Abstract
Aim of the Study
This study investigates the anti-inflammatory effects of
Methods
The anti-inflammatory effect was studied using LPS-induced inflammation in RAW 264.7 macrophages. EEPS extract was prepared using 70% ethanol, and LPS-induced pro-inflammatory factors such as nitric oxide (NO), iNOS, and COX-2 were analyzed using the Griess reagent and immunoblotting. The pro-inflammatory cytokines expressions IL-1β, IL-6, and TNF-α were studied by qPCR. Intracellular ROS and Ca2+ accumulation induced by LPS were assessed using DCF-DA and Fluo-3 AM fluorescent dyes. The LPS-induced NF-κB and MAPK pathways were also studied by immunoblotting. The active compounds present in this EEPS were isolated, and their structures were identified using NMR techniques.
Results
EEPS significantly inhibited these effects. EEPS also reduced LPS-induced expression of pro-inflammatory cytokines IL-1β, IL-6, and TNF-α. Immunoblot data revealed that EEPS suppressed NF-κB activation by preventing the phosphorylation of p65 and IκB-α as well as inhibited the phosphorylation of MAPK proteins ERK, p38, and JNK. Furthermore, EEPS reduced LPS-induced intracellular accumulation of ROS and Ca2+ in RAW 264.7 macrophages. From EEPS, we isolated 7 compounds. Among these, the saponin derivatives exhibited significant anti-inflammatory activity, reducing NO production by 60%∼70% in RAW 264.7 cells compared to LPS-treated cells.
Conclusion
EEPS exhibits strong anti-inflammatory effects in LPS-induced inflammation in RAW 264.7 cells by inhibiting the release of NO, iNOS, and COX-2 and preventing the activation of the NF-κB and MAPK pathways. This study is the first to highlight the anti-inflammatory potential of
Introduction
Inflammation is a common protective immune response induced by immune cells in response to various harmful factors, such as bacterial infections, toxic chemicals, and stressful insults. It serves as a defense mechanism initiated by immune cells to protect and maintain the homeostasis of body cells and tissues. 1 However, prolonged and excessive activation of the inflammatory response can cause more tissue damage than protection and serving as a primary contributor to various diseases, such as arthritis, diabetes, sepsis, allergies, multiple sclerosis, cancers and various neurodegenerative diseases.2–4 Inflammation is a highly coordinated cellular process involving the regulation of multiple signaling pathways, including NF-κB and STAT3 signaling, activation of pro-inflammatory cytokines, and MAPK pathways. 5
The NF-κB signaling pathway plays a major role in activating various pro-inflammatory cytokines and signaling pathways. NF-κB is a heterodimer protein and functions as a transcription factor. Under normal conditions, it remains inactive in the cytoplasm bound to its inhibitor protein, IκB. IκB prevents NF-κB from translocating to the nucleus and is regulated by phosphorylation. During an inflammatory reaction, IκB becomes phosphorylated, leading to its dissociation from NF-κB. 6 Subsequently, NF-κB itself undergoes phosphorylation, translocates to the nucleus, and activates the transcription of pro-inflammatory cytokines such as IL-1, IL-6, and TNF-α, as well as proteins like iNOS and COX-2 proteins.7,8 In parallel, the MAPK signaling pathway is also activated during inflammation. The MAPKs, including ERK, p38, and JNK, are regulated through phosphorylation, further contributing to the inflammatory response.9–11 Furthermore, inflammatory signals can disrupt intracellular calcium homeostasis, leading to elevated calcium concentrations that affect the NF-κB pathway and stimulate the release of inflammatory cytokines, such as TNF-α. 12 Additionally, calcium imbalance induces intracellular oxidative stress, which triggers the activation of various stress signaling pathways, including MAPKs, and may also initiate apoptotic signaling. 13
Macrophages play a key role in chronic and pathological inflammation due to their rapid release of large amounts of pro-inflammatory cytokines in response to inflammatory stimuli. Lipopolysaccharide (LPS), derived from gram-negative bacteria, is a potent trigger of severe inflammatory responses in macrophages.5,10 It induces a rapid inflammatory response in RAW 264.7 macrophage cells and is widely used as an
Materials and Methods
General Information
The 1H- and 13C-NMR were recorded using a Bruker Avance III 600 spectrometer (1H at 600 MHz and 13C at 150 MHz, Bruker, Karlsruhe, Germany) with tetramethylsilane as the internal standard. ESI-MS analyses were conducted employing a Shimadzu LC-MS/MS system equipped with a DAD detector (Shimadzu, Tokyo, Japan). Silica gel 60 (60-200 μm, Merck, Darmstadt, Germany), and RP-18 (120 Å, 150 μm, Merck, Darmstadt, Germany) were used for column chromatography (CC). Thin layer chromatography (TLC) was performed on Merck pre-coated 60 F254 silica gel plates (Merck, Darmstadt, Germany) and compounds were visualized by spraying the dried plates with 50% H2SO4, followed by heating. Organic solvents (grade ≥95%, republic of Korea) for CC were purchased from a local company.
