Abstract
Keywords
Introduction
Idiopathic pulmonary fibrosis (IPF) is a lung disease of unknown etiology. The condition is confined to the lungs in chronic, progressive, fibrotic interstitial pneumonia, and is the most common type of idiopathic interstitial pneumonia. 1 In addition to genetic and environmental factors, 2 radio-pulmonary lesions are also a high-risk factor for the development of pulmonary fibrosis. 3 The incidence of IPF has increased in recent years, and the average survival period is just 2 to 3 years after diagnosis. Thus, due to its high incidence, low survival rate, and very poor prognosis, IPF now represents a serious threat to public health.4,5 Studies have shown that nintedanib and pirfenidone, which are currently recommended in clinical practice, are effective in reducing the expected decline in forced vital capacity (FVC) and slowing disease, but do not halt or reverse its pathological course. In addition, long-term use of these drugs can cause damage to liver function and the gastrointestinal tract.6–8 Therefore, further research on the pathogenesis of IPF and exploration of safe and effective therapeutic drugs are key directions of current research in the respiratory community.
Traditional Chinese Medicine (TCM) is a system of medical science with a long history, unique theories, and techniques. It has significant advantages in treating symptoms and improving the quality of life for IPF patients. Moreover, it is often considered safer and more cost-effective compared to many conventional medications. According to TCM, IPF belongs to the categories of “Feiwei” and “Feibi” and is associated mechanistically with deficiency of the lung and loss of moistening of the fluid. Coughing and sticky sputum are caused by heat that cauterizes the fluid while coughing and sputum with blood are caused by dry heat that cauterizes the lung channels, which is associated with deficiency of lung yin, internal incandescence of deficiency fire, and the loss of the dispersing and descending function of the lung. The etiology of this disease is complex, and “deficiency, phlegm, stasis and toxicity” are the main causative factors. Therefore, the treatment should be based on “tonifying lung deficiency, clearing heat and phlegm, and activating blood circulation to remove blood stasis.” 9
Chinese herbal medicine primarily consists of botanical substances derived from plants (roots, stems, leaves, and fruits) and mineral components. This article specifically refers to compound formulations composed of 2 or more Chinese herbal ingredients. As a classic TCM formula, Qingzao Jiufei Tang decoction (QZJFD) is derived from Yu Jiayan's “Medical Gate Law” according to The World Flora Online database (http://www.worldfloraonline.org) or MPNS (http://mpns.kew.org) and the Pharmacopoeia of the China (2020 Edition). 10 QZJFD is consists of Morus indica L. (Sang Ye), CaSO4·2H2O (Shi Gao), Glycyrrhiza uralensis Fisch. (or Glycyrrhiza inflata Bat., or Glycyrrhiza glabra L.) (Gan Cao), Panax ginseng C. A. Mey. (Ren Shen), Sesamum indicum L. (Hu Ma Ren), Equus asinus L. (E Jiao), Ophiopogon japonicus (Linn. f.) Ker-Gawl. (Mai Dong), Prunus armeniaca L.var.ansu Maxim. (or Prunus sibirica L., or Prunus mandshurica (Maxim.) Koehne, or Prunus armeniaca L.) (Xing Ren), and Eriobotrya japonica (Thunb.) Lindl. (Pi Pa Ye). This classic herbal prescription is used to treat IPF. The Sesamum indicum L. (Hu Ma Ren) component of QZJFD is recorded in Shennong's Herbal Classic Herbal and Bencaojing Jizhu11,12 as black sesame, which was used in this study. Clinical studies have shown that QZJFD plus and minus has an overall efficiency of 90.6% in the treatment of pulmonary fibrosis, alleviating the symptoms and improving the quality of life of patients. 13 A study of collateral disease theory revealed that the total effectiveness rate of modified QZJFD in the treatment of IPF was 95.24%, which was significantly higher than that of the group treated with conventional medicine (79.27%, P < .05). QZJFD was also shown to effectively alleviate clinical symptoms, promote the improvement of pulmonary function and arterial blood gases, alleviate inflammation, and inhibit pulmonary fibrosis. 14 Modern pharmacological research shows that QZJFD has antioxidant activity and can improve pulmonary fibrosis. 15 In animal models, QZJFD has been shown to improve lung function by reducing lung inflammation, decreasing Mycoplasma pneumonia (MP) toxin content, and increasing the levels of active substances in lung tissue. 16 However, the therapeutic effect, main active ingredients, and mechanism of action of QZJFD on IPF are still unclear.
Network pharmacology is a novel research discipline that involves the systematic construction of network models to reveal drug pharmacological mechanisms and guide clinical treatment. This approach also facilitates new drug development by predicting the active ingredients of herbs and their formulations, effective targets, and signaling pathways for specific diseases17,18 Molecular docking simulation is used to predict intermolecular interactions and identify the binding mechanism and patterns between small molecules and target proteins. 19
To explore the safety and effectiveness of QZJFD for the prevention and treatment of IPF and clarify the main active components as well as the underlying mechanism of action, we employed a network pharmacology approach combined with macromolecular docking analysis and experimental verification. This information will provide a scientific and effective basis for clinical diagnosis and treatment of IPF.
Results
Network Pharmacology Predictions
Identification of the Main Active Ingredients of QZJFD and Target Prediction
A total of 187 active compounds were retrieved from the Traditional Chinese Medicine Systems Pharmacology (TCMSP) database. The active compounds were mainly derived from Gan Cao (92 compounds), Pi Pa Ye (18 compounds), Sang Ye (29 compounds), Ren Shen (22 compounds), Xing Ren (19 compounds), and Hu Ma Ren (7 compounds); some of the main active ingredients are shown in Table 1.
Basic Information for Some Active Ingredients of QZJFD Identified in the TCMSP Database.

