgesic, and anti-inflammatory properties of Curcuma longa rhizomes; isolate and characterize its compounds using GC-MS, as well as dock these compounds with cyclooxygenases-1&2 (COX-1, COX-2), phosphodiesterase-4B (PDE-4B), superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPX) enzymes in order to discover the lead compounds for optimization; hence, the provision of new perspectives on the therapeutic potentials of bioactive compounds from Curcuma longa for the management oxidative stress and inflammation being the mediators of most non-communicable diseases.
Methods
Collection, Identification, and the Extraction of Curcuma longa Rhizomes
The rhizomes of Curcuma longa were obtained in March 2023 from a botanical farm in Arthur Jarvis University, Akpabuyo, Cross River State, Nigeria. Akpabuyo has an area of 1241 km2 land mass, coordinate of 5oN 8.4oE, and a population of 271295 based on 2006 census. A plant taxonomist, Dr Echeng, Department of Botany, University of Calabar, Nigeria identified the rhizome and identity number UCH512G was deposited. The rhizomes were washed, chopped, and dried under shade. Thereafter, they were pulverized into coarse particles. The coarse material was weighed (356.2 g) and macerated in 2.5 L of dichloromethane (DCM) solvent; kept for seventy-two hours; filtered, and concentrated using a rotary evaporator at 40 °C. The process yielded 49.71% of turmeric extract (103.7 g), which was used for the study. The study was carried out following ARRIVE guidelines. 25
Gas Chromatography-Mass Spectrometry Analysis of Dichloromethane Extract of Curcuma longa
The GC-MS analysis was carried out with a Clarus 500 Perkin Elmer gas chromatograph outfitted with an Elite-5 capillary column (5% phenyl, 95% dimethyl polysiloxane) (30 nm × 0.25 mm ID × 0.25 m df) and a mass detector turbo-mass gold of the company that was operated in EI mode. The injector worked at 290 °C, and the oven temperature was controlled in the following order: 50 °C at 8 °C/min to 200 °C (5 min) at 7 °C/min to 290 °C (10 min). Helium served as the medium for the gas at the rate of flow of 1 ml/min. The National Institute of Standards and Technology (NIST) database, which contains more than 62,000 patterns, was used for the interpretation of the mass spectrum. The names and molecular weights of the extract's constituents were determined using the retention time and peak area acquired from the mass spectra of the unknown compounds and those of the known compounds stored in the library. 25
The Assessment of the Physicochemical and Pharmacokinetics Characteristics of the Bioactive Compounds
The drug-likeness and ADME characteristics of the bioactive compounds were determined using previously reported methods26–28 whereas the canonical strings of the various compounds were retrieved from the PubChem website (https://www.ncbi.nlm.nih.gov/pccompound) in 3D-configuration. The online programme Predicting Small-Molecular Pharmacokinetic and Toxicity Properties (PKCSM) software was used to evaluate the absorption, distribution, metabolism, and excretion (ADME) properties of the ligands whereas the SWISS Target Prediction tool enhanced the determination of the targets of the different ligands. All the parameters including Lipinski's rule of five, Veber, and the Ghose parameters are reported in Table 4.
Target Proteins’ Properties and Molecular Docking of the Compounds
The protein-ligand interaction in this study was determined through the use of Auto Dock Vina 4.2 tool. Six target proteins (CAT, SOD, GPX, COX-1, COX-2, PDE-4B) with PDB ID codes 7VD9, 3HFF, 2F8A, 2OYE, 6COX, and 4WCU, respectively were downloaded in PDB format from the Research Collaboratory for Structural Bioinformatics (RCSB) protein data bank. The 2F8A, 3HFF, and 7VD9 proteins’ default binding sites for binding centers and size were (centre_x = 4.941100, centre_y = 15.834500, centre_z = 21.572900, size_x = 19, size_y = 19, size_z = 19); (centre_x = 15.122917, centre_y = 16.911167, centre_z = 21.13.198583, size_x = 17, size_y = 17, size_z = 17); (centre_x = 102.055000, centre_y = 109.644000, centre_z = 97.221400, size_x = 27, size_y = 27, size_z = 27), respectively. However, the default binding sites for binding centers and size for 2OYE, 6COX, and 4WCU proteins’ were (centre_x = 238.249182, centre_y = 102.324273, centre_z = 21.000000, size_x = 21, size_y = 21, size_z = 21); (centre_x = 48.994857, centre_y = 25.739439, centre_z = 52.441714, size_x = 24, size_y = 24, size_z = 24); (centre_x = 22.330000, centre_y = -23.983875, centre_z = -3.299000, size_x = 25, size_y = 25, size_z = 25), respectively. The selected ligands were docked against their target proteins. First, the water molecules in the proteins were removed. The proteins were further prepared by adding hydrogen atoms using 3D protonation, and any incomplete charges were corrected. The energy of the proteins was minimized. Next, the Site Finder MOE module located the allosteric site previously determined using the Allosite Pro online server. Biovia discovery studio software was used for visualization of the interaction. The molecular docking between the ligands and the proteins was performed using the AutoDock Vina 4.2 tool. For comparison, diclofenac and vitamin C were used as anti-inflammatory and antioxidant drugs, respectively; their binding affinities and 2D-visual interactions were documented.
