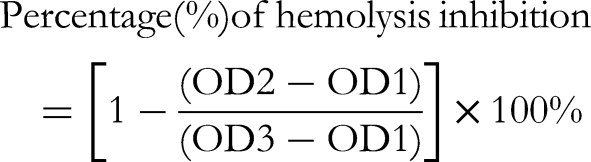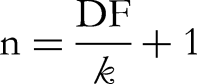Abstract
Objectives
This study aimed to investigate the chemico-pharmacological properties of the leaves methanolic extract (ME) of Zanthoxylum rhetsa (Roxb.) and its various solvent fractions, focusing on its antioxidant, cytotoxic, thrombolytic, anti-inflammatory, analgesic, hypoglycaemic, and antidiarrheal effects.
Methods
Phytochemicals in the ME were identified and characterized using gas chromatography and mass spectrometry (GC-MS). Antioxidant activity was measured via DPPH (2, 2-diphenyl-1-picrylhydrazyl) scavenging assay and measuring total phenolic content (TPC). Anti-inflammatory, thrombolytic, and cytotoxic effects were evaluated using membrane stabilization, clot lysis, and brine shrimp bioassays, respectively. In vivo evaluations of hypoglycaemic, antidiarrheal, and analgesic activities of the ME (200, 400 and 600 mg/kg bw) were conducted in mice model.
Results
A total of 65 phytoconstituents were identified in the ME, where 13-docosenamide (Z) (17.78%) and phytol (10.64%) were the most abundant. The aqueous soluble fraction (AQSF) fraction exhibited the highest TPC (181.875 mg GAE/gm of extractive) and antioxidant efficacy (IC50 = 4.78 μg/mL). The AQSF also exhibited promising antithrombotic properties, showing over 88% clot lysis capacity. Furthermore, chloroform soluble fraction (CSF) displayed 93.2% haemolysis inhibition under heat-induced conditions, while the inhibition of haemolysis was 46.8% in hypotonic conditions. All the tested three doses showed dose-dependent statistically significant (p < 0.05) analgesic and antidiarrheal efficacy, where only 200 and 400 mg/kg bw doses exhibited significant hypoglycaemic effects. In acute oral toxicity test, the ME exhibited median lethal dose greater than 4000 mg/kg bw.
Conclusions
Z. rhetsa leaf extractives demonstrated promising antioxidant, cytotoxic, antithrombotic, anti-inflammatory, analgesic and antidiarrheal effects, supporting the traditional uses of the plant species. Further extensive investigation, particularly on the AQSF fraction, is recommended to identify the responsible lead compounds.
Introduction
Synthetic drugs are often scrutinized due to potential side effects. 1 Conversely, traditional herbal remedies are becoming more popular for their diverse bioactive compounds, eco-friendly, and less likely to cause adverse reactions.2–5 Historically, around 80,000 medicinal plants have been used in Indian traditional medicine 6 and about 80% of the global population relies on plant-based medicines.4,7,8 In low-income countries, natural sources are considered as cost-effective and culturally accepted healthcare alternative. 9 In contemporary research, natural products and their structural diversity and novelty contribute substantially to advancing modern drug discovery and development. According to a recent report published by Newman and Cragg, 3 the identification and development of final therapeutic entities from natural compounds and/or synthetic versions, utilizing their new structures, is still an important and active technique in modern pharmaceutical research. For example, in the field of oncology, between 1946 and 1980, 40 of the 75 small molecules (53.3%) approved by the United States Food and Drug Administration (FDA) were either unaltered natural products or their derivatives. Between 1981 and 2019, 62 (33.5%) of the 185 FDA-approved small molecules were from natural products. This proportion rises to 64.9% when 58 synthetic medications are combined with natural product pharmacophores or synthetic mimics of natural products. 3
The Zanthoxylum genus, part of the Rutaceae family with over 160 genera and 2000 species, found globally, especially in Asia, America, and Africa, is well-known for being traditionally used to various ailments.10,11 Particularly, their decoctions are commonly used to treat infections like leishmaniasis, malaria, and various kinds of parasitic infection. 10 Furthermore, they demonstrated efficacy against several fatal ailments such as tumor, and sickle cell anaemia, in addition to fungal, viral, and bacterial infections. 10 They are also effective to prevent numerous insects especially mosquitos which are responsible deadly diseases such as malaria. 10 Secondary metabolites from Zanthoxylum such as alkaloids, coumarins, flavonoids, amides, lignoids, sterols, and terpenes exhibit antioxidant, analgesic, anti-inflammatory, and effects against dementia, obesity, and diabetes.10,12
Zanthoxylum rhetsa (Roxb.) DC (Synonym: Zanthoxylum budrunga; common Bengali name: Kantahorina), a prominent species of Zanthoxylum genus, is traditionally used in various medicinal preparations, especially its different parts such as roots, leaves, and barks. 13 For example, a paste from the spines of Z. rhetsa relieves pain and enhances lactation.13,14 Bark can alleviate chest and stomach aches, while they also exert their potentiality against snake bite. 12 Additionally, seeds and fruits can be employed to treat toothache, malaria, urinary diseases, bloating, rheumatism, and dizziness. 12 Insecticide properties have also been noted, as the decoction of leaves showed effectiveness against intestinal worm infections. 12 The new branches and leaves are consumed as foods and utilized as condiments, particularly among the diverse ethnicities of Arunachal Pradesh, India. 15 Furthermore, the root as a chewing stick has been traditionally used in Nigeria to treat teethache and dental decay because of the fragrant, warm flavor and the numbing effect when chewed. 16 Cholinergic, hypoglycemic, and spasmolytic effects are all exhibited by root bark. 13 Fruits and stem bark stimulate circulation, induce perspiration, and have antirheumatic and carminative properties. Fruits are also beneficial for illnesses like cholera, bronchitis, asthma, and toothaches. 13
Z. rhetsa is rich in bioactive compounds across its various parts, notably the bark, trunk, seeds, and roots. The bark holds numerous phytochemicals, including alkaloids like columbamine and furoquinoline derivatives, as well as terpenoids and phenolic compounds, underscoring its therapeutic potential.12,17 Roots are primarily a source of unique alkaloids, such as chelerybulgarine, 18 while terpenoids like lupeol are abundant in the bark and fruits. 19 The trunk contains nitidine, an alkaloid with strong antifungal and cytotoxic properties. 20 Essential oils, including sabinene and α-pinene, were identified in the fruit pericarp, and a gas chromatography-mass spectrometry (GC-MS) analysis of roots and bark revealed dibutyl phthalate as a major component.21,22
Despite extensive research on the Rutaceae family, particularly on the phyto-pharmacological properties of various Z. rhetsa parts like fruits, bark, and seeds, studies on the phytochemistry and in vivo efficacy of this leaves of the plant species are limited. Therefore, this study aimed to address this gap by analyzing the methanolic extract of Z. rhetsa leaves through GC-MS/MS to detect diverse secondary metabolites. Additionally, this study assessed the in vivo antidiarrheal, hypoglycemic, and analgesic activities, as well as in vitro cytotoxic, antioxidant, anti-inflammatory, and thrombolytic properties of the leaves of the Z. rhetsa species. Furthermore, an oral acute toxicity test was conducted to assess the safety profile of the plant species, as this area remains largely unexplored in prior studies. Through comprehensive phytochemical screening, the research may provide new insights into the chemical composition and therapeutic potential of Z. rhetsa leaves.