Basic Information for Some Active Ingredients of QZJFD Identified in the TCMSP Database.

Abbreviations: QZJFD, Qingzaojiufei decoction; TCMSP, Traditional Chinese Medicine Systems Pharmacology database and analysis platform.
“Shi Gao, Mai Dong, E Jiao,” which were not retrieved from the TCMSP, were also searched in the Bioinformatics Analysis Tool for Molecular mechANism (BATMAN) database. Gypsum was also searched using the corresponding InChI in PubChem. This search yielded 22 active ingredients in E Jiao (3 compounds), Mai Dong (18 compounds), and Shi Gao (1 compound); the main active ingredients are shown in Table 2.
Basic Information for Some Active Ingredients of QZJFD Identified in the BATMAN Database.

Abbreviations: QZJFD, Qingzaojiufei decoction; BATMAN, Bioinformatics Analysis Tool for Molecular mechANism.
Searches of the Online Mendelian Inheritance in Man (OMIM) and GeneCards databases using the keywords “idiopathic pulmonary fibrosis” yielded 3875 disease-related targets. As shown in Figure 1, 308 common targets were identified in the intersection of the QZJFD active ingredients and the IPF-related targets.

Venn diagram of Qingzaojiufei decoction (QZJFD) and disease-related targets.
Visual Analysis of the TCM-Component-Target-Disease Network
The TCM-component-target-disease network is shown in Figure 2. The network topology parameters were analyzed using a network analyzer plug-in to derive the degree values of the nodes in the above network, which refers to the number of edges or target nodes associated with the nodes. Finally, 5 compounds with the highest degree values were obtained as potential bioactive ingredients in this study, namely quercetin, (-)-epicatechin acid ester, lysine, stigmasterol, and kaempferol (Table 3).

The Traditional Chinese Medicine (TCM)-component-target-disease network.
Potential Core Components and Degree Values of IPF for QZJFD Control.

Abbreviations: QZJFD, Qingzaojiufei decoction; IPF, idiopathic pulmonary fibrosis.
Visualization of the TCM-component-target-disease network using Cytoscape 3.9.0. Green circles represent TCM nodes, yellow/orange arrowheads represent the active component nodes, and blue boxes represent the gene nodes.
Construction of Protein-Protein Interaction (PPI) Networks
As shown in Figure 3, the PPI network showed a strong correlation between targets, with 308 nodes identified as the core targets of QZJFD in IPF therapy. Among these, albumin (ALB), serine/threonine protein kinase 1 (AKT1), tumor necrosis factors (TNF), interleukin-6 (IL-6), and tumor protein p53 (TP53) were identified as pivotal genes (Table 4).

Protein-protein interaction (PPI) network diagram.
Selected Information for Core Targets of QZJFD in IPF Therapy.

Abbreviations: QZJFD, Qingzaojiufei decoction; IPF, idiopathic pulmonary fibrosis; AKT1, recombinant protein; ALB, albumin; IL-6, interleukin-6; TNF, tumor necrosis factors; TP53, tumor protein p53; MAPK, mitogen-activated protein kinase.
Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) Enrichment Analyses
GO functional enrichment analysis was then performed to further explore the mechanism underlying the effects of QZJFD on IPF. In total, genes were enriched in 2668 biological processes (BP), 120 cellular components (CC), and 273 molecular function (MF) categories. The top 10 entries were selected based on P < .05 and are shown in Figure 4. These data suggested that the biological processes involved in the effects of QZJFD on IPF are mainly related to responses to drugs, lipopolysaccharides, and molecules of bacterial origin, while membrane rafts and membrane microdomains are the main cellular components and carboxylic acid binding and oxidoreductase activity are the main molecular functions.

