Abstract
Introduction
Tannic acid (TA) is a naturally occurring polyphenol commonly found in tea, fruits, and wine.1–5 The molecular structure of TA consists of a central glucose unit linked to 10 gallic acid moieties (Figure 1). This compound exhibits several unique properties and so has been used to develop a variety of interesting biomaterials. As an example, TA is able to form strong bonds with metals so as to generate stable surface coatings that improve the physical and chemical properties of various implants as a means of increasing implant success rates. 4 In addition, because TA exhibits exceptional biocompatibility and good antibacterial and antioxidant characteristics, this compound has applications in cosmetics and pharmaceuticals. However, limited research has been conducted regarding the complexation of TA with phospholipid aggregates, such as liposomes, which are widely used in biomaterials due to their high biocompatibility.6–10 On this basis, the present work examined the hybridization of TA with liposomes composed of Technol PG (Figure 1). Technol PG comprises a mixture of C16 and C18 fatty acids having 0 (C18-0), 1 (C18-1), 2 (C18-2), or 3 (C18-3) double bonds, with each fatty acid connected to a phosphatidylglycerol moiety. Traditionally, phosphatidyl glycerol derivatives have been obtained by organic synthesis11,12 which sometimes takes cost. Since Technol PG is less expensive than dioleoylphosphatidyl glycerol, a representative phosphatidyl glycerol, the complex between TA and Technol PG evaluated in this work is considered to be more practical. Although the authors previously developed Technol PG-based nanocarriers,13–17 the assembly of this mixture of compounds into liposomes has not yet been demonstrated. The present study indicates that Technol PG liposomes can be prepared using a gentle hydration method that takes advantage of the solubility of this mixture of compounds in methanol and ethanol. Subsequent to this, a highly dispersible complex can be prepared by mixing TA with the Technol PG liposomes.

Molecular structures of tannic acid and phosphatidyl glycerol and the composition of Technol PG.
Results
In preparation for the formation of Technol PG liposomes using a common hydration process, the dispersibility of Technol PG in various organic solvents was assessed. In each trial, 20 mg of Technol PG was dispersed in 1 mL of acetone, chloroform, ethanol, methanol, or water. The dispersions in acetone (Figure 2a), chloroform (Figure 2b), and water (Figure 2e) were found to be cloudy. In contrast, ethanol (Figure 2c) and methanol (Figure 2d) gave transparent dispersions, indicating that Technol PG was highly soluble in these solvents. On the basis of these results, Technol PG liposomes were prepared using a gentle hydration technique in conjunction with ethanol. Technol PG films were prepared by first evaporating ethanol dispersions of Technol PG (Figure 2f), after which water was added to generate a cloudy dispersion (Figure 2g). Phase-contrast microscopy of these dispersions confirmed that Technol PG liposomes had been formed (Figure 2h).

