Abstract
Keywords
Introduction
The Asteraceae Bercht. & J. Presl is the largest plant family with around 33,000 accepted species. 1 Wyethia Nutt. belongs to the family Asteraceae, subfamily Asteroideae (Cass.) Lindl., tribe Heliantheae Cass., subtribe Engelmanniinae Stuessy. 1 Within the Engelmanniinae subtribe are ten genera, Agnorhiza (Jeps.) W.A. Weber, Balsamorhiza Hook., Berlandiera DC., Borrichia Adans., Chrysogonum L., Engelmannia A.Gray ex Nutt., Lindheimera A.Gray & Engelm., Silphium L., Vigethia W.A. Weber, and Wyethia. 1 There are 11 recognized species of Wyethia, 2 which are found only in western North America. 3 Wyethia amplexicaulis (Nutt.) Nutt. is a perennial forb found in eastern Washington and eastern Oregon, most of Idaho, western Montana, western Wyoming, and south into Nevada and Utah (Figure 1). The plant grows to 20 to 50 cm tall, with deep green leathery, glossy leaves. The heads are large with yellow ray flowers (Figure 2). Wyethia helianthoides Nutt. ranges from eastern Oregon, across southern Idaho, and into southwestern Montana, northwestern Wyoming, and northeastern Nevada (Figure 1). The plant is 30 to 50 cm tall, with dull gray-green leaves with a covering of soft hairs. The head is around 3 cm broad with cream-colored or white ray flowers (Figure 3). 3

Western United States showing the range of Wyethia amplexicaulis ( ), the range of Wyethia helianthoides (
), the range of Wyethia helianthoides ( ), based on Weber,
3
and the approximate location of the collection site (★).
), based on Weber,
3
and the approximate location of the collection site (★).

Wyethia amplexicaulis (Nutt.) Nutt. (A) Photograph of the plant (K. Swor). (B) Scan of pressed plant (W.N. Setzer).

Wyethia helianthoides Nutt. (A) Photograph of the plant (K. Swor). (B) Scan of pressed plant (W.N. Setzer).
Wyethia species have proven to be a rich source of flavonoids.4,5 However, there are apparently no previous reports on essential oils from Wyethia species. The purpose of this study is to examine the volatile chemicals present in W. amplexicaulis and W. helianthoides collected in the foothills near Boise, Idaho.
Results and Discussion
Essential Oil Composition
Leaves of three individual W. amplexicaulis and three individual W. helianthoides were collected during the flowering stage from locations near Prairie, Idaho. The leaves were hydrodistilled to give pale yellow essential oils in yields of 0.090% to 0.136% and 1.49% to 1.77%, respectively. The essential oils were analyzed by gas chromatography with flame ionization detection (GC-FID) and gas chromatography coupled with mass spectrometry (GC-MS). A total of 125 compounds were identified in the leaf essential oils of W. amplexicaulis accounting for 89.6% to 95.6% of the compositions. The GC analysis of the essential oils of W. helianthoides allowed 76 compounds to be identified (89.3%-92.4% of the compositions). The essential oil components are compiled in Table 1.
Chemical Compositions (%) of the Leaf Essential Oils of Wyethia amplexicaulis and Wyethia helianthoides from Southwestern Idaho.

RIcalc is the retention index determined with respect to a homologous series of n-alkanes on a ZB-5 ms column using the linear equation of van den Dool and Kratz. 6 RIdb is the reference retention index obtained from the databases.7–10 tr = trace (< 0.05%).
MS(EI): 220(5%), 205(7%), 177(69%), 159(100%), 149(35%), 135(38%), 121(46%), 107(80%), 93(94%), 91(59%), 81(50%), 55(40%), 43(57%), 41(48%).
MS(EI): 220(7%), 205(5%), 202(8%), 177(38%), 159(100%), 135(36%), 107(35%), 105(36%), 93(65%), 91(59%), 79(34%), 55(30%), 43(32%), 41(42%).
