Abstract
Objectives
In the present study, we explored the antiarthritic potential of Eupatilin on complete Freund's adjuvant (CFA)-induced arthritis rats.
Materials and Methods
In order to induce arthritis in the Sprague-Dawley rats, CFA vaccination was used, and Eupatilin was given to the rats at dose of 5, 10, and 20 mg/kg. Various biochemical and other parameters were studied to define the pharmacological benefit of Eupatilin.
Results
The study's findings revealed that Eupatilin increases rats’ body weight and significantly reduces hind paw volume. It also improves the antioxidant status of CFA rats by lowering malondialdehyde levels and increasing the concentration of endogenous antioxidants superoxide dismutase and GPx. Eupatilin also improved hematological markers, which were demonstrated to be altered in CFA rats as compared to the control. In the Eupatilin-treated group, the level of pro-inflammatory cytokines was found to be reduced as compared to CFA rats. In a molecular docking analysis, it showed favorable interaction with the active site of cyclooxygenase-2 (COX-2) by the formation of numerous nonbonded interactions by interacting with Cys32, Leu138, Pro 139, Val141, Ala142, and Cys145. It also showed a Vina score of −9.4 and interacts with the cavity volume of 4435 Å3.
Conclusion
Eupatilin showed favorable antiarthritic benefits in the CFA-induced arthritis model, as evidenced by reductions in paw volume, significant weight loss prevention, and maintenance of the healthy hematological and biochemical profile may be through inhibiting COX-2.
Introduction
The symptoms of rheumatoid arthritis (RA), a chronic, systemic inflammatory disease, include joint swelling, discomfort, and the deterioration of bone and cartilage. 1 The disorder might become more severe and result in a considerable disability, which would reduce life expectancy and raise mortality.2,3 Antirheumatic drugs, nonsteroidal anti-inflammatory medicines, and corticosteroids make up the majority of the current therapeutic care for RA.1,4 These drugs offer symptomatic relief and are often associated with serious adverse effects. Therefore, the search for an effective agent to combat RA is worthwhile.
Due to their multifaceted pharmacological effect, natural compounds including curcumin, 5 resveratrol, 6 gingerol, 7 corynoline, 8 rhoifolin, 9 and others have shown substantial benefits against RA. These compounds showed potent biological activity against numerous human diseases—including cancer, diabetes, cardiovascular disorders, and infectious and viral diseases. The class of natural chemicals known as flavonoids and isoflavonoids has earned a prominent role in the drug discovery process. 10 However, due to their altered pharmacokinetics, their pharmacological activity was frequently compromised. 11 The bioavailability of flavonoids is typically poor and varies greatly between flavonoid classes and even within a class between individual molecules. The bioavailability of flavonoids is affected by Phase 2 metabolism, which frequently occurs in humans. Methylation of these flavonoids has been shown to boost their potential as pharmacological agents, which could lead to new uses. 12 The metabolic stability and membrane transport of flavonoids is greatly improved through the methylation of their free hydroxyl groups or C atom, resulting in greatly improved oral bioavailability.
Eupatilin is one of the O-methylated flavonols that have been identified from the plant genus Ipomopsis aggregate. 13 Previous research has shown that it has a strong anticancer14,15 and an antiasthmatic effect. 16 Since no previous research had explored the pharmacological importance of Eupatilin against RA, the current study sought to discover whether or not Eupatilin had an antiarthritic effect on Complete Freund’s adjuvant (CFA)-induced arthritis in Sprague-Dawley (SD) rats.
Material and Methods
Animals
Sprague-Dawley rats were obtained from the institutional animal home and kept in a clean environment at room temperature and with unlimited access to food and water before the experiment.
