Abstract
TaohongSiwu decoction (THSWD), a traditional Chinese recipe, has been widely used to treat hypertension since ancient times. However, the mechanisms of its action are still unclear. Herein, we aimed to explore the gut microbial activity of THSWD in high-salt diet-induced hypertensive mice. Eight percent high-salt (NaCl) diet was used to induce hypertension for 4 weeks in a mouse model. Meanwhile, THSWD was used to intervene in the high-salt diet-induced mice, and the efficacy was evaluated by different parameters. Here, we found that THSWD significantly restored blood pressure compared with the model group. Moreover, THSWD effectively protected endothelial function by significantly upregulating the level of nitric oxide (NO) and downregulating the level of endothelin-1 (ET-1), angiotensin I (AngI), and vascular endothelial growth factor (VEGF) in serum compared with the model group. Notably, THSWD significantly upregulated the relative abundance of Dubosiella and downregulated that of Cyanobium_PCC-6307 and DNF00809 at the genus level compared with the model group. The results of PCA and microbial distance calculation further exhibited that THSWD treatment resulted in significant regulation of the microbial community. Furthermore, compared with the model group, THSWD increased the level of vitamin k2 (VK2) in serum. These findings indicate that THSWD could protect blood pressure and endothelial function by regulating gut microbiota and promoting microbial metabolite VK2. These results show the important role of THSWD in regulating the gut microbiota in response to high-salt diet-induced mice.
Introduction
Hypertension is characterized by elevated systemic circulatory arterial pressure, which is caused by the combination of genetic and environmental factors. 1 Hypertension is a risk factor leading to stroke, coronary heart disease, and other related diseases in modern society. 2 Because of the high incidence rate and serious associated complications of hypertension, there is always a great demand for drugs for treating this condition. 3
TaohongSiwu decoction (THSWD) has been widely utilized in the treatment of hypertension since ancient times, with the functions of improving blood circulation and reducing blood stasis. 4 Furthermore, our previous pharmacological study has revealed that quercetin, kaempferol, myricanone, luteolin, β-sitosterol, and baicalein in THSWD are related to hypertension and are significantly associated with many signaling pathways. 5 However, although the chemical compounds related to hypertension of THSWD have been studied previously, the mechanisms of THSWD in the alleviation of hypertension have not been investigated.
Studies in recent years have revealed substantial evidence for a link between gut microbiota and hypertension.6,7 As a whole, gut microbiota can interact with other tissues and organs directly or indirectly. In most cases, the effect is linked with microbial metabolites that regulate the tissues and organs in the body functions.8,9 Gut microbiota can help with digestion and absorption, regulate mucosal barrier development and function, govern food intake and metabolism, promote immunological tissue maturation, alter human body tolerance to intestinal content antigens, and prevent pathogenic germs from spreading.6,10,11 Even, the gut microbiota is considered to metabolize and transform the Chinese herbs through a specified mechanism and then be absorbed by the human body to achieve the therapeutic effect, which reveals the close relationship between the gut microbiota and the efficacy of Chinese herbs.12,13 Moreover, the mechanisms of many herbs have been investigated based on their link and effect on the gut microbiota.14–16 Therefore, this study aimed to explore the gut microbial mechanisms of THSWD on hypertension to provide an evidential basis for further research.
Methods
Study Design and Grouping
The ethics committee at Jinan University approved the animal experiment (Nos: IACUC-20190726-04), which was carried out in the university's animal facility. The experimental animal center at Guangzhou University of Chinese medicine provided a total of 24 male pathogen-free (SPF) grade C57BL/6J mice (10-12 weeks). The mice were randomly divided into 3 groups after 10 days of adaptive feeding: control, model, and THSWD, each with 8 mice.
Duplicate Mice Model
In this work, a mouse model was given an 8% high-salt (NaCl) diet for 4 weeks to develop hypertension in the model and THSWD groups. 17 The nutritional formula for the high-salt diet is shown in Supplemental Table 1. Nantong Troffer Feed Technology Co., Ltd provided the high-salt feed (production license: [2014] 06092), which was then sterilized by Nantong Michael Irradiation Co., Ltd. A normal diet was served to the control group.