Go functional enrichment analysis of core targets of QZJFD in IPF therapy.
The KEGG enrichment analysis revealed 197 pathways in the IPF-related targets of QZJFD treatment, including lipid and atherosclerosis, chemical carcinogenesis-receptor activation, mitogen-activated protein kinase (MAPK), calcium (Ca), advanced glycosylation end-product receptor (AGE-RAGE), TNF, and IL-17 signaling pathways. The top 20 signaling pathways were selected based on P < .05 and are shown in Figure 5.
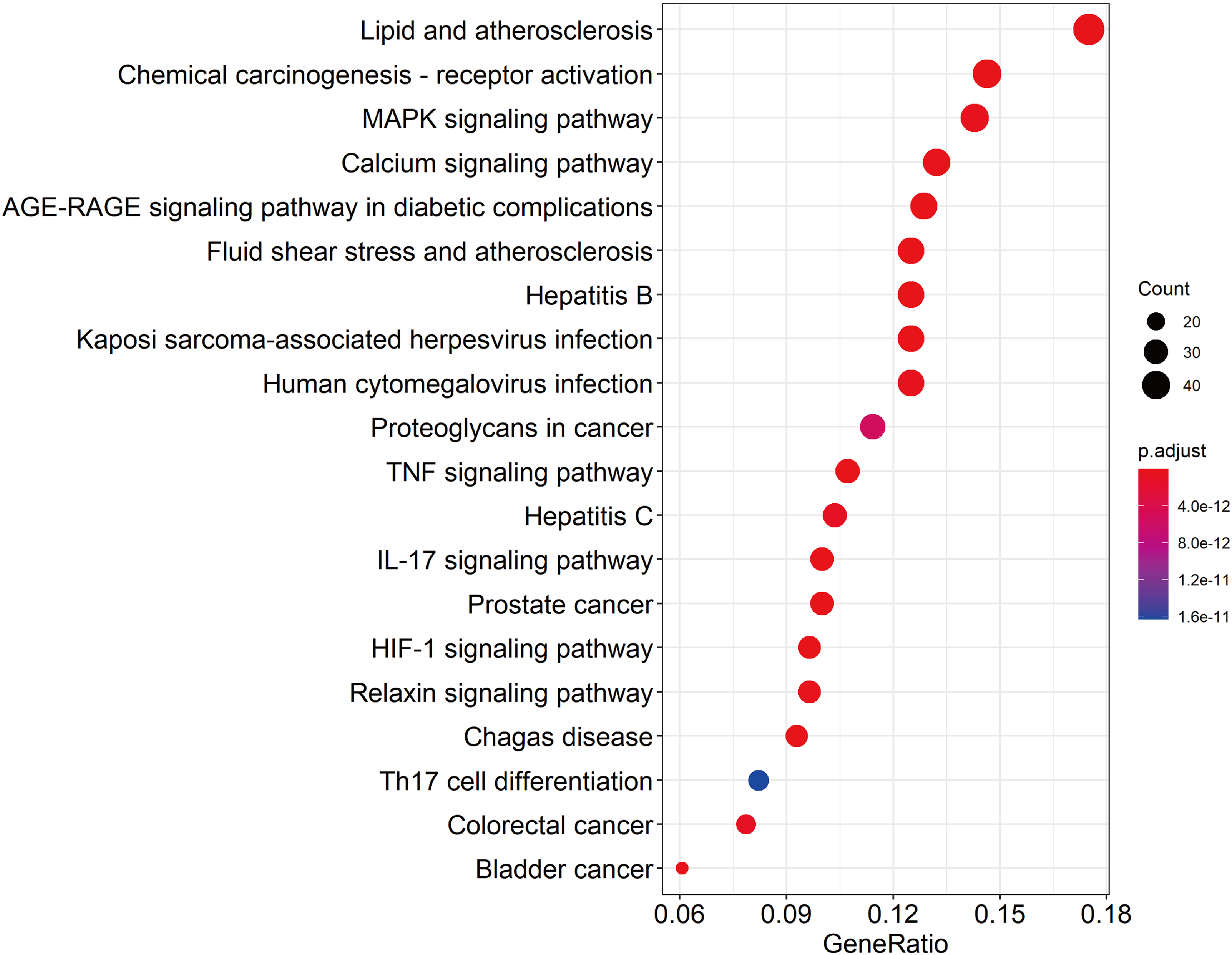
KEGG functional enrichment analysis of key signaling pathways involved in the effects of QZJFD in IPF therapy.
Molecular Docking Simulation
In general, lower binding energy (kcal/mol) reflects a higher affinity of the active substance for the target protein and therefore, greater stability of the interaction, with binding energy < −5 kcal/mol regarded as strong binding activity. The binding energy of each compound is shown in Table 5, and its molecular docking thermal energy diagram is shown in Figure 6.

Molecular docking binding energy thermogram.
Docking Binding Energy for the Top 10 Compounds.

Abbreviations: AKT1, recombinant protein; ALB, albumin; IL-6, interleukin-6; TNF, tumor necrosis factors; TP53, tumor protein p53.
All key compounds in the network had a strong affinity for ALB. The small molecules naringenin (MOL004328) and kaempferol (MOL000422) showed a strong binding association with ALB. Naringenin formed 3 hydrogen bonds with amino acid residues LEU-135, GLY-189, and LYS-190, and kaempferol formed 2 hydrogen bonds with amino acid residues LEU-154 and TYR-138, resulting in the stable complexes formed by naringenin and kaempferol with ALB (Figure 7).