Acute Toxicity Determination
Using the method described by Lorke, the acute toxicity test of the dichloromethane extract of Curcuma longa rhizomes administered to mice was evaluated by the use of sixteen rats. 29 However, the Animal House in the Faculty of Natural and Applied Sciences, Arthur Jarvis University, Nigeria provided seventy-two (72) albino mice (22-25 g) used for the experiment. The NIH protocols for the use, handling, and care of laboratory animals were judiciously followed. Throughout the experiment, the mice had access to water, a regular pelleted feed, and housing at room temperature. The experimental protocol was strictly followed and authorized by the faculty's animal ethics committee (approval number: AJUFNAS/0042).
Evaluation of in Vitro Antioxidant Activities of Curcuma longa Rhizomes
Hydrogen Peroxide Scavenging Activity
This was estimated by using the method of Ruch et al. 30 Plant extract (4 ml) was prepared in distilled water at various concentration, mixed with 0.6 ml of 4 mM solution prepared in phosphate buffer (0.1 M pH 7.4) and incubated for 10 min. The absorbance of the solution was taken at 230 nm against blank solution containing no plant extract. The amount of hydrogen peroxide radical inhibited by the extract were calculated using the following equation: radical scavenging activity = {(Abs control — Abs sample) /(Abs control)}, Where Abs control is the absorbance of radical + methanol; Abs sample is the absorbance of H2O2 radical + sample extract or standard.
Ferrous ion Chelating Activity
The chelating effect of ferrous ions by the prepared extract was estimated by the method of Carter 31 with slight modifications. Briefly, 100 μL of each test sample (1 mg/ml) was taken and raised to 3 mL with methanol. 740 μL of methanol was added to 20 μL of 2 mM FeCl2. The reaction was initiated by the addition of 40 μL of 5 mM ferrozine into the mixture, which was then left at room temperature for 10 min and then, the absorbance of the mixture was determined at 562 nm.
Total Antioxidant Capacity
This method according to Re et al, 32 2, 2 – Azino-bis (e-ethylbenzothiazoline-6-solfonic cation radical (ABTS+) reduction was measured. ABTS+ was prepared by mixing 0.1 mM ABTS, 0.0125 mM horse radish peroxidase and 1 mM H2O2 in a 50 mM phosphate buffer (pH 6.0). After 15 min, 10 µL diluted leaf extract was added to 190 µL ABTS+ solution and conversion of a cation radical into colorless ABTS was followed by as decrease in absorption at 651 nm.
Vitamin C
This was determined colorimetrically using the method by Klein and Perry. 33 The extract (100 mg) was extracted with 10 mL of 1% metaphosphoric acid for 45 min at room temperature and filtered through Whatman No. 4 filter paper. The filtrate (1 mL) was mixed with 9 ml of 2, 6-dichlorophenolimfophenol and the absorbance was measured within 30 min at 515 nm against a blank. Contents of vitamin C were calculated on the basis of the calibration curve of authentic L-ascorbic acid (0.020-0.12 mg/mL). The assays were carried out in triplicate; the results were expressed as mg of ascorbic acid/g of extract.
Ferric Reducing Antioxidant Power
This was done according to the method by Yen and Chen. 34 Volumes of 1.0 mL of the extract were prepared in distilled water and BHT (Butylated hydroxytoluene), vitamin C, and vitamin E (0-0.5 mg/mL), were mixed individually to the mixture containing 2.5 mL of 0.2 M phosphate buffer (pH 6.6) and 2.5 mL of potassium ferricyanide (K3Fe(CN)6) (1% w/v). The resulting mixtures were incubated at 50 °C for 20 min, followed by the addition of 2.5 mL of trichloroacetic acid (10% w/v), which were then centrifuged at 3000 r/min for 10 min. The upper layers of the solution (2.5 mL) were mixed with 2.5 mL of distilled water and 0.5 mL of ferrous chloride (0.1%, w/v). Their absorbance was measured at 700 nm against blank samples.
2,2-Diphenyl-1-Picrylhydrazyl (DPPH)-Radical Scavenging Activity
The method of Liyana-Pathiranan and Shanhidi 35 was used. A solution of 0.135 mM DPPH in methanol was prepared and 1.0 mL of this solution was mixed with 1.0 mL of the extract prepared in methanol containing 0.025–0.5 mg of the plant extracts and standard drugs separated (BHT and rutin). The reaction mixture was vortexed thoroughly and left in a dark at room temperature for 30 min. The absorbance of the mixture was measured spectrophotometrically at 517 nm. The ability of the plant extract to scavenge DPPH radical was calculated by the equation: DPPH radical scavenging activity = {(Abs control — Abs Sample)/(Abs control)} x 100, Where Abs control is the absorbance of DPPH radical + methanol; Abs sample is the absorbance of DPPH radical + sample extract or standard.
Assessment of the Anti-Inflammatory Properties of Curcuma longa Rhizomes
Carrageenan–Induced Edema in Mice