Preparation of THSWD
In order to facilitate repeated quality control, all granules of the Chinese herbs in THSWD included Prunus davidiana (CarriŠre) Franch. (family Rosaceae), Angelica sinensis (Oliv.) Diels (family Apiaceae), Carthamus tinctorius L. (family Asteraceae), Ligusticum striatum DC. (family Apiaceae), Rehmannia glutinosa (Gaertn.) DC. (family Orobanchaceae), and Paeonia delavayi Franch. (family Paeoniaceae), which were purchased from the first affiliated hospital of Jinan University. The production batch numbers of granules of the herbs were 1902003W, 1905001S, 1905005C, 1905001S, 1906001S, and 1905003S, respectively, which were produced by China Resources Sanjiu Pharmaceutical Co., Ltd. The equivalent dose was converted depending on the body surface area of humans and mice. The granules of all the herbs (3:3:2:2:4:3) were mixed and diluted with water to prepare the THSWD solution and then stored in a 4 °C refrigerator. According to the equivalent dose obtained from the conversion relationship between human and mouse body surface area and combined with the results of different dose screening in the previous study, we chose a 10-time equivalent dose of THSWD as 66.3 g/(kg·d). Finally, each mouse in the THSWD group was intragastrically administered THSWD once a day, and the other 2 groups received the same volume of normal saline.18,19 The details of THSWD are listed in Supplemental Table 2.
UPLC–Q/TOF-MS Analysis of THSWD
One milliliter THSWD granules solution was centrifuged at 4 °C for 25 min at 13000 rpm, and 100 μL of the supernatant was placed in the injection bottle for testing. The target compounds were separated by UPLC–Q/TOF-MS. Phase A was an aqueous phase containing 25 mmol/L ammonium acetate and 25 mmol/L ammonia, and phase B was acetonitrile. With a flow rate of 0.5 mL/min, the gradient elution system was set up as follows: 0 to 0.5 min, 95% B; 0.5 to 7 min, 95% to 65% B; 7 to 8 min, 65% to 40% B; 8 to 9 min, 40% B; 9 to 9.1 min, 40% to 95% B; 9.1 to 12 min, 95% B. The MS/MS spectra on an information-dependent basis (IDA) during an LC/MS experiment were acquired through the TripleTOF 6600 mass spectrometer (AB Sciex). Then, the full scan survey MS data were continuously evaluated using the acquisition software (Analyst TF 1.7, AB Sciex) according to the acquisition of MS/MS spectra depending on preselected criteria in this mode. MS raw data (.wiff) files were converted to the mzXML format by ProteoWizard, and processed by R package XCMS (version 3.2). The process included peak deconvolution, alignment, and integration. Minfrac and cutoff were set as 0.5 and 0.3, respectively. Finally, the compounds were identified using an in-house MS2 database.
According to the top 10 base peak intensities in our study, the results of previous network pharmacology research of THSWD, 5 the convenience of obtaining standard products in our laboratory, and the experience of related experimental operations, amygdalin, 3,4-dihydroxyphenylacetic acid, kaempferol, and adenine were selected for quantitative analysis by UPLC–Q/TOF-MS. Amygdalin, 3,4-dihydroxyphenylacetic acid, and kaempferol stock solutions (5 mg/mL) were prepared with methanol, and adenine (5 mg/mL) with a methanol: 20% formic acid solution. Ten microliter of the test solutions were injected, and the resulting chromatograms were recorded.
The previous supernatant of the THSWD granules was tested by UPLC–Q/TOF-MS analysis. Phase A was a water phase with 20 mmol/L sodium dihydrogen phosphate solution. The pH was adjusted to 2.5 with phosphoric acid. Phase B was pure methanol. A gradient elution system was used at a flow rate of 1 mL/min: 0 to 6 min, 98% A; 6 to 20 min, 98% to 30% A; 20 to 30 min, 30% A; 30 to 30.1 min, 30% to 98% A; 30.1 to 35 min, 98% A. Finally, the chromatogram collection and integration of the compounds were processed by Chemstation software.
Measurement of Blood Pressure in Mice by Tail Artery Manometry
The mouse platform’s temperature was pre-set at 37 °C. According to the operation instructions, it was conducted in 4 parts: test, temperature setting, measurement, and recording. Each mouse was tested 15 times in each round, and the average was recorded as the systolic blood pressure (SBP) of each mouse. The measurement time was set at 3 pm every week, and each step of the operation was completed using the blood pressure analysis program (BP2000, USA) by the same operator.
Detection of Vascular Endothelial Function-Related Factors in Serum by ELISA
To reduce pain, the procedures on the mice were conducted under sodium pentobarbital anesthesia. After that, blood was drawn from the eyeball and placed in an aseptic blood collection vessel. It was centrifuged for 5 min at 3000/min after 2 to 4 h. Then, the supernatant was collected in a sterilized 1.5-mL EP tube for detection of NO, ET-1, AngI, and VEGF using kits provided by MEIMIAN and following the operation instructions.
