Abstract
Mixing surfactants with phospholipid membrane is a useful approach to control the assembling structures for various applications such as nuclear magnetic resonance (NMR) alignment medium. This work aims to investigate the effects of surfactants on the assembly behavior of Technol PG, a mixture of anionic phosphatidyl glycerols. 31P, 2H, and 1H NMR spectroscopies were utilized for the evaluations. Among the surfactants, cholic acid, an anionic surfactant, was effective for decompose of liposomal assembly of Technol PG.
The assembly structure of phospholipids can be controlled by complexation with surfactants.1–8 For example, liposome, which is a hollow phospholipid aggregates, undergo a structural transition to form micellar structures 1 or disc-shaped bicelle4–8 in the presence of surfactants. Self-assemblies of anionic phospholipids such as phosphatidyl glycerol have also attracted attention because they may show physical properties differently from phosphatidyl choline due to electrostatic repulsion. Recently, we have developed nanomaterials using Technol PG (Figure 1).9–15 Technol PG, a mixture of anionic phosphatidyl glycerols, is commercially available and less expensive than conventional anionic phosphatidyl glycerols.

Molecular Structure of Phosphatidyl Glycerol and Components of Technol PG, Triton-X, 3-[(3-Cholamidopropyl)Dimethylammonio]-2-Hydroxy-1-Propanesulfonate (CHAPSO), Sodium Cholate (SC), and Cationic Hexadecyltrimethylammonium Bromide (CTAB).