Albino mice (18) comprising both males and females were divided into three separate groups, each consisting of six mice. These groups were utilized for the experiment following a 24 h of fasting and deprivation of water. To induce inflammation, a 0.1 mL dose of freshly prepared carrageenan suspension (1%) in normal saline was injected into the sub-planar surface of their hind paw. The linear circumference of the injected paw was measured both before and at 0.5, 1, 2, 3, and 4 h after the administration of the inflammation-inducing substance. The increase in paw circumference, measured with a vernier caliper, was used as the parameter to assess inflammation, as documented36,37 whereas after the experiment, some of the mice died naturally, others recovered and survived. Edema was evaluated by calculating the difference in paw circumference between the control group and the measurements taken at 0.5, 1, 2, 3, and 4 h after the carrageenan injection, as documented. 38 Prior to the induction of inflammation, a group of mice received an oral dose of Curcuma longa extract (500 mg/kg) one hour in advance. The negative control mice were given 10 ml/kg of distilled water, while the positive control group was administered aspirin (100 mg/kg).
Egg Albumin-Induced Inflammation in Mice
Albino mice (18) consisting of both males and females underwent a 24-h fasting period prior to the experiment. Subsequently, they were divided randomly into three distinct groups, each consisting of six mice. One group of mice received an oral dose of Curcuma longa extract (500 mg/kg) an hour before the initiation of inflammation, while the negative control group was given 10 ml/kg of distilled water. The mice were subjected to the experiment after 24 h of fasting and water deprivation. Inflammation was induced by injecting 0.1 mL of freshly prepared egg-albumin into the sub-planar tissue of the right hind paw of the mice, as described in. 37 Paw circumference measurements using a vernier caliper were taken both before and at 0.5, 1, 2, 3, and 4 h following the administration of the inflammation-inducing substance. The assessment of inflammation was based on the difference in paw circumference between the control group and the measurements obtained at 0.5, 1, 2, 3, and 4 h, as outlined in 38 whereas after the experiment, some of the mice died naturally, others recovered and survived.
Evaluation of Analgesic Activity of Curcuma longa Rhizomes
Hot Plate Test Method
Albino mice (18) were subjected to a 24-h fasting period prior to the experiment. Subsequently, they were divided randomly into three groups, each consisting of six mice. One group received an oral administration of 500 mg/kg of Curcuma longa extract one hour before the induction of pain, while the negative control group was given 10 ml/kg of distilled water. To assess their response to pain, the mice were placed on a heated surface (45 °C) inside a glass beaker with a diameter of 50 cm. The time it took for the mice to start licking their paws after placement was recorded 39 whereas, after the experiment, some of the mice died naturally, while others recovered, and survived. The response was observed through paw licking and shaking. The percentage increase in reaction time, or the inhibition of pain threshold, was calculated using the formula: % Elongation (increase in latency) = (Latency (test) − Latency (control))/ Latency (test) × 100.
Acetic Acid-Induced Writhing Response
Albino mice (18) regardless of their gender were divided into three groups, each consisting of six mice. Prior to the experiment, these mice underwent a 24-h period of fasting and water deprivation. To induce pain, each mouse was administered an intra-peritoneal (i.p.) injection of 0.1 ml/10 g acetic acid solution (equivalent to 10 ml/kg body weight). The positive control group of animals received an oral pretreatment of aspirin (100 mg/kg, i.p.), while the other group was given Curcuma longa extract (500 mg/kg) 25 min before the administration of acetic acid. Five minutes after the induction of pain, the number of writhing and stretching movements exhibited by the mice was recorded whereas, after the experiment, some of the mice died naturally, while others recovered and survived. The level of analgesic activity was quantified as the reduction in abdominal constrictions in comparison to control animals treated with distilled water. This reduction was expressed as a percentage, calculated using the formula: % Analgesic activity = (mean writhing count of control group - mean writhing count of treated group) / mean writhing count of control group × 100. 39
Statistical Analysis
The analysis of the results was conducted utilizing Statistical Package for Social Sciences (SPSS) software version 21. The outcomes were expressed as the mean along with the standard error of the mean (SEM) for the responses. To assess statistical significance, we employed One-way Analysis of Variance (ANOVA), followed by a Turkey post hoc test to compare differences among the groups, with alpha value set at 0.05. The data were subsequently displayed using tables and graphs.
Results
GC-MS Analysis of Curcuma longa Extract
Twenty bioactive compounds (Table 1) were characterized, the constituents in the rhizome included but were not limited to curcumin, γ-sitosterol, α-tocopherol, isocurcumenol, Ar-tumerone, adamantane, curdione, phenol, 1-hexadecene, 3-oxallobetulane, isopulegol acetate, α-bisabolene epoxide, bicyclo [2.2.1] heptane, geranyl-α-terpinene, and carbamic acid phenyl ester.
GC-MS Analysis of Dichloromethane Extract of Curcuma longa Rhizomes.