Molecular docking simulation of albumin (ALB)-naringenin and molecular docking simulation of ALB-kaempferol.
The Effects of QZJFD in a Rat Model of Lung Fibrosis
Serum Inflammatory Factor Levels
As shown in Figure 8, the serum levels of IL-6, TNF-α, and IL-1β of rats in the model group were significantly higher than those in the blank group (P < .001). Furthermore, the serum levels of IL-6, TNF-α, and IL-1β of rats in the pirfenidone (positive control) and the QZJFD mid- and high-dose groups were significantly lower than those in the model group (P < .01).
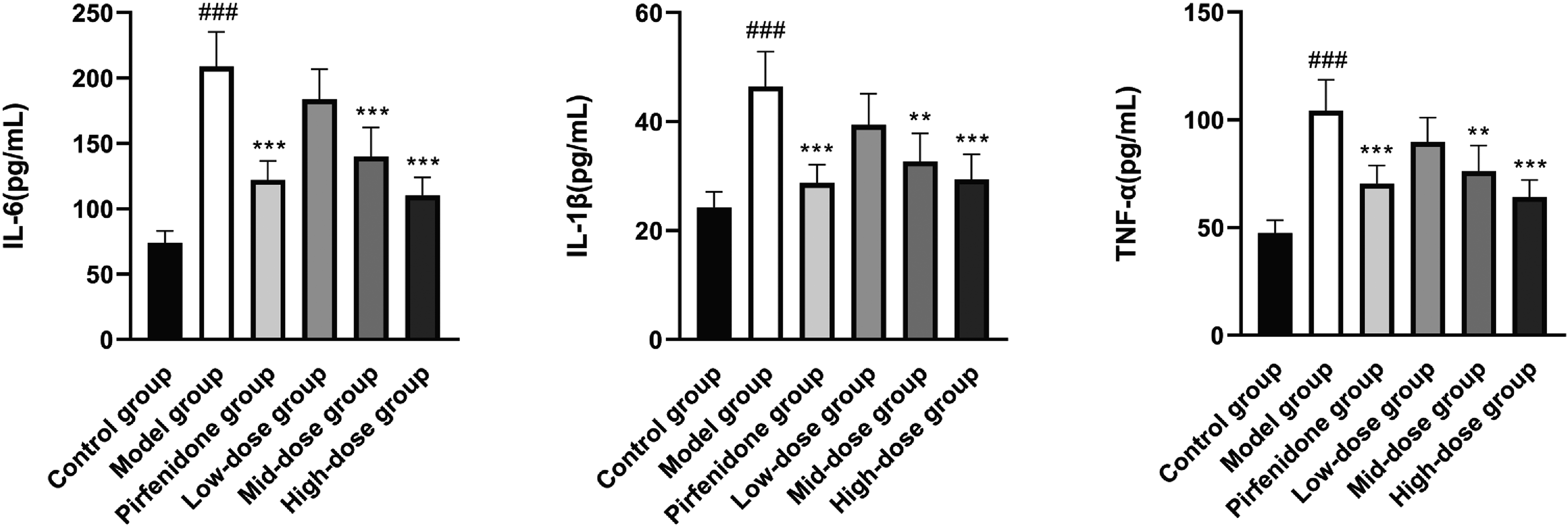
The effects of Qingzaojiufei decoction (QZJFD) on serum inflammatory factor levels in a rat model of lung fibrosis.
Histomorphological Evaluation of the Lung
Hematoxylin and eosin (HE) staining (Figure 9) confirmed that the structure of the lung tissue in control rats was normal. The alveolar cavity was clearly visible without hemorrhage or inflammatory cell infiltration. In contrast, the lung tissue structure in the model group was disorganized, the alveolar structure was destroyed (some alveoli had disappeared), and the alveolar wall was obviously thickened. In addition, the interstitial cavity was infiltrated by a large number of inflammatory cells. Compared with the model group, the alveolar structure of the lung tissue was improved in the rats in the pirfenidone (positive control) and the QZJFD mid- and high-dose groups, and the inflammatory cell infiltration in the interstitial cavity were reduced; these effects appeared to be dose-dependent.

