Abstract
Titanium dioxide (TiO2) is widely used in cosmetics and self-cleaning materials because of its high refractive index and photocatalytic activity. In the present work, the surfaces of TiO2 nanoparticles were coated with sodium cholate and the commercially available anionic phospholipid mixture Technol PG. These surface-coated TiO2 nanoparticles could be homogeneously dispersed in water and subsequently produced films on glass substrates after drop casting.
Titanium dioxide (TiO2; Figure 1a) is widely used in pigments, paints, ointments sunscreens, and toothpaste. TiO2-based materials also have applications in photovoltaics and photocatalysis based on the discovery of photocatalytic water splitting in 1972 via the so-called Honda-Fujishima effect.1,2 As such, much effort has been devoted to the study of these materials. This work has led to many promising uses in areas ranging from photocatalysis and photovoltaics to photo-/electrochromics and sensors. Surface modification of TiO2, especially using biocompatible molecules, is an especially important aspect of designing TiO2-based materials.3,4 Coating of TiO2 surfaces with neutral phospholipids such as 1,2-dipalmitoyl-sn-glycero-3-phosphocholine has been reported, 5 although the use of commercially available phospholipids would be preferable with regard to establishing practical applications.

(a) Structure of titanium dioxide (TiO2). (b) Molecular structure of PG and composition of Technol PG. (c) Molecular structure of sodium cholate (SC).
The present study examined the feasibility of using Technol PG for the surface coating of TiO2, employing model TiO2 nanoparticles with diameters of less than 100 nm. Technol PG (obtainable from YOKOZEKI) is less expensive than conventional anionic phosphatidyl glycerol compounds. Previously, our group determined that mixtures of Technol PG (Figure 1b) and sodium cholate (SC; Figure 1c) can effectively disperse bioactive compounds such as resveratrol 6 and fullerene. 7 These prior studies also established that TiO2 nanoparticles can be dispersed by a mixture of Technol PG and SC. In a typical procedure for surface coating of TiO2 nanoparticles (Figure 2a), quantities of Technol PG powder (5.0 wt%), SC (2.0 wt%), and TiO2 nanoparticles (0.1 wt%) were dispersed in water with ultrasonication for 2 min. Without the Technol PG and SC, the nanoparticles were observed to precipitate (Figure 2b). In contrast, mixing with Technol PG and SC allowed the TiO2 nanoparticles to remain dispersed in the solution (Figure 2c). In addition, TiO2 nanoparticles were dispersed, even with a diluted mixture of Technol PG (0.5 wt%) and SC (0.2 wt%) (Figure 2d). The sizes of the nanoparticles were assessed using dynamic light scattering (DLS). Although the size of bare TiO2 nanoparticles generated a peak at approximately 460 nm (Figure 2e), the average size of these nanoparticles was reduced to 120 nm after mixing with Technol PG and SC (Figure 2f). We also confirmed by phase-contrast microscopy that TiO2 nanoparticles were well dispersed in solution after mixing with Technol PG and SC (Figure 2g and h). This result indicated that the aggregation of TiO2 nanoparticles was suppressed by the surface coating. Surface modification of TiO2 nanoparticles is required to produce TiO2-based self-cleaning films. In the case that uncoated TiO2 nanoparticles were drop-cast onto a glass substrate, a somewhat transparent TiO2 film was obtained (Figure 3a and b), but the TiO2 nanoparticles were easily dislodged simply by manual rubbing (Figure 4a). Conversely, a similar film fabricated from surface-coated TiO2 nanoparticles exhibited greater transparency (Figure 3c and d). In addition, the TiO2 remained on the substrate when the surface was rubbed (Figure 4b). These results suggest that surface modification with Technol PG and SC could be used to prepare robust TiO2 nanoparticle films on substrates.
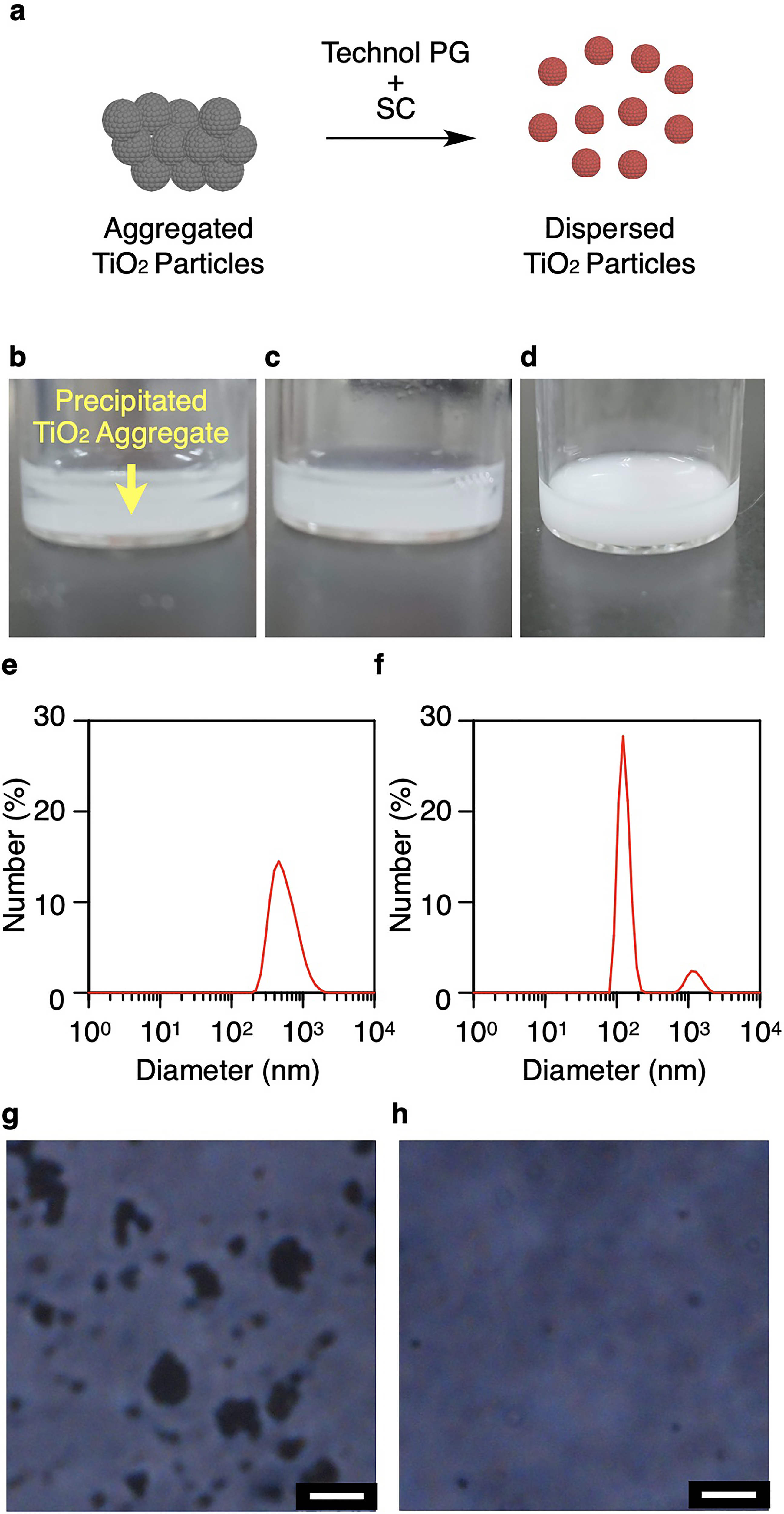
(a) Diagram showing dispersion of titanium dioxide (TiO2) nanoparticles using Technol PG and sodium cholate (SC). Photographic images of dispersions of TiO2 nanoparticles (b) without and (c) with Technol PG (5.0 wt%) and SC (2.0 wt%) or (d) with Technol PG (0.5 wt%) and SC (0.2 wt%). DLS profiles of dispersions of TiO2 nanoparticles (e) without and (f) with Technol PG (5.0 wt%) and SC (2.0 wt%). Phase-contrast microscopy of TiO2 nanoparticles (g) without and (h) with Technol PG (5.0 wt%) and SC (2.0 wt%).