Chemicals and Reagents
The common laboratory chemicals for the experiments were purchased from Sigma-Aldrich (St. Louis, Missouri, United States). DMEM high glucose media was purchased from Cytiva (#SH30243.01, Wilmington, MA, USA), fetal bovine serum (FBS, #12483020, Gibco, Waltham, MA, USA) and penicillin/streptomycin were from Gibco (#15140-122, Waltham, MA, USA), dexamethasone was purchased from MedChemExpress (#HY-14648, New Jersey, USA). Ez-Cytox reagent was purchased from DoGenBio (#EZ-1000, Seoul, Republic of Korea), and all antibodies were purchased from cell signaling technology (Danvers, MA, USA, supplement information, Table S2) and BioLegend (San Diego, CA, USA).
Preparation of EEPS Extract
The roots of
Isolation of Single Compounds from EEPS Extract
The roots of
Cell Culture and Cell Viability Assay
The RAW 264.7 cells were cultured using DMEM high-glucose media supplemented with 10% heat-inactivated FBS and 1% penicillin/streptomycin at 37 °C under 5% CO2 conditions. For cell viability assays, 30 000 cells were seeded in a 96-well plate and incubated for 24 h. Subsequently, the media was removed, and EZ-Cytox reagent was added. After 30 min of incubation, the absorbance was measured at 450 nm.
Nitric Oxide (NO) Assay
The RAW 264.7 cells (30 000 cells/well) were seeded in a 96-well plate with 100 μL of media per well. The plates were then incubated for 24 h at 37 °C with 5% CO2. Then, the cells were treated with EEPS (100 µg/mL, 50 µg/mL, and 25 µg/mL). After 2 h, LPS (1 µg/mL) was added, and the cells were further incubated for 24 h. Following this incubation, the supernatant media was collected, and an equal volume of Griess reagent was added. The absorbance OD was measured at 540 nm. Simultaneously, cell viability was measured to compare with NO production.
Western Blot
The EEPS and LPS were treated in RAW 264.7 cells for appropriate duration in a 6 cm dish (Corning, New York, USA). Subsequently, the media was removed, and the cells were washed with cold DPBS. Then, cell extraction buffer supplemented with protease inhibitor cocktail (Roche, Basel, Swiss), and phosphatase inhibitor cocktail (Roche, Basel, Swiss) was directly added to the dish, and lysate was collected using a scraper. The lysate was then spun down at 14 000 rpm for 20 min. The supernatant was collected, and BCA assay (Thermo Fisher, Waltham, MA, USA) was carried out to quantify the total protein. Subsequently, SDS-PAGE was performed with 20 μg of protein at 100 volts. The resolved proteins were transferred to PVDF membranes at 100 volts for 90 min. Afterwards, the membranes were blocked with 3% BSA prepared in tris-buffered saline for 30 min. Then, primary antibody treatment was carried out overnight at 4°C with gentle rocking. Subsequently, the membranes were washed and treated with the respective secondary antibody at room temperature for 90 min with gentle rocking. Finally, the membranes were washed, and protein bands were developed using a chemiluminescent reagent (Thermo Fisher, Waltham, MA, USA). The precision plus protein western C protein ladder (Bio-Rad, Hercules, CA, USA) was used and treated with StrepTactin-HRP conjugate for 15 min (Bio-Rad, Hercules, CA, USA) to develop ladder bands in the membrane.