He and Masson's trichrome staining of lung tissue (200 × magnification).
The results of Masson staining (Figure 9) also showed that the alveolar structure of control rats was intact, with no obvious collagen (blue) deposition in the alveolar interstitium. In the model group, the alveolar structure was destroyed, with the deposition of large amounts of collagen and obvious fibrosis in the alveolar interstitium. In the rats in the pirfenidone (positive control) and the QZJFD mid- and high-dose groups, the collagen deposition and fibrosis in the alveolar interstitium were reduced or lower than the levels in the model group, and the improvement was more obvious with increasing dose.
Expression of α-SMA, TGF-β1, p-JNK, JNK, p-P38 MAPK, P38 MAPK, Collagen I, and FN1 in Lung Tissues
The expression levels of α-SMA, TGF-β1, p-JNK, p-P38 MAPK, collagen I, and FN1 in lung tissue were evaluated by immunofluorescence labeling and western blot analysis (Figures 10 and 11, respectively). The levels of these proteins were significantly higher in the lung tissues of rats in the model group compared with those in the blank group (P < .001). The expression levels of TGF-β1 in the lung tissues of model rats were significantly lower in the pirfenidone and QZJFD high-dose groups (P < .01), while the expression levels of α-SMA, p-JNK, p-P38 MAPK, collagen I, and FN1 in the lung tissues of rats in the pirfenidone and QZJFD mid- and high-dose groups were significantly lower (P < .05); the effects of QZJFD were dose-dependent.

Immunofluorescence labeling of α-SMA, TGF-β1, p-JNK, JNK, p-P38 MAPK, P38 MAPK, collagen I, and FN1 in lung tissue (200 × magnification).