Induction of arthritis in rats using CFA and sample collection
The 1 mL of CFA contains 1 mg of Mycobacterium tuberculosis (H37Ra, ATCC 25177), heat killed and dried, 0.85 mL paraffin oil and 0.15 mL mannide monooleate (Sigma Aldrich). It was subcutaneously administered to SD rats (0.1 mL) into the footpad of the left hind paw. The rats were further administered 0.1 mL of CFA on the same day, and the next day into the tail for a booster dose. The treatment group was administered Eupatilin (≥98%, Sigma Aldrich) at various dosages orally as a suspension with 10% carboxy methylcellulose as the suspending agent. The administration was done using the oral gavage technique with a flexible plastic feeding tube. On day 28nd, after the research protocol was accomplished, the rats were anesthetized with an intraperitoneal injection of 80 mg/kg ketamine and 10 mg/kg xylazine. The blood samples collected from the retro-orbital plexux and subjected to centrifugation at 3000 rpm for 10 min at 4°C for serum harvesting for biochemical analysis. The rats were sacrificed by cervical dislocation to harvest tissue from the knee joints and preparation homogenate for further biochemical estimations. 17
Treatment group
The groups will be as follows:
8
Group 1: Control (Normal Saline for 28 days) Group 2: CFA (administered with CFA with no treatment) Group 3: Eupatilin (5 mg/kg for 28 days) Group 4: Eupatilin (10 mg/kg for 28 days) Group 5: Eupatilin (20 mg/kg for 28 days)
The subsequent treatment group also receives CFA for the induction of arthritis prior to the administration of Eupatilin as shown above.
Determination of hind paw volume
The progression of the hind paw volume (HPV) in SD rats to investigate the severity of the arthritis condition was investigated using the plethysmometer at 0, 7, 14, 21, and 28 days after induction.
Preparation of Tissue Homogenate
The knee joint tissues of rats were rinsed in ice-cold normal saline and homogenized in Tris-HCl buffer (0.01 M, pH 7.4) at 10,000 rpm at 4°C for 5 min using an electric homogenizer (Ningbo Scientz Biotechnology Co., Ltd) to obtain 10% homogenates. The homogenates were subsequently centrifuged at 10,000 × g at 4°C for 10 min, and the resultant supernatants were collected for further biochemical estimations.
ELISA analysis
The serum levels of tumor necrosis factor alpha (TNF-α) (cat. no. DTA00D, R&D Systems, Inc.), interleukin (IL)-1β (cat. no. RLB00; R&D Systems, Inc.), IL-8 (cat. no. RA20553; Bioswamp; Wuhan Bienle Biotechnology Co., Ltd), CRP (cat. no. SU-B30078, Quanzhou Ruixin Biotechnology Co., Ltd), and anti-CCP antibody (cat. no. B37304, Quanzhou Ruixin Biotechnology Co., Ltd) were determined using commercially purchased ELISA kits to determine the concentration of each cytokine. In total, a 100-µL aliquot of the supernatant from each well was collected for measurement. The concentration of each cytokine in the supernatant was standardized to the cell protein concentration in the respective well. The absorbance was measured at 450 nm using a microplate reader (Bio-Rad Laboratories, Inc.).
Estimation of superoxide dismutase and malondialdehyde