MS(EI): 207(8%), 204(10%), 189(5%), 163(7%), 149(19%), 107(19%), 95(40%), 81(22%), 67(15%), 59(100%), 43(114%), 41(14%).
MS(EI): 220(2%), 105(8%), 204(11%), 177(41%), 159(100%), 135(33%), 121(44%), 119(38%), 107(50%), 93(73%), 91(47%), 81(49%), 55(56%), 43(98%), 41(58%).
MS(EI): 220(36%), 189(16%), 150(24%), 135(100%), 133(46%), 107(55%), 105(81%), 91(95%), 79(59%), 67(34%), 55(44%), 41(54%).
MS(EI): 121(11%), 120(100%), 107(10%), 83(12%), 77(8%), 55(27%).
MS(EI): 121(12%), 120(100%), 107(8%), 83(35%), 77(8%), 55(16%).
MS(EI): 236(13%), 221(14%), 167(55%), 126(29%), 109(30%), 107(35%), 98(86%), 95(62%), 91(43%), 79(54%), 69(99%), 55(85%), 43(100%), 41(76%).
MS(EI): 202(12%), 177(10%), 159(34%), 132(18%), 83(100%), 55(55%), 43(16%), 41(15%).
There were several components in relatively high concentrations that were common to both W. amplexicaulis and W. helianthoides. These include α-pinene (2.4%-7.5% and 6.3%-7.3%, respectively), myrcene (0.3%-32.2% and 8.6%-12.4%), (E)-β-ocimene (0.2%-7.6% and 10.0%-14.3%), (E)-β-caryophyllene (3.7%-6.9% and 1.1%-4.1%), germacrene D (0.3%-6.4% and 16.5%-21.5%), 15-copaenol (5.3%-11.4% and 4.3%-6.0%), and carotol (2.8%-10.0% and 3.5%-5.9%). On the other hand, silphiperfolan-7β-ol was abundant in W. amplexicaulis (5.5%-10.2%) but was a minor constituent in W. helianthoides. Likewise, caryophyllene oxide showed 0.3% to 5.0% in W. amplexicaulis but was not observed in W. helianthoides.
Sample #3 of W. amplexicaulis seems to be a notable outlier in the W. amplexicaulis essential oils. Monoterpene hydrocarbons were very low (only 7.9%) compared to samples #1 (47.4%) and #2 (45.0%). Likewise, oxygenated sesquiterpenoids were high in sample #3 (64.1%) compared to samples #1 (27.9%) and #2 (29.0%). The reason for the large difference in compositions is not clear. The three samples were collected from the same location on the same day. The three W. helianthoides essential oils, on the other hand, are very similar to one another.
Multivariate analyses were carried out to reveal the similarities between the six essential oil samples. Hierarchical cluster analysis (HCA) showed three well-defined groups (Figure 4). W. amplexicaulis #1 and #2 form one group with > 90% similarity, W. helianthoides #1, #2, and #3 form another group with > 90% similarity, and W. amplexicaulis #3 is the outlier and is not similar to the other essential oil samples. Principal component analysis (PCA) verifies the groupings observed in the HCA (Figure 5). W. amplexicaulis samples #1 and #2 correlate strongly with myrcene; W. helianthoides #1, #2, and #3 correlate strongly with germacrene D and (E)-β-ocimene; and W. amplexicaulis #3 correlates strongly with caryophyllene oxide.