Western blot analysis of expression levels of α-SMA, TGF-β1, p-JNK, JNK, p-P38 MAPK, P38 MAPK, collagen I, and FN1 in lung tissue.
Discussion
The poor prognosis of IPF has prompted the search for safer and more effective drugs that will provide improved outcomes. Because of its unique advantages, TCM has played an important role in clinical diagnosis, treatment, and prevention. To further study the pathogenesis of IPF and explore safe and effective prevention and treatment drugs, this study aims to verify the main active ingredients and therapeutic effects of QZJFD on idiopathic pulmonary fibrosis through network pharmacology, molecular docking analysis, and animal experiments, while also investigating its mechanism of action. QZJFD is a classic prescription for the treatment of lung injury syndrome caused by dryness evil. Mechanistically, QZJFD promotes Qing and reduces moistening to remove evil and support righteousness. In modern Chinese medicine, QZJFD is clinically effective in treating chronic cough of lung flaccidity and Yin deficiency dryness and heat.9,20 Relevant studies have shown that QZJFD has antioxidant activity, 21 which can significantly improve pulmonary inflammation in MP-infected rats and reduce lung index and pulmonary scores. 22 To further clarify the mechanism by which QZJFD prevents and treats IPF, we used network pharmacology, molecular docking, experimental verification, and other methods to build a “TCM compound – disease” network, systematically explore the mechanism by which TCM compounds in QZJFD act through different pathways and targets to treat IPF and provide a more powerful theoretical basis for clinical treatment.
Although the pathogenesis of IPF has not yet been fully elucidated, it is generally accepted that this condition is caused by repeated damage and abnormal repair of alveolar epithelial cells, occurring mainly during the process of epithelial-mesenchymal-like transition (EMT). 23 Pulmonary fibroblasts are important effector cells in the fibrosis stage of IPF, 24 and myofibroblasts are important cells that promote the development of pulmonary fibrotic disease. 25 α-SMA is an important marker of differentiation of fibroblasts to myofibroblasts. 26 TGF-β, which plays a key role in the excessive inflammatory response in IPF, 27 by inducing fibroblast to myofibroblast differentiation via the Smads signaling pathway. 28 FN1 is an extracellular matrix protein secreted mainly by fibroblasts. 29 It facilitates cell adhesion, survival, migration, and differentiation, playing a crucial role in processes like tissue healing and embryonic development. 30 Studies have shown that PF1 overexpression can effectively reduce the expression of α-SMA induced by TGF-β1 and block differentiation into myofibroblasts. 31 Type I collagen and Type III collagen are the primary components of collagen in the extracellular matrix of lung tissue. 32 The extracellular matrix of lung tissue is the main factor in the development of pulmonary fibrosis. After lung tissue injury, excessive deposition of the extracellular matrix occurs in the lung interstitium, causing thickening of the alveolar membrane, leading to structural and functional damage to the lungs and resulting in respiratory failure. 33 The MAPK signaling pathway also plays an important role in the development of pulmonary fibrosis. 34
In this study, we identified quercetin, (-)-epicatechin acid ester, lysine, dousterol, and kaempferol as the 5 core effective components of QZJFD for the treatment of IPF through network pharmacological analysis. Studies in a rat model of IPF induced by bleomycin have shown that quercetin reduces the concentration of pro-fibrotic mediators such as IL-13, platelet-derived growth factor-β (PDGF-β), and TNF-α to achieve antifibrotic effects while enhancing antioxidant responses, reducing inflammatory factors and increasing nuclear factor erythroid 2-related factor 2 (Nrf2) activity.35,36 (-)-Epicatechin acid esters achieve therapeutic effects of IPF through the regulation of cholesterol-related genes. 37 Lysine-specific demethylase 1 (LSD1) is a target for the treatment of pulmonary fibrosis and exerts a pro-fibrotic regulatory effect through its ability to activate the transforming growth factor-β1/recombinant SMAD family member 3 (TGF-β1/Smad3) signaling pathway. 38 Soysterol acts as a powerful immunomodulator by inhibiting lipopolysaccharide-induced innate immune responses in mice to protect against liver and lung injury. 39 Kaempferol induces inhibition of the mammalian target of rapamycin (mTOR), which leads to downregulation of protein synthesis in the lung tissue, thereby alleviating pulmonary fibrosis and suppressing inflammatory factors. 40 Kaempferol also ameliorates the pathological damage in a rat model of lung ischemia-reperfusion injury by inhibiting the release of inflammatory factors and reducing oxidative stress, which further exerted a protective effect by regulating the sirtuin 1/high mobility group protein B/nuclear factor kappa-B (SIRT1/HMGB/NF-κB) axis. 41 Our study provides a theoretical basis for the screening of core components capable of preventing and treating IPF by network pharmacological analysis and further suggests that these core components may be the pharmacodynamic material basis of the therapeutic effects of QZJFD in IPF.
The core targets ALB, AKT1, TNF, IL-6, TP53 and other proteins identified in the PPI analysis are closely related to the main inflammatory mediators involved in the functions of TNF-α, IL-1, and IL-6 in IPF development. 27 AKT1, TNF, and TP53 were found to be enriched targets in the MAPK signaling pathway. Molecular docking simulation showed that the core targets have good binding affinity for the 5 core components, indicating that QZJFD may prevent IPF through its interaction with these core targets. ALB, which is a target of oxidation and protects other proteins from oxidative damage, is not only a peripheral blood biomarker for IPF, 42 but also a possible candidate biomarker for pulmonary fibrosis. 43 Furthermore, ALB can inhibit pulmonary fibrosis through its antioxidant effects. 