Abstract
Background
Anthriscus caucalis is a non-native plant in North America with invasive potential. As part of our interests in essential oils of the Great Basin, a wild-growing sample of A. caucalis was collected in southwestern Idaho.
Methods
The essential of A. caucalis was obtained by hydrodistillation and analyzed by gas chromatographic methods (GC–MS, GC–FID, and chiral GC–MS).
Results
The major components in the essential oil were cis-chrysanthenyl acetate (42.3%), myrcene (20.4%), cis-chrysanthenol (6.7%), α-pinene (4.2%), and (E)-β-farnesene (4.2%). (+)-α-Thujene, (+)-α-pinene, (+)-sabinene, (+)-limonene, (−)-β-phellandrene, (−)-bornyl acetate, (−)-(E)-β-caryophyllene, and (−)-germacrene D were the predominant enantiomers in the essential oil.
Conclusion
The essential oil composition of A. caucalis from Idaho is similar to those from Europe, but markedly different from A. caucalis from China. The enantiomeric distributions are comparable to those reported in other essential oils of the Apiaceae.
Introduction
Anthriscus caucalis M. Bieb. (Apiaceae), bur chervil, is native to Europe, the Mediterranean region, northwest Africa and, apparently, northeastern Argentina, but has been introduced and naturalized into North America, Chile, Kirgizstan, Tajikistan, Japan, Korea, New Zealand, and Tasmania. 1 The plant was introduced to the Snake River grasslands by 1965. 2 The leaves of A. caucalis are triangular in shape, pinnately dissected, beset with little bristles. The inflorescences are umbels of flowers with 3–5 white rays (Figure 1). There is a high potential for invasiveness of A. caucalis where it has been introduced outside its natural range.2‐5 The purpose of this study is to examine the essential oil composition of A. caucalis growing in the wild in the Great Basin region of Idaho.

Anthriscus caucalis M. Bieb. (Apiaceae) from southwestern Idaho. Photograph by K. Swor.
Results and Discussion
The colorless aerial parts essential oil of A. caucalis was obtained in 1.414% yield by hydrodistillation. The essential oil was analyzed by GC‐MS and GC‐FID (Table 1). A total of 51 compounds were identified, accounting for 98.8% of the total composition. The major components in the essential oil were cis-chrysanthenyl acetate (42.3%), myrcene (20.4%), cis-chrysanthenol (6.7%), α-pinene (4.2%), and (E)-β-farnesene (4.2%).
Chemical Composition (%) of the Aerial Parts Essential Oil of Anthriscus caucalis from Southwestern Idaho.

The stem, leaf, and fruit essential oils of A. caucalis from two locations in Vienna, Austria, have been reported. 10 The compositions of the Austrian samples are comparable to the Idaho sample. cis-Chrysanthenyl acetate dominated the tissues of the Austrian samples (52.8‐73.6%), followed by cis-chrysanthenol (2.1‐16.1%), (E)-β-farnesene (1.6‐9.7%), dodecanal (0.9‐5.2%), myrcene (0.0‐5.0%), and α-pinene (0.0‐3.7%). Interestingly, the compositions of the essential oil samples from Idaho and Austria essential oils are very different from a sample from Guizhou province, China. 11 The sample from China was devoid of cis-chrysanthenyl acetate, cis-chrysanthenol, dodecanal, myrcene, and α-pinene, and (E)-β-farnesene was found in a small amount (0.5%). In contrast, sesquiterpene hydrocarbons dominated the A. caucalis essential oil from China with β-bisabolene (28.4%), germacrene D (18.9%), (Z,E)-α-farnesene (16.7%), and γ-muurolene (7.3%). While germacrene D was observed in the essential oils from Idaho and Austria, neither β-bisabolene, (Z,E)-α-farnesene, nor γ-muurolene were detected. (E,E)-α-Farnesene, however, was found in both the essential oil samples from Idaho and Austria. The Flora of China lists only one species of Anthriscus in China (Anthriscus sylvestris (L.) Hoffm.). 12 There have been reports on the essential oil composition of A. sylvestris from the Netherlands, 13 Türkiye, 14 and Iran, 15 and the chemical compositions of these essential oils are also very different from the A. caucalis essential oil from China. It is difficult to rationalize the very different compositions of A. caucalis essential oil of the Idaho and Austria samples compared to the China sample. It may be that they are very different chemotypes.