First, we evaluated how the assembling behavior of Technol PG is affected by the freeze-thaw process 16 to optimize the preparation method for wide applications of Technol PG. 31P nuclear magnetic resonance (NMR) spectroscopy5,17–20 was adopted for this purpose, since it is commonly used for evaluating phospholipid assembly structures. Technol PG assemblies were prepared from powder samples, and then the effects of the freeze-thaw process on the assembly state of Technol PG were evaluated. When Technol PG powder (9 wt%) was dispersed in water, two NMR peaks were observed around 0 ppm and −10 ppm (Figure 2a, bottom), which are assignable to mixture of the non-orientable edge part and the magnetically aligned phospholipid membrane due to the formation of liposomes. 21 After one (Figure 2a, middle) and three (Figure 2a, upper) freeze-thaw cycles, the intensity of the peak at −10 ppm decreased as the number of the cycles increased. Since the peak at −10 ppm was attributed to phospholipid membranes that were oriented under the magnetic field, the decrease of the peak indicated that the size of the phospholipid assemblies changed due to the freeze-thaw cycles. When the Technol PG dispersion was diluted from 9 to 1 wt% and 6 freeze-thaw cycles were performed in the same way, no significant difference was observed in 31P NMR spectra (Figure 2b), suggesting that the assembly behavior did not change throughout the freeze-thaw process at the lower concentration.
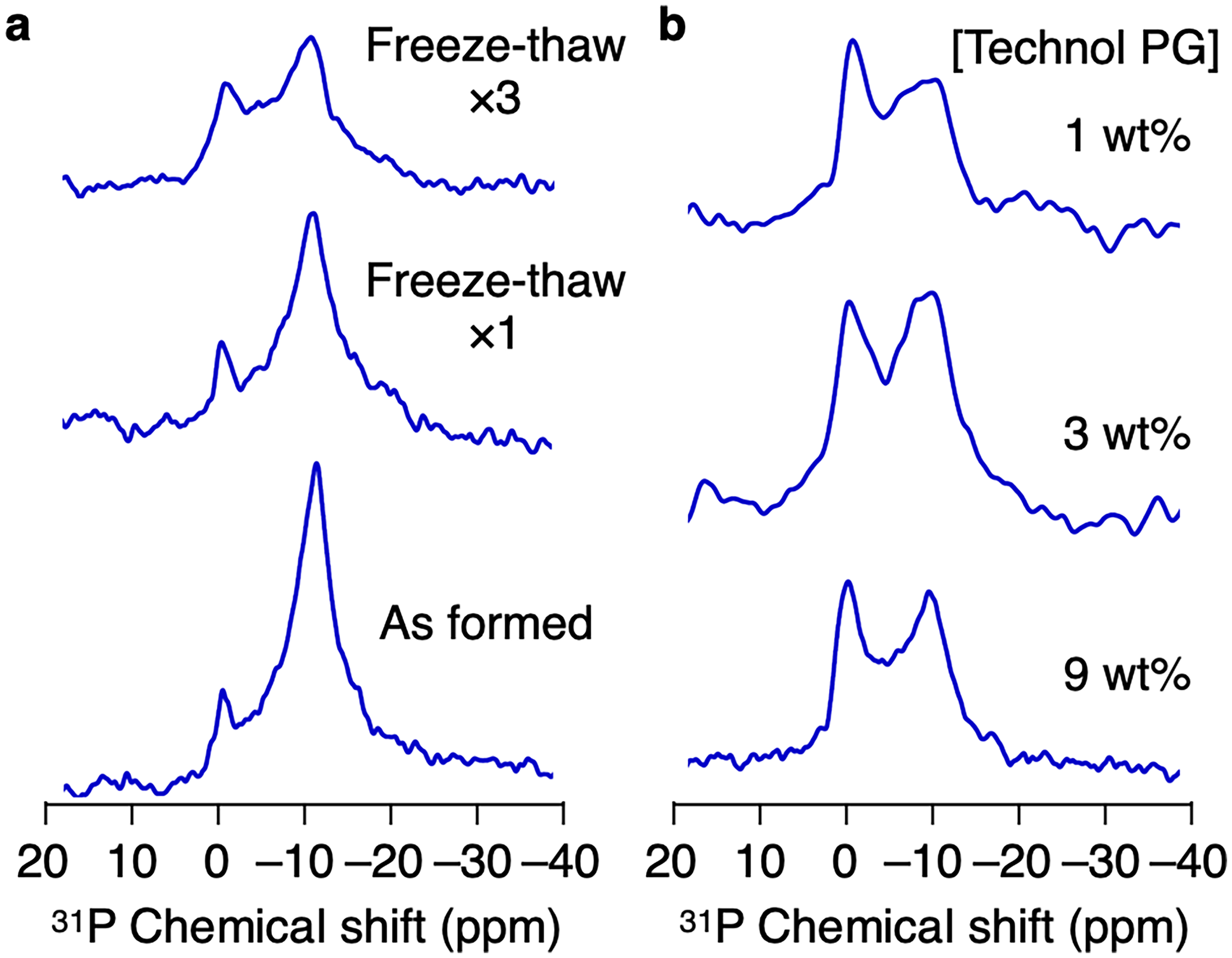
(a) 31P NMR Spectra of Aqueous Dispersion of Technol PG (9 wt%) at 25 °C After 0 (lower), 1 (middle), and 3 (upper) Freeze-Thaw Cycles. (b) 31P NMR Spectra of Aqueous Dispersion of Technol PG at 1 wt% (upper), 3 wt% (middle), and 9 wt% (lower) after 6 Freeze-Thaw Cycles.