44 AKT1 is a serine/threonine protein kinase that has roles in regulating cell proliferation, metabolism, and angiogenesis. Nie et al 45 showed that AKT1-induced IL-13 production by macrophages regulates pulmonary fibrosis and can block the fibrogenic process in IPF. TNFs such as TNF-α and TNF-β can cause hemorrhagic necrosis in tumor tissues. TNF-α is mainly produced by activated monocytes/macrophages and has a role in regulating the immune response and promoting cell growth and differentiation. As an important cytokine in the inflammatory response, IL-6 promotes the growth and differentiation of primary bone marrow-derived cells and enhances the lytic function of natural killer cells (NK cells). TNF-α and IL-6 have been shown to play an important role in IPF formation, with TNF-α promoting myofibroblast differentiation of lung mesenchymal stem cells (LR-MSCs) by activating the nuclear factor kappa-B (NF-κB) signaling pathway, 46 while IL-6 promotes the proliferation and migration of primary human fibroblasts by inducing signal transducer and activator of transcription 3 (STAT3) phosphorylation. 47 According to the GeneCards database, the TP53 gene on chromosome 17 encodes an oncoprotein containing transcriptional activation, deoxyribonucleic acid (DNA) binding, and oligomeric structural domains. The functions of p53 are cell-specific. In human alveolar epithelial cells (AEC), p53 accelerates apoptosis, induces senescence, and promotes the development of pulmonary fibrosis. In contrast, in human lung fibroblasts (HPFs), p53 promotes apoptosis, exacerbates oxidative stress, and slows the development of pulmonary fibrosis. 48 Thus, it can be speculated that QZJFD acts on the core targets of IPF to inhibit the formation of pulmonary fibrosis via different pathways to achieve a therapeutic effect.
GO and KEGG enrichment analyses further elucidated the molecular mechanism underlying the therapeutic effects of QZJFD in IPF. GO enrichment analysis suggested that the biological processes involved in the treatment of IPF by QZJFD are mainly related to the response to drugs, lipopolysaccharide response, and molecules of bacterial origin. KEGG enrichment analysis indicates that key targets are associated with immune responses, inflammatory reactions, and disease processes. Concerning involvement in disease processes, KEGG pathway categories include lipid and atherosclerosis, as well as the age-RAGE signaling pathway in diabetic complications. In terms of immune and inflammatory responses, KEGG pathways are divided into chemical carcinogenesis-receptor activation and the “MAPK signaling pathway.” In recent years, numerous studies have shown that lipid and atherosclerosis and chemical carcinogenesis-receptor activation represent redundant pathways49,50 and broad pathways,51–53 showing a relatively poor correlation with IPF. However, the MAPK signaling pathway can regulate inflammatory factors, fibrosis, and the development of cancer, playing a crucial role in the formation of pulmonary fibrosis.41,54 TNF can mediate the MAPK signaling pathway involved in lung physiological and pathological cell differentiation processes as well as the induction of cell apoptosis and inflammation. 55 TGF-β1 is a key molecule in activating the fibrotic program, mainly by regulating myofibroblast differentiation, proliferation, and extracellular matrix (ECM) deposition. 56 It has been shown that p38 MAPK is effective in ameliorating pulmonary fibrosis 57 and that activation of p38 MAPK and JNK may allow TGF-β1 to induce EMT in AECs, thereby effectively inhibiting the formation of pulmonary fibrosis. 58 QZJFD effectively delays lung cancer development and metastasis by reducing the expression of TGF-β1 and Smad2 and decreasing p38 MAPK protein activity.59,60 On the other hand, the effective component Quercetin in QZJFD not only inhibits the Smad and MAPK pathways, reducing collagen production and extracellular matrix (ECM) deposition, but it also exerts an anti-fibrotic effect by inhibiting MMT and regulating TGF-β-Smad2/3 signal transduction.61,62 Based on this, we speculate that the MAPK signaling pathway might be a crucial target for QZJFD in treating IPF. Our experiments further validated this speculation.
After treatment with pirfenidone and QZJFD at the medium and high doses, serum IL-6, TNF-α, and IL-1β levels in rats were significantly lower than those in the model group. Furthermore, the expression levels of α-SMA, p-JNK, p-P38 MAPK, TGF-β1, collagen I, and fibronectin-1 (FN1)in the lung tissues of rats in the pirfenidone and QZJFD high-dose groups were significantly lower than those in the model group, and the fibrosis status of lung tissues was improved. Studies indicate that the combination of QZJFD and pirfenidone can effectively alleviate the degree of pulmonary fibrosis in IPF patients, suppress the levels of airway inflammatory factors, and inhibit tumor invasion and metastasis. In clinical applications, it can effectively alleviate clinical symptoms and improve the syndrome of lung dryness due to Yin deficiency.63,64 Sang Ye extracts from Fang Zhongzhong have potential anti-inflammatory and antioxidant effects, inhibiting the proliferation of non-small cell lung cancer and inducing the apoptosis of lung cancer cells.65,66 Sang Ye aqueous extract and ginseng saponin in QZJFD exert anti-inflammatory effects by inhibiting the release of pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α.67,68 Shi Gao can ameliorate inflammation by regulating body temperature and inhibiting IL-1β overexpression. 69 Pi Pa Ye triterpene acid can significantly reduce the lung index and the content of hydroxyproline (HYP) in rat lung tissue, enhance the activity of superoxide dismutase (SOD) in rat serum, improve the lung tissue structure of pulmonary fibrosis model rats, and reduce the degree of pulmonary fibrosis hyperplasia. 70 Gan Cao flavonoids and Pi Pa Ye extract exert anti-inflammatory effects by downregulating p38 MAPK and JNK protein expression.71,72 The Gan Cao hypo acid derivative-TY501, Xing Ren glycosides, and Mai Dong polysaccharides exert antifibrotic effects by blocking TGF-β expression and inhibiting the TGF-β/smad pathway.73,74 Liu et al 75 found that Equus asinus L. (E Jiao) can reduce infiltration by macrophages, maintain the alveolar structure, and inhibit the production of pro-inflammatory cytokines (TNF-α and IL-1β), while increasing the production of IL-10 in the lung, and regulating the metabolic pathway disorder of model rats with lung injury induced by 2.5-micrometer particulate matter (PM2.5), thereby protecting against lung damage. Studies have shown that ellagic acid downregulates the expression of TGF-β1, α-SMA, and collagen (types I and II), improving respiratory function in mice with bleomycin-induced pulmonary fibrosis, and reducing collagen deposition in lung tissue. 76 Thus, our findings, combined with other related studies suggest that QZJFD inhibits fibrosis and the inflammatory response induced by IPF by downregulating MAPK signaling pathway-mediated TGF-β expression. Furthermore, our data suggest that the therapeutic effect of QZJFD is positively correlated with the dose.