Evaluation of VK2 Level in Serum
A VK2 ELISA kit provided by MEIMIAN (Yancheng, China) was used to analyze the level of VK2 in serum, according to the manufacturer's instructions.
16S rRNA Gene Sequencing of Intestinal Contents in Mice
DNA extraction and PCR amplification
The E.Z.N.A. soil DNA Kit (Omega Bio-Tek) was used to extract microbial DNA according to the manufacturer's instructions. A NanoDrop 2000 UV-vis spectrophotometer (Thermo Scientific) was used to assess the final DNA concentration and purification, and DNA quality was confirmed using 1% agarose gel electrophoresis. With primers 338F (5′-ACTCCTACGGGAGGCAGCAG-3′) and 806R (5′-GGACTACHVGGGTWTCTAAT-3′), the V3-V4 hypervariable portions of the bacterium 16S rRNA gene were amplified by a thermocycler PCR system (GeneAmp 9700, ABI). The PCR products were extracted from a 2% agarose gel, purified with a AxyPrep DNA Gel Extraction Kit (Axygen Biosciences), and quantified with QuantiFluorTM-ST (Promega) according to the manufacturer's instructions. 19
Illumina miseq sequencing and biological information analysis
The Majorbio Bio-Pharm Technology Co. Ltd pooled purified amplicons in an equimolar ratio and sequenced them (2 × 300) on an Illumina MiSeq platform (Illumina) (Shanghai, China). The operational taxonomic units (OTUs) were clustered with a 97% similarity criterion using UPARSE (version 7.1 http://drive5.com/uparse/) with a unique “greedy” approach that performs chimera filtering and OTU clustering simultaneously after raw fastq files were quality-filtered and merged. Finally, the taxonomy of each 16S rRNA gene sequence was compared to the Silva (SSU123) 16S rRNA database using the RDP Classifier algorithm (http://rdp.cme.msu.edu/) with a confidence threshold of 70%.
Statistical Analysis
All data were expressed as mean ± standard error (SE) and analyzed by GraphPad Prism 7. T tests were used to determine whether there were any significant differences between groups. If the P value was less than .05, the difference was considered significant. The 16S rRNA gene sequencing data were analyzed using the Majorbio I-Sanger Cloud Platform, which is available for free online (www.i-sanger.com).
Results
Identification of the Chemical Composition of THSWD
UPLC–Q/TOF-MS analysis of THSWD was used for quality control of THSWD. The chemical composition of the top 10 ingredients (sucrose, pro-Trp, choline, 3,4-dihydroxyphenylacetic acid, adenosine, ketoprofen, raffinose, adenine, amygdalin, and l-arginine) were identified from THSWD according to base peak intensity (Supplemental Figure 1, Supplemental Table 3). Then, amygdalin, 3,4-dihydroxyphenylacetic acid, kaempferol, and adenine were selected for quantitative analysis by UPLC–Q/TOF-MS analysis using the standard controls. The chemical base peak intensity chromatogram of the chemical ingredients of THSWD granules, as shown in Supplemental Figure 2, gave the composition as adenine 0.12242 µg/mg, 3.4-dihydroxyphenylacetic acid 0.04025 µg/mg, amygdalin 3.34079 µg/mg, and kaempferol 0.01974 µg/mg. The detailed constituents are shown in Supplemental Table 4. These results provided quality control for THSWD for the follow-up experimental research.
Effects of THSWD on SBP of High-Salt Diet-Induced Mice
The SBP of mice was restored after administration of THSWD for 4 weeks (Figure 1). Compared with the control group, the SBP of the model group increased with time after high salt induction (P < .01), while that of the THSWD group did not; the difference was statistically significant when compared with the model group (P < .01). Moreover, the weight of the mice decreased after high salt induction for 1 week, but there was no statistically significant difference after 4 weeks (Supplemental Figure 3).

Effects of THSWD on systolic blood pressure (SBP) of high-salt diet-induced mice (n = 7-8). All data are shown as mean ± standard error (SE). **P < .01 versus control, ##P < .01 versus model.
Effects of THSWD on Vascular Endothelial Function-Related Factors in Serum
As shown in Figure 2, THSWD significantly upregulated the content of NO (P < .01) and downregulated the level of ET-1, AngI, and VEGF in the serum compared with the model group (Figure 3A-D; P < .01), which shows that THSWD can promote the level of vascular endothelial function-related factors and restore endothelial function.

