Abstract
Scutellaria (skullcap) are important medicinal plants. Scutellaria baicalensis and S.barbata have been used in Chinese traditional medicine, while S. incana and S. lateriflora were used as herbal medicines by Native Americans. In this work, the essential oils of Scutellaria baicalensis Georgi, Scutellaria barbata D. Don, Scutellaria incana Biehler, and Scutellaria lateriflora L. were obtained from plants cultivated in south Alabama and analyzed by gas chromatographic techniques, including chiral gas chromatography. The most abundant components in the Scutellaria essential oils were 1-octen-3-ol (31.2% in S. incana), linalool (6.8% in S. incana), thymol (7.7% in S. barbata), carvacrol (9.3% in S. baicalensis), (E)-β caryophyllene (11.6% in S. baicalensis), germacrene D (39.3% in S. baicalensis), (E)-nerolidol (10.5% in S. incana), palmitic acid (15.6% in S. barbata), phytol (19.7% in S. incana), and linolenic acid (8.0% in S. barbata). These analyses of the essential oil compositions and enantiomeric ratios of predominant aromatic molecules add to our understanding of the medicinal phytochemistry of the genus Scutellaria.
Keywords
Scutellaria baicalensis Georgi, Scutellaria barbata D. Don, Scutellaria incana Biehler, and Scutellaria lateriflora L. were cultivated in southern Alabama where two of the species, S. incana and S. lateriflora, are natives. These herbs, commonly and collectively referred to as ‘skullcap’, belong to the mint family (Lamiaceae) and are found across the globe in temperate regions on almost every continent.
This is part of a larger ongoing study of essential oils from the medicinal plants traditionally used by Native American tribes of the United States, in particular the Cherokee of the southeastern United States. 1 -14 The Cherokee have a rich tradition of using plants as medicines including many of the aromatic species native to the southeastern U.S. that are rich in essential oils and aromatic compounds. Many ethnobotanical accounts have been given in the anthropologies of the Cherokee, and these works were studied to determine which plants to examine in further detail. 15 -22 Two of the species of Scutellaria studied here were used by the Cherokee (S. incana and S. lateriflora) for at least hundreds of years, while the other 2 have been used in traditional Asian phytotherapies for several millennia (S. baicalensis and S. barbata) . 23 All four were easily cultivated in the climate of south Alabama, United States, and are growing as perennials as they are being observed sprouting new growth during the spring of their second year.
The genus Scutellaria encompasses over 470 species with more than 70 of them being used traditionally as plant medicines. 24,25 Scutellaria incana, or ‘downy skullcap,’ is a perennial herb preferring medium-wet to dry conditions and full sun to partial shade. It is native to the central and eastern United States, found from Michigan and New York in the north, to the southern tip of Florida in the south, and can be found in bloom between July and September. 26,27 Scutellaria lateriflora, ‘blue skullcap, mad-dog skullcap, or American skullcap’, is also native to the United States and is the most common species of Scutellaria found in North America. It can be found in zones 2‐7 blooming in July through September across most of the North American continent, from Florida to Canada and California to New York, preferring wetlands and full sun. 28,29 The other 2 species, S. barbata and S. baicalensis, are also perennial herbs, both native to China, with S. barbata, ‘barbed skullcap,’ being prominent throughout southeast Asia as well as north into the mountains of Nepal. Wetlands and the edges of streams and rice fields are common places to find S. barbata and it usually flowers from April to June. 30,31 Commonly called ‘baikal’ and found across eastern Europe and Russia and across Mongolia and China and in Korea and Japan, S. baicalensis is the fourth herb studied here. It can be found blooming usually between July and August along roadsides and in disturbed and cultivated areas as a weed, as well as covering grassy hillsides in the wild. 32