Conclusions
In this study, we identified the core components of QZHFD as quercetin, (-)-epicatechin ester, lysine, stigmasterol, and Kaempferol. We also identified a strong correlation between these components and key IPF targets, including ALB, AKT1, TNF, IL6, and TP53. Furthermore, our findings indicate that the anti-fibrosis and anti-inflammatory effects of QZHFD are mediated by the downregulation of MAPK signaling pathway-related proteins. Thus, our study provides the first insights into the mechanism of action of QZJFD on IPF, further expanding the consideration of TCM as an option for the prevention and treatment of IPF and providing a scientific and effective theoretical basis for the clinical diagnosis and treatment of this condition. This study only conducted correlation analysis on the 5 important components of QZHFD water decoction. However, it remains unclear which specific core compounds among the aforementioned 5 main active ingredients have an effect on IPF. In order to further elucidate the specific mechanisms by which QZHFD affects IPF and to provide an effective theoretical foundation for clinical treatment, we will continue to supplement our research with subsequent animal and cell experiments.
Materials and Methods
Animals
All rats were housed in a quiet, well-ventilated environment with daily lighting (12-h light/12-h dark), relative humidity of 50% to 60%, temperature of 25 °C, and free access to food, water, and drinking water. This study was approved by the Experimental Animal Ethics Committee of Ningxia Medical University, China (ethical approval number IACUC-NYLAC-2022-151).
Medications
QZJFD (Medical Gate Law): Sang Ye 9 g (Lot No. 201102), Shi Gao 8 g (Lot No. 2010041), Gan Cao 3 g (Lot No. 200904), Ren Shen 2 g (Lot No. 20092901, Hu Ma Ren 3 g (Lot No. 2203484), E Jiao 3 g (Lot No. 2101001), Mai Dong 4 g (Lot No. 2011068), Xing Ren 2 g (Lot No. 20102701), and Pi Pa Ye 3 g (Lot No. 20010801), all components were provided by the Ningxia Traditional Chinese Medicine Hospital and Institute of Traditional Chinese Medicine, and identified by Professor Gao Xiaojuan, School of Medicine, Ningxia Medical University. QZJFD was conventionally decocted twice, filtered, and concentrated to 1.12 g/mL. Using the human/rat body surface area-based dose conversion method, the rat dosages were calculated at 6.3 times the human dosage. This conversion yielded rat gastric administration amounts of 12.257, 6.137, and 3.069 g·kg−1 as the high, medium, and low QZJFD dosage groups, respectively. 77 Bleomycin hydrochloride and pirfenidone were obtained from MedChemexpress (HY-17565A) and Beijing Continent Pharmaceuticals (Lot No. 20210319).
Reagents and Instruments
Rat IL-6, TNF-α, and IL-1β ELISA kits (Multi Sciences Biotech, lot numbers: EK306, EK382, and EK301BHS, respectively). Rabbit fluorescent secondary antibody-green, rabbit fluorescent secondary antibody-red (Beijing Zhongsun Golden Bridge Biotechnology, Lot Nos. ZF0511, and ZF0516, respectively). Goat anti-rabbit secondary antibody and goat anti-mouse secondary antibody (Beijing Zhongsun Golden Bridge Biotechnology, Lot Nos: ZB2301 and ZB2305, respectively). Transforming growth factor-β (TGF-β), α-smooth muscle actin (α-SMA), protein-38 (P38), p-P38, Jun N-terminal kinase 1 (JNK1), p-JNK1, glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Affinity, Lot Nos. AD1027, AF1032, AF6456, AF4001, AF6318, AF3318, and AF7021, respectively). LC-MS grade methanol (MeOH) was purchased from Fisher Scientific (Loughborough, UK). 2-Amino-3-(2-chloro-phenyl)-propionic acid was obtained from Aladdin (Shanghai, China). Ultrapure water was generated using a Milli-Q system (Millipore, Bedford, USA). LC-MS grade acetonitrile (ACN) was purchased from Fisher Scientific (Loughborough, UK). Formic acid was obtained from TCI (Shanghai, China). Ammonium formate was obtained from Sigma-Aldrich (Shanghai, China). Weighing scales (BP310P type, Sartorius, Germany). Low-temperature high-speed centrifuge (5417R type, Eppendorf, Germany). Nucleic acid-protein quantification instrument (DS-11, Denovix ultra-micro UV–visible spectrophotometer, USA). Electrophoresis apparatus (DYCZ-24DN type, Beijing Liuyi Company). High-speed centrifuge was obtained from Hunan Xiangyi Experiment Equipment Co., Ltd (Hunan, China). The centrifugal vacuum evaporator was from Eppendorf China Ltd (Shanghai, China). The vortex mixer was obtained from Haimen Kylin-bell Lab Instruments Co., Ltd (Haimen, China). Microporous membrane filters (0.22 µm) were purchased from Tianjin Jinteng Experiment Equipment Co., Ltd (Tianjin, China). HPLC/UHPLC (Thermo, Vanquish, USA).
Network Pharmacology
QZJFD Active Ingredient Collection and Target Acquisition
The targets of the active ingredients of QZJFD were obtained by searches of The Chinese Medicine Systems Pharmacology database and analysis platform (TCMSP, http://tcmspw.com/tcmsp.php) using “Sang Ye, Shi Gao, Gan Cao, Ren Shen, Hu Ma Ren, E Jiao, Mai Dong, Xing Ren, Pi Pa Ye” as search terms. Shi Gao, Mai Dong, and E Jiao were not identified in the TCMSP. Oral bioavailability (OB) ≥ 30% and drug-likeness (DL) ≥ 0.18 were set as the key indicators for screening to obtain the active ingredient data set. 78 The target genes of the active ingredients were predicted by the DrugBank database. The Chinese herbal medicines “Shi Gao, Mai Dong and E Jiao” were searched in The Bioinformatics Analysis Tool for Molecular mechANism of Traditional Chinese Medicine (BATMAN, http://bionet.ncpsb.org.cn/). Shi Gao was searched using the corresponding InChI in PubChem database (https://pubchem.ncbi.nlm.nih.gov), and the names of the other Chinese medicines were converted into pinyin for searches to identify the active ingredients and their targets; target genes with scores > 20 were selected. The genes encoded by the proteins identified as the targets of the active ingredients of