Antioxidant Activities of Curcuma longa Extract
The results of the DPPH-radical scavenging activity, ferrous ion chelating activity, ferric reducing antioxidant power, and total antioxidant capacity of the extract (Figures 1 to 5) were better than the ascorbic acid at 80 and 100 μg/ml concentrations. However, the hydrogen peroxide scavenging activity of the vitamin C was better than the extract at all the concentrations. The variation in antioxidant activities was in concentration dependent manner.

DPPH scavenging activity of dichloromethane extract of Curcuma longa rhizomes.

Ferrous ion chelating activity of dichloromethane extract of Curcuma longa rhizomes.
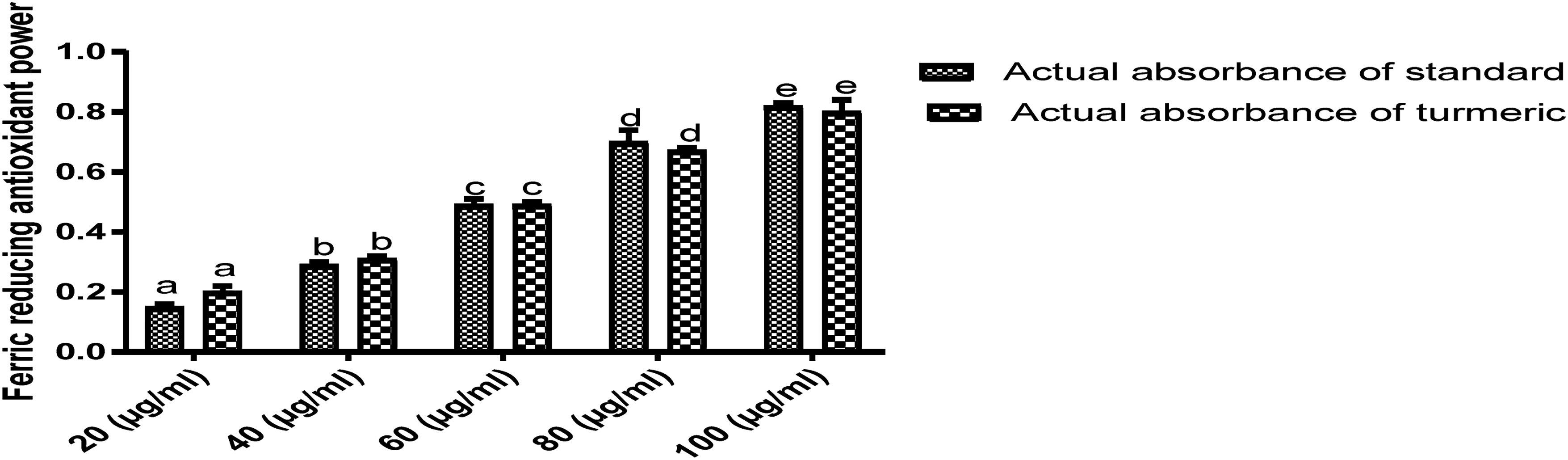
Ferric reducing antioxidant power of dichloromethane extract of Curcuma longa rhizomes.

Total antioxidant capacity (using linoleic acid as standard) of dichloromethane extract of Curcuma longa rhizomes.
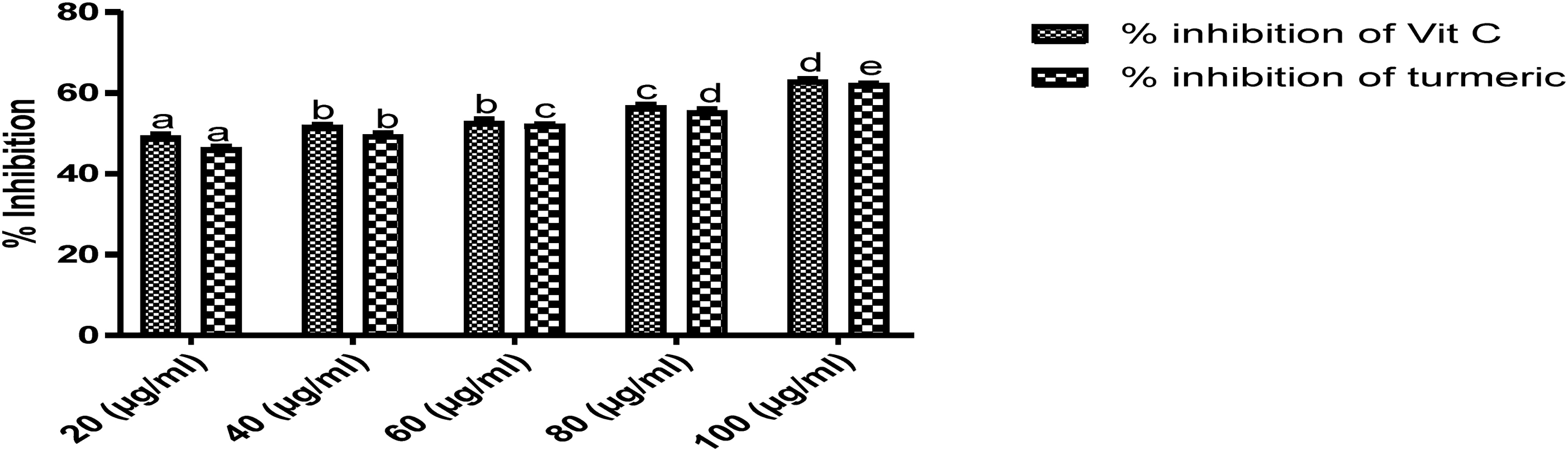
Hydrogen peroxide scavenging activity of dichloromethane extract of Curcuma longa rhizomes.
Anti-Inflammatory Activities of Curcuma longa Extract
Both models assessed the potential anti-inflammatory effects of Curcuma longa rhizomes in both acute and sub-acute phases of inflammation. In the experiments, mice were subjected to sub-planar injections of 0.05 ml of 1% carrageenan and 0.1 mL of freshly prepared egg-albumin in their hind paws. This led to a gradual increase in paw thickness, reaching its peak after 60 min in the negative control group (as shown in Figures 6 and 7) and after 30 min in the other groups. Notably, the other groups (as depicted in Figures 6 and 7) exhibited statistically significant inhibition (p < 0.05) of paw thickness starting from 30 min, and this effect persisted for up to four hours after carrageenan and egg albumin inductions when compared to the negative control group. Importantly, the extract's ability to reduce paw edema was comparable to that of Aspirin.