Next, we evaluated the effects of various surfactants on the assemblies of Technol PG without the freeze-thaw process. In this study, we selected non-ionic Triton-X, zwitterionic 3-[(3-cholamidopropyl)dimethylammonio]-2-hydroxy-1-propanesulfonate (CHAPSO), anionic sodium cholate (SC), and cationic hexadecyltrimethylammonium bromide (CTAB) as model surfactants (Figure 1). When each surfactant was mixed with a Technol PG dispersion (9 wt%) at a concentration of 0.2 wt%, 31P NMR spectra exhibited peaks around 0 and −10 ppm, as in the case without surfactant, suggesting that the liposome structure was maintained (Figure 3a,c,e,g). However, when the concentration of the surfactants was increased to 1 wt%, significant differences were observed. Namely, the liposome structure was maintained upon addition of Triton-X (Figure 3b) and CTAB (Figure 3h), while addition of CHAPSO (Figure 3d) and SC (Figure 3f) caused a decrease in the peak at −10 ppm and the peak at 0 ppm became the main peak. The peaks at 0 ppm are possibly due to the micellar assemblies that did not orient in the magnetic field, 22 suggesting that the assembly structure of Technol PG was greatly affected by CHAPSO and SC. In order to investigate the changes in the assembly structures of Technol PG in detail, we performed 2H NMR measurements on the dispersions. When magnetically alignable phospholipid assemblies such as bicelles exist in solution, 2H NMR spectroscopy shows quadrupolar splitting due to the orientation of deuterium oxide (D2O) molecules. 