Many tribes of Native Americans were known to use Scutellaria as a medicinal plant. The Cherokee used Scutellaria incana roots as an abortifacient and for diarrhea treatment. They also used a decoction of the plant to soothe the nerves. They made a compound from the plant which was used topically to aid in the expelling of afterbirth as well as a kidney medicine and for breast pains. This could refer either to mastitis or breast tumors as these Scutellaria species have been studied for their ability to act as anti-inflammatory agents and have potent cytotoxic anti-tumor effects. 33 The roots of S. lateriflora were used in an infusion to calm diarrhea and to encourage menstruation. A decoction of the roots was taken as an emetic and to expel afterbirth. It was also made into a compound to be applied to help expel afterbirth. According to Mooney, in his 1885 research, the Scutellaria species were used to bring about menstruation and to counteract the perceived negative effects of eating food cooked by a woman who was menstruating. 17 The Iroquois Native Americans of New York and Ontario used an infusion of dried, ground roots of S. lateriflora to prevent smallpox and for throat problems. 22 The Miwok of modern-day central California used multiple species of Scutellaria in a decoction for cold and cough remedy and as an eye wash.. 34
In traditional Chinese medicine (TCM) S. baicalensis is known as ‘huang qin’ and is used to treat inflammations and fevers. 32 Russian research into phytomedicinals has shown that S. baicalensis has antibacterial, anthelmintic, anti-schlerotic, astringent, digestive stimulant, hemostatic, and anti-allergen properties. 35 The common name in TCM for S. barbata is ‘ban zhi lian’ and it is traditionally used as both an internal and external anti-inflammatory. Both S. barbata and S. baicalensis have also been used as anxiolytics and sedatives. 30 -32,36
Flavonoids, diterpenes, alkaloids, glycosides, and volatile essential oil components have all been studied from Scutellaria species, with over 295 compounds having been characterized by 2010. 37 The presence of neo-clerodane diterpenoid alkaloids with cytotoxic activity in Scutellaria barbata has been confirmed experimentally, 38,39 numerous other neo-clerodane diterpenoids as well as flavonoids, 23 and aromatic terpenoids and hydrocarbons have been analytically revealed. This study examines the volatile composition and enantiomeric distribution of components of the essential oils of these 4 scutellaria species and compares these findings with previous literature reports of their essential oil compositions from other geographic locations as well as discusses the relevance of the terpenoids that were found in relation to their ethnobotanical uses.
Results and Discussion
In this study, the essential oils of 4 species of Scutellaria most commonly used in traditional medicine were obtained through hydrodistillation and analyzed by gas chromatographic techniques. The following table (Table 1) reports the yields of essential oils obtained from the dried plant matter of the 4 scutellaria species.
Essential Oil Yields After Hydro-Distillation.

a Scutellaria barbata #1 sample was harvested on June 7, 2020
b Scutellaria barbata #2 sample was harvested on July 7, 2020.
Table 2, below, compares the essential oil components from the Scutellaria spp. as identified by gas chromatographic analysis.
Essential Oil Chemical Composition of Scutellaria Species Cultivated in South Alabama. Retention Indices, RI (Cal), Were Calculated Using a Homologous Series of N-Alkanes on a ZB-5ms Column. RI (Db) Refers to Reference Database Indices.

a Scutellaria barbata #1 sample was harvested on June 7, 2020.
b Scutellaria barbata #2 sample was harvested on July 7, 2020.
Table 3 lists the enantiomeric distribution percentages for eight of the most prevalent terpenoids found in these Scutellaria species. For all the species (–)-linalool was the predominant enantiomer, (+)-(E)-β-caryophyllene was the only enantiomer found in all samples, α-terpineol was found in about a 50/50% split with (–)-α-terpineol being favored by a less than one percent margin. Germacrene D was found only in its (+)-enantiomeric form and it was absent from S. incana altogether, δ-cadinene was only found as its (–)-enantiomer and it was not found in S. incana or S. lateriflora.
Chirality Data for the Enantiomeric Distribution of Eight of the Most Prevalent Terpenoids in These 4 Scutellaria Species Here Examined.