Intracellular ROS Detection by DCF-DA
The RAW 264.7 cells were seeded in a 6 cm dish and incubated at 37 °C with 5% CO2 conditions for 24 h. Subsequently, the cells were treated with EEPS and LPS for 18 h. Following this treatment, DCFDA (5 μM) was added and incubated for 20 min. The cells were then harvested, washed with DPBS twice, and analyzed using flow cytometry (FACS Verse, BD, Franklin Lakes, NJ, USA) with the FITC channel.
For the imaging experiment, RAW 264.7 cells were seeded in a 96-well plate (Greiner Bio-One, Kremsmünster, Austria). Then, EEPS and LPS were treated for 18 h. Following this treatment, DCFDA (5 μM) was added and, after 20 min, the cells were washed twice with DPBS. The images were then taken using the Operetta high-content imaging system (Perkin Elmer, Waltham, MA, USA)
RNA Isolation and qRT-PCR Analysis
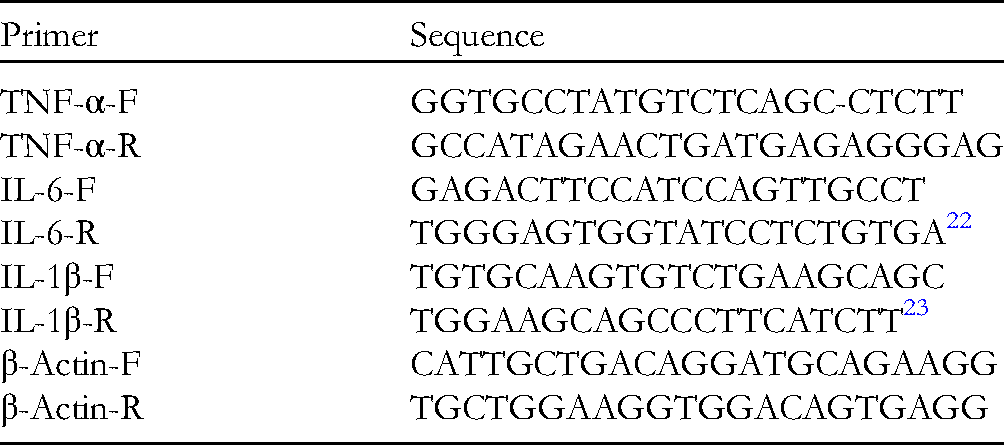
The RAW 264.7 cells were treated with EEPS and LPS for 12 h, and total RNA was isolated using the AccuPrep universal RNA Extraction Kit (K-3141; Bioneer, South Korea) according to the manufacturer's protocol. Total RNA was reverse-transcribed using the AccuPower cyclescript RT premix kit (K-2044; Bioneer, South Korea) according to the manufacturer's recommendations. Then, qPCR was performed using the AccuPower 2X greenstar qPCR master mix (K-6254; Bioneer, South Korea) on a QuantStudio 5.0 thermocycler (Thermo Fisher Scientific, Waltham, MA, USA). The following gene primer sequences were used in this study:
Intracellular Ca2+ Detection by Fluo-3 AM
The RAW 264.7 cells were seeded in a 96-well plate (Greiner Bio, USA) and incubated at 37 °C with 5% CO2 conditions for 24 h. Then, EEPS and LPS were treated for 18 h. Following this treatment, fluo-3 AM (2 μM) was added and, after 20 min, the cells were washed twice with DPBS. The images were then taken using the Operetta high-content imaging system (Perkin Elmer, Waltham, MA, USA)
Data Analysis
The GraphPad prism 10 (GraphPad, Boston, USA) software was used to make all the graphs and data are presented as mean ± SD of three independent experiments. Statistical significance was calculated using one-way ANOVA and
Results
EEPS Treatment Reduces the LPS-Induced Inflammation in RAW 264.7 Macrophages
The root samples of six
The different concentrations of EEPS were treated on RAW 264.7 cells for 24 h to assess the cytotoxicity, and the results showed that neither LPS nor EEPS exhibited any toxicity in RAW 264.7 cells (Figure 1A). Next, we tested EEPS for anti-inflammatory properties using the same concentrations. LPS treatment at 1 µg/mL for 24 h strongly induced nitric oxide secretion in RAW 264.7 cells, but EEPS pre-treatment significantly reduced nitric oxide secretion in a concentration-dependent manner at 25 µg/mL, 50 µg/mL, and 100 µg/mL (Figure 1B). Dexamethasone (Dexa) was used as a positive control for the NO assay. This assay clearly demonstrated that EEPS significantly prevents NO secretion in RAW 264.7 cells.