The enantiomeric distributions of chiral terpenoid compounds in A. caucalis essential oil have been determined using chiral GC‐MS (Table 2). The chiral gas chromatogram is available as Supplemental material (Supplementary Figure S1). The dextrorotatory enantiomers were dominant for α-thujene, α-pinene, camphene, sabinene, and limonene, whereas the levorotatory enantiomers were the exclusive stereoisomers for β-phellandrene, bornyl acetate, (E)-β-caryophyllene, and germacrene D.
Enantiomeric Distributions (% Enantiomer) of Chiral Terpenoid Components in the Aerial Parts Essential Oil of Anthriscus caucalis from Southwestern Idaho.
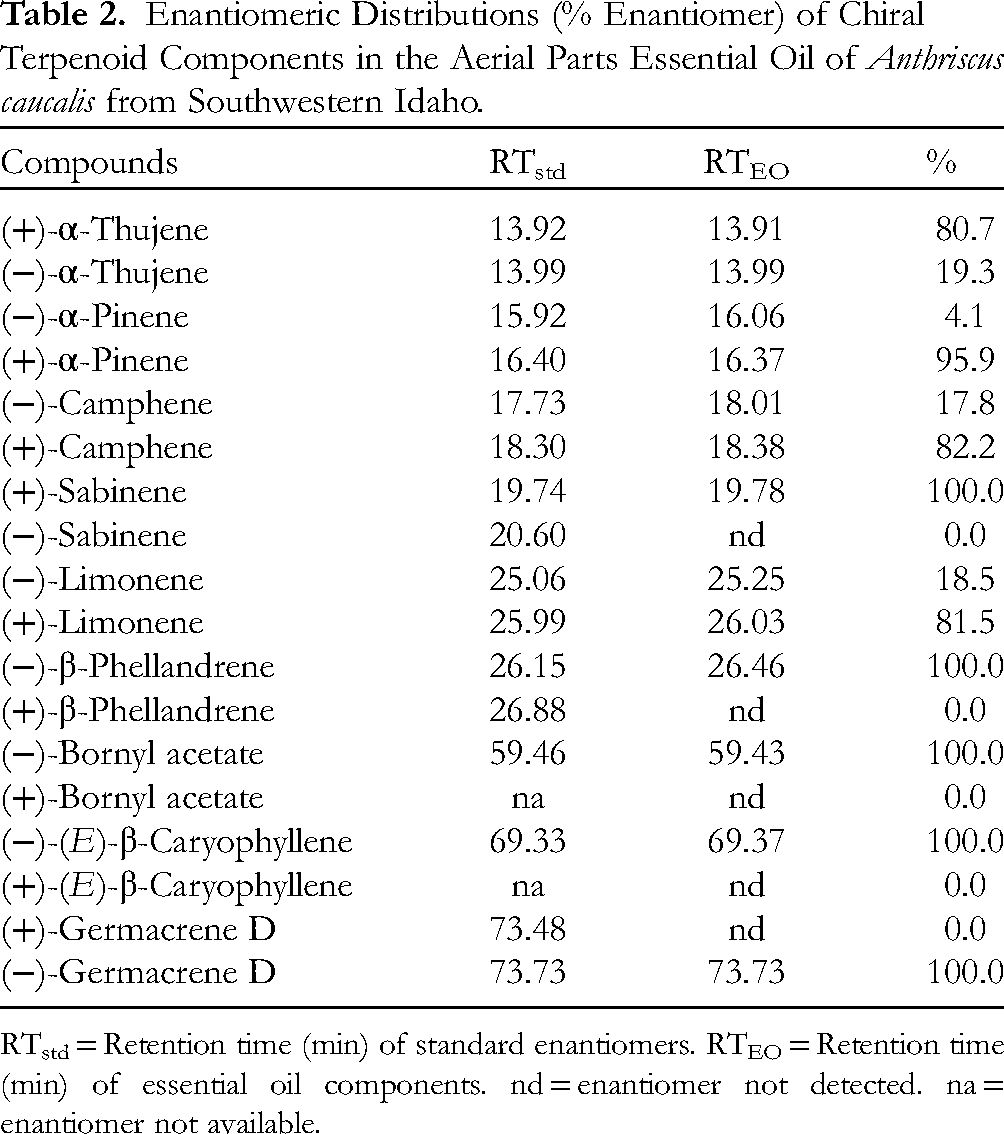
RTstd = Retention time (min) of standard enantiomers. RTEO = Retention time (min) of essential oil components. nd = enantiomer not detected. na = enantiomer not available.