Photographic images of dispersions of Technol PG (2 wt%) in (A) acetone, (B) chloroform, (C) ethanol, (D) methanol, and (E) water. Photographic images of (F) a Technol PG film and (G) a dispersion of Technol PG liposomes in water. (H) Phase-contrast microscopy image showing the dispersion of Technol PG liposomes. The scale bar is 5 μm.
The hybridization of these Technol PG liposomes was subsequently attempted. Adding a 1 wt% aqueous solution of TA (Figure 3c) to the Technol PG liposomes (Figure 3a) gave a transparent dispersion of the resulting complex (Figure 3b). The absorption spectrum of the complex formed between the TA and Technol PG liposomes (Figure 3d, red) showed a change in the intensity (Figure 3d, blue), suggesting an interaction between the liposomes and the TA. 18 In addition, absorption signal around 600 nm in the liposome dispersion, derived from scattering (Figure 3d, green), was less intense following complexation with the TA. This result confirmed that the dispersibility of the Technol PG liposomes was improved. Phase-contrast microscopy of the TA/liposome mixture confirmed good dispersion of the liposomes (Figure 4a). In addition, analyses by dynamic light scattering indicated that the average particle size was decreased following complexation with TA (Figure 4b), where the polydispersity index increased from 0.22 to 0.66. Zeta potential of the Technol PG liposome samples without and with TA were −54.5 ± 1.6 mV and −49.8 ± 3.6 mV, respectively. Moreover, the size of Technol PG (478 nm in diameter) liposome hardly changed even after incubation for overnight (519 nm in diameter) confirming the long-time stability, while it increased in the presence of NaCl (100 mM) (Figure 5, 837 nm in diameter) indicating that aggregation behavior can be controlled by reducing repulsive force between liposomes.
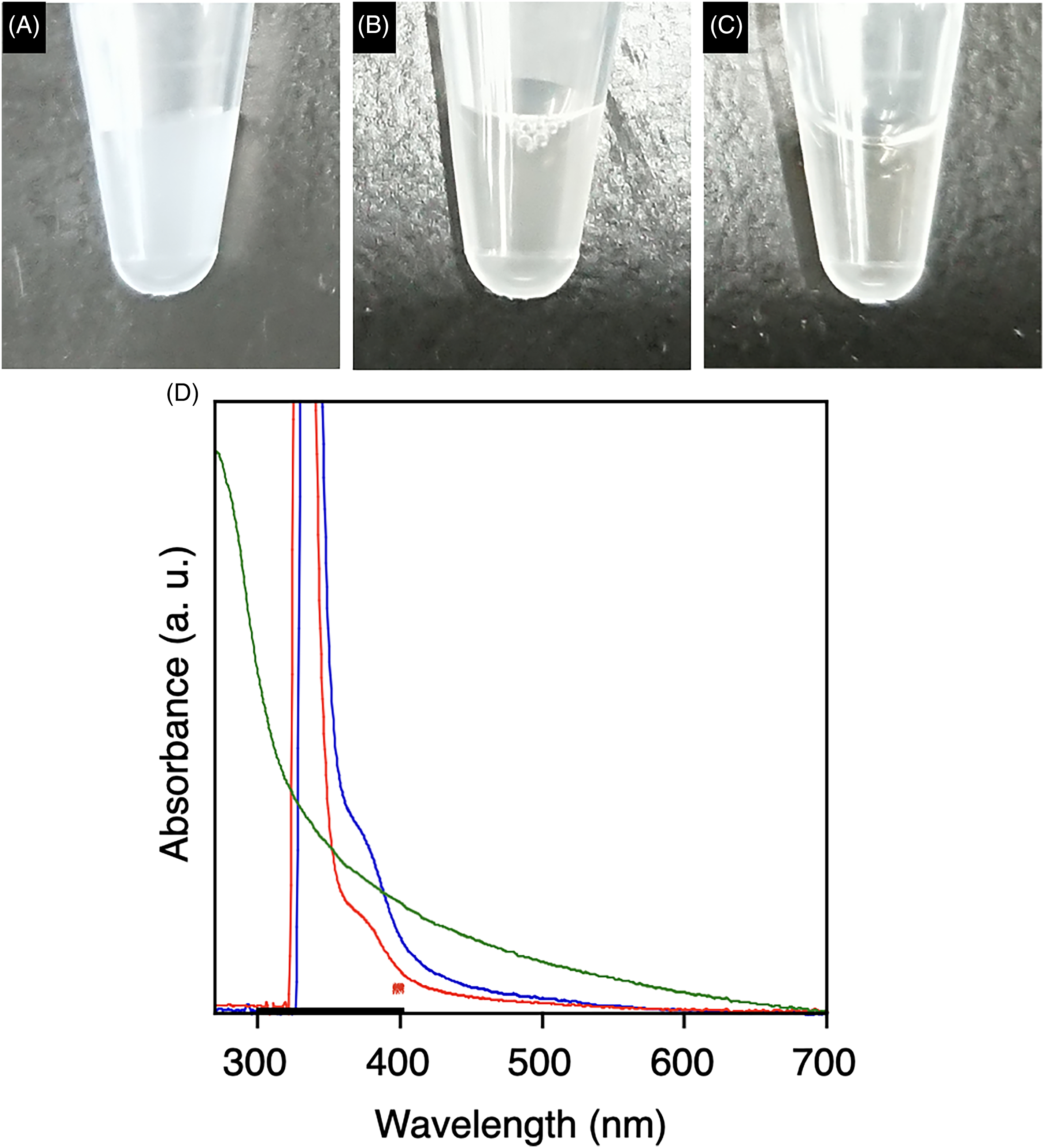
Photographic images of a dispersion of Technol PG liposomes (A) before and (B) after the addition of tannic acid and of (C) a solution of tannic acid. (D) Absorption spectra of Technol PG liposomes before (green) and after (blue) the addition of tannic acid, and of a solution of tannic acid (red).

(A) A phase-contrast microscopy image of an aqueous solution of Technol PG liposome (2 wt%) following hybridization with TA (1 wt%). The scale bar is 5 μm. (B) DLS data for the dispersion of Technol PG liposome before (red) and after (blue) the hybridization with TA.

DLS profiles of Technol PG liposome in the presence of NaCl (100 mM).
Discussion
In this study, we have demonstrated the preparation of Technol PG liposome and their complexation with TA. Technol PG showed high solubility in methanol and ethanol, and Technol PG liposome was prepared by gentle hydration method for the film after the evaporation of the solvent. Since ethanol is a highly biocompatible dispersant, Technol PG liposome has potential for applications in cosmetics, etc. By mixing with TA, the dispersibility of Technol PG liposome was improved. The change in absorption spectrum suggested a possible interaction between the phenolic groups in TA and phosphate anion of the phospholipid, suggesting that this method could be widely applied to phospholipid-based materials. Together with the fact that TA has unique properties such as astringent effect, this study would provide a guideline for the design of new class of phospholipid-based biomaterials.
Experimental
General: Technol PG was obtained from Yokozeki and tannic acid was purchased from Kanto Chemical. Acetone, chloroform, methanol, and ethanol were purchased from Wako.
Instruments: Microscopic observation was performed with an Olympus IX-73 microscope. Zeta potential and DLS measurements were performed by using a Malvern model Zetasizer Nano ZSP zeta potential analyzer. UV/Vis absorption spectra were recorded on a Thermo scientific model NanoDrop One spectrophotometer.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grant-in-Aid for Scientific Research (C) 23K04927 (NU), National Research and Development Agency, Intensive Support for Young Promising Researchers 23W1M041 (NU), Japan Association for Chemical Innovation (NU), Moritani Foundation (NU), Tanaka Foundation (NU), Kose Cosmetology Foundation (NU), Asahi Glass Foundation (NU), Lotte Shigemitsu Prize (NU), Izumi Foundation (NU), Sumitomo Foundation (NU), and Tateishi Foundation (NU).