Materials and Methods
Plant Materials
The Z. rhetsa leaves had been gathered from the Habiganj Hill tracts in February 2022. The collected leaves were of good quality and were green. Based on the plant sample's physical characteristics, a taxonomist and scientific officer from the Bangladesh National Herbarium (BNH) in Dhaka, Bangladesh, recognized and verified it. A voucher specimen has been formally catalogued and stored at the BNH for future use. It was identified and authenticated with the accession number DACB 87252. The various parts of this plant Z. rhetsa species and its identification and accession number information were represented in the supplementary Figure S1.
Reagents and Chemicals
In this study, analytical-grade reagents and chemicals were utilized. Solvents, tert-butyl-1-hydroxytoluene (BHT), gallic acid, Folin–Ciocalteau reagent, and Tween 80 were sourced from Merck (Germany). 2,2-Diphenyl-1-picrylhydrazyl (DPPH) and Sephadex LH-20 were obtained from Sigma-Aldrich, (USA). Normal saline solution, loperamide, and glibenclamide were provided by Square Pharmaceuticals Ltd, Dhaka, Bangladesh.
Drying, Grinding and Extraction
On the day of collection, the freshly gathered leaves were immediately cleaned with clean water to get rid of any dirt or undesired items. Following a thorough cleaning, the leaves were allowed to air dry naturally for a few days in the warm winter sun until they were sufficiently brittle to break apart and be ground into powder. In order to protect heat-sensitive chemicals and stop their deterioration, the drying process was meticulously carried out at room temperature below 30 °C. The dried leaves were then ground into a coarse powder using a high-capacity grinding machine. Then the powdered sample (300 gm) had been macerated with 3.0 L of methanol in a closed amber container for 15 days with occasional stirring. A new cotton plug and Whatman filter paper No. 1 were used to filter the solvent mixture. A Buchi Rotavapor (BUCHI Labortechnik AG, Flawil, Switzerland) was used to obtain the concentrated extract. The consequent filtrate was then concentrated with the rotary evaporator at 35°C and under low pressure to generate a viscous crude extract (28.9% yield).
Fractionation
The modified Kupchan partitioning technique 23 was used to fractionate 5 grams of crude methanolic extract (ME). The extract was mixed with 10% aqueous methanol and consecutively extracted through pet-ether, dichloromethane, chloroform, and distilled water, producing four distinct fractions. This process was repeated twice to obtain more extractives. Each fraction was then evaporated to dryness, yielding approximately 19% pet ether soluble fraction (PESF), 27% dichloromethane soluble fraction (DCMSF), 25% chloroform soluble fraction (CSF), and 19% aqueous soluble fraction (AQSF) fractions.
GC-MS/MS Analysis
The detection of secondary metabolites, especially the volatile compounds, of the Z. rhetsa leaves methanolic extract was conducted with a Shimadzu auto-sampler connected to a GC-MS-QP2010 ultra (Japan). A 5 MS/HP column (30 m length, 0.25 mm wide, and 0.25 μm thickness) with ultra-pure helium (99.99%) as the carrier gas at a linear flow rate of 39 cm/s and a circulation rate of 1.12 mL/min was operated. The oven temperature has been raised uniformly at a rate of 10 °C per minute, starting from 110 °C and reaching up to 280 °C. The injector temperature was fixed at 250 °C, while 50 µL sample was introduced in splitless mode (10:1 ratio). Regarding spectra identification, an electron ionization energy approach was chosen with a substantial ionization power of 0.94 kV where 0.2 s was scanning duration, and fragments from GC-MS were obtained throughout an area of (m/z) 85 to 500 at an average speed of 10,000 u/s. The ion source and MS transfer line temperatures were maintained at 200 °C and 250 °C, respectively. The composition of phyto-molecules included in the test materials was detected in comparing the length of retention (min), peak region, peak intensity, and matching the mass spectral patterns of compounds according to the National Institute of Standards and Technology (NIST) library.
Antioxidant Activity
Cytotoxicity
The cytotoxic effects of extractives were assessed using the brine shrimp lethality test technique.
26
Dimethyl sulfoxide (DMSO) was utilized as the negative control and vincristine sulfate (VS) as the positive control.
27
To generate different concentrations, a serial of dilutions technique were applied (400, 200, 100, 50, 25, 12.5, 6.25, 3.125, 1.563, and 0.781 μg/mL), which were subsequently dissolved with DMSO before application. Each test tube holding the test samples, positive control or negative control was filled with twenty live nauplii (Artemia salina), which were then remained for 24 h at ambient temperature and light. Using a magnifying lens, the amount of surviving nauplii in each test tube was measured at post-incubation. The following formula was used to get the nauplii mortality rate.
Thrombolytic Activity
The thrombolytic potential of all extractives was determined using the technique described in prior investigation, with streptokinase (SK) as the standard and water as the negative control.
28
An Eppendorf tube containing 15,000,000 IU of lyophilized Alteplase (Streptokinase) was utilized. This was dissolved through vigorously mixing 5 mL of sterilized distilled water, with 1 mL (30,000 I.U.) serving as the positive control. Then, 1 mg of each extractive has been mixed in 1 mL of distilled water in different vials. Aliquots of venous blood (5 mL) were obtained from healthy volunteers, divided into ten separate sterile Eppendorf tubes, and incubated at 37 °C for 45 min. Each tube contains 0.5 milliliters of blood. When a clot was formed, the serum was extracted without damaging itself. 100 µL of testing materials, standard, and control were added to separate vials and left for 90 min at 37○C to clot lysis. After this period, the lysis clot was removed, and the residual clot weight was determined using the equation below.
Anti-Inflammatory Activity
The extractives of Z. rhetsa were evaluated for their anti-inflammatory effects using the membrane stabilization technique through two methods: heat and hypotonic induced hemolysis techniques. A Shimadzu UV spectrophotometer set to 540 nm was used to measure the absorbance of supernatant fluids, enabling the examination of membrane stabilizing effects in both methods. 29
Experimental Animals
Healthy (around 30 gm), and 4–5 weeks Swiss albino mice from both gender were collected from the animal division at the International Centre for Diarrheal Diseases and Research in Bangladesh (ICDDR,B). These animals had been kept in polypropylene cages under regulated settings (25 ± 2 °C, 55% humidity, and 12-h light/dark cycle). The rats were housed in metal cages with a 12-h light/dark cycle and a temperature control of 21 ± 2 °C. To assure ideal conditions, the space was regularly cleaned and had adequate ventilation. The rats were given unlimited access to a typical laboratory meal during their 14-day acclimation period before the experimental procedures began. 30
Analgesic Activity
Hypoglycemic Activity
The oral glucose tolerance test (OGTT) was used to evaluate the hypoglycemic effects of ME.34,35 All of the animals were fasted for eighteen hours before the experiment. One-touch glucometer (Bioland G-423S) was employed to determine the blood glucose level at zero hours. After that, each group was given their specified medication orally. Group II had the standard medication glibenclamide (10 mg/kg b.w.) whereas control group (Group I) had taken 1% Tween 80 solution in normal saline (10 mL/kg b.w.). All groups received an oral 10% glucose solution (2 mg/kg b.w.) after an hour. To measure blood glucose levels, blood samples were taken from each mouse's tail vein 30, 60, and 90 min after the glucose was administered. The hypoglycemic activity was quantified by calculating the percentage reduction in blood glucose levels. The % reduction was determined using the following formula and contrasted to both negative and positive control groups:
Antidiarrheal Activity
The antidiarrheal effect of ME was assessed by employing the castor oil-induced diarrhea technique.