Superoxide dismutase (SOD) and malondialdehyde (MDA) levels in joint tissues were measured using specific kits from different manufacturers: a SOD kit from Cayman Chemicals, USA (cat no. 706002) and an MDA kit from Nanjing Jiancheng Bioengineering Institute, China (cat no. A003). The joint tissue was weighed, placed in physiological saline, and homogenized with a vortex mixer. The supernatants were collected by centrifuging at 4000 revolutions per minute for 5 min. The SOD activity was measured using the abovementioned assay kit as per the manufacturer's procedure. This method uses a tetrazolium salt to detect superoxide radicals generated by xanthine oxidase and hypoxanthine. The assay measures the 3 types of SOD (Cu/Zn, Mn, and FeSOD). The SOD activity values are expressed as unit U/mg protein. One unit of SOD is defined as the amount of enzyme needed to exhibit 50% dismutation of the superoxide radical. Moreover, the concentration of MDA was determined using the thiobarbituric acid technique as per the procedure obtained with above mentioned kit. During the measurement process, a standard tube, standard blank tube, and measuring tube were utilized. A 4 mL of the working solution was added to each of the 3 tubes. Additionally, 100 μL of the standard sample was added to the standard tube. Simultaneously, 100 μL of anhydrous alcohol was put to the standard blank tube, and 100 μL of the sample was introduced to the measurement tube. Each tube's mixed solution was homogenized using a vortex mixer and then incubated for 80 min at a water temperature of 95 °C. The samples were cooled using running water and then centrifuged at 3500 RPM for 10 min. A 200 μL of supernatant was collected from each tube. The absorbance of each sample was determined with a microplate reader at a wavelength of 532 nm. The calculation of each sample was done using the conversion formula.18,19
Estimation of glutathione
GSH (Glutathione) was assessed according to the procedure of the GSH assay kit (cat. No. 703002, Cayman Chemicals). In order to assay the concentration of GSH, the reaction between GSH and 5,5′-dithio-bis-2-nitrobenzoic acid (DTNB) was performed. Then, the absorbance (at 405-412 nm) of the sample containing the product of this reaction (5-thio-2-nitrobenzoic acid [TNB]), was measured. The rate of TNB production was directly proportional to the concentration of GSH (nm/mg protein) in the sample.18,19
Docking analysis
The docking of Eupatilin was performed with cyclooxygenase-2 (COX-2) (3ln1.pdb) using the default setting of the CB-Dock tool. CB-Dock is a protein-ligand docking method that automatically identifies the binding sites, calculates the center and size, customizes the docking box size according to the query ligands, and then performs the molecular docking with AutoDock Vina (version 1.1.2). 20
Statistical analysis
The data were analyzed using GraphPad Prism version 6.0. Descriptive statistics were used to present the data as mean ± SEM (standard error of the mean). Behavioral parameters were analyzed using a 2-way analysis of variance (ANOVA) followed by tacchi test Bonferroni's test for post hoc comparisons. Biochemical factors were analyzed using 1-way ANOVA with P < .05 considered statistically significant.
Results
Effect of Eupatilin on HPV