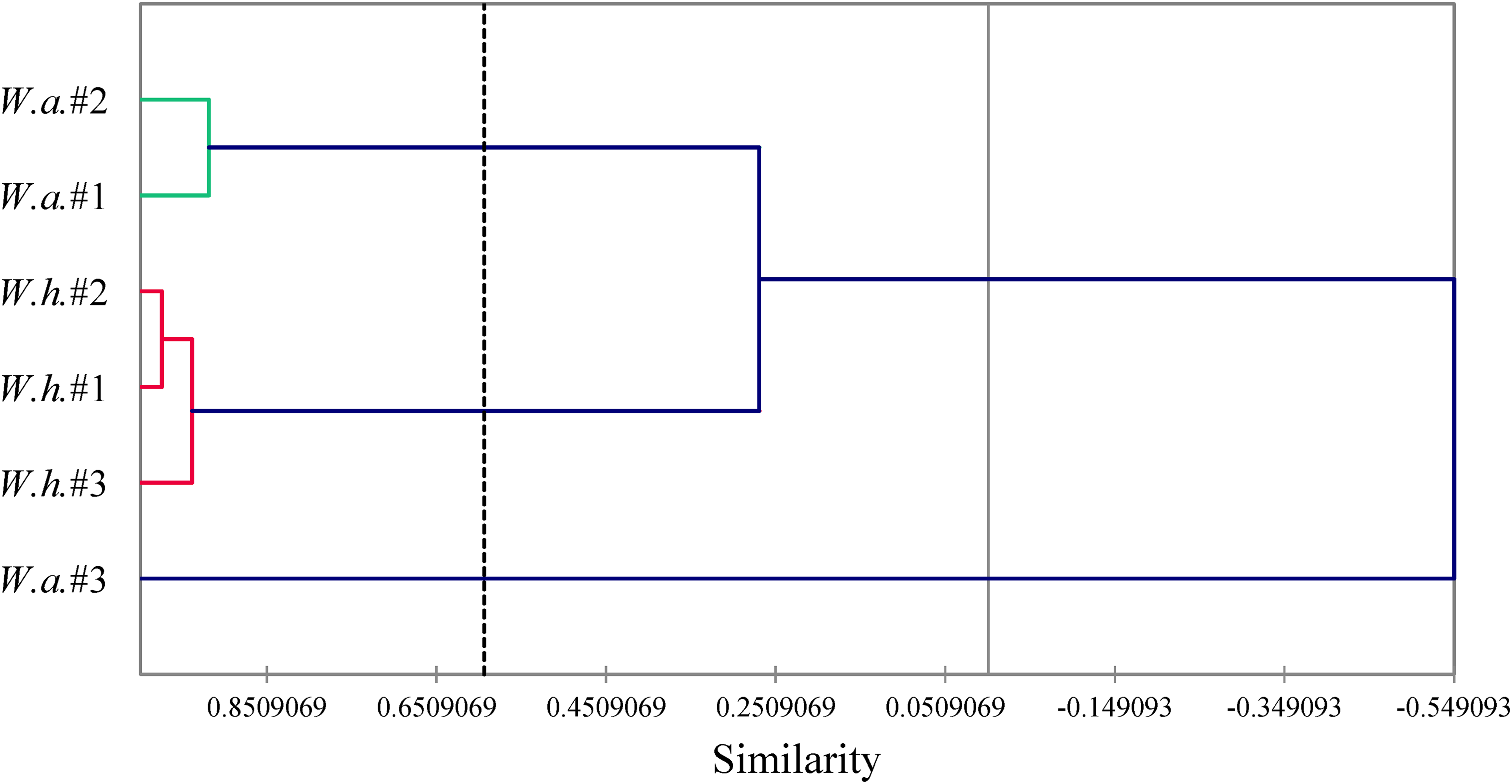
Dendrogram obtained by hierarchical cluster analysis (HCA) of Wyethia amplexicaulis (W.a.) and Wyethia helianthoides (W.h.) leaf essential oils.