Anti-inflammatory effects of EEPS in RAW 264.7 cells. The EEPS (25, 50, 100 µg/mL) and positive control dexamethasone (50 µM) was pre-treated for 2 h and then LPS 1 µg/mL was treated for 24 h, (A) cytotoxicity of EEPS in RAW 264.7 cells, (B) EEPS prevents NO secretion induced by LPS, and suppressed the iNOS (C, D) and COX-2 (C, E). Data are presented as the mean ± standard deviation (SD) of three independent experiments (
Further, we also tested other inflammatory factors induced by LPS, such as iNOS and COX-2, by western blot. LPS strongly induced iNOS and COX-2 levels, but EEPS pre-treatment diminished their levels in a concentration-dependent manner. EEPS significantly decreased iNOS and COX-2 levels, similar to the positive control dexamethasone, at 100 µg/mL (Figure 2D–F). These data clearly demonstrate the anti-inflammatory properties of EEPS against LPS in RAW 264.7 macrophages.

Effects of EEPS on pro-inflammatory cytokines gene expression induced by LPS. RAW 264.7 cells were treated with dexamethasone 50 µM and EEPS (25, 50 and 100 µg/mL) for 2 h, and then stimulated with LPS (1 μg/mL) for 12 h. Then mRNA expression levels of IL-1b (A), IL-6 (B), TNF-α (C) were measured by real-time qRT-PCR. (D-E) The intracellular Ca2+ levels were measured using Fluo-3 dye after 18 h LPS and EEPS treatment. Scale bar 100 µm. White bar: non-treated group, black bar: LPS treated group, gray bar: sample treated group. Data are presented as the mean ± standard deviation (SD) of three independent experiments (
EEPS Reduces the pro-Inflammatory Cytokines Induced by LPS in RAW 26.7 Cells
LPS treatment induces the expression of various pro-inflammatory cytokines in RAW 264.7 cells, activating various inflammatory responses.7,8 Therefore, we tested whether EEPS suppresses the expression of pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α. The qPCR data show that LPS treatment profoundly induces the expression of these cytokines, but EEPS suppressed their expression, similar to the positive control, in RAW 264.7 cells (Figure 2A–C). Additionally, intracellular Ca2+ levels were also monitored using Fluo-3 dye, as inflammatory reactions are known to elevate intracellular Ca2+ levels, which are linked to the regulation of pro-inflammatory cytokines like TNF-α.12,13 LPS treatment increased the intracellular calcium levels to three times the control, but EEPS treatment significantly reduced Ca2+ levels in a dose dependent manner (Figure 2D and E, Figure S2). These data indicate that EEPS reduces the expression of pro-inflammatory cytokines in RAW 264.7 macrophages.