The HPV of the rats used in the CFA-induced arthritis model was initially used to gauge the antiarthritic effects of Eupatilin. The HPV levels in the CFA-treated rats were found to be noticeably greater than those in the control group, as shown in Figure 1A. In contrast, the paw edema brought on by CFA was substantially decreased by Eupatilin at the indicated dosages (5, 10, and 20 mg/kg). This hypothesis suggests that Eupatilin may have had a dose-dependent antiarthritic effect in rats.
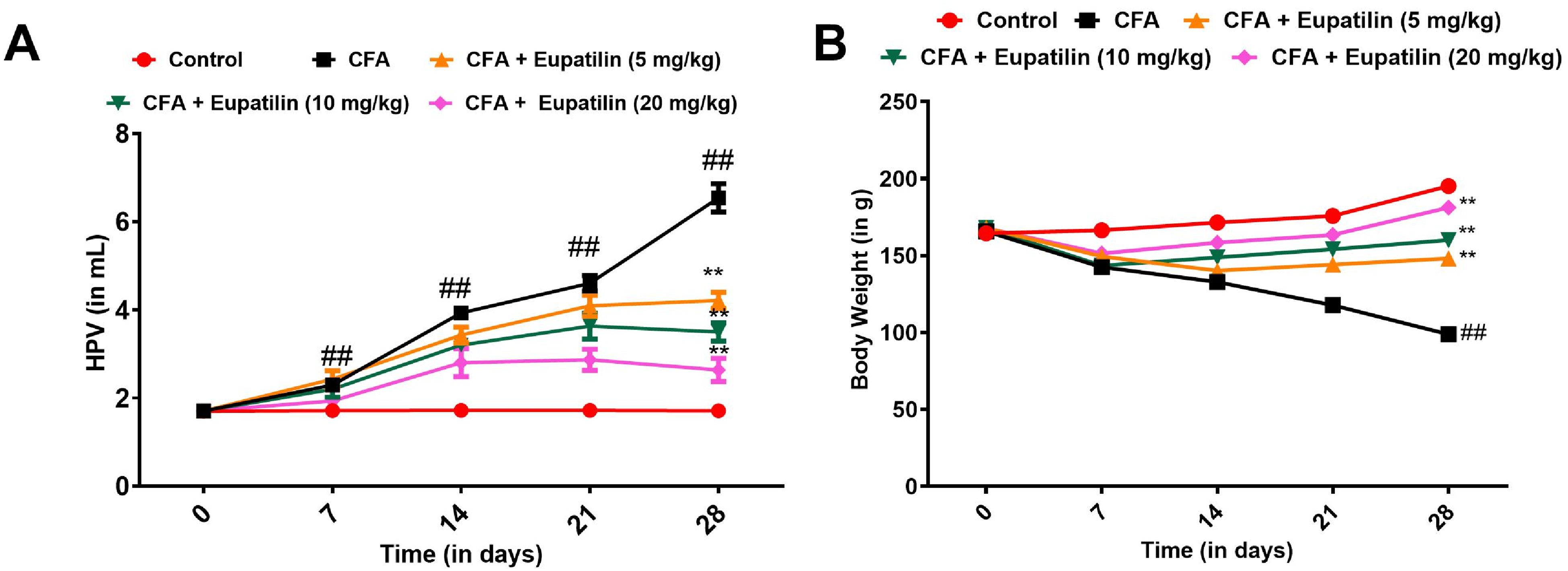
Effect of Eupatilin on the (A) hind paw volume (HPV) and (B) body weight of the Sprague-Dawley (SD) rats. ##P < .05 versus control and **P < .01 versus CFA group. Data are presented as means ± SEM.
Effect of Eupatilin on the Body Weight
To support the preventative effect of Eupatilin against CFA-induced arthritis, the rat's body weight was determined and results were presented in Figure 1B. The weights of the CFA-treated rats dramatically declined by the time the experiment ended. Whereas the body weight in the Eupatilin-treated rats improved dose-dependently, peaking in the 20 mg/kg treatment group. It has been suggested that Eupatilin dramatically decreased arthritis in CFA rats as a result.
Effect of ESG on the Hematological Parameters
As shown in Table 1, CFA-induced arthritis in rats causes significant differences in the hematological parameters as compared to the control. It has been found that CFA-induced rats showed decreased levels of HGB, PCV, and RBC with an increase in WBC and ESR. However, the administration of Eupatilin to these rats causes restoration of these biomarkers near normal. It was speculated that Eupatilin's antiarthritic properties are what caused these parameters to return to normal.
Effect of Eupatilin on the Hematological Parameters of SD Rats.

Abbreviations: CFA, Complete Freund’s adjuvant; SD, Sprague-Dawley.
Effect of Eupatilin on the CRP and CCP Antibody Levels
The most well-known RA diagnostic markers, including CRP and anti-CCP-antibodies level, were assessed from serum samples of all experimental animals in order to further establish the antiarthritic potential of Eupatilin. As shown in Figure 2, results demonstrated that disease-control animals had higher levels of CRP with reduced levels of CCP-antibody. The level of these diagnostic markers was found substantially decreased in all animals administered with 3 different dosages of Eupatilin, in contrast to CFA-arthritis control, as demonstrated.

Effect of Eupatilin on the (A) CRP and (B) CCP antibody levels. #P < .05 versus control and **P < .01 versus CFA group. Data are presented as means ± SEM.
Effect of Eupatilin on the Pro-Inflammatory Cytokines
Next, we sought to determine how Eupatilin affected the levels of numerous pro-inflammatory cytokines (such as TNF-α, IL-1β, and IL-6) present in rats’ blood. According to Figure 3, the CFA rats had noticeably greater levels of the cytokines mentioned above than the control group. However, compared to the arthritis control CFA group, the Eupatilin-treated rats demonstrated a substantial decrease in the blood level of these cytokines. This finding, which may be related to a reduction in cytokines that are pro-inflammatory, highlights the anti-inflammatory effects of Eupatilin in CFA rats.