18 Quadrupolar splitting was not observed under any conditions in the presence of 1 wt% surfactants (Figure 4b,d,f,h). However, noticeable quadrupolar splitting of 2.9 Hz was observed when SC was added at 0.2 wt% (Figure 4e), while other mixtures did not show any quadrupolar splitting (Figure 4a,c,g). These results indicated that SC caused a change in the assembled structures, making it easier for the molecules in the solution to be oriented. The reason why SC caused the significant change may be because electrostatic repulsion of SC facilitated the dispersion of the phospholipid assemblies. In addition, we demonstrated the alignment media of for measurement of 1H NMR of uridine (4 wt%). As a result, the peak evenly splitting around 5.8 ppm (Figure 5, red) became unevenly split in the presence of Technol PG (9 wt%) and CA (0.2 wt%) (Figure 5, blue), which may be due to the orientation of uridine molecules. Capitalizing on this phenomenon, the mixture of Technol PG and SC could be utilized as an alignment medium to observe angle-dependent NMR signals such as residual dipole coupling of molecules dispersed in solution. 7
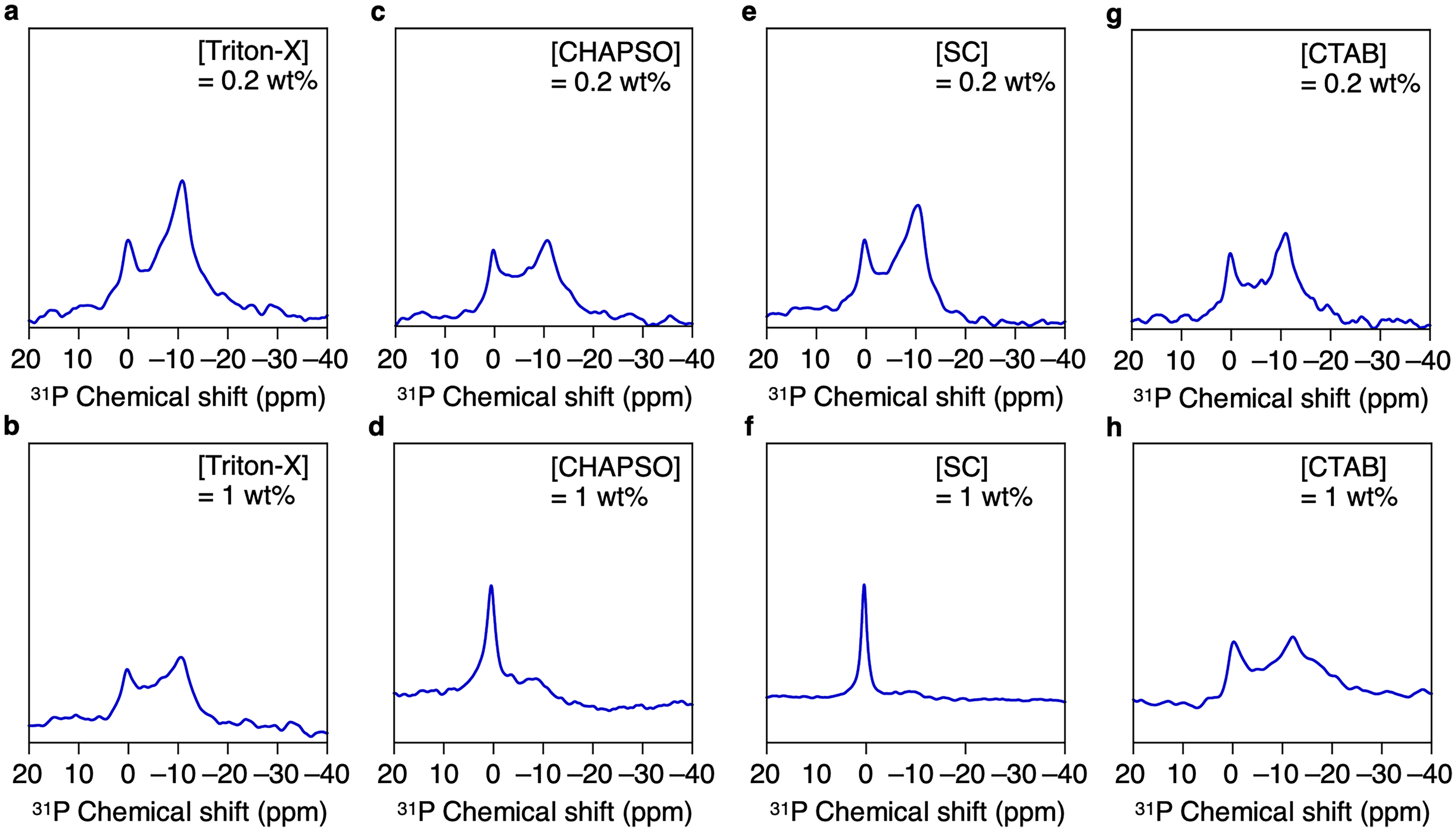
31P NMR spectra of Aqueous Dispersion of Technol PG (9 wt%) with 0.2 wt% of Triton-X (a), CHAPSO (c), SC (e), and CTAB (g), or with 1 wt% of Triton-X (b), CHAPSO (d), SC (f), and CTAB (h) at 25 °C.
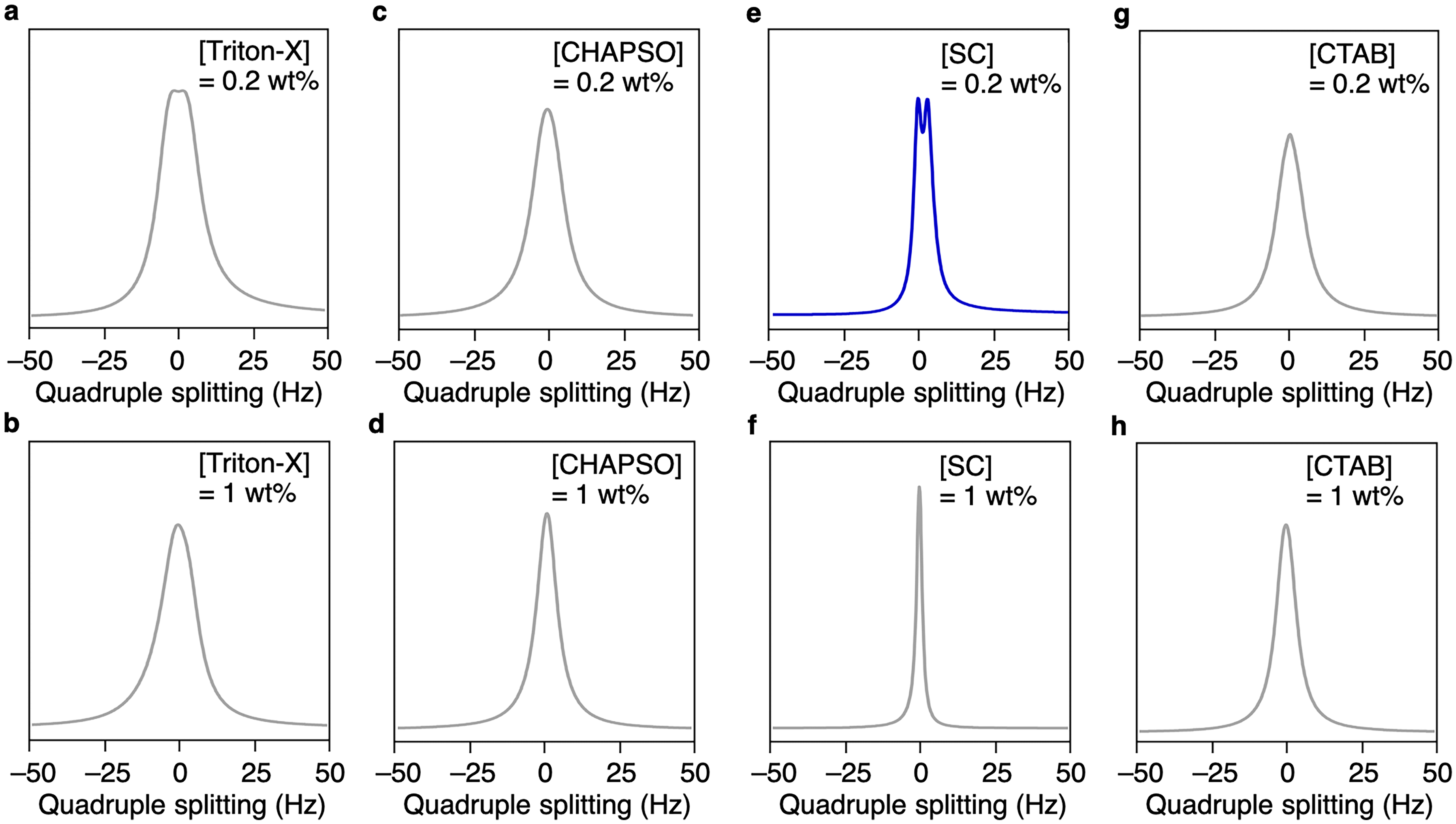