EEPS Suppresses the Activation of NF-κB Signaling Pathway Induced by LPS
NF-κB is a transcription factor that translocates to the nucleus upon activation by inflammatory signals, such as LPS. 24 The activated NF-κB plays a major role in activating the expression of various pro-inflammatory cytokines, including NO, iNOS, IL-1b, IL-6, and TNF-α. 25 The data show that LPS treatment in RAW 264.7 cells induced the activation of p65 by phosphorylation; p65 is a subunit of NF-κB. EEPS treatment significantly reduced the phosphorylation of p65 (p-p65) and decreased its levels in a concentration-dependent manner (Figure 3A and B).

Effects of EEPS on NF-κB pathway induced by LPS. RAW 264.7 cells treated with dexamethasone 10 µM and EEPS (25, 50, 100 μg/mL) for 2 h and then stimulated with lipopolysaccharide (LPS; 1 μg/mL) for 30 min. Then, the immunoblot was carried out for p-p65, p65, p-IκB-α, IκB-α (A). The EEPS prevents the phosphorylation of p65 (A, B) and IκB-α (A, C) and induces the IκB-α (A, D) levels in RAW 264.7. (-) non-treated group, (+): LPS-treated group. Data are presented as the mean ± standard deviation (SD) of three independent experiments (
Next, we assessed the status of the NF-κB inhibitor protein IκB-α in RAW 264.7 cells after LPS treatment. LPS treatment significantly increased the phosphorylation status of p-IκB-α and decreased the total IκB-α levels in LPS-treated samples. However, co-treatment with EEPS gradually decreased the levels of p-IκB-α, thereby retaining NF-κB in the cytoplasm and blocking its activation (Figure 3A and C). EEPS also increased the total form of IκB-α levels at 100 µg/mL, similar to the positive control, dexamethasone (Figure 3A and D). These data support that EEPS exhibits anti-inflammatory effects by suppressing the activation of the NF-κB signaling pathway.
EEPS Treatment Reduces the LPS-Induced MAPK Signaling Pathway in RAW 264.7 Cells
The MAPK signaling pathway is also involved in the release of various pro-inflammatory cytokines during the inflammatory reaction process. LPS treatment induces the phosphorylation of MAPK family members ERK, p38, and JNK thereby activating them during the inflammatory process.24,25 Therefore, we tested whether EEPS treatment prevents the activation of the MAPK signaling pathway. LPS treatment activated the MAPK proteins via phosphorylation and increased the levels of p-ERK, p-p38, and p-JNK in RAW 264.7 cells. However, EEPS treatment suppressed the activation of MAPKs in a concentration-dependent manner, more effectively than the positive control dexamethasone (Figure 4A–D).

EEPS effects on MAPK signaling pathway induced by LPS. RAW 264.7 cells were treated with dexamethasone 50 µM and EEPS (25, 50, 100 μg/mL) for 2 h and then stimulated with lipopolysaccharide (LPS; 1 μg/mL) for 30 min. Then, immunoblot was carried out for MAPK proteins. The EEPS has significantly reduced the phosphorylation of ERK (A, B), JNK (A, C) and p38 (A, D) in RAW 264.7 cells. Data are presented as the mean ± standard deviation (SD) of three independent experiments (
The inflammatory process also leads to the accumulation of intracellular ROS, thereby enhancing oxidative stress. 26 Therefore, we tested the oxidative stress levels after LPS treatment by measuring intracellular ROS level in RAW 264.7 cells. LPS treatment significantly induced the accumulation of intracellular ROS as analyzed using the DCF-DA fluorescent dye via flow cytometry and imaging. The data show that LPS treatment drastically increased the DCF-DA fluorescent intensity, indicating the accumulation of intracellular ROS (Figure 5A–D, Figure S2). However, EEPS treatment has gradually reduced the accumulation of intracellular ROS levels in RAW 264.7 macrophages (Figure 5A–D, Figure S3).