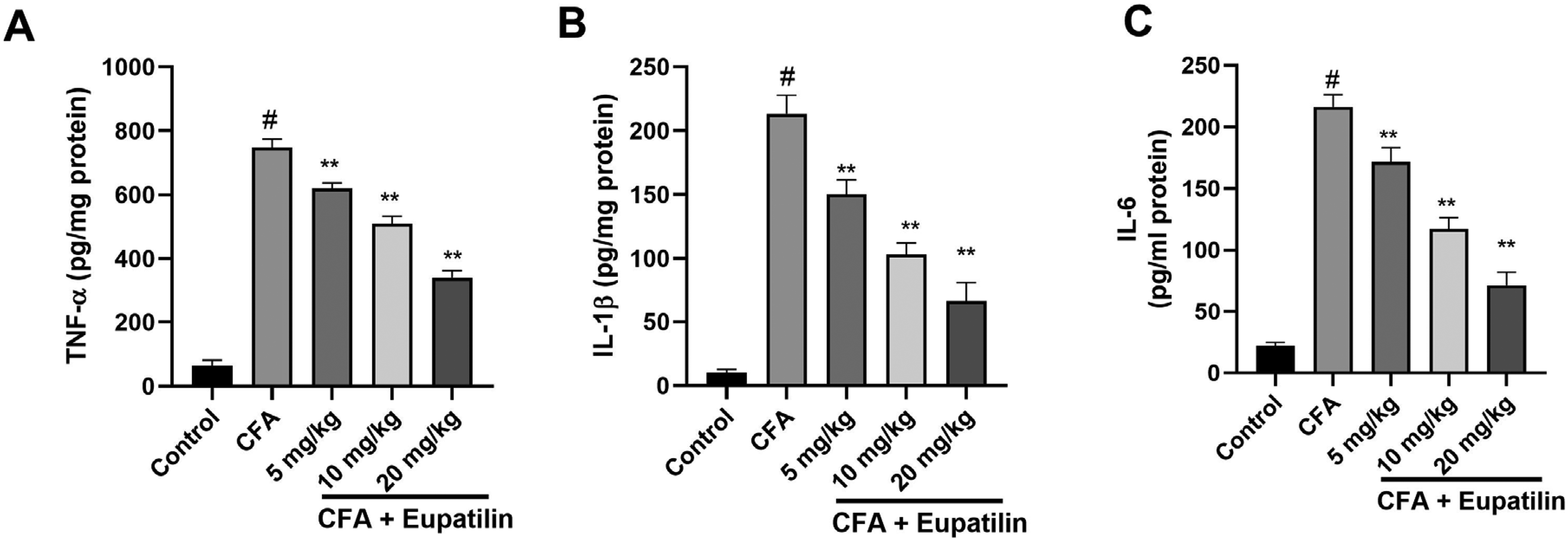
Effect of Eupatilin on the levels of pro-inflammatory cytokines in Sprague-Dawley (SD) rats. #P < .05 versus control and **P < .01 versus CFA group. Data are presented as means ± SEM.
Effect of Eupatilin on the Oxidative Stress status
The effects of Eupatilin on oxidative stress in rats were evaluated using CFA rats, and the findings are shown in Figure 4. The CFA-treated rats had greater levels of MDA and lower levels of GSH and SOD when compared to the control-treated animals. Furthermore, it was shown that after consuming Eupatilin, the level of these examined biomarkers had practically returned to normal, supporting the antioxidant effect of Eupatilin.

Effect of Eupatilin on the oxidative stress biomarkers. #P < .05 versus control and **P < .01 versus CFA group. Data are presented as means ± SEM.
Docking Analysis of Eupatilin With COX-2
The docking analysis as shown in Figures 5 and 6 demonstrated that the Eupatilin was excellently docked into the active site of COX-2. It showed the formation of various H-bonds with Glu451, Gln447, and Asn24. It also showed the formation of numerous nonbonded interactions by interacting with Cys32, Leu138, Pro 139, Val141, Ala142, and Cys145 as shown in Table 2. Thus, it was believed that Eupatilin might exert an anti-inflammatory effect via interacting with COX-2.

3D docked interaction of Eupatilin with cyclooxygenase-2 (COX-2).