The enantiomeric distributions observed for A. caucalis in this study are largely consistent with enantiomeric distributions previously reported for other essential oils of the Apiaceae. Kapetanos and co-authors have carried out a broad study of several members of the Apiaceae from the Central Balkan Peninsula (Aegopodium podagraria L., Bupleurum praealtum L., Bupleurum sibthorpianum S. S. var. diversifolium (Roch.) Hay, Chaerophyllum aureum L., Chaerophyllum hirsutum L., Chaerophyllum temulum L., Cnidium silaifolium (Jacq.) Simk. ssp. orientale (Boiss.) Tutin, Laser trilobum (L.) Brokh., Laserpitium siler L., Libanotis montana Cr., Orlaya grandiflora (L.) Hoffm., Pastinaca hirsuta Pancic., Pastinaca sativa L., Peucedanum alsaticum L., Peucedanum austriacum (Jacq.) Koch, Peucedanum cervaria (L.) Cuss., Peucedanum longifolium W. et K., Peucedanum officinale L., Peucedanum oreoselinum (L.) Moench, Physospermum cornubiense (L.) DC., Pimpinella serbica (Vis.) Bentham & Hooker, Tordylium maximum L., and Torilis anthriscus (L.) Gmel.), including chiral GC‐MS. 16 These workers found (+)-α-pinene and (+)-sabinene to be the predominant enantiomers in most samples, except for P. alsaticum, P. austriacum, P. cervaria, P. longifolium, P. officinale, and P. oreoselinum; limone enantiomeric distributions varied widely; borneol and bornyl acetate, when observed, were exclusively the (−)-enantiomer. Interestingly, (+)-(E)-β-caryophyllene was the exclusive enantiomer observed in 15 essential oils, while (−)-(E)-β-caryophyllene was the exclusive enantiomer in 8 samples. According to the Dictionary of Natural Products, (−)-(E)-β-caryophyllene is the common enantiomer in higher plants, while (+)-(E)-β-caryophyllene is observed in liverworts. 17
An investigation of Anethum graveolens L., Apium graveolens L., Carum carvi L., Cuminum cyminum L., Petroselinum crispum (Mill.) Fuss, and Trachyspermum ammi Sprague essential oils revealed that, when observed, (+)-α-pinene and (+)-limonene were the exclusive enantiomers. 18 The enantiomeric distributions of α-pinene and sabinene in Coriandrum sativum L. fruit and herb were dominated by the (+)-enantiomers, (−)-borneol was the exclusive enantiomer observed, and limonene showed a slight excess of (+)-limonene over (−)-limonene. 19
Conclusions
This is the first report on the chemical characterization of the essential oil of Anthriscus caucalis growing wild in Idaho. The composition is qualitatively similar to that reported from Austria, but profoundly different from a sample from China. The enantiomeric profiles of chiral terpenoid components of A. caucalis essential oil, reported herein for the first time, are comparable to other members of the Apiaceae.
Materials and Methods
Plant Material
Aerial parts of A. caucalis were collected from a single flowering individual on 16 May 2022 near Lucky Peak Lake (43°31ʹ40ʺ N, 116°2ʹ51ʺ W, 945 m elevation). The plant was identified by W.N. Setzer by comparison with specimens in the New York Botanical Garden. 20 A voucher specimen (WNS-Ac-5324) has been deposited in the University of Alabama in Huntsville herbarium. The fresh aerial parts of A. caucalis (223.5 g) were hydrodistilled for 3 h using a Likens-Nickerson apparatus to give a colorless essential oil (3.161 g).
Gas Chromatographic Analysis
The A. caucalis essential oil was analyzed by GC‐MS, GC‐FID, and chiral GC‐MS as previously described. 21 Retention indices were calculated based on a homologous series of n-alkanes on a ZB-5 ms column. 22 Each essential oil component was determined by comparison of its retention index and its mass spectral fragmentation pattern (>85% reliability index), which were available in the databases.6‐9 Enantiomers of terpenoid components were identified by comparison of retention times with standards (Sigma-Aldrich, St. Louis, MO, USA); the enantiomer percentages were calculated based on peak areas.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231187699 - Supplemental material for The Essential Oil Composition of Anthriscus caucalis M. Bieb. (Apiaceae) Growing Wild in Southwestern Idaho
Supplemental material, sj-docx-1-npx-10.1177_1934578X231187699 for The Essential Oil Composition of Anthriscus caucalis M. Bieb. (Apiaceae) Growing Wild in Southwestern Idaho by Kathy Swor, Prabodh Satyal and William N Setzer in Natural Product Communications
Footnotes
Acknowledgements
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