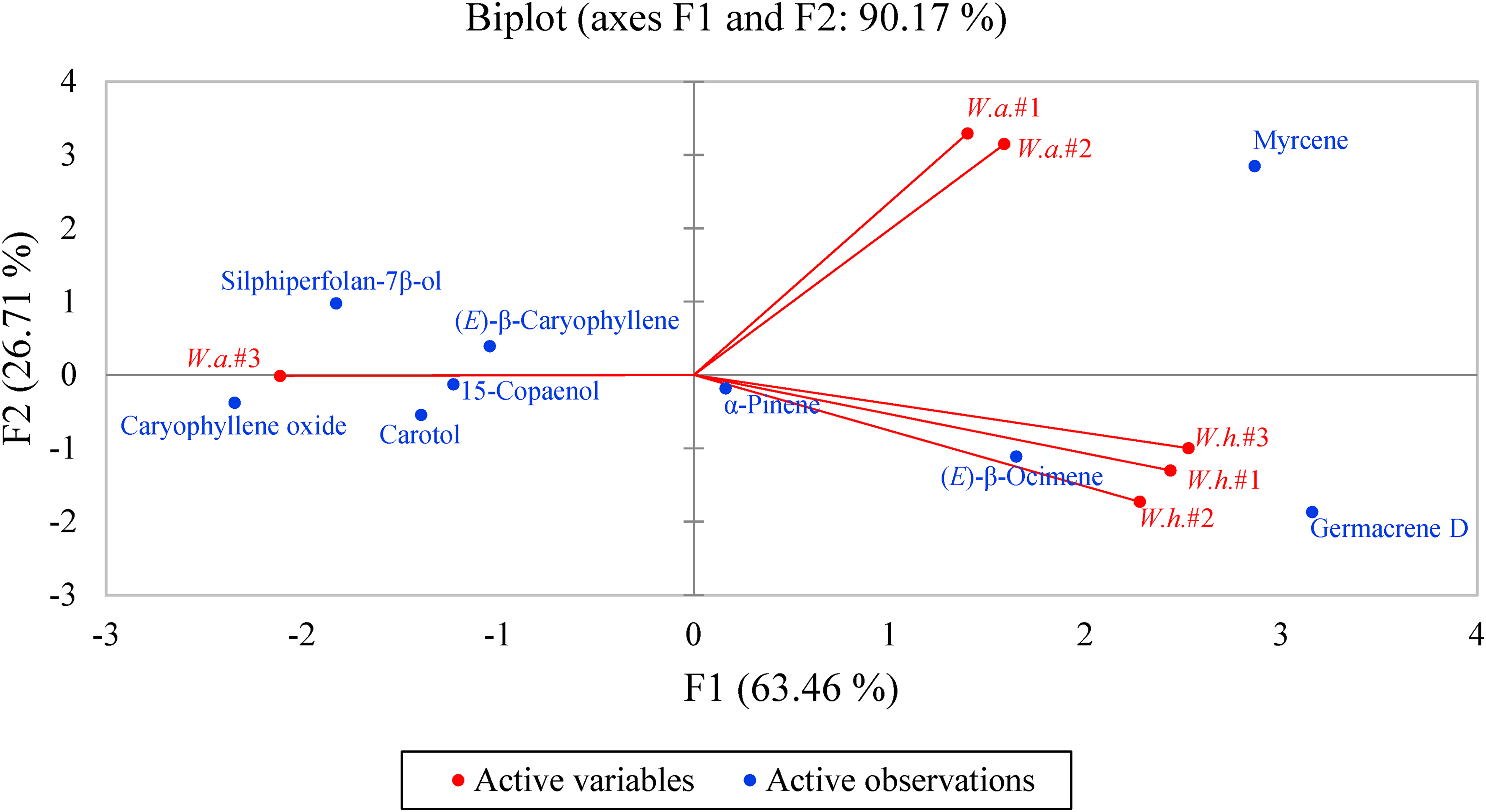
Principal component analysis (PCA) of Wyethia amplexicaulis (W.a.) and Wyethia helianthoides (W.h.) leaf essential oils.