Effects of EEPS on intracellular ROS accumulation induced by LPS. RAW 264.7 cells were treated with dexamethasone (Dexa) 50 µM and EEPS (25, 50, 100 µg/mL) for 2 h and then LPS 1 µg/mL was treated and incubated for 18 h. Then, cells were stained with 5 µM DCF-DA for 15 min. Then, fluorescent images (A, B) and flow cytometry (C, D) was carried out. scale bar = 100 µm. The data are presented as means ± SD of three independent experiments (
Isolation and Identification of Single Compounds from EEPS Extract
The EEPS extract was suspended in H2O and partitioned with
Compound

The structures of single compounds isolated from EEPS extract.
Names of Single Compounds Isolated from EEPS.

Compound
Similarly, compounds
Compound
Compound
Compound
Anti-Inflammatory Effects of Single Compounds Isolated from EEPS Extract
The anti-inflammatory effect of single compounds isolated from EEPS root extract was studied in RAW 264.7 macrophages. The seven tested single compounds showed anti-inflammatory effects against LPS-induced NO release (Figure 7). Among them, the compound PS1 exhibited strong cytotoxicity, while the other compounds demonstrated anti-inflammatory effects at the tested concentrations (Figure 7). Quercitrin,30,31 hydroxymethylfurfuryl,
32
and afzelin
33
are already known to have anti-inflammatory effects and suppress the release of pro-inflammatory factors. Notably, PS2 (Polyscioside A), PS3.1, and PS3.2 (Polyscioside D) showed potent anti-inflammatory activity in LPS induced RAW 264.7 cells. The anti-inflammatory activity of these three compounds has not been reported so far. These three structures were identified in
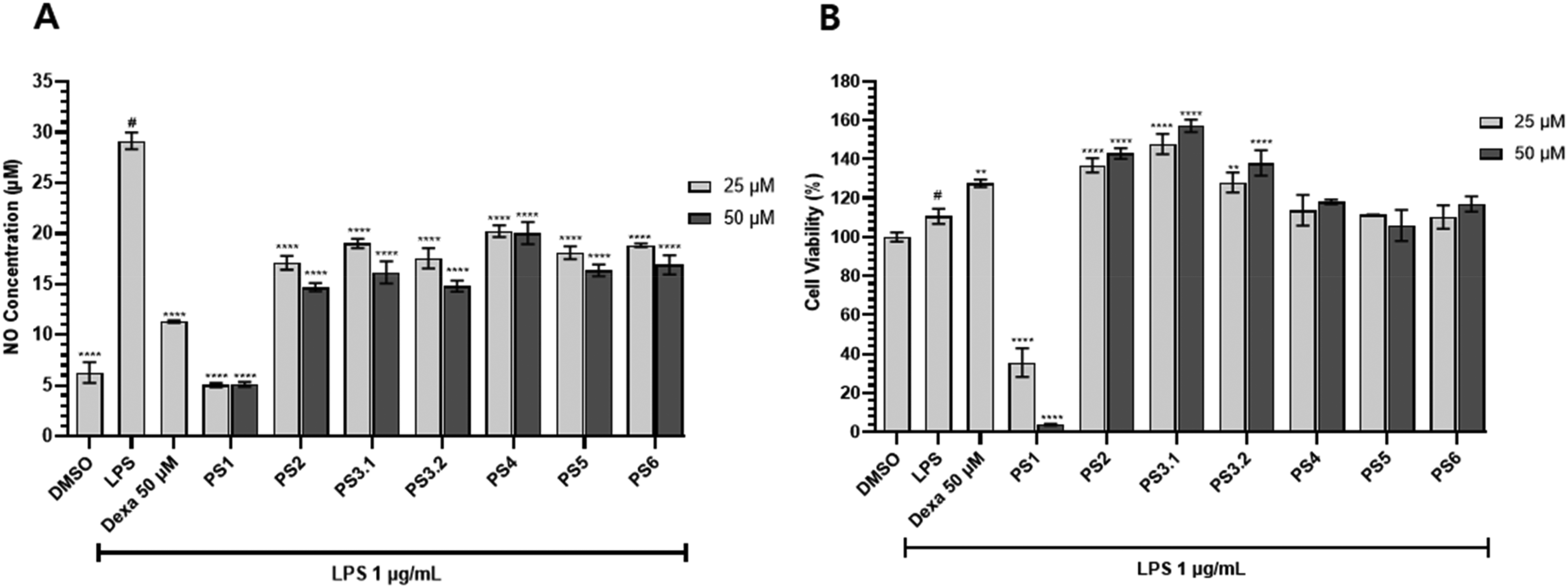
Anti-inflammatory effects of EEPS single compounds. (A) RAW 264.7 cells treated with dexamethasone 50 µM or compounds for 2 h, and then stimulated with LPS (1 µg/mL) for 24 h. Cell supernatants were collected, and (A) NO assay was carried out and the (B) cytotoxicity measured by EZ-cytox cell viability assay. White bar: non-treated group, black bar: LPS treated group, gray bar: sample treated group. (
Discussion
The inflammatory reaction is associated with pathophysiology of various neurodegenerative diseases, cancers, stroke, diabetes and arthritis etc.3,34 Suppressing inflammatory reactions has become a therapeutic strategy for treating inflammation-mediate diseases.