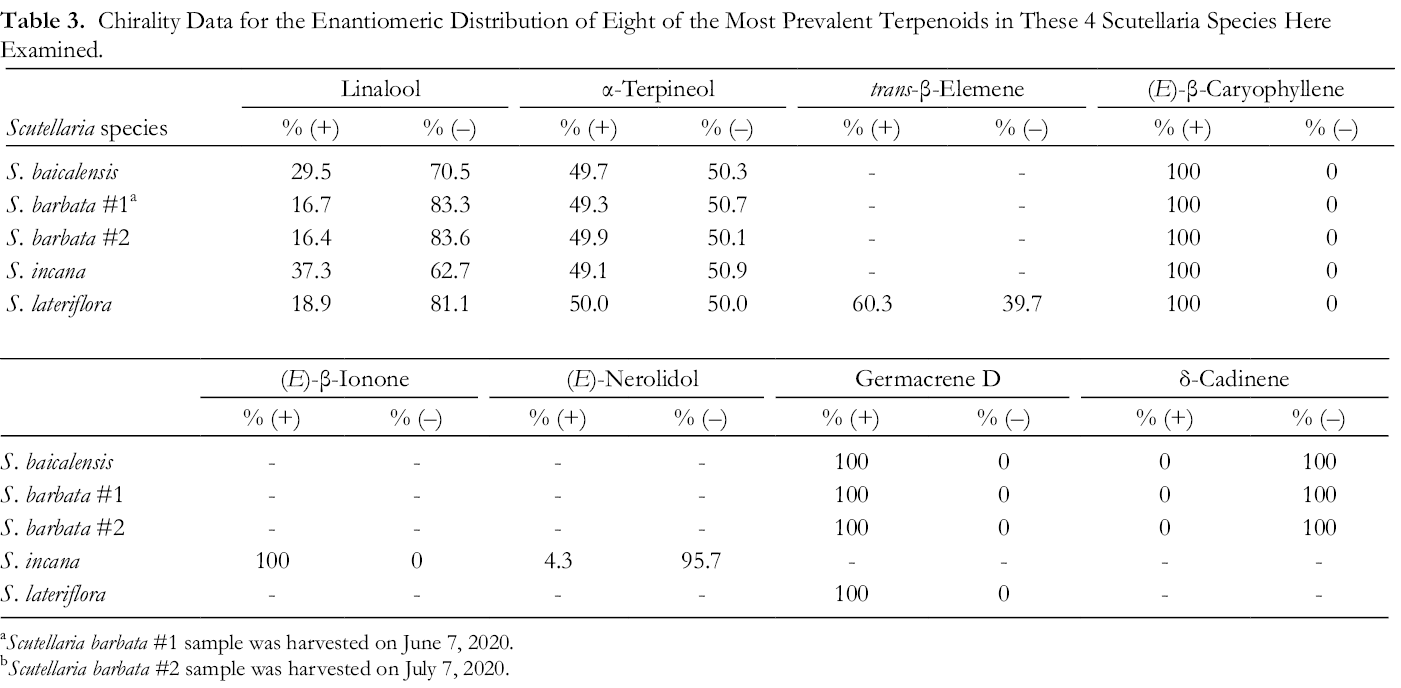
a Scutellaria barbata #1 sample was harvested on June 7, 2020.
b Scutellaria barbata #2 sample was harvested on July 7, 2020.
The volatile phytochemical makeup of the essential oils of these 4 scutellaria species were collectively dominated by 1-octen-3-ol, thymol, carvacrol, (E)-β-caryophyllene, germacrene D, (E)-nerolidol, palmitic acid, phytol, linalool, β-bourbonene, and linolenic acid. Studies of the essential oil from S. barbata from the Hunan region of China showed a predominance of 1-octen-3-ol, hexadecanoic acid (palmitic acid), and (E)-β-caryophyllene, 40 however most of the other key aromatic molecules such germacrene D, carvacrol, thymol, phytol, and linalool found in our research were not reported in their analysis. A previous study from the University of Wuhan, China showed the essential oil of S. barbata to exhibit more similar molecular makeup to our samples, with hexahydrofarnesylacetone, 3,7,11,15-tetramethyl-2-hexadecen-1-ol, menthol, and 1-octen-3-ol being the dominant by percentage, but also in significant quantities (>1%) were linalool, β-bourbonene, β-himachalene, and thymol. 41
In their 2010 review of several Scutellaria essential oil studies, Shang et al, 37 noted high concentrations of (E)-β-caryophyllene and linalool. Samples of essential oils from wild-harvested Scutellaria species from Uzbekistan also contained linalool, (E)-β-caryophyllene, thymol, and germacrene D, 42 similar to our findings of these volatile components in the essential oils of Alabama-grown Scutellaria.