There have been no previous reports on essential oil compositions of Wyethia species for comparison. However, several members of the Engelmanniinae subtribe of the Asteraceae have been examined. Silphium perfoliatum L. leaf essential oil showed relatively high concentrations of α-pinene (5.9% and 5.4%), (E)-β-caryophyllene (4.8 and 4.0%), germacrene D (6.4 and 24.3%), and caryophyllene oxide (34.7 and 8.5%). 11 However, neither myrcene, (E)-β-ocimene, nor carotol were observed. Likewise, Silphium integrifolium Michx. and Silphium trifoliatum L. leaf essential oils were rich in α-pinene (7.3%-8.8% and 1.2%-6.0%), (E)-β-caryophyllene (2.8%-4.8% and 6.7%-14.9%), germacrene D (18.7%-28.4% and 8.3%-16.1%), and caryophyllene oxide (6.1%-12.4% and 25.4%-29.3%), but neither myrcene, (E)-β-ocimene, nor carotol were observed. 12 Another sample of S. integrifolium leaf essential oil, however, showed high concentrations of α-pinene (58.6%) and myrcene (9.7%), as well as (E)-β-caryophyllene (2.5%) and germacrene D (3.0%), but low concentrations of (E)-β-ocimene (0.4%) and caryophyllene oxide (0.5%). 13 Borrichia frutescens (L.) DC. leaf essential oil had high concentrations of α-pinene (3.8%-8.3%), myrcene (0.6%-7.8%), and germacrene D (0.7%-18.0%), but low concentrations of (E)-β-caryophyllene (0.2%-1.3%), carotol (0.0%-0.9%), and caryophyllene oxide (0.0%-0.1%), and (E)-β-ocimene was not observed. 14
Enantiomeric Distribution
In order to more fully characterize the volatile phytochemistry of Wyethia, the essential oils were analyzed by chiral GC-MS. The enantiomeric distributions of chiral terpenoid components are listed in Table 2.
Enantiomeric Distributions of Chiral Terpenoid Components in Wyethia amplexicaulis and Wyethia helianthoides Leaf Essential Oils.
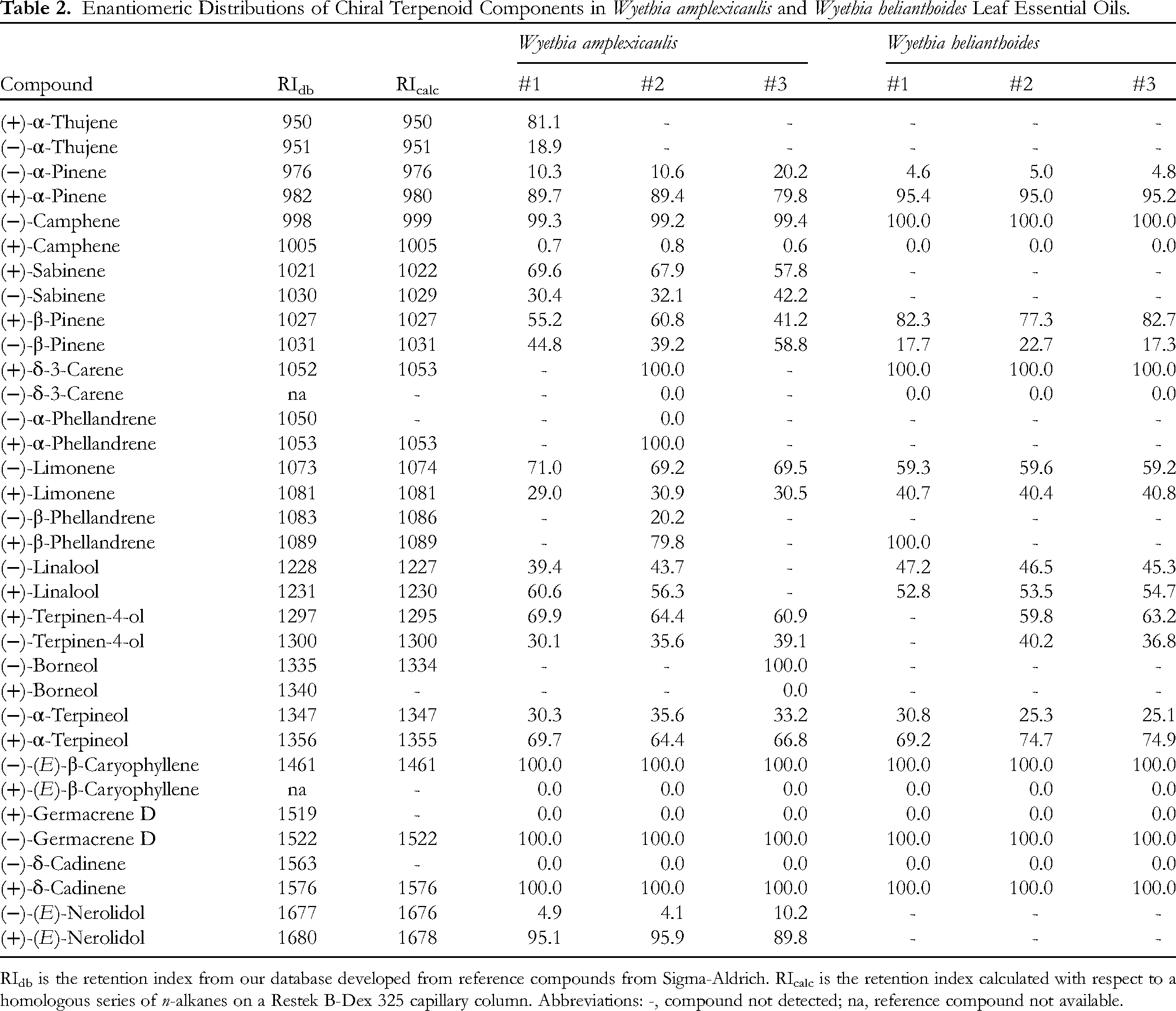
RIdb is the retention index from our database developed from reference compounds from Sigma-Aldrich. RIcalc is the retention index calculated with respect to a homologous series of n-alkanes on a Restek B-Dex 325 capillary column. Abbreviations: -, compound not detected; na, reference compound not available.