Anti-inflammatory effect of dichloromethane extract of Curcuma longa rhizomes (carrageenan -induction).

Anti-inflammatory effect of dichloromethane extract of Curcuma longa rhizomes (egg albumin-induction).
Effect of Curcuma longa on Acetic Acid-Induced Writhing Assay and Thermal Stimulation Response
As shown in Figures 8 and 9, both the aspirin and extract groups demonstrated a significant reduction (P < 0.05) in acetic acid-induced writhing in mice compared to the negative control group. Notably, the pain-relieving effect of Curcuma longa extract surpassed that of aspirin. Furthermore, the Curcuma longa extract significantly (P < 0.05) prolonged the response time of mice to hot plate thermal stimulation, with the most remarkable outcome observed for the extract when compared to aspirin.

Analgesic effect of dichloromethane extract of Curcuma longa rhizomes (acetic acid-induced writhing model).

Analgesic effect of dichloromethane extract of Curcuma longa rhizomes (hot plate thermal stimulation model).
Ligands Docking with Superoxide Dismutase, Catalase, and Glutathione Peroxidase
The docking analysis (Table 2) and (Figures 10 to 12) of the compounds with 3HFF, 7VD9, and 2F8A revealed their 2D-visual interactions, amino acid sequences, and binding affinities. Further analysis with superoxide dismutase and comparison with ascorbic acids (–4.3 kcal/mol) showed the binding affinities of the best five compounds in decreasing order as follows: curcumin (–5.5 kcal/mol), γ-sitosterol (–5.2 kcal/mol), isocurcumenol (–5.2 kcal/mol), Ar-tumerone (–5.0 kcal/mol), and adamantane (–4.9 kcal/mol). Also, the best five docking results based on their binding affinities with catalase and comparison with vitamin C (–6.4 kcal/mol) were curcumin (–8.7 kcal/mol), α-tocopherol (–8.7 kcal/mol), γ-sitosterol (–8.5 kcal/mol), Ar-tumerone (–8.0 kcal/mol), and adamantane (–7.3 kcal/mol). Moreover, γ-sitosterol (–6.9 kcal/mol), isocurcumenol (–6.0 kcal/mol), Ar-tumerone (–5.9 kcal/mol), curcumin (–5.8 kcal/mol), and α-tocopherol (–5.1 kcal/mol) were the best five compounds based on their binding affinities with glutathione peroxidase as compared with vitamin C (–5.0 kcal/mol).

Ligands interaction with superoxide dismutase (3HFF).

Ligands interaction with catalase (7VD9).

Ligands interaction with glutathione peroxidase (2F8A).
Binding Affinities of Curcuma longa ‘s Bioactive Compounds Docked with Superoxide Dismutase (3HFF), Catalase (7VD9), and Glutathione Peroxidase (2F8A) Proteins.