36
Prior to the trial, all animals were starved for 18 h. The standard medication loperamide (50 mg/kg b.w.) and crude extracts (ME) were given orally, whereas the Group I got a 1% Tween 80 solution in normal saline (10 mL/kg b.w.). After one hour, all of the mice received fresh castor oil (1 mL) to stimulate diarrhea. Each mouse was remained in individual crates coated with paper towels. The total count of diarrheal feces of every mouse was recorded after four hours of castor oil administration. The findings of the experiment were compared to both negative and positive control groups. The percentage of reduction of diarrheal feces was determined through the below formula:
in Vivo Oral Acute Toxicity
Twenty male Swiss Albino mice, weighing between 28–34 gm and aged 8 to 10 weeks, were used in the acute oral toxicity test of the crude methanolic extract of Z. rhetsa. The experiment followed the protocol described by Rahman et al 31 and adhered to the guidelines set forth by the Organization for Economic Co-operation and Development (OECD 423). The mice were administered single oral doses of the leaf extracts at concentrations ranging from 1000 to 4000 mg/kg, while the control group received a vehicle solution. Before administration, the mice were fasted overnight. Observations were conducted over a subsequent seven-day period. Group 1 (n = 5) received 1000 mg/kg bw of leaf extract, Group 2 (n = 5): 2000 mg/kg b.w. of leaf extract, Group 3 (n = 5): 3000 mg/kg bw of leaf extract, and Group 4 (n = 5): 4000 mg/kg b.w. of leaf extract. Each mouse was provided with standard food and water for sixty minutes after administering the extract. The mice that received the drug were observed for thirty minutes following administration, then hourly for eight hours, and daily for the subsequent seven days. During the observation period, detailed visual assessments were conducted, including monitoring for mortality, behavioral changes (such as locomotion, sleep, or convulsions), alterations in physical appearance (such as immobility or body tremors), and signs of injury.
Statistical Analysis
The in vivo pharmacological test results have been displayed as mean ± SEM (n = 4) or in percentage. The in vivo analysis was performed in GraphPad Prism (version 9.3.1), following Dunnett's multiple comparisons and one-way ANOVA statistical analysis. The level of statistical significance was assessed in comparison to the normal control group and expressed the level of significance as “*”, “**”, “***”, and “****”, which denoted p < 0.05, p < 0.01, p < 0.001, and p < 0.0001, respectively. Moreover, because the data were considered from a single in vitro experiment, no formal statistical evaluation was carried out for the in vitro testing; instead, the findings were compared numerically. However, all the data was processed, and all the images were generated by using GraphPad Prism (version 9.3.1) software.
Results
Phytochemical Analysis: GC–MS/MS Analysis
A total of 65 phytoconstituents were predominantly found in the ME based on their GC-MS analysis. The spectrum obtained from GC-MS analysis is available as Figure 1. Among the detected compounds, 13-docosenamide, (Z)- (17.78%) was the most abundant compound. After that, phytol was predominant (10.64%) (Table 1). Furthermore, hexadecanoic acid, methyl ester (9.66%), 9,12,15-octadecatrienoic acid, methyl ester (Z, Z, Z)- (8.2%), glycerin (5.56%), and squalene (5.2%) were discovered in extensive amounts. In contrast, 3H-3,10a-methano-1,2-benzodioxocin-3-ol, octahydro-7,7-dimethyl-, (3α,6αβ,10αβ)- (0.22%), 4-[α-[2-hydroxyethyl]ethylamino]-4-toluidino]-7-chloroquinoline (0.22%), 2(5H)-furanone (0.22%), octadecanamide (0.2%) were the least amount compounds (Table 1). Though some polar compounds were also identified in this investigation, most of the detected phytocompounds were non-polar cyclic molecules. Several frequent diterpenoids, and triterpenoids such as phytol, ergosterol, retinal, tocopherol, and squalene were also identified. Some aromatic compounds such as (hexahydro pyridine, 1-methyl-4-[4,5-dihydroxyphenyl ]-, 1-(1-Hydroxy-1-methylethyl)-3-methoxymethyl benzene, 4-[α-[2-hydroxyethyl]ethylamino]-4-toluidino]-7-chloroquinoline, benzofuran, 2,3-dihydro-, 2-methoxy-4-vinylphenol, acetamide, 2-amino-N-phenyl-2-thioxo) were also recorded. Finally, all identified compounds’ names, PubChem CIDs, retention time, molecular formula, % area, molecular weight, and the literature reported pharmacological activities of them were tabulated in Table-1.