Effects of THSWD on vascular endothelial function-related factors in serum (n = 7-8). (A-D) Level of NO, ET-1, AngI, and VEGF, respectively. All data are expressed as mean ± standard error (SE). ** P < .01.
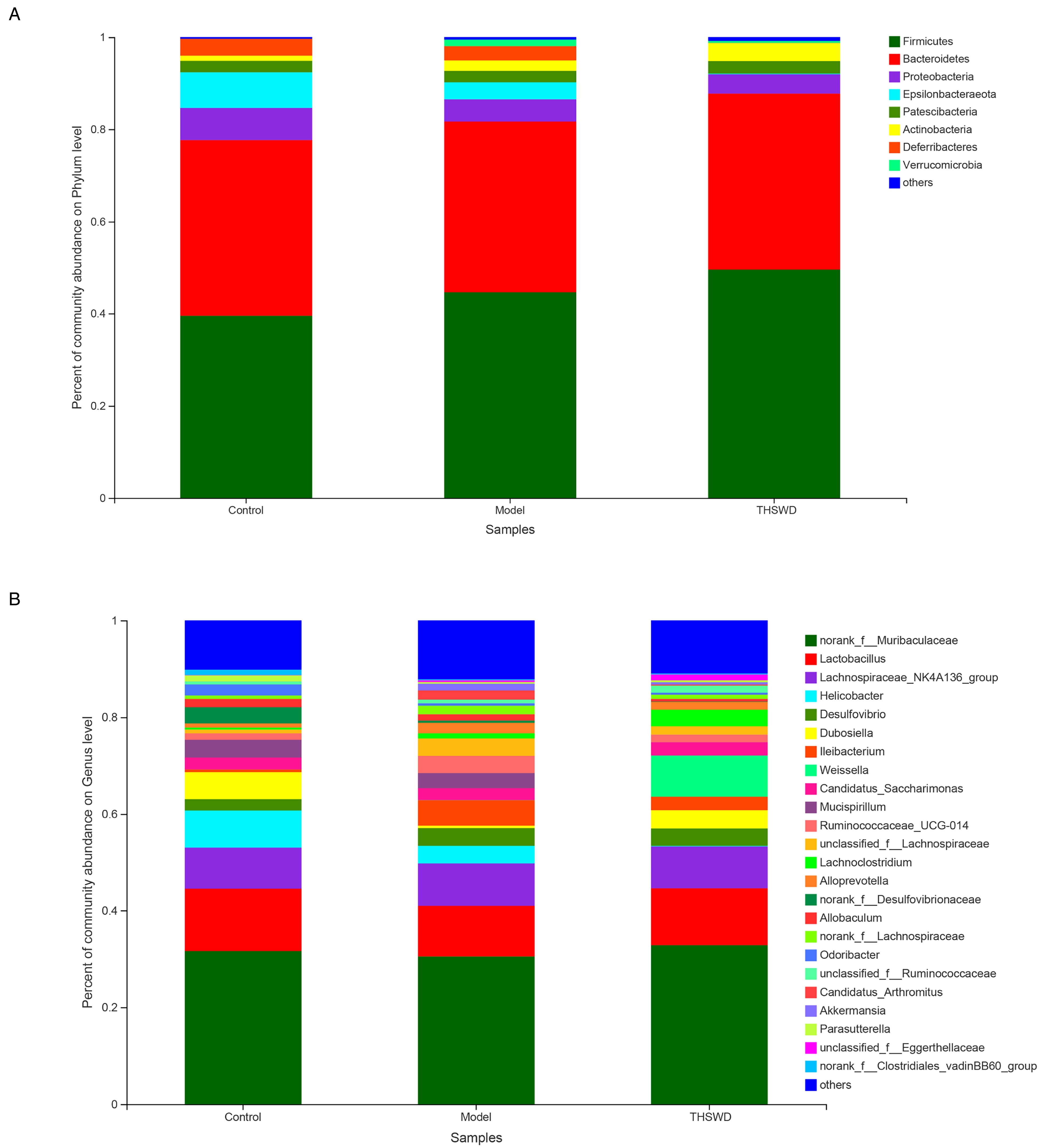
Effect of THSWD on gut microbial community abundance (n = 6). (A) Phylum-level abundance as a percentage of total community abundance. (B) The top 25 in community abundance in terms of genera percent.