In the Wyethia essential oils, (+)-α-pinene, (−)-limonene, (+)-terpinen-4-ol, and (+)-α-terpineol were the major enantiomers; (−)-camphene was the dominant (> 99%) enantiomer, and (+)-δ-3-carene, (−)-(E)-β-caryophyllene, (−)-germacrene D, and (+)-δ-cadinene were the only enantiomers observed. (+)-Sabinene and (+)-(E)-nerolidol were the major enantiomers in W. amplexicaulis essential oil, but neither sabinene nor (E)-nerolidol were observed in W. helianthoides. (+)-β-Pinene was the major enantiomer in W. helianthoides, but β-pinene was virtually racemic in W. amplexicaulis. Linalool was nearly racemic in the Wyethia essential oils with 52.8% to 60.6% (+)-linalool.
Consistent with these enantiomeric distributions, the leaf essential oil of a closely related species, Balsamorhiza sagittata (Pursh) Nutt.3,15 showed comparable distributions with (−)-camphene, (−)-(E)-β-caryophyllene, (−)-germacrene D, (+)-δ-cadinene, and (+)-(E)-nerolidol as exclusive enantiomers; (+)-sabinene, (−)-limonene, and (+)-terpinen-4-ol as major enantiomers; while α-pinene and β-pinene were virtually racemic. 16 Interestingly, the enantiomeric distributions are reversed for several components identified in Ericameria nauseosa (Pursch) G.L. Nesom & G.I. Baird (Asteraceae). 17 The major enantiomers in this plant were (−)-α-pinene, (−)-sabinene, (−)-β-pinene, (−)-terpinen-4-ol, and (−)-α-terpineol.
Conclusions
This work reports, for the first time, the leaf essential oil compositions of 2 species of Wyethia collected from the foothills near Boise, Idaho. W. amplexicaulis essential oils are generally dominated by myrcene, while W. helianthoides shows germacrene D, and (E)-β-ocimene, as well as myrcene as major essential oil components. A limitation of this particular study is that only three individuals of each species were sampled from only one geographical location. Further research is needed to examine additional individuals from other locations. In addition, research on other Wyethia species is needed to help characterize the volatile components of this genus.