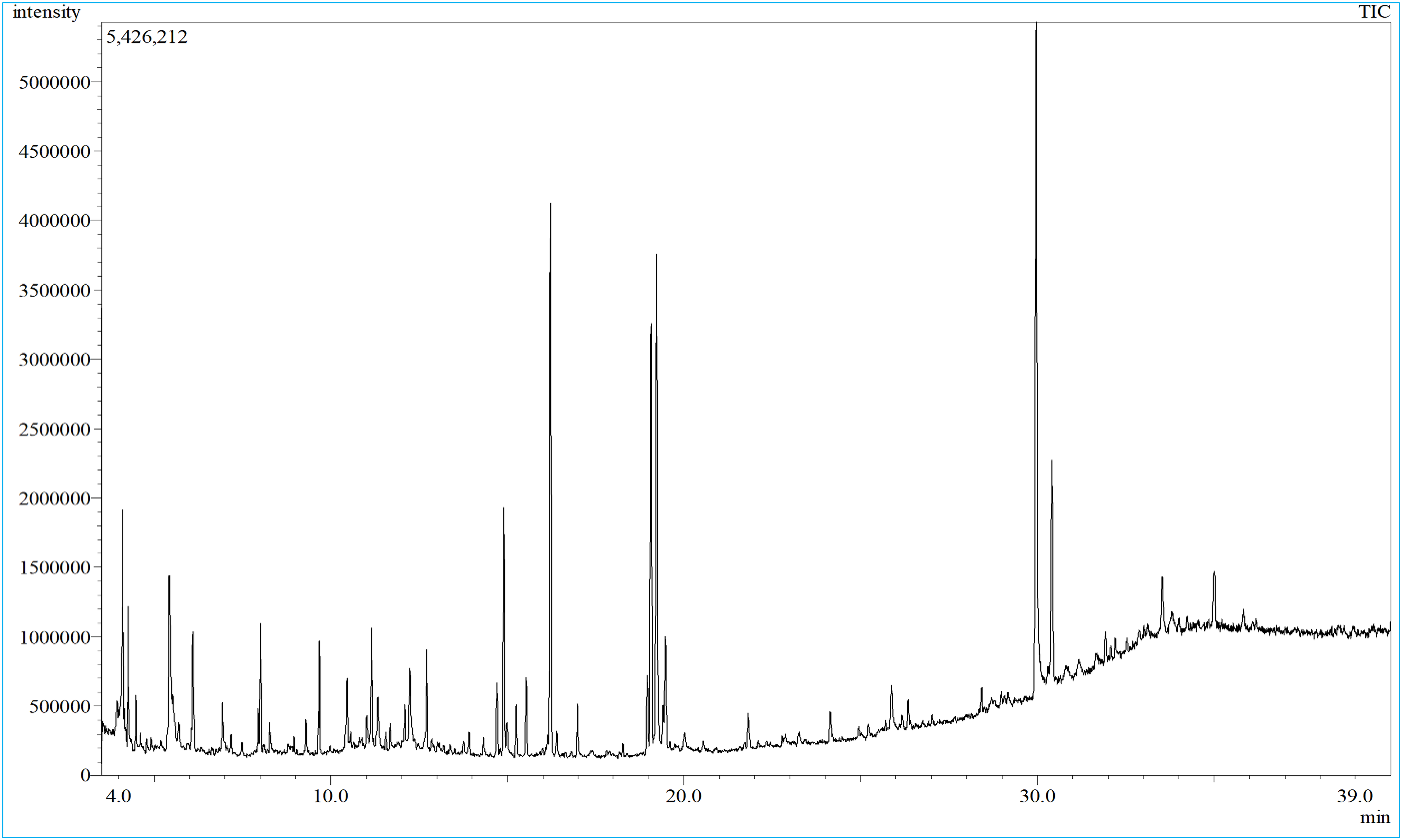
The total ionic chromatogram obtained from GC-MS/MS analysis of methanolic crude extratc of leaf of Z. retsha.
A Total of 65 Phytocompounds Characterized Through GC-MS Analysis from the Z. rhetsa Leaves Methanolic Extract. The Pharmacological Effects Reported in Literature with the Relevant Citations Were Also Added in the Table.

Antioxidant Potency

Estimation of total phenolic content and antioxidant effects of different fractions and ME of Z. rhetsa leaves. (A) The standard curve generation of gallic acid (GA) where the absorbance at 760 nm was used to create the curve. (B) The measured TPC (mg of GAE/gm of extractives) of the methanolic extract and its various solvent extractives. (C) The representation of DPPH free radical scavenging activity of the methanolic extract (ME) and its various solvent fractions. The inhibition of free radicals demonstrated a concentration-dependent relationship, indicating that higher extract concentrations correlated with greater scavenging capacity. (D) The half-maximal inhibitory concentration (IC50) values, expressed in μg/mL, were determined for the methanolic crude extract, various partitioned fractions of Zanthoxylum rhetsa leaves, and standard reference compounds found from the DPPH free radical scavenging assay. The abbreviations ME, PESF, DCMSF, CSF, and AQSF refer to the methanolic extract, petroleum ether soluble fraction, dichloromethane soluble fraction, chloroform soluble fraction, and aqueous soluble fraction, respectively. The standard compounds BHT and AA represent butylated hydroxytoluene and ascorbic acid, respectively. The GraphPad Prism program was employed to make the graphs (A-D).
Cytotoxicity
The relationship between the logarithmic values of the sample concentrations used in the brine shrimp lethality bioassay and the mortality % is shown in Figure 3A. These results provide information on the cytotoxic potential of the samples as assessed by the brine shrimp lethality test. Among the all extractives, ME, PESF, and DCMSF exerted potential cytotoxic effects in brine shrimp lethality assay. Their median lethal concentration (LC50) values were belong to 11.88 to 26.6 µg/mL, while standard vincristine sulphate showed 0.451 µg/mL (Figure 3B). However, another two fractions CSF (LC50 = 150.29 µg/mL), and AQSF (LC50 = 145.35 µg/mL) demonstrated moderate level cytotoxic effect (Figure 3B).

The cytotoxic potential of the crude methanolic leaf extract of Zanthoxylum rhetsa and its various solvent fractions. (A) Illustration of the dose-response relationship, depicting the correlation between the percentage of mortality and the logarithmic transformation of the sample concentrations. (B) The median lethal concentration (LC50) values of methanolic extract and its various fractions of Z. rhetsa leaves obtained from brine shrimp lethality assay. The abbreviations used in this figure are defined as follows: ME refers to the methanolic extract, PESF denotes the petroleum ether soluble fraction, DCMSF represents the dichloromethane soluble fraction, CSF indicates the chloroform soluble fraction, and AQSF corresponds to the aqueous soluble fraction. The standard compound, vincristine sulfate, is abbreviated as VS. The graphical representations of the data were generated using the GraphPad Prism software, which facilitated the statistical analysis and visualization of the results.
Thrombolytic Activity
In comparison with standard drug streptokinase (64.2% clot lysis), AQSF demonstrated a higher thrombolytic activity (88.5% clot lysis) (Figure 4A). Rest of extractives such as ME and DCMSF revealed minor thrombolytic properties, while PESF and CSF showed moderate level of thrombolytic activity (Figure 4A).