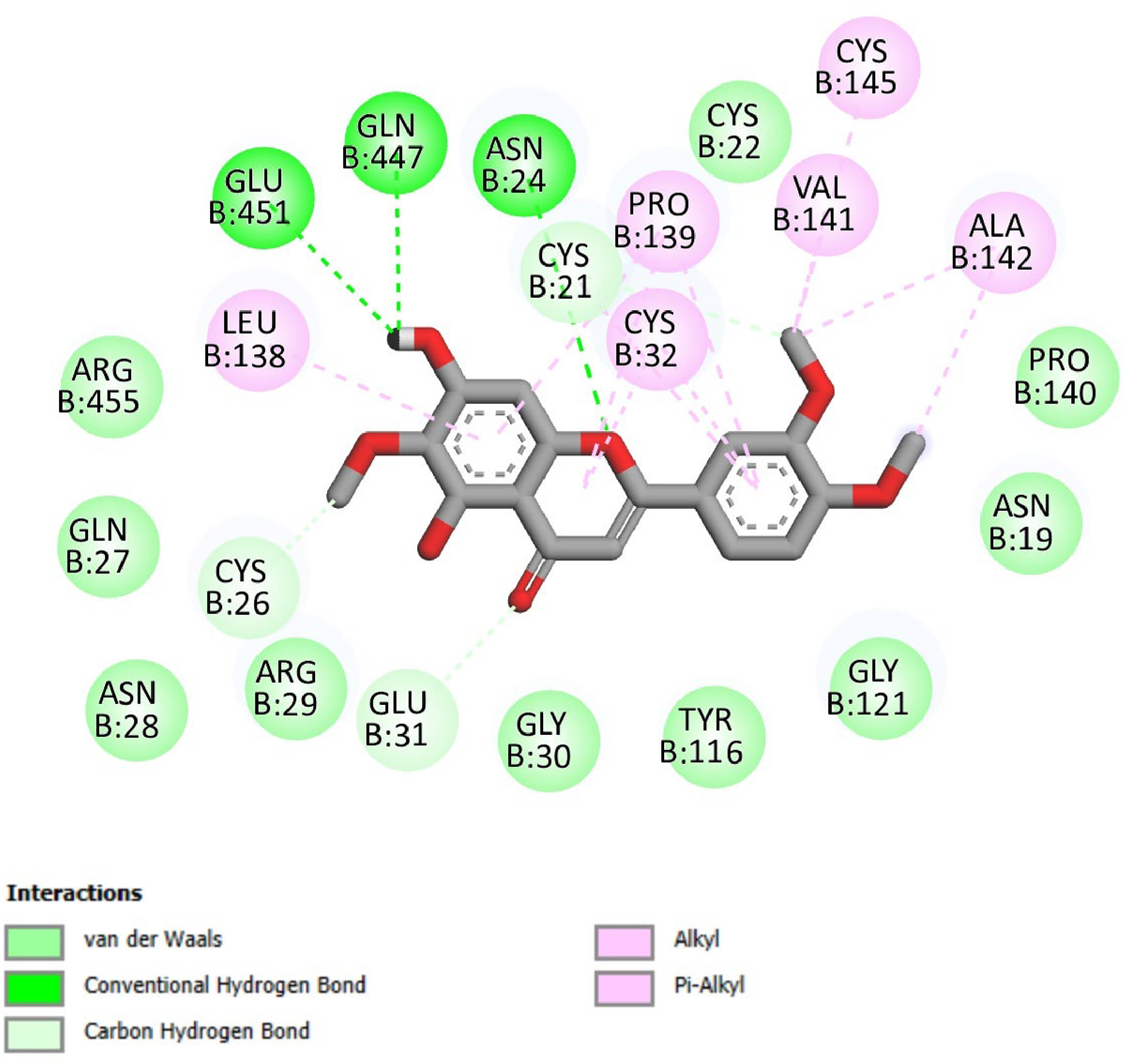
2D docked interaction of Eupatilin with cyclooxygenase-2 (COX-2).
Docking Score of Eupatilin With COX-2.