Ligands Docking with Phosphodiesterase-4B, Cyclooxygenase-1, and Cyclooxygenase-2
The docking analysis (Table 3) and (Figures 13 to 15) of the compounds with 4WCU, 2OYE, and 6COX revealed their 2D visual interactions, amino acid sequences, and binding affinities. The interaction of the ligands with phosphodiesterase-4B as compared with diclofenac revealed the best five compounds as follows: curcumin (–8.3 kcal/mol), γ-sitosterol (–7.7 kcal/mol), isocurcumenol (–7.6 kcal/mol), Ar-tumerone (–7.6 kcal/mol), and 1-(3-Isopropylidene-5,5-dimethyl-bicyclo[2.1.0]pent-2-yl)-ethanone (–7.6 kcal/mol) whereas ligands docking with COX-1 showed that it was only γ-sitosterol (–8.7 kcal/mol) that had a better binding affinity than diclofenac (–8.4 kcal/mol). Moreover, γ-sitosterol (–8.6 kcal/mol), α-tocopherol (–8.1 kcal/mol), isocurcumenol (–7.2 kcal/mol), curdione (–7.2 kcal/mol), and curcumin (–8.9 kcal/mol) were better than ascorbic acid (–6.3 kcal/mol) in terms of their binding with COX-2.
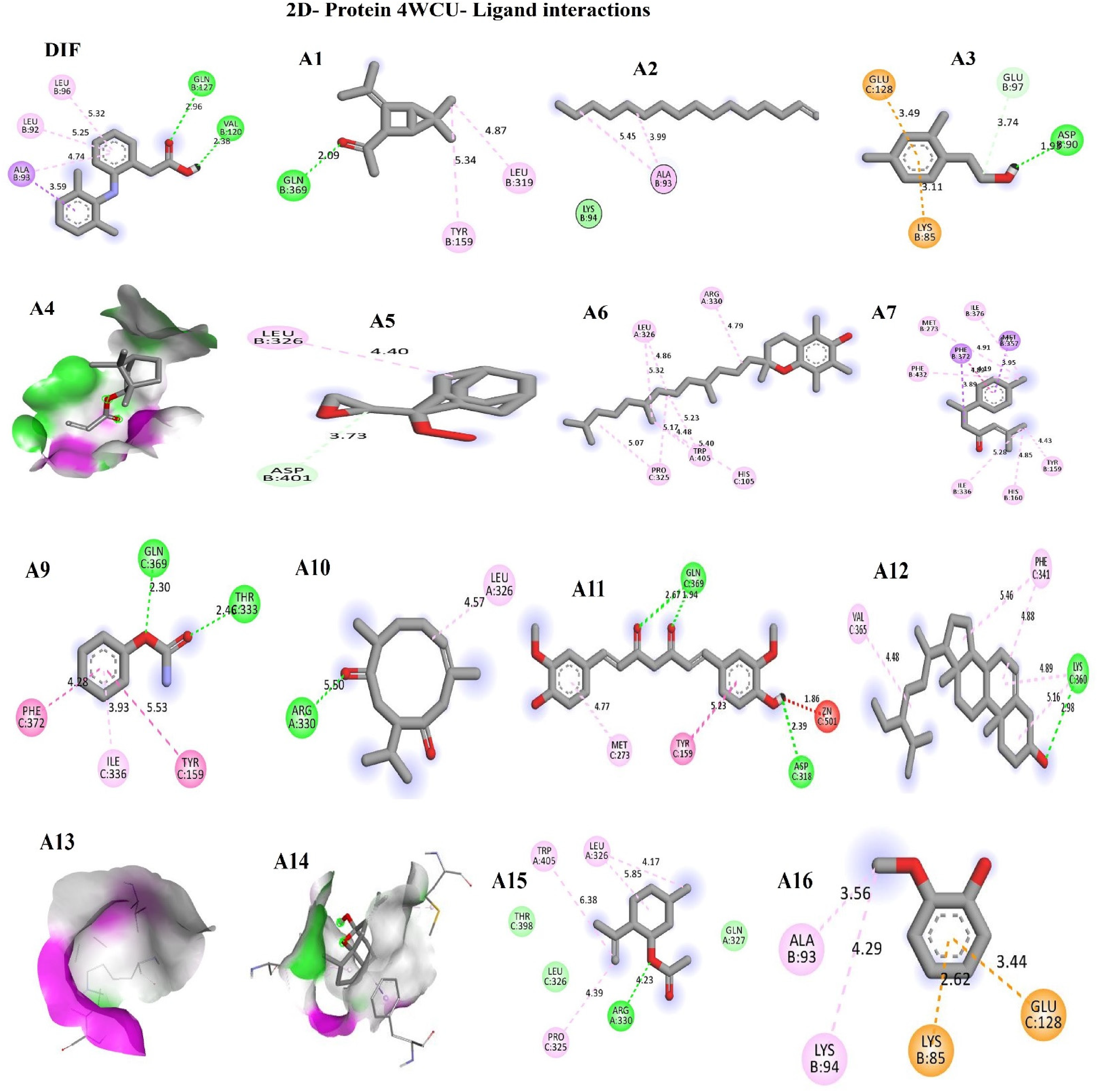
Ligands interaction with phosphodiesterase-4B (4WCU).

Ligands interaction with cyclooxygenase-1 (2OYE).

Ligands interaction with cyclooxygenase-2 (6COX).
Binding Affinities of Curcuma Longa's Bioactive Compounds Docked with Cyclooxygenase-1 (2OYE), Cyclooxygenase-2 (6COX), and Phosphodiesterase-4B (4WCU

Physicochemical, Drug-Likeness, and ADME Properties of Turmeric's Bioactive Compounds
According to Table 4, out of the thirteen (13) compounds screened, the lead compounds (curcumin, α-tocopherol, γ-sitosterol, Ar-tumerone, isocurcumenol, and adamantane) revealed a high rate of absorption in the GIT and inabilities to permeate the blood-brain barrier (BBB) as well as not being substrate of permeation glycoprotein. Further analysis of the predictors of drug metabolism for possible inhibition of the different mono-oxygenases revealed non-inhibition of CYP1A2, CYP2C19, CYP2C9, CYP2D6, and CYP3A4. All (100%) of the screened compounds obeyed Lipinski's rule of five with the highest of only one violation for the compounds whereas the molecular weights of the lead compounds were less than 500 Daltons.
Physicochemical, Drug-Likeness, and ADME Properties of Curcuma Longa's Bioactive Compounds.