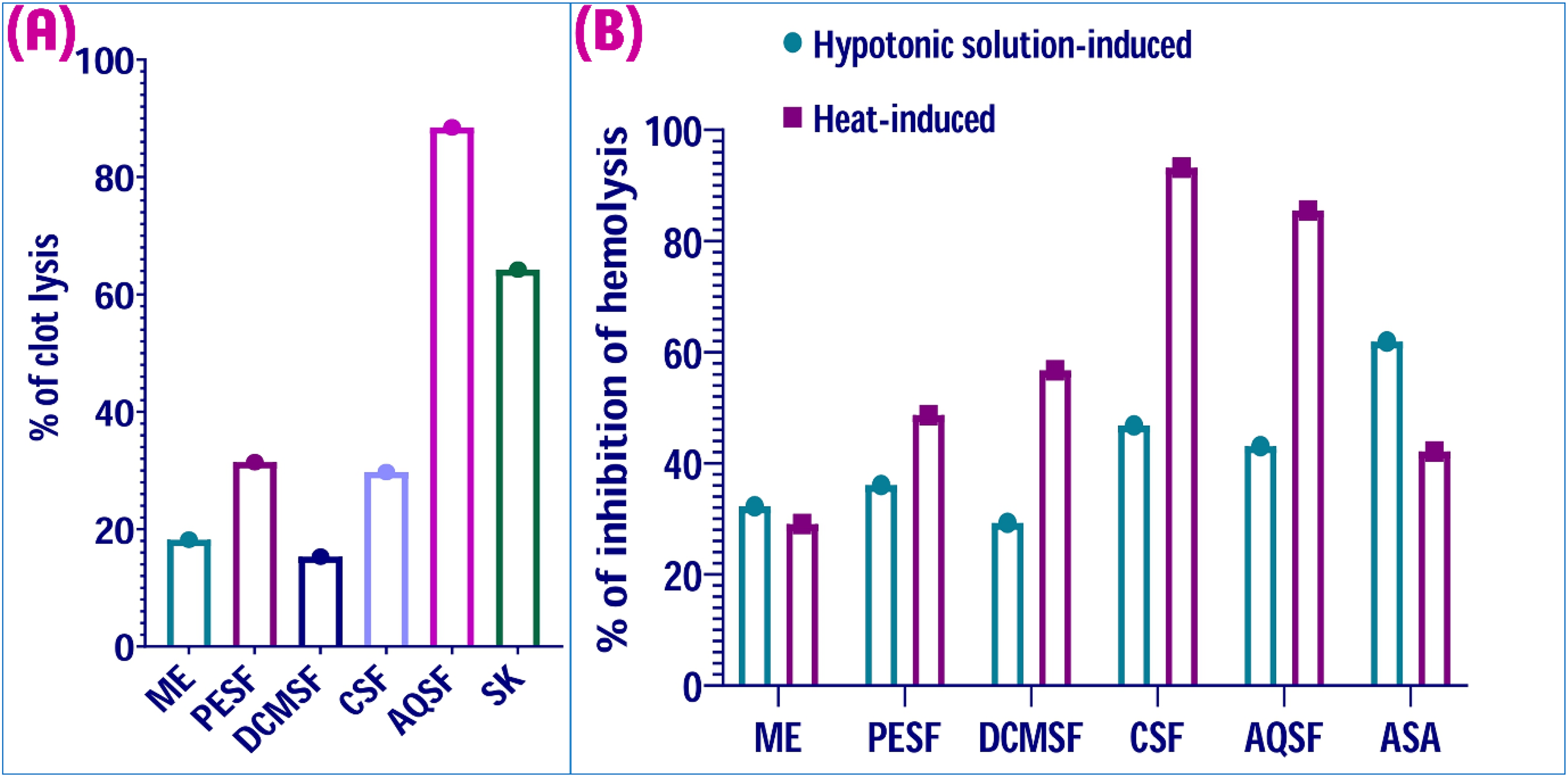
Antithrombotic and anti-inflammatory effects of the methanolic extract and its various solvent fractions of the leaves of Zanthoxylum rhetsa species. (A) Thrombolytic activity in terms of percentage of clot lysis of methanolic extract and its various solvent fractions of Z. rhesta leaves. (B) Inhibition of hemolysis (%) of different extractives of leaves Z. rhetsa in hypotonic solution-induced and heat-induced conditions. The following definitions apply to the abbreviations used in this figure: The terms ME, PESF, DCMSF, CSF, and AQSF stand for methanolic extract, petroleum ether soluble fraction, dichloromethane soluble fraction, chloroform soluble fraction, and aqueous soluble fraction, respectively. SK and ASA stand streptokinase and acetyl salicylic acid, respectively.
Anti-Inflammatory Activity
Under heat-induced conditions, except ME (29.1%), all the fractions such as PESF (48.7%), DCMSF (56.7%), CSF (93.2%), and AQSF (85.5%) showed higher hemolysis inhibition than the standard drug ASA (42.0%) (Figure 4B). In hypotonic solution-induced conditions, no samples surpassed the standard ASA (61.9%), though CSF (46.8%) and AQSF (43.1%) exhibited moderate membrane-stabilizing effects (Figure 4B).
Central Analgesic Activity
The results from the central analgesic test in terms of tail immersion time (sec) were expressed in the Figure 5A. Following the administration of the loading doses of the samples, significant increases in tail immersion times were observed at 30, 60, and 90 min across all tested doses of the extracts compared to the normal control group. It is noteworthy to mention that following the administration of the loading dose, a 400 mg/kg dose of the crude extract (ME) demonstrated significantly (p < 0.05) enhanced efficacy at 30 min compared to the 200 mg/kg dose. Furthermore, the 600 mg/kg dose exhibited a significantly (p < 0.05) higher pain reaction time at 90 min compared to the 200 mg/kg dose, revealing a dose-dependent pharmacological effects (Figure 5A).

Analgesic property of the methanolic leaf extract of Zanthoxylum rhetsa.
Peripheral Analgesic Activity
The data obtained from the acetic-induced peripheral analgesic test in terms of average number of writhing (mean ± SEM) are expressed in the Figure 5B, and the percentage of writhing inhibition has been represented in the supplementary Figure S2. This in vivo experiment found that all the tested doses of the ME displayed significant (p < 0.0001), dose-dependent peripheral analgesic activity compared to the normal control group (Figure 5B). Furthermore, the 600 mg/kg dose exhibited a significantly (p < 0.05) lowered the number of the writhing response compared to the 200 mg/kg dose, revealing that a higher dose showed better analgesic property (Figure 5B).
Hypoglycemic Activity
The results obtained from OGTT showed that the 200 and 400 mg/kg doses ME of Z. rhetsa leaves, and the standard drug glibenclamide exhibited significant (p < 0.05) blood glucose lowering capacity in vivo compared to the normal control group at the consideration of 60 min of the experiment (Figure 6A). At 90 min of the experiment, only the lowest dose (200 mg/kg bw) ME and the standard drug glibenclamide showed significant (p < 0.05) hypoglycemic property compared to the normal control group. No statistically dose-response relationship among the tested doses was observed in this experiment.