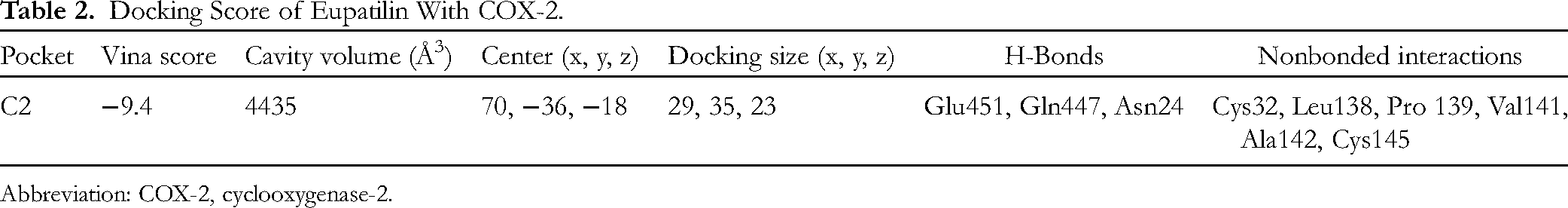
Abbreviation: COX-2, cyclooxygenase-2.
Discussion
Rheumatoid arthritis is a common source of disability that impacts billions of people globally. 21 We must find a more effective medicine that can help a range of people because of the potential negative consequences of present medications. Natural products have gained more attention recently for improving and treating various diseases. 22 The current study examined the effects of Eupatilin on rats with arthritis due to its remarkable anti-inflammatory properties reported in several research investigations.
We used the CFA-induced arthritis rodent model to produce arthritis in rats for our investigation. 23 Because of its close resemblance to the clinical characteristics of human arthritis, such as synovial hyperplasia and cartilage deterioration, this method is widely utilized for studying chronic polyarthritis.
Multiple studies have shown that chronic inflammation in people with RA led to significant weight loss. 24 Rats administered CFA to induce arthritis saw a decrease in body weight according to comparable studies.8,25,26 We examined the impact of varying dosages of Eupatilin on the body weight and inflammation of CFA rats. The study's results suggest that Eupatilin significantly decreased the inflammation associated with CFA, evidenced by a notable reduction in HPV. Moreover, it showed an enhancement in the rats’ body weight. It is hypothesized that Eupatilin reduced inflammation caused by CFA, resulting in an increase in the body weight of rats. The results confirmed that Eupatilin has antiarthritic capabilities against CFA-induced arthritis.
Considering the connection between the inflammatory and immunological aspects of RA with blood disorders such as anemia, neutropenia, and lymphopenia, it is crucial to evaluate how a new treatment could address these issues in developing an innovative RA therapy strategy.27,28 The study demonstrated the impact of Eupatilin on many hematological parameters including HGB, PCV, RBC, WBC, and ESR. Eupatilin effectively restored the evaluated parameters to near-normal levels in a dose-dependent manner, with the highest efficacy shown in rats administered with 20 mg/kg.
Cachexia has been seen as the distinguishing characteristic of RA.29,30 Numerous factors contribute to the development of this RA-related muscle wasting, but it is well-recognized that high TNF-α levels can result in cachexia.31–33 Tumor necrosis factor-α stimulates energy consumption by altering the metabolism of proteins and lipids. This causes a decrease in calorie intake by inducing anorexia, which ends up in cachexia. It plays a significant role in joint degeneration by causing inflammation by acting directly on a variety of tissues. This leads to the induction of other cytokines, including IL-1, IL-8, and IL-6. As a result, blocking TNF-α avoids not only the terrible consequences it causes but also the activity of other inflammatory mediators. 34 Our study demonstrated a significant increase in the levels of TNF-α, IL-1β, and IL-6 in the paw tissues of CFA-rats, as compared to the control. However, the Eupatilin-treated group showed a significant reduction in the level of these cytokines. Therefore, reduction of these elevated pro-inflammatory cytokines in the flared paw region demonstrates that Eupatilin successfully prevents inflammation of joints in CFA SD rats.