Discussion
Oxidative stress plays a role in the onset, progression, and complications of diabetes mellitus, neurogenerative, and cardiovascular disorders because the free radicals generated during inflammatory diseases, cell transformation, and metabolism react with macromolecules in the cells; thereby, resulting in mutations and cell apoptosis. However, the neutralization of oxidant species and free radical scavenging have been proposed as the possible mechanism of action of some compounds. 40 According to (Figures 1 to 5), the DPPH-radical scavenging activity, ferrous ion chelating activity, ferric reducing antioxidant power, and total antioxidant capacity of Curcuma longa extract were better than vitamin C used for comparison in the study. This result corroborated the report that the interaction between antioxidant compounds and free radicals often resulted in scavenging through hydrogen ion donation.41,42 Also, metal chelating compounds are known for iron detoxification in lipid membranes through the stabilization of their reduced ions; 42 therefore, Curcuma longa's high chelating power implies it could be used as a chelation therapy to avert iron toxicity. Moreover, the astounding total antioxidant capacity of the extract when compared with ascorbic acid revealed the rhizome's rich antioxidant compounds; this result was consistent with the report. 43 Furthermore, the better ferrous reducing antioxidant power of the extract as compared with vitamin C in a concentration-dependent manner is an indication of the rhizome's reductive and excellent antioxidant activity. Conversely, vitamin C's hydrogen peroxide scavenging activity was better than the extract suggesting that they could have donated electrons to H2O2 to generate water and oxygen molecules. From the GC-MS analysis (Table 1), the following compounds characterized from the extract have already been reported to possess good antioxidant activities; they include, curcumin, 44 α-bisabolene epoxide, 45 geranyl-α-terpinene, 46 1-hexadecene, 47 isopulegol acetate, 48 γ-sitosterol, 49 and α- tocopherol. 50 In addition, Stirac et al 51 reported that the compound, adamantane is potent in drug delivery and surface recognition; thereby, enhancing the lipophilicity of drugs across liposomes and lipid bilayers.
It is worthy of note that superoxide dismutase as the most powerful detoxification enzyme in the cells catalyzes the dismutation reaction of O2* to H2O2 that is further catalyzed by CAT and GPx 52 ; however, catalase enhances the reduction of H2O2 to H2O and O2 in the cell's peroxisomes, 53 whereas glutathione peroxidase as an intracellular enzyme that catalyzes the detoxification of H2O2 generated during fatty acid oxidation in the mitochondria and cytosol and is implicated in cancer and cardiovascular diseases. 54 Consequently, the binding affinities (Table 2) of curcumin, α-tocopherol, γ-sitosterol, Ar-tumerone, isocurcumenol, and adamantine through their docking with superoxide dismutase, catalase, and glutathione peroxidase affirmed their strong interaction (activation) of these proteins via hydrophobic linkages with the various amino acid residues in their active sites and suggested that these compounds could have mediated their free radical scavenging through the neutralization of reactive oxygen species and possibly reducing the generated hydrogen peroxide to water and oxygen molecules.
The assessment of Curcuma longa extract's anti-inflammatory properties in both the acute and sub-acute stages of inflammation, as shown in Figures 6 and 7, demonstrated its effectiveness in reducing inflammation. Carrageenan, a non-antigenic pro-inflammatory substance, was used as a control agent in acute phase inflammation due to its consistent and reproducible effects without significant systemic impacts. 55 Consequently, we induced acute localized inflammation by indirectly releasing various endogenous inflammatory mediators in a biphasic sequential manner. The initial stage of inflammation, occurring at 0 and 2 h after carrageenan induction, was marked by enhanced vascularization in the damaged tissue, primarily driven by the release of serotonin, bradykinin, and histamine. 56 In contrast, the later phase (2-4 h) was sustained by the excessive production of COX-2 and its pro-inflammatory prostaglandin products, leading to the infiltration of polymorphonuclear leukocytes. Our findings, as shown in Figure 6, revealed that both the extract and aspirin significantly (p < 0.05) reduced edema formation starting from the first hour, and this effect persisted until the fourth hour of observation. These results suggest that the compounds identified in the extract, known for their established anti-inflammatory properties (curcumin, 44 isopulegol acetate, 48 bicyclo[2.2.1]-heptane, 57 isocurcumenol, 58 curdione), 59 could have mitigated both phases of acute inflammation by interfering with the release of chemical mediators. The high % edema inhibition (Figure 6) further revealed the astounding anti-inflammatory effect of the compounds against endogenous inflammatory mediators such as COX; therefore, they also played key roles in the later inflammation phase.19,60 The above result further corroborated the better binding affinities (Table 3) for curcumin, γ-sitosterol, isocurcumenol, Ar-tumerone, and 1-(3-Isopropylidene-5,5-dimethyl-bicyclo[2.1.0]pent-2-yl)-ethanone; moreover, the amino acid residues in the active sites of PDE-4B interacted (through inhibition) within their respective complex in every possible orientation and geometry, an important indication of stability and excellent interaction with the ligands implying that these compounds are potential PDE-4B inhibitors that could have acted by blocking the degradation of cAMP and further prevented the mobilization of immune cells and pro-inflammatory mediators; hence their anti-inflammation mechanism of action.