Effects of THSWD on the Gut Microbiota in High-Salt Diet-Induced Mice
Effect of THSWD on gut microbial community abundance
The gut microbiota included 978 OTUs, 20 phyla, and 249 genera. The control group contained 713 OTUs, the model group 854, and the THSWD group 750. The phyla and genera were selected to conduct further analysis (Figure 3). As shown in Figure 3A, the top 8 phyla are displayed in the control group, model group, and THSWD group. There were 39.37% Firmicutes in the control group, 45.8% in the model group, and 49.8% in the THSWD group. There were 6.96% Proteobacteria in the control group, 4.93% in the model group, and 3.99% in the THSWD group. In Figure 3B, the top 25 genera are displayed in the control, model, and THSWD groups. For example, there were 31.86% norank_f__Muribaculaceae in the control group, 29.63% in the model group, and 32.72% in the THSWD group, whereas there were 12.79% Lactobacillus in the control group, 12.10% in the model group, and 11.76% in the THSWD group. These results indicate that THSWD can regulate the gut microbial community abundance, even upregulating Firmicutes and norank_f__Muribaculaceae, while downregulating Proteobacteria and Lactobacillus.
Effects of THSWD on the Firmicutes/Bacteroidetes (F/B) ratio and significant genera of the microbial community
To investigate further the gut microbial composition, the effect of THSWD on the Firmicutes/Bacteroidetes (F/B) ratio and significant genera of the microbial community are evaluated, as shown as Figure 4. In Figure 4A, the F/B ratio of the model group was higher than that in the control group, but apparently less than that in the THSWD group, although no statistically significant differences were observed. Moreover, there was a significantly lower relative abundance of Dubosiella in the model group than in the control group (P = .005), while there was a significantly higher relative abundance in the THSWD group than that in the model group (Figure 4B; P = .007). There was a significantly higher relative abundance of Cyanobium_PCC-6307 in the model group than in the THSWD and control groups (Figure 4C; P = .046). Additionally, there was a significantly higher relative abundance of DNF00809 in the model group than in the control group (P = .015) and was also significantly higher in the model group than in the THSWD group (Figure 4D; P = .031). These findings reveal that THSWD can significantly upregulate the relative abundance of Dubosiella and downregulate the relative abundance of Cyanobium_PCC-6307 and DNF00809 in the gut microbiota.

Effects of THSWD on the Firmicutes/Bacteroidetes (f/b) ratio and significant members of the microbial community (n = 6). (A) Effects of THSWD on F/B. (B-D) Effects of THSWD on significant genera. All data are shown as mean ± standard error (SE). NS represented no significance. P < .05 was considered as statistically different.
Beta diversity to reveal the effect of THSWD on the microbial community
Principal component analysis (PCA) was selected to calculate beta diversity based on the OTU level, and Adonis was used to show a significant difference. The microbial population of the control group differed significantly from that of the control, model, and THSWD groups (R2 = 0.2092, P = .004; Figure 5). These results reveal that THSWD can regulate the microbial community. PC1 explained 11.88% of the total variation in the OTU level, and PC2 accounted for 11.53% (Figure 5).

PCA plot to reveal the effect of THSWD on the microbial community. The closer the 2 sample points are, the more similar the composition of the 2 sample species. Different colored points and shapes indicate samples from different groups.
Microbial distance calculated on the OTU level to reveal grouping difference
ANOSIM analysis was performed to explore the significant difference in 3 groups. As shown in Supplementary Figure 4, a significant difference was observed in the microbial structure based on the results of the ANOSIM analysis (R = 0.5267, P = .001). These findings further show that THSWD can regulate the microbial community.

Effects of THSWD on the level of vk2 (n = 7-8). All data are shown as mean ± standard error (SE). **P < .01.
Effects of THSWD VK2 Serum Level
The level of VK2 in serum was analyzed to study the effect of THSWD on microbial metabolites. As shown in Figure 6 a significantly lower expression of VK2 was found in the model group than that in the control group (P < .01), and a significantly higher expression of VK2 was found in the THSWD group than that in the model group (P < .01). These results indicate that THSWD can upregulate VK2 in the serum.