The development of several diseases, such as RA, which leads to the breakdown of lipids, proteins, and DNA in joint tissues, has been linked to reactive oxygen species (ROS).35,36 The body's several antioxidant defense mechanisms typically regulate the production of ROS. Past studies have connected inflammation and joint deterioration in arthritic animals and RA patients to oxidative stress. Animals with arthritis have shown higher levels of ROS generation and markers of protein and lipid oxidation. The brain, liver, and vascular tissues of rats with experimental arthritis in the murine model showed significant oxidative stress, as found by researchers. The levels were also discovered to be altered in the serum of patients with RA. 35 In this work, we have examined the impact of Eupatilin on different biomarkers related to oxidative stress. The study revealed that Eupatilin effectively reduces ROS generation in CFA-rats by enhancing their oxidative defense system, leading to elevated SOD and GPx levels and reduced MDA levels.
Patients with RA have higher levels of COX-2 expression in their synovial tissues.37,38 In most tissues, COX-2 expression is tightly controlled and rapidly induced in response to inflammatory stimuli. It has been discovered that the pro-inflammatory cytokines IL-1 and TNF-α are primarily responsible for the elevated COX-2 expression in synovial tissues. These cytokines activate transcription factors including NF-κB and c/EBP, which in turn drive COX-2 transcription.
Molecular docking has emerged as a crucial part of computer-aided drug discovery. Using this method, we can make atomic-level predictions about how a tiny chemical will interact with a protein. 39 This allows scientists to investigate the biochemical process underlying the interaction between tiny compounds like nutraceuticals and their target proteins. The method relies on structural information about the target protein, which can be collected using x-ray crystallography, NMR spectroscopy, or cryo-electron microscopy. In a previous study, Min et al showed significant inhibitory effect on the on carrageenan-induced inflammation in mice via inhibition of COX-2. 40 In addition, it remarkably inhibits LPS-induced expression of COX-2 in J774A.1 cells in a concentration-dependent manner. 41 However, none of these studies have enumerated the interaction behavior of Eupatlin against COX-2. Thus, to further confirm the Eupatilins's ability to interact with COX-2, a molecular docking analysis was conducted using CBDock, a web-based platform. The present docking experiment tries to elucidate the nature of interaction and information on the interacting residues and bonding pattern for Eupatlin against COX-2. Results of the study suggested that Eupatilin was found excellently docked into the active site of COX-2 by interacting with Glu451, Gln447, Asn24, Cys32, Leu138, Pro 139, Val141, Ala142, and Cys145 via the formation of H-bonds and Pi-Pi-interactions. The interactions shown by Eupatilin was discovered to be analogous to the interaction induced by celecoxib in the active site of COX-2. 42 Thus, it was suggested that, Eupatilin was proposed to interact with COX-2 in a manner similar to celecoxib by binding to the same active site.
Moreover, it would be prudent to assess the efficacy of Eupatilin on a genetically induced arthritis model in addition to the CFA-induced arthritis model, since the former somewhat resembles RA and serves as the study's primary limitation. Another significant long-term goal is the discovery of the safest and most effective human doses of Eupatilin and the creation of targeted therapies for RA.
Conclusion
The aforementioned data suggest that Eupatilin may have shown potential antiarthritic properties. It has a strong and dose-dependent antiarthritic impact by suppressing both inflammation and oxidative stress. This effect may be attributed to COX-2 inhibition. Considering all, it can be said that Eupatilin may qualify for RA therapy. To get insight into this medication's precise mechanism of action, further study through mechanistic screening must be done.
Footnotes
Authors’ Contributions
LW: data processing, collection, perform experiment, analysis and interpretation of results, draft manuscript preparation, and visualization. JL: data processing, collection, performing experiment, analysis, and interpretation of results; HLZ: analysis and interpretation of results, draft manuscript preparation, visualization; GC: study conception, critical revision or editing of the article, supervision, and funding acquisition. All authors approved the final version to be published.
Authors’ Note
Data Availability: The datasets used and/or analyzed during the present study are available from the corresponding author upon reasonable request. Ethical Approval: The study has been approved by the Institutional Animal Ethical Board for Biomedical Experiments of the Cangzhou Hospital of Integrated Traditional Chinese and Western Medicine (SOP/001/01.0).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Thankful to the Hebei Provincial Department of Science and Technology (162777137).