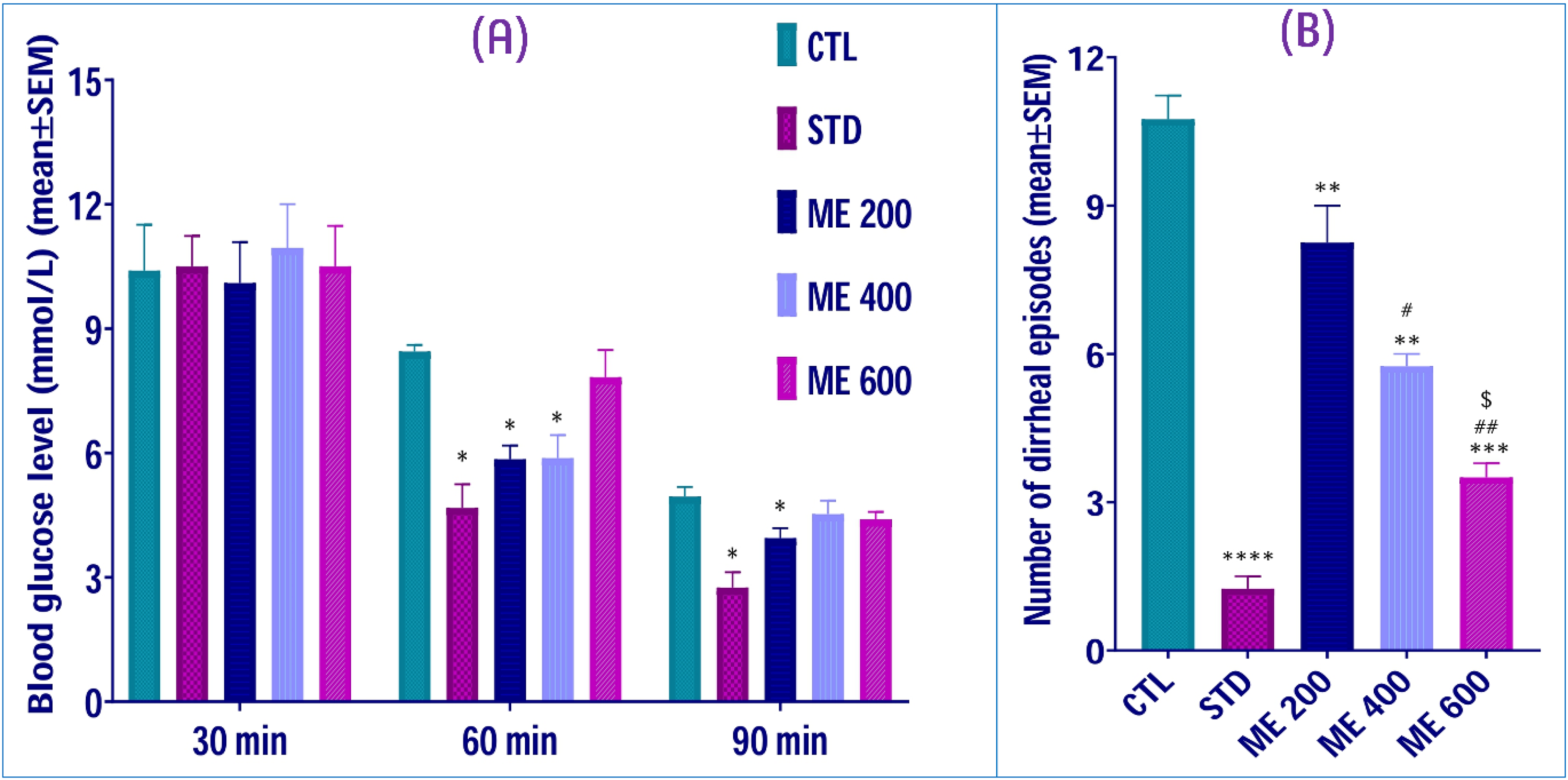
Hypoglycemic and antidiarrheal property of the leaf extract of Z. rhetsa species. (A) Effects of crude methanolic extract of Z. rhetsa leaves on blood glucose level (mmol/L) of mice in oral glucose tolerance test. (B) Effects of crude methanolic extract of Z. rhetsa leaves on number of diarrheal episodes of mice in castor oil-induced antidiarrheal test. Data are represented as mean ± SEM for n = 4, “*”, “**”, “***”, and “****” denote p < 0.05, p < 0.01, p < 0.001, and p < 0.0001 versus control group, respectively. “#” and “##” denote p < 0.05 and p < 0.01 versus ME 200 group, respectively. “$” shows the level of significance as p < 0.05 versus ME 400 group.
Antidiarrheal Activity
In this experiment, all the tested doses of the ME, including the standard drug loperamide, showed very statistically significant (p < 0.01) antidiarrheal effects compared to the control group (Figure 6B). The tested crude extracts exhibited a dose-response relationship as the 400 and 600 mg/kg doses lowered significantly (p < 0.05) the diarrheal episodes of the mice compared to the ME 200 mice group, which were administered 200 mg/kg bw. In addition, the ME 600 group exerted significantly (p < 0.05) higher antidiarrheal capacity compared to the ME 400 group (Figure 6B). Furthermore, the supplementary Figure S3 exhibits the percentage of the inhibition of diarrheal episodes of the three tested doses.
Acute Toxicity Assessment
When the extract of Z. rhetsa was administered orally at levels between 1000 and 4000 mg/kg, there were no side effects or deaths associated with the treatment. The animals in the control group and those exposed to the extract did not exhibit any discernible changes in behavior, breathing patterns, cutaneous reactions, water intake, food consumption, or body temperature during either of two periods of observation: four hours or seventy-two hours. With the estimated LD50 exceeding 4000 mg/kg, these observations point to the safety at a 4000 mg/kg dose of the methanolic crude extract.
Discussion
The present study examined the chemico-pharmacological profiles of Z. rhetsa leaves methanolic extract. Photochemical analysis found that leaves of Z. rhetsa is the treasure trove of various types of phytochemicals. The current experiment expressed the 65 phytoconstituents and most of them have antioxidant, antibacterial, and anticancer effects. Organic acid (5-bromopentanoic acid, dihydroxybenzoic acid), esters (5-bromo pentanoic acid, 4- methylpentyl ester, methyl ester-7-hexadecenoic acid), aliphatic hydrocarbons (squalene, 3,3-dimethyl-hexane, caryophyllene, neophytadiene), alcohols (2,4- hexadien-1-ol, glycerin, vinyl phenol, d-allose, d-mannitol, α-tocopherol), amines and amides (13(Z)-docosenamide, octadecanamide, allocryptopine), ketones (4-hydroxy-3-methyl- 2-butanone, 4-cyclopentene-1,3-dione, 2(5H)-furanone, cyclohexanone), benzopyrone (loliolide, and coumarine), anhydride derivatives ((+)-sesamin retinal, ethyl-2,2-difluoroheptacosanoic acid, and ergostane- 3,5,6,12,25-pentol) are notable (Table 1). These findings underscore the diverse array of bioactive compounds present in different parts of Z. rhetsa, which may contribute to its medicinal and therapeutic potential. Previous reports of Z. rhetsa indicated that alkaloids are more prominent and the barks are the major reservoir of several phytochemicals, including alkaloids (columbamine, 8-methoxy-N-methylflindersine, dictamnine, skimmianine, rutaecarpine, canthin-6-one, and evodiamine), terpenoids (lupeol, and 3,5-dimethoxy-4-geranyloxycinnamyl alcohol), coumarin (xanthyletin) and phenolic compounds.12,17,19,75,76 In a separate study, Ahsan (2014) isolated and reported alkaloids such as chelerybulgarine, simulanoquinoline, 2-episimulanoquinoline, and rhetsidimerine from the roots of the plant species. 18 Furthermore, lignans such as yangambin, kobusin, and sesamin, alongside a phenolic compound syringaresinol were also noted.17,18,75 Various GC-MS analyses of different parts of Z. rhesta (roots barks and fruit pericarp) revealed that dibutyl phthalate, sabinene, β-phyllandrene, α-pinene, 4-terpineol, γ-terpinene, and α-elemene were the main constituents of the plant species.21,22 Furthermore, some anti-inflammatory molecules such as 2,3-pinanediol, and terpinen-4-ol were identified from seeds, fruits and pericarp of the plant species. 77
Oxidative stress is caused by an excess generation of free radicals in the body compared to its capacity to eliminate them. This aberrant oxidative stress can develop degenerative and chronic diseases like rheumatoid arthritis, autoimmune disorders, ageing, cataracts, neurological diseases, and cancer. The body produces antioxidants or they can be taken in the form of supplements or food. 3 Plant phenolic compounds act as antioxidants and are vital in defense response because they can act as anti-aging, anti-inflammatory, and anti-proliferative activities. 78 The current study revealed that the fractions and ME, except PESF and DCMSF, had significantly larger amount of TPC (122.5 mg of GAE/gm to 181.87 mg of GAE/gm). Previous study also showed that Z. rhetsa had TPC content (15.5 mg of GAE/100 gm).79,80 A recent study showed that ethanolic root-bark extract expressed notable DPPH scavenging activity (IC50 = 42.65 μg/mL), 21 while essential oil from Z. rhetsa seeds (at concentration 1200 μg/ml) exhibited 78.5% and 75.5% activity in comparison to standard (α–tocopherol). 81 Another study demonstrated that the leaves of Z. rhetsa indicated 92.5% inhibition property at DPPH free radical scavenging test. 80 Moreover, in Zohora et al study, the AQSF (IC50 = 55.25 μg/mL) showed the highest antioxidant activity among the other fractions. 