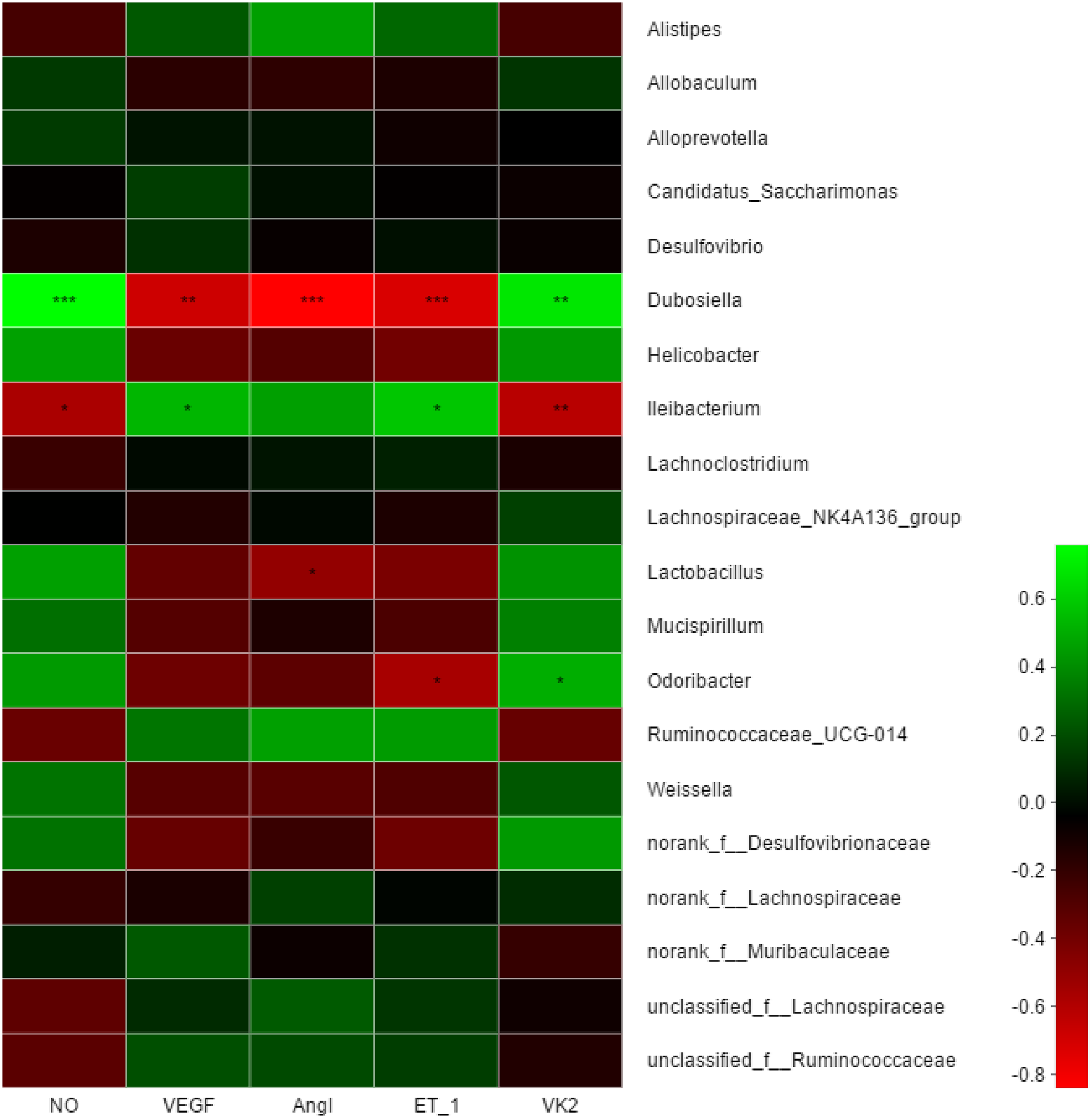
Spearman correlation analysis. *P < .05, **P < .01, ***P < .001.
Correlation Analysis
Spearman correlation analysis was used to assess the correlation between microbial classification, vascular endothelial function-related factors, and VK2 in mice. Twenty genera were used to perform an in-depth analysis. As shown in Figure 7, Dubosiella was positively correlated with VK2 and NO (P < .01) but was negatively correlated with ET-1, AngI, and VEGF (P < .001). Ileibacterium was negatively correlated with NO (P < .05) and VK2 (P < .01) and positively correlated with VEGF and ET-1 (P < .05). Odoribacter was negatively correlated with ET-1 (P < .05) and positively correlated with VK2 (P < .05). In addition, Lactobacillus was negatively related to AngI (P < .05).
Discussion
Accumulating evidence shows that gut microbiota, microbial metabolites, and probiotics can affect immune and endocrine functions and participate in the prevention and treatment of hypertension.6,20–22 The effects of Chinese herbs in terms of their mechanism of affecting the gut microbiota are crucial.14,15,23 In the current study, the effects of THSWD on blood pressure and the mechanisms related to gut microbiota were investigated in high-salt diet-induced mice. For the first time, we found that THSWD improved the gut microbial balance and increased the serum level of the microbial metabolite VK2 in high salt-diet mice, restoring blood pressure and endothelial function.