The aromatic terpenoids of herbs and flowers have a long history of aromatherapeutic and internal uses as teas and decoctions to treat an extremely wide range of disease states. Terpenes have been shown to be effective antibacterial, antifungal, anti-viral, anti-inflammatory, neuro-protectives, anti-tumor and anti-proliferative as well as uses for dental and dermatological treatments and other uses 43 including bacteriostatic surface cleaners 44 and pesticides, 45 in the case of thymol. Interestingly, 1-octen-3-ol, the most prevalent aromatic compound in all except one out of these four essential oils (S. baicalensis) is known for being a chemical feeding attractant for certain varieties of mosquito while acting as a repellant to other strains of mosquito. 46 It is also known as the “mushroom acid,” and is the most prevalent aromatic compound in most mushrooms (including truffles), molds, and fungi and is the primary olfactory irritant in rotting wood. Over-exposure to this aromatic compound has been shown to cause conjunctivitis, rhinitis, and respiratory inflammation. 47 This compound is also found throughout nature, in raspberries, elder flowers, in the molded rind of Camembert cheese, in certain unwanted wine-molds, and is expressed as a pheromone by certain millipedes during distress. 48 The two enantiomers of this compound are known to have different aromas and may exhibit differing effects when it comes to inflammatory response. Furthermore, 1-octen-3-ol has shown antibacterial and antifungal activities. 49
In this case it is significant to note the finding of a significant percentage of phytol in both S. lateriflora and S. incana, the American herbs, but not the Asian species. Phytol has shown promise for controlling the neglected tropical disease schistosomiasis 50,51 as well as showing antibacterial properties against Staphylococcus aureus 52 and Mycobacterium tuberculosis. 53 Phytol has also been shown to be an anti-allergen and when applied topically in murine studies. 54 Antioxidant and antinociceptive properties of phytol have also been observed. 55
A high percentage (>5%) of (E)-nerolidol and linalool was unique to S. incana. Studies of Greek varieties of Scutellaria also showed linalool and nerolidol to be the main constituents of the essential oil and testing showed moderate antimicrobial activity. 56 (E)-Nerolidol has shown low toxicology with unspecified increased respiratory function as well as cytotoxic effects again hepatic carcinomas. 57 Plant essential oils with high percentages of nerolidol have been found to kill Schistosoma 58 and have antileishmanial activity. 59 (E)-Nerolidol has shown promise as an antifungal in guinea pig testing by healing fungal skin lesions, 60 and as an antiulcer agent as well. 61 Linalool has been experimentally shown to be antimicrobial, cytotoxic, antioxidant, anti-inflammatory, and have insect repellant properties while showing almost no toxicity or allergic response and near zero skin irritant traits. 62
Linalool also has been shown in-vivo to have significant sedative 63 and anti-convulsant effects 64 and is one of the main aromatic compounds in lavender essential oils. 65 Linalool has been found to be a potent acetylcholinesterase inhibitor and has potential as an Alzheimer’s therapeutic essential oil. 66
The native Asian species showed relatively high percentages of carvacrol and thymol where the native American species did not. The largest percentage of any single compound in the essential oils examined herein was the presence of germacrene D at 39% in S. baicalensis. Scutellaria baicalensis essential oil from the aerial parts in this study was rich in (+)-germacrene D (39.3%), (+)-(E)-β-caryophyllene (11.6%), carvacrol (9.3%), thymol (7.5%), and 1-octen-3-ol (6.1%). A headspace volatile analysis of S. baicalensis flowers showed the floral volatiles to be dominated by sesquiterpene hydrocarbons, particularly (E)-β-caryophyllene (22.3 to 41.5%) and germacrene D (12.4 to 27.5%), but carvacrol, thymol, or 1-octen-3-ol were not reported. 67 The root essential oil of S. baicalensis was reported, but the constituents were not quantified. 68 Based on the gas chromatogram, the root oil had acetophenone, (E)−4-phenyl-2-butanone, palmitic acid, and oleic acid as major components; (E)-β-caryophyllene was detected, but in low concentration; and neither carvacrol nor thymol were detected. The analgesic and anti-inflammatory properties of (E)-β-Caryophyllene have been shown experimentally 69 -74 . Both thymol and carvacrol has shown notable antimicrobial activities against pathogenic microorganisms 75 -78 and thymol has shown a wide array of pharmacological properties. 79 The high concentrations of these compounds may contribute to the biological activities and traditional uses of S. baicalensis.
As far as we are aware, this study is the first to report the essential oil composition of S. incana. The aerial parts essential oil was rich in 1-octen-3-ol (31.2%), linalool (6.8%), (E)-nerolidol (10.5%), and phytol (19.7%). The (–)-enantiomers predominated for both linalool (63%) and (E)-nerolidol (96%). The essential oil of S. lateriflora cultivated in Alabama was also rich in 1-octen-3-ol (27.5%) and phytol (14.8%) in addition to (E)-β-caryophyllene (8.8%). The chemical composition of S. lateriflora cultivated in Alabama is in marked contrast to the composition reported for S. lateriflora from Iran, which was dominated by sesquiterpenes (78.3%) including τ-cadinene (27.0%), calamenene (15.2%), and β-elemene (9.2%). 80 Scutellaria lateriflora has been shown in rat experiments to have anticonvulsant capabilities in its flavone component extract, 81 and linalool, found in all four of these Scutellaria species, has been shown to be a sedative and anticonvulsant in murine experiments, 63,64 thus supporting and explaining the pharmacology of the traditional use of this plant for such purposes.