Materials and Methods
Plant Material
Leaves of W. amplexicaulis and W. helianthoides, three individuals each, were collected near Prairie, Idaho, on 25 May 2023. The plants were identified in the field by W.N. Setzer and confirmed by comparison with samples from the C.V. Starr Virtual Herbarium. 18 Voucher specimens (WNS-Wa-7104 and WNS-Wh-7114) have been deposited with the University of Alabama in Huntsville herbarium. The fresh leaves were stored frozen (–20 °C) until processing. For each plant sample, the fresh-frozen leaves were chopped and then added to a 500-mL flask and enough distilled water was added to cover the plant materials. The leaves were hydrodistilled, once per plant sample, for 4 h using a Likens-Nickerson apparatus19–21 with continuous extraction of the distillate with dichloromethane to give pale-yellow essential oils (Table 3).
Collection and Hydrodistillation Details for Wyethia.

Gas Chromatographic Analyses
GC-MS was used to analyze the compositions of the Wyethia essential oils. A Shimadzu Model GC-MS-QP2010 Ultra (Shimadzu Scientific Instruments) equipped with a ZB-5 ms fused silica capillary column (60 m, 0.25 mm; 0.25 μm film thickness) as stationary phase was used (Phenomenex). The helium carrier gas was adjusted to 1.0 mL/min at 208.2 kPa head pressure. The injection of diluted essential oils in dichloromethane (5% w/v) were carried out in the split mode (1:24.5). The injector temperature was 260 °C. The programed oven temperature was 50 to 260 °C at a rate of 2 °C/min). The mass selective detector was operated in the EI mode with electron energy = 70 eV. The scan range was 40 to 400 amu, and scan rate was 3.99 scans/s. the ion source and interface temperatures were 260 °C. Identification of the oil components was based on their retention indices, determined by reference to a homologous series of n-alkanes using the linear equation of van den Dool and Kratz, 6 and by comparison of their mass spectral fragmentation patterns with those reported in the Adams, 7 FFNSC 3, 8 NIST20, 9 and Satyal 10 libraries.
GC-FID, was carried out on each essential oil sample using a Shimadzu GC 2010 with FID detector (Shimadzu Scientific Instruments), ZB-5 GC column (60 m × 0.25 mm × 0.25 μm film thickness. (Phenomenex) using the same operating conditions as above for GC-MS. The percent compositions were determined from raw peak areas without standardization.
Enantioselective GC-MS were GC-FID analysis was carried out using a Shimadzu GC-MS-QP2010S instrument (Shimadzu Scientific Instruments) fitted with a Restek β-Dex 325 column, 30 m × 0.25 mm diameter × 0.25 μm film thickness, Restek Corp.). The injector and detector temperatures were 240 °C. Helium was the carrier gas with a flow rate of 1.00 mL/min and a column head pressure of 53.6 kPa. The GC oven temperature program was initial temperature of 50 °C, which was held for 5 min, then increased to 100 °C at a rate of 1.0 °C/min, then increased to 220 °C at a rate of 2 °C/min. Essential oil samples (0.3 μL of a 5% (w/v) essential oil solution in dichloromethane) were injected using a splitting mode of 1:24. The enantiomers were determined by comparison of calculated retention indices with those of authentic samples (Sigma-Aldrich). Enantiomeric ratios were calculated from GC-MS (TIC) peak areas.
Hierarchical Cluster Analysis and Principal Component Analysis
HCA was carried out to examine the similarity of oil samples based on the percentages of nine major components (myrcene, germacrene D, (E)-β-ocimene, 15-copaenol, α-pinene, carotol, silphiperfolan-7β-ol, (E)-β-caryophyllene, and caryophyllene oxide). The six essential oil samples were treated as operational taxonomic units. Pearson correlation was used to measure similarity, and the unweighted pair-group method with arithmetic average was used to define the clusters. Principal component analysis (PCA, type Pearson Correlation) was used to determine the interrelationship of essential oil components and to verify the previous HCA analysis. Both the HCA and PCA analyses were carried out using XLSTAT v. 2018.1.1.62926 (Addinsoft).
Footnotes
Acknowledgments
Data Availability Statement
All the data for this study are available in the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