Vascular endothelial dysfunction has been related to the pathogenesis of hypertension. 21 The main manifestations of vascular endothelial dysfunction are either a decrease of endothelium-dependent relaxation function or the enhancement of endothelium-dependent contraction function.24,25 The imbalance of vasodilator and systolic factors released by endothelial cells aggravates the development of hypertension.26,27 Convincing evidence has shown that Chinese herbs can help protect blood pressure by regulating endothelial function.28,29 We found similar results where blood pressure was lowered in the THSWD group. Cytokines are small polypeptides or glycoproteins synthesized and secreted by a variety of tissue cells that mediate cell–cell interactions, such as regulating endothelial function. 30 Our results were consistent with the findings that THSWD could protect blood pressure and significantly upregulate the content of NO and downregulate the level of ET-1, AngI, and VEGF in the serum compared with the model group. This shows that THSWD can protect blood pressure and restore endothelial function.
Chinese herbs have an obvious regulatory effect on gut microbiota.14,15 On the one hand, flavonoids, polysaccharides, and saponins, the main functional components of Astragalus membranaceus, can maintain the homeostasis of the intestinal microenvironment by affecting the population structure, metabolism, and intestinal cell function of gut microbiota. 31 On the other hand, gut microbiota also participate in the absorption, metabolism, and transformation of these active components in the body, which has an impact on the performance of their functions, which is an essential role in the mechanism of the function of bioactive substances. 32 Previous studies have indicated that the gut microbiota might be either the target or an important way for Chinese medicines to exert their efficacy. 33 In a recent study, the Chinese herb Fei-Xi-Tiao-Zhi-Fang (FTF) was found to alleviate the symptoms of recurrent respiratory tract infection (RRTI) by recovering gut microbiota and improving immunity. 13 Another study revealed that Hetiao Jianpi decoction prevented the occurrence of antibiotic-associated diarrhea (AAD) by regulating the microbial structure. 34 The results were consistent with these findings that THSWD could regulate the gut microbial community abundance, upregulating Firmicutes and norank_f_Muribaculaceae, while downregulating Proteobacteria and Lactobacillus. THSWD was found to regulate the most dominant microbial genera and phyla and enhance gut microbial activity. The F/B ratio, which is a marker and often used to assess the balance of gut microbiota, is associated with blood pressure. 35 In this study, although no statistically significant differences in the F/B ratio were observed, we found that THSWD can upregulate the F/B ratio in the gut microbiota in a trend that suggested that THSWD may have the function of regulating and tending to balance the disturbed gut microbiota. The results also reveal that THSWD can significantly upregulate the relative abundance of Dubosiella and downregulate the relative abundance of Cyanobium_PCC-6307 and DNF00809 in gut microbiota. So, they may play the role of biomarkers related to hypertension and may be the microbial targets of THSWD. A previous study revealed that Dubosiella could be beneficial in obesity. 36 However, there is no evidence that Cyanobium_PCC-6307 and DNF00809 are related to the diseases, as they were found elevated in the hypertensive mice group. They are supposed to be the pathogenic bacteria of hypertension in this study, but further research is still required. For in-depth gut microbiota analysis, PCA was selected to calculate beta diversity based on the OTU levels and using Adonis. A previous study indicated that PCA has been widely used to show a significant difference between groups. 37 The results reveal that there exists a significant difference between the THSWD and hypertensive mice microbial communities. Moreover, the results of microbial distance among the 3 groups calculated on OTU levels further confirmed that THSWD can regulate the microbial community structure.
The gut microbiota influences body processes and health through metabolites secreted in the gut.21,38 Even more, it was found that microbial metabolites played an essential role in hypertension, affecting endothelial function and blood pressure.21,39 VK2 is mainly produced by intestinal microbial metabolism, which has shown its vital role in inhibiting vascular calcification.40,41 Studies have shown the therapeutic effect of VK2 on various cardiovascular diseases.41,42 Our results revealed that THSWD can significantly upregulate the level of VK2 in serum. Therefore, it is demonstrated that THSWD restores blood pressure and endothelial function in high salt–induced hypertensive mice by regulating VK2 microbial biosynthesis.