Conclusions
This horticulture, distillation, GC-MS, GC-FID, and chirality analysis of the popular Asian medicinals, S. barbata and S. baicalensis, as well as the native Scutellaria species to Alabama and the southeastern United States, S. incana and S. lateriflora, aims to better characterize the expected volatile composition of these Scutellaria species. With the ability to compare the terpenoid distributions one can make a more well-informed decision as to the true identity of commercially available essential oils. This will also help users of phytotherapies to discern the relevance of the traditional Asian medicine and Native American medicinal claims for the uses of these herbs for inhalation therapies, baths, and teas which can, in certain instances, be supported simply by the appearance of certain aromatic terpenes like thymol, linalool, nerolidol, and β-caryophyllene, with their experimentally supported wide-ranging medicinal uses.
Experimental
Plant Material
Scutellaria baicalensis seeds (www.Amazon.com—Outsidepride seed store), Scutellaria barbata seeds (Plants with a Purpose —www.plantswithapurpose.net), Scutellaria incana seedlings (Prairie Moon Nursery—www.prairiemoon.com), and Scutellaria lateriflora seeds (Palm Beach Medicinals—www.palmbeachherbs.com) were grown by Kirkland Gardens LLC., in Newville, Alabama. The seeds were germinated in trays of organic peat moss outdoors in March 2020, then when approximately six inches tall they, along with the S. incana seedlings, were transplanted to a shady location in the garden under a tree with partial sunlight. The plants were cultivated either in one- or two-gallon black nursery pots with a mix of peat moss, perlite, and cow manure growing medium or in the ground with the same amendments/growing media. The plants were fertilized with chicken manure, bone meal, and kelp meal during transplanting and then watered regularly with well water by drip emitter system. Plants were harvested mid-flowering period, which was the week of June 7, 2020. A second harvesting of S. barbata occurred July 7, 2020. The aerial parts of the plants were photographed then cut at ground level and identifications verified with comparison to online plant databases 24,26 -32 and S. incana and S. lateriflora were positively identified by Kirk Lawson using the Manual of the Vascular Flora of the Carolinas. 82 The plant material was allowed to dry indoors in an air-conditioned area at approximately 23 °C for 10 days. Then, the dried aerial parts were vacuum-sealed and shipped to the University of Alabama in Huntsville. Over the weeks of July 24–August 26, 2020, they were and hydro-distilled with a Likens-Nickerson apparatus with continuous extraction with dichloromethane (DCM) for approximately 4 hours each. The DCM extracts were collected, and the small amount of solvent evaporated under a flow hood. The essential oils (EO’s) were then stored at –20 °C until further analysis.
Photographs of the plants during flowering and before collection were taken by the author, Kirk Lawson (Figures 1 -4). These photographs are stored for reference on multiple hard drives including all of the authors of this manuscript and the digital herbarium at the University of Alabama in Huntsville.

Scutellaria baicalensis photographed at Kirkland Gardens just before harvest. Photograph by S. K. Lawson, June 2020.

Scutellaria barbata, photographed in May, 2020, by Kirk Lawson.

Scutellaria incana. Photograph taken by S. K. Lawson immediately prior to harvest in June 2020.

Scutellaria lateriflora. Photographed by S. K. Lawson immediately prior to harvest in June 2020.
Gas Chromatography
The essential oils were analyzed by gas chromatography–mass spectrometry (GC-MS), gas chromatography with flame ionization detection (GC-FID), and chiral GC-MS as previously reported. 83 For GC-MS the Shimadzu GCMS-QP2010 Ultra was utilized, with a ZB-5ms GC column, GC oven temperature of 50 °C – 260 °C (2 °C/min), 1 µL injection of 5% solution of EO in dichloromethane (split mode, 30:1). The retention indices (RIs) were determined by using a series of reference n-alkanes. The compounds listed in Table 2 were identified by comparing the mass spectrometry data and retention indices with those in the databases. 84 -87 Gas Chromatography–Flame Ionization Detection was conducted using a Shimadzu GC 2010 with FID detector, ZB-5 GC column, GC oven temperature 50 °C– 260 °C (2 °C/min). The percent compositions were determined from raw peak areas without standardization. Chiral Gas Chromatography–Mass Spectrometry was carried out with a Shimadzu GCMS-QP2010S. A Restek B-Dex 325 column was utilized with an initial GC oven temperature of 50 °C, which was increased gradually to 120 °C at a rate of 1.5 °C/minute. Then the temperature was increased to 200 °C at the rate of 2.0 °C/minute. Exactly 0.1 µL injection of 5% solution of essential oil samples in dichloromethane (split mode, 45:1) was injected. The enantiomeric distributions were determined by comparison of retention times with authentic samples obtained from Sigma-Aldrich (Milwaukee, WI, USA). The relative enantiomer percentages were calculated from peak areas.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