2H NMR Spectra of Deuterium Oxide (D2O; > 90%) Solutions of Technol PG (9 wt%) with 0.2 wt% of Triton-X (a), CHAPSO (c), SC (e), and CTAB (g), or with 1 wt% of Triton-X (b), CHAPSO (d), SC (f), and CTAB (h) at 25 °C.
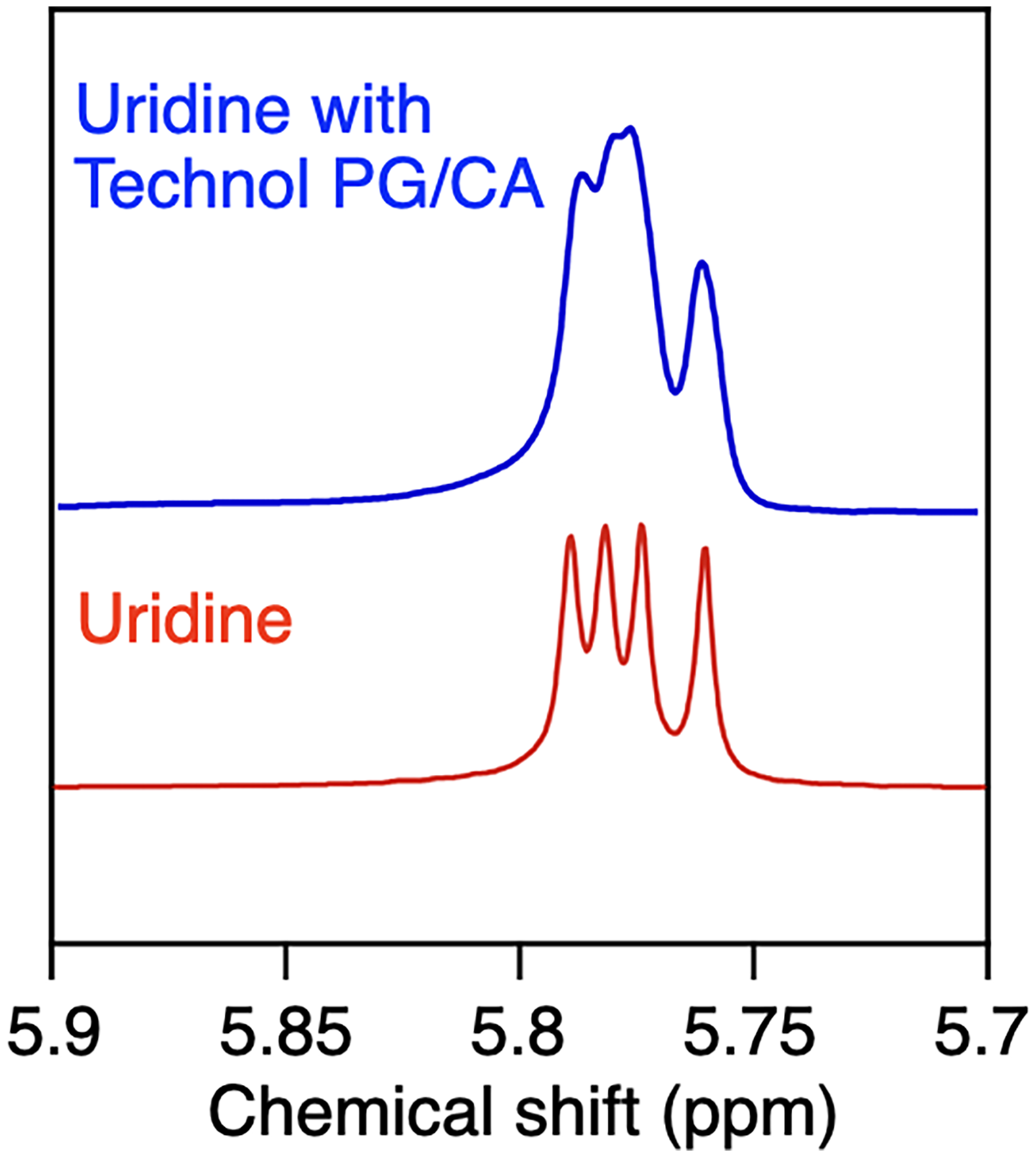
1H NMR Spectra of Uridine (4 wt%) without (Red) and with (Blue) Technol PG (9 wt%) and CA (0.2 wt%) in D2O at 25 °C.
Conclusions
In this study, we evaluated the effects of surfactants on the assembly structure of Technol PG using NMR spectroscopy. The surfactant SC had a significant effect on the assembly structure of Technol PG, thus it has potential to be used as an alignment medium for NMR measurements. To date, Pf1 bacteriophage 23 has been utilized for NMR measurements, but one issue has been its high cost. The alignment medium composed of Technol PG, which is available at a low price, and SC has the potential to become a practical alignment medium for future NMR studies.
Experimental
General
Technol PG was provided by Yokozeki. Triton-X (100% purity) was purchased from Nacalai Tesque. CHAPSO (>96% purity), D2O (99.8% purity), and uridine were purchased from FUJIFILM Wako. SC (>97% purity) and CTAB (>98% purity) were purchased from TCI.
Typical Preparation of Technol PG-Based Assemblies
For the preparation of Technol PG liposomes, Technol PG (9 wt%) was dispersed in 1 mL of water and then sonicated for 30 s using AS ONE model ASU-3 (80 W). To evaluate the effect of surfactants, 0.2 wt% or 1 wt% of Triton-X, CHAPSO, SC, or CTAB was added to the Technol PG solution which was then mixed by pipetting.