82 The same potent antioxidant activity has been noted in this current investigation. As like as Zohora et al, 82 AQSF fraction in this research showed the strongest antioxidant activity even more than standard drug BHT. Unsaturated or phenolic compounds found in the leaf of the Z. rhetsa plant species may be responsible for the antioxidant property. Phytoconstituents detected through GC-MS techniques, including 9,12-octadecadienoic acid, 9,12,15-octadecatrienoic acid, methyl ester, hexadecenoic acid, 2-hydroxy-1-(hydroxymethyl)ethyl ester, carbonic acid might be the responsible leads for such antioxidant activities.83–86 The most abundant compound extracted from the leaves of Z. rhetsa was 13-(Z)-docosenamide, which has shown promising antioxidant activities. 66 Another two molecules, phytol and squalene, have antioxidant characteristics.87,88 It is noteworthy that the antioxidant properties of squalene are primarily attributed to its unique molecular characteristics and its ability to interact with free radicals, where squalene not only neutralizes free radicals but also inhibits the generation of reactive oxygen species (ROS). 29
Very limited research has been conducted to find out the cytotoxic activity of the leaves of Z. rhetsa. In this study, the extractives ME, PESF, and DCMSF showed potential cytotoxic activity at brine shrimp lethality assay (LC50 = 11.88 to 26.6 µg/mL). Another two fractions CSF (LC50 = 50.29 µg/ml), and AQSF (LC50 = 145.35 µg/ml) revealed a moderate level of cytotoxic effect, while prior research exerted the four fractions of methanolic extract of root-bark had remarkable cytotoxic effects (LC50 = 0.52 to 1.52 µg/ml). 82 Furthermore, another investigation demonstrated the average LC50 of ethanolic extract of root-bark was round to 22 μg/mL in brine shrimp lethality test. 89 These data denoted the evidence that leaves of Z. rhetsa has cytotoxic effect. Various phytochemicals identified through GC-MS techniques such as 2(5H)-furanone, caryophyllene, 2,3-dihydro-benzofuran, D-allose, 10-O-(t-butyloxy)-dihydroartemisinin, 3,7,11,15-tetramethyl-2-hexadecen-1-ol, 3-methyl-4-(phenylthio)-2-prop-2-enyl-2,5-dihydrothiophene 1,1-dioxide, (+)-sesamin may be accountable for this extensive cytotoxic effects of Z. rhetsa.43,47,48,52,63,73 One previous study reported that the cytotoxic effects of nitidine isolated from the trunk of Z. rhetsa showed low cytotoxicity against the normal cell line Vero, (IC50 = 140.65 µM) but strong inhibitory action against the KB, LU-1, HepG2, LNCaP, and MCF7 cell lines (IC50 = 0.28, 0.26, 0.27, 0.25, and 0.28 µM, respectively). 20 The findings of the current study also support the anti-cancer efficacy reported in the previous studies.
Although modern therapeutics are effective in alleviating inflammation-induced pain, their use is often accompanied by adverse effects. Herbal therapy is considered as a safe and alternative to conventional treatments for inflammation management that may not have many adverse effects. 90 In current study, the CSF and AQSF derived from Z. rhetsa leaves demonstrated higher anti-inflammatory activity compared to the standard drug ASA by preventing erythrocyte hemolysis under heat-induced conditions. Several anti-inflammatory phytomolecules, such as loliolide, 2-methoxy-4-vinylphenol, D-allose, (1α,2β,3α,5β)-1,2,3,5-cyclohexanetetrol, 2,6-dihydroxybenzoic acid, 3-methyl-4-(phenylthio)-2-prop-2-enyl-2,5-dihydrothiophene 1,1-dioxide, squalene, allocryptopine and 1-decylsulfonyl-D-mannitol were detected and might be responsible for this response.43,46,48,53 These outcomes indicated that Z. rhetsa leaves may have the same degree of anti-inflammatory compared to the pharmacological properties of the bark part of this plant species. Santhanam et al 91 reported that the compound hesperidin isolated from the methanolic bark extract of the plant species considerably reduced the generation of inflammatory moieties such as TNF-α, IL-1β, and IL-6. Another study conducted by Santhanam et al 92 further revealed that the crude methanolic extract of the bark and its fractions (hexane, chloroform, ethyl acetate, and butanol) reduced cytokine levels in a dose-dependent manner, while chloroform fraction showed the most significant inhibition on pro-inflammatory cytokines in LPS-stimulated RAW 264.7 macrophages. This effect was linked to the prevention of IκBα degradation and suppression of NF-κB signaling targets, blocking NF-κB activation, and subsequent pro-inflammatory gene transcription. 92 Moreover, the ethanolic : hexane (95:1) extract from the pericarp of the Z. rhetsa plant species, along with the essential oil, exhibited the strongest inhibition of NO production (IC50 = 11.99 μg/ml, and 15.33 μg/ml, respectively). They also demonstrated the significant anti-inflammatory effects on TNF-α (IC50 = 36.08 μg/ml, and 34.90 μg/ml, respectively). 77
One of the main causes of morbidity and mortality in a variety of vascular conditions is thrombosis. 93 Patients with blocked veins or arteries are given fibrinolytic drugs, which activate tissue plasminogen activator (t-PA) and have a thrombolytic effect. 94 To find out the potential thrombolytic agent, one prior roots-barks investigation of Z. rehtsa reported that carbon-tetra chloride, chloroform, and aqueous fractions had noticeable thrombolytic activity expressed as 41.13%, 43.77%, and 50.47%, respectively. 82 Furthermore, almost all of the Z. rhetsa leaves extractives used in the present investigation showed notable thrombolytic action. Though extensive preliminary studies on the thrombolytic activity of Z. rhetsa are still missing, this finding indicated that further investigations are needed that may aid in the development of new thrombolytic agents from this plant species.