Furthermore, the egg albumin-induced paw edema model serves as a method to assess the potential anti-arthritic and anti-inflammatory properties of natural substances during the sub-acute phase of inflammation. 55 In the negative control group (as depicted in Figure 7), the induction of edema was marked by heightened leucocyte and phagocyte migration, infiltration of neutrophils and macrophages, and the proliferation of fibroblasts in the vicinity of the injured area. 61 Therefore, according to Figure 7, the edema inhibition observed for the turmeric extract at the fourth hour was better than aspirin. This could be attributed to the bioactive compounds mediating their activities through the inhibition of neutrophils and macrophages infiltrations, as well as the mitigation of leucocytes and phagocytes migration into the area of inflammation. 61 Chatterjee et al 62 and Kaneria et al 63 reported that terpenoids such as geranyl-α-terpinene (Table 1) exert their activities through the inhibition of TNF-α production, iNOS, and COX-2 expressions. In addition, curcuminoids such as curcumin and isocurcumenol (Table 1), demonstrate their anti-inflammatory effects by reducing the production of inflammatory cytokines. 60 This, in turn, led to the suppression of both COX and ROS activity. The results (Tables 3 and 4) on the pharmacokinetic, drug-likeness, physicochemical, and the binding affinities of curcumin, γ-sitosterol, isocurcumenol, Ar-tumerone, and 1-(3-Isopropylidene-5,5-dimethyl-bicyclo[2.1.0]pent-2-yl)-ethanone against PDE-4B revealed that the ligands interacted (inhibition) and formed complexes with the proteins’ active sites through hydrogen and hydrophobic bonds, as well as ionic bonds. These reports corroborated the fact that these lead compounds are PDE-4B inhibitors with good anti-inflammatory activities and pharmacological potentials for lead optimization; the result was in line with the findings on the anti-inflammatory activities of medicinal plants. 64
Acetic acid-induced writhing and thermal analgesia tests are pharmacological models used in the study of peripheral and central analgesia to evaluate the analgesic potentials of compounds. According to Figure 8, Curcuma longa extract significantly reduced (p ≤ 0.05) acetic acid-induced writhing relative to the negative control group and the result was better than aspirin. The induction of pains with acetic acid normally lead to capillary permeability and the reduction of nociceptive threshold because of the stimulation of nociceptive fibers at the nerve terminals 65 or through the release of PGE2 and PGF2α at the peritoneal receptors. 66 So, the extract and aspirin (Figure 9) demonstrated considerable time and dose–dependent analgesia against thermal-induced pains in mice; therefore, the delayed latency time for both extract and aspirin implied nociceptive reaction against the thermal stimuli; thereby, unraveling opiate analgesic property for turmeric's bioactive compounds.1,39 In addition, carbamic acid phenyl esters (Table 1) have analgesic properties, 67 this compound characterized from the plant may have acted as a centrally acting analgesic compound. Moreover, COX-2 is known for the production of prostaglandins that mediate pains and inflammations; so, the better binding affinities of these compounds (γ-sitosterol α-tocopherol, isocurcumenol, curdione, and curcumin) as compared to diclofenac drug, their improved pharmacokinetic, drug-likeness, and physicochemical properties as well as the observation that the ligands interacted (inhibition) and formed complexes with the proteins active sites through hydrogen and hydrophobic bonds, as well as ionic bonds. Therefore, these amino acid residues interacted within their respective complex in every possible orientation and geometry, an important indication of stability and excellent interaction with the ligands 68 that corroborate their analgesic potentials through the inhibition of COX-2; thereby, decreasing vascular permeability, reducing histamine and serotonin secretions, blood flow, and plasma exudation, as well as down regulation of homeostatic functions; hence their prospect for lead optimization.
However, the present study is prone to potential limitations. First, the study had fewer study sample size. Secondly, in silico results would need further in vivo confirmations.
Conclusion
Seventy-two mice were used for the studies, aspirin and Curcuma longa both performed favorably when it came to the percentage of edema inhibition and analgesic potentials in relation to acute and subacute inflammations, as well as chemically and thermally produced pains. While the plant's antioxidant properties outperformed those of vitamin C, the extract's characterization led to the unraveling of 20 compounds with previously documented analgesic, antioxidant, and anti-inflammatory properties. Furthermore, it was discovered through in silico studies that the lead compounds (adamantane, α-tocopherol, γ-sitosterol, Ar-tumerone, isocurcumenol, and curcumin) docked with their respective target proteins with better binding energies and good pharmacokinetic properties than vitamin C and diclofenac; therefore, the interaction of these ligands with the antioxidant enzymes led to their activation and subsequent neutralization of the reactive oxygen species whereas the ligands mediated COX-1&2, and PDE-4B interaction through inhibition. Furthermore, adamantane, α-tocopherol, and γ-sitosterol reported for the first time in the plants rhizome could mitigate oxidative stress and inflammation being the activators of apoptosis, cytotoxic impaired autophagy, DNA damages, and mitochondrial dysfunction that contributes to the progression of non-communicable diseasess (NCDs).
Footnotes
Acknowledgments
We appreciate Mrs Ekaette Umoh and Mr Ukpong for their professional guidance on this study.
Availability of Data
All datasets generated in this study are available from the corresponding author on request.
Authors’ Contributions
The conceptualization and methodology was designed by EEA. SE, HUO, EAU, and AJ carried out the statistical analysis whereas the docking analysis was done by EEA, CME, and UOE. The financial resources were provided by EEA, GC, DNO, PBE, and VAO; then, the drafting and proofreading were done by EEA, NEU, HUO, OAE, AN, and JEO. The manuscript has been read and approved by all the authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The experimental protocol was strictly followed approved by the Faculty of natural and Applied Science animal Ethics Committee, Arthur Jarvis University with approval number AJUFNAS/0042. The study was carried out in accordance with ARRIVE guidelines.
Statement of Animal Rights
All the experimental procedures involving mice were conducted in accordance with the International Animal Care guidelines of Arthur Jarvis University, Nigeria and approved by Ethics Committee Faculty of Natural and Applied Science.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Consent for Publication
Not applicable