The correlation between microbial community classification and various associated factors was conducted by Spearman correlation analysis. 43 In the study, it was found that Dubosiella was positively correlated with VK2 and NO and negatively correlated with ET-1, AngI, and VEGF. The relationships between gut microbiota, vascular endothelial function-related factors, and metabolites are complex. The results indicate that Dubosiella may function as the regulator of protecting endothelial function and promoting microbial metabolism of VK2 in hypertensive mice. Furthermore, it is further supposed that Dubosiella may play the role of biomarker related to hypertension and may be the microbial target of THSWD. These findings, interestingly, provide clues for further study and needed long-term research.
Chinese medicines are often digested and absorbed by the intestine before they can produce efficacy. There is a close relationship between gut microbiota and the efficacy of Chinese medicines. Our previous study has revealed that there are many chemical compounds in THSWD related to hypertension. 5 In the present study, the potential of amygdalin, 3,4-dihydroxyphenylacetic acid, kaempferol, and adenine was selected and verified by UPLC–Q/TOF-MS analysis based on network pharmacology research of THSWD. 5 Nie et al concluded that quercetin could reduce atherosclerotic lesions by altering the gut microbiota and reducing metabolites. 44 Koper et al showed that luteolin played a role in immunity and homeostasis of the gut barrier and contributed to tryptophan metabolites. 45 Additional studies also showed that the core components of THSWD were related to the gut microbiota. 22 There is currently no report that 3,4-dihydroxyphenylacetic acid is derived from any single herb of THSWD, and so could be produced during the preparation of THSWD decoction (Supplemental Table 3). Lofft et al found that 3,4-dihydroxyphenylacetic acid could reverse miRNAs through inflammatory and cancer pathways. 46 Interestingly, 3,4-dihydroxyphenylacetic acid has vasorelaxant effects on the mesenteric artery, which was confirmed to decrease blood pressure when given either as a bolus or by slow infusion in vivo. 47 Aa et al demonstrated that the high level of kaempferol in the gut regulated the gut microbiota and was a potential mechanism for anti-arthritic activities. 48 Therefore, all of this evidence provide quality control for THSWD. However, the specific pharmacological mechanism of THSWD still needs to be further studied.
Conclusion
In conclusion, the current research highlighted that treatment with THSWD resulted in protecting blood pressure and endothelial function in hypertensive mice. The mechanism of action was demonstrated here to be associated with regulating gut microbiota and promoting microbial metabolic VK2. These data indicate the important role of THSWD in controlling the gut microbiota of high-salt diet-induced mice. However, further studies are required to investigate further the detailed mechanism with a complete metabolic profile, and then linking and associating that with health and disease conditions.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221118199 - Supplemental material for The Important Role of TaohongSiwu Decoction in Gut Microbial Modulation in Response to High-Salt Diet-Induced Hypertensive Mice
Supplemental material, sj-docx-1-npx-10.1177_1934578X221118199 for The Important Role of TaohongSiwu Decoction in Gut Microbial Modulation in Response to High-Salt Diet-Induced Hypertensive Mice by Tian-hao Liu, Ting Xie, Zhen-yu Bai, Qiu-er Liang, Peng-cheng Xie, Yu-zheng Xue, Ya Xiao and Li-guo Chen in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X221118199 - Supplemental material for The Important Role of TaohongSiwu Decoction in Gut Microbial Modulation in Response to High-Salt Diet-Induced Hypertensive Mice
Supplemental material, sj-docx-2-npx-10.1177_1934578X221118199 for The Important Role of TaohongSiwu Decoction in Gut Microbial Modulation in Response to High-Salt Diet-Induced Hypertensive Mice by Tian-hao Liu, Ting Xie, Zhen-yu Bai, Qiu-er Liang, Peng-cheng Xie, Yu-zheng Xue, Ya Xiao and Li-guo Chen in Natural Product Communications
Footnotes
Acknowledgements
All of the experimental procedures in this study involving animals were conducted under the control of the Committee for Control and Supervision of Experiments on Animals and approved by the Animal Ethics Committee of Jinan University.
Authors’ Contribution
Tian-hao Liu, Li-guo Chen, and Ya Xiao participated in the study design. Peng-cheng Xie, Qiu-er Liang, Zhen-yu Bai, and Ting Xie searched databases and conducted animal experiment operations. Li-guo Chen, Tian-hao Liu, Yu-zheng Xue, and Ya Xiao drafted the manuscript. Ya Xiao and Tian-hao Liu carried out the statistical analysis of the data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Sciences Foundation of China (81673848, and 82174148), and Wuxi Municipal Health Commission Scientific Research Fund Youth Project (Q202106).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