LPS treatment in RAW 264.7 cells induce various inflammatory reactions and triggers the secretion of inflammatory factors such as NO, iNOS, and COX-2. The immunoblot data showed the induction of these factors in RAW 264.7 cells and EEPS treatment significantly reduced their secretion (Figure 1). iNOS mediates the synthesis of NO, while COX-2 mediates the synthesis of prostaglandins. 35 During the acute inflammation they produced in high concentrations, which can be harmful to cell survival. The overproduction of NO and prostaglandins is associated with several diseases such as Alzheimer's, brain ischemia, and multiple sclerosis. 36 Therefore, the suppression of NO and COX-2 production holds significant therapeutic potential for the treatment of these diseases.
The expression of pro-inflammatory cytokines is induced during the inflammatory response. LPS-induced inflammation drastically increased the expression of pro-inflammatory cytokines such as TNF-α, IL-1b, IL-6. But, the EEPS treatment gradually reduced the expression of these inflammatory cytokines in RAW 264.7 cells (Figure 2A–C). The continuous secretion of these inflammatory factors is associated with many inflammations-related diseases, such as Alzheimer's. In human brain the continuous release of these factors due to neuro-inflammation aggravates the Alzheimer's disease progression by contributing to the abnormal aggregation of amyloid beta and tau protein phosphorylation.37,38 Additionally, TNF-α, IL-1b, and IL-6 play major roles in osteoarthritis, leading to the destruction of cartilage tissues. 39 Therefore, controlling the expression of these inflammatory cytokines could significantly reduce the progression of inflammation-related pathophysiology.
The NF-κB pathway is implicated in many autoimmune inflammatory diseases such as atherosclerosis, rheumatoid arthritis (RA), multiple sclerosis, and inflammatory bowel diseases. This pathway mediates the expression of various pro-inflammatory cytokines and chemokines.4,6 It is regulated by the inhibitor protein IκB-α. NF-κB signaling induces the expression of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1b, thereby aggravating the progression of inflammatory diseases. The intracellular Ca2+ levels increased after the LPS treatment, but co-treatment with EEPS significantly reduced these levels (Figure 2D and E).