Diagrams showing preparation of titanium dioxide (TiO2) nanoparticle films (a) without and (c) with surface coating with Technol PG and sodium cholate (SC). Corresponding photographic images of glass substrates covered with (b) uncoated and (d) coated TiO2 nanoparticles.

Photographic images of glass substrates covered with titanium dioxide (TiO2) nanoparticles (a) without and (b) with modification with Technol PG and sodium cholate (SC) after rubbing. The rubbed areas are indicated by the yellow dashed lines.
In summary, this work demonstrated the coating of TiO2 nanoparticle surfaces with Technol PG and SC. The coated nanoparticles were readily dispersed and formed films on glass substrates. Because TiO2 has practical applications with regard to self-cleaning surfaces, this technique for coating TiO2 nanoparticles could lead to the design of a variety of self-cleaning surfaces8–13 in the future.
Experimental
General
Technol PG was obtained from YOKOZEKI, SC from TCI, TiO2 nanoparticles (rutile-type, <100 nm in diameter) from Sigma-Aldrich, and the glass plate from MATSUNAMI.
Surface Coating of TiO2 Nanoparticles
Surface coating of TiO2 nanoparticles was performed by mixing Technol PG (5.0 wt%) and SC (2.0 wt%) with TiO2 (0.1 wt%) in water with ultrasonication for 2 min. For DLS analysis of the particles, a Malvern model Zetasizer Nano ZSP was used.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