Due to low side effects, herbal pain relievers are being investigated as possible substitutes for synthetic medications. 95 The results of this study indicate that the ME of the plant caused a significant decrease in central analgesic pain reaction. Similarly, peripheral analgesic evaluations revealed a significant reduction the acetic acid-induced pain perception in mice model. The presence of high concentration of antioxidant compounds may be the responsible of their analgesic effects. Oxidative stress and inflammation are closely related pathophysiological processes of pain. 95 Therefore, the presence of antioxidant molecules may reduce the pain of the experimental mice, especially in the peripheral analgesic investigation technique. Any extensive analgesic investigation of Z. rhetsa could not be found in the literature, suggesting that this may be the first investigation into its analgesic effects.
The metabolic disease diabetes mellitus is a global public health concern. 96 Consequently, there is a growing demand for safe and efficient anti-diabetic compounds made from natural sources due to their low toxicity, low cost, and widespread availability. 95 The current investigation showed that, at 60 min into the OGTT, the methanol extract (ME) of Z. rhetsa leaves at doses of 200 and 400 mg/kg body weight significantly reduced blood glucose levels in vivo when compared to the normal control group. Only the lower dose of the extract (200 mg/kg body weight) continued to have a significant hypoglycemic effect at 90 min in comparison to the normal control group. A prior research of the Z. rhetsa root-bark ethanolic extract found considerable hypoglycemic efficacy in an in vivo mice model (alloxan-induced diabetes). An in vitro experiment on pancreatic α-amylase found that the extract had significant antidiabetic properties (IC50 = 81.45 μg/mL). 21 Due to the limited research on the hypoglycemic properties of leaves of this plant species, it is highly recommended to conduct comprehensive in vivo and in vitro analyses using various methods to accurately determine the antidiabetic efficacy of Z. rhesta leaves.
The three primary mechanisms responsible for castor oil-induced diarrhea include the production of nitric oxide, an increase in calcium permeability of the gastrointestinal membrane, and the synthesis of prostaglandins. These processes collectively result in an elevation of fluid and electrolytes within the digestive tract, as well as the stimulation of peristalsis. It has been shown that ricinoleic acid, the primary component of castor oil, can cause diarrhea by stimulating peristaltic movement and producing prostaglandins, which ultimately damage the intestinal barrier. However, prostaglandin inhibition is significantly induced by antioxidant compounds.97,98 In this current study, ME of Z. rhetsa leaves showed extremely significant antidiarrheal potency in a dose-dependent manner. One previous investigation demonstrated that methanolic extract of Z. rhetsa stem bark oral doses of 250 and 500 mg/kg significantly suppressed acetic acid-induced intestinal movements and castor oil-induced diarrhea in mice model. 99 Although, no notable anti-diarrheal compounds were identified, several antioxidant compounds were detected from this GC-MS analysis (Table 1), which may be responsible for the inhibition of castor oil-induced gut inflammation diarrhea effects.
In addition to the therapeutic potential and efficacy of Z. rhetsa leaves extract, its toxicity and safety profile must be carefully assessed. However, the safety and toxicity analysis of this plant were not performed extensively. The current study found no noticeable side effects or fatalities, while the calculated LD50 was found to be above 4000 mg/kg, placing the extract in the safe category. These results align with those of a previous study, which also reported no signs of morbidity, toxicity, or significant disruption in behavioral or motor functions in female Swiss albino mice. 100 Moreover, the previous study also reported that the key organ health, serum biochemistry, and hematological parameters remained unchanged compared to the control group, and the calculated LD50 was over 5000 mg/kg. 100 These findings are in line with toxicological studies of other Zanthoxylum species, such as Z. armatum, whose fruit and leaf extracts were shown to be safe at 2000 mg/kg body weight. 101
Limitations and Future Studies
In the present study, the identification and characterization of compounds potentially present in the methanolic crude extract of the Z. rhetsa leaves were conducted exclusively using the GC-MS technique. However, a comprehensive phytochemical investigation employing advanced chromatographic methods is currently ongoing and will be reported in future studies. Each in vitro bioactivity result is based solely on a single assay, which is another significant flaw in the research. Although such data is sometimes considered inadequate primary evidence for drawing definitive scientific conclusion because of potential biases, errors, or variability, these tests offer essential initial insights for developing hypotheses in subsequent research. Additionally, several factors such as the harvesting season, time of collection (day or night) of plant species were not considered in this study. The biological investigation of the extractives conducted was preliminary in this report. Therefore, more comprehensive biological studies are needed to confirm the pharmacological potential of this plant species. In addition, computational chemistry could also provide valuable insights into the mechanisms of action of the bioactive compounds to support these pharmacological actions.
Conclusions
Employing GC-MS approach, the phytochemical evaluation of Z. rhetsa leaf methanolic extract identified and characterized over 60 secondary metabolites. These molecules could have therapeutic advantages. According to pharmacological testing, the aqueous fraction showed promising in vitro thrombolytic, antioxidant properties. Additionally, the chloroform and aqueous fractions showed notable heat-induced membrane-stabilizing properties. All three tested doses (200, 400, and 600 mg/kg bw) of the methanolic extract demonstrated dose-dependent statistically significant (p < 0.05) in vivo analgesic and antidiarrheal activities. However, significant hypoglycemic effects were observed only for the 200 and 400 mg/kg bw doses. In the acute oral toxicity assessment, the median lethal dose (LD50) of the ME was found over 4000 mg/kg bw. However, further extensive studies, especially the phytochemical isolation, are required to confirm the lead compounds of the leaves of the plant species responsible for these pharmacological activities.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251319206 - Supplemental material for Unveiling Phytochemicals and Antioxidant, Cytotoxic, Antithrombotic, Anti-Inflammatory, Analgesic, Hypoglycemic and Antidiarrheal Activities from the Leaves Extract of Zanthoxylum rhetsa (Roxb.) DC
Supplemental material, sj-docx-1-npx-10.1177_1934578X251319206 for Unveiling Phytochemicals and Antioxidant, Cytotoxic, Antithrombotic, Anti-Inflammatory, Analgesic, Hypoglycemic and Antidiarrheal Activities from the Leaves Extract of Zanthoxylum rhetsa (Roxb.) DC by Md. Jamal Hossain, Md. Abdus Samadd, Sharmin Akter, Md. Shohel Hossen, Abdul Kuddus, Md. Azizur Rahman Tamim, Marufa Maharin Mitu and Mohammad A. Rashid in Natural Product Communications
Footnotes
Acknowledgments
The authors appreciate the scientific equipment support received from the Department of Pharmacy, State University of Bangladesh.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Clearance
The State University of Bangladesh's Animal Ethics Committee thoroughly examined and approved the investigation's ethical rules and protocols (2022-07-20/SUB/A-ERC/008).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This experiment subsequently followed the standards for ethical management of animals as defined by the Federation of European Laboratory Animal Science Associations (FELASA).
Statement of Informed Consent
Since there are no human subjects’ involvement, consent permission is irrelevant.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.