LPS treatment has been shown to drastically induce NF-κB activation by suppressing the inhibitor protein IκB-α (Figures 3A and 4B). However, co-treatment with EEPS significantly decreased the activation of NF-κB by preventing the phosphorylation of the NF-κB subunit p65 and IκB-α (Figure 4A and B). Furthermore, the MAPK pathway is also activated by inflammatory signals such as LPS, and intracellular ROS accumulation increases due to the toxic inflammatory reaction. 40 Co-treatment with EEPS significantly reduced the activation of MAPK proteins (ERK, p38, and JNK) (Figure 4A–D) and intracellular ROS in RAW 264.7 macrophages (Figure 5A–D). Literature indicates that MAPK inhibitors, such as SB220025 and SB203580, appear to reduce the progression of collagen-induced arthritis.41,42
The single compounds were isolated from the EEPS and their structures were identified. The isolated compounds showed anti-inflammatory activity in RAW 264.7 cells (Figure 7). These compounds are the major active components responsible for the strong anti-inflammatory activity of EEPS. Among the isolated compounds, quercitrin, 5-hydroxymethyl-2-furaldehyde, and afzelin are known to suppress inflammation in RAW 264.7 cells by reducing ROS accumulation, preventing the release of pro-inflammatory cytokines, and inhibiting the NF-κB pathway.30,32,33 Among the saponin derivatives, including PS1, PS2, PS3.1, and PS3.2, high concentrations of PS2, PS3.1, and PS3.2 demonstrated anti-inflammatory activity, reducing NO production by 60%–70% in RAW 264.7 cells compared to LPS-treated cells. These active components are the key ingredients responsible for the biological activity of EEPS. Each compound (PS2, PS3.1, and PS3.2) exhibited relatively weak or similar anti-inflammatory effects compared to EEPS. We initially assumed that a single compound would exhibit more potent anti-inflammatory activity than an extract. The strong anti-inflammatory activity of EEPS suggests a potential synergistic effect when compared to treatment with a single compound. Almeida and colleagues have reported a similar synergistic effect of extracts in anti-inflammatory activity.43,44
These experiments were conducted using in vitro method and cell lines. To further validate the findings and evaluate their potential relevance in more complex biological systems, future research will involve studies using animal models.
The previous report demonstrated the anti-inflammatory effect of the lipophilic components derived from
Overall, EEPS appears to contain active components with anti-inflammatory properties. By strongly suppressing the activation of NF-κB, MAPK, and intracellular ROS accumulation, EEPS exhibits strong anti-inflammatory activity in RAW 264.7 cells.
Conclusions
This study explored the anti-inflammatory potential of EEPS in RAW 264.7 cells. Inflammation in RAW 264.7 macrophages was induced by LPS. EEPS significantly reduced LPS-induced inflammatory effects by suppressing the release of NO and the activation of iNOS and COX-2. Furthermore, it reduced the expression of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β. Additionally, EEPS reduced the activation of the NF-κB and MAPK signaling pathways, which play major roles in the expression and release of various inflammatory cytokines and chemokines. The study also isolated single compounds and assessed their anti-inflammatory activities. These findings suggest that EEPS is a promising candidate for the development of anti-inflammatory therapies.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251324006 - Supplemental material for The Ethanol Extract of Polyscias scutellaria (EEPS) Shows Anti-Inflammatory Signaling Pathway Against LPS-Induced Inflammation in RAW 264.7 Macrophages
Supplemental material, sj-docx-1-npx-10.1177_1934578X251324006 for The Ethanol Extract of
Footnotes
Acknowledgements
The authors would like to express their appreciation for the facilities offered by Korea Institute of Science and Technology, Gangneung Institute, Republic of Korea, for this study.
Abbreviations
EEPS (ethanol extract of
Author Contributions
N.T.A.N., S.Q.N.N. and V.T.O. performed this research. H.T.V. and L.H.O. conducted experiments. H.L. and Q.L.T. analyzed the data and interpreted the results. B.S., P.T.T. and S.H.J. drafted the manuscript. J.W.L. drafted and revised the paper. All authors read and approved the final version of the manuscript.
Data Availability
The data will be provided upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is supported by the National Research Foundation of Korea (No. NRF-2019K1A3A1A82113697), KIST intermural grant (No. 2Z07113) and Ministry of Science and Technology of Vietnam (No. NDT.90.KR/20).
Statement of Human and Animal Rights
Not applicable
Statement of Informed Consent
Not applicable
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