NMR Analysis
31P, 2H, and 1H NMR spectra were recorded on Bruker AVANCE-600 spectrometers. Size of fid (TD), number of dummy scans (DS), number of scans (NS), loop count for ‘td0’ (TD0), acquisition time (AQ) in the 31P NMR measurements are 65536, 4, 1024, 1, and 0.3408, respectively. TD, DS, NS, TD0, and AQ in the 2H NMR measurements are 65536, 2, 16, 1, and 2.7263, respectively. TD, DS, NS, TD0, and AQ in the 1H NMR measurements are 65536, 2, 1024, 1, and 2.7263, respectively. The chemical shifts were adjusted depending on reference peaks. All samples were measured in D2O solutions.
Footnotes
Acknowledgements
This work was supported by the following funding sources: Grant-in-Aid for Scientific Research (C) JP23K04927 (to NU); New Energy and Industrial Technology Development Organization, Intensive Support for Young Promising Researchers 23W1M041 (to NU); the Japan Association for Chemical Innovation (to NU); the Urakami Foundation (to NU); Toyota Riken Foundation (to NU); and the Lotte Foundation (to NU). JP23K04927 and 23W1M041 were used to purchase surfactants such as SC, CTAB, and CHAPSO in this work, and used to purchase tannic acid in previous our work (Nat Prod Commun 2024;19. doi: 10.1177/1934578X241253719).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science, (grant number Grant-in-Aid for Scientific Research (C) JP23K0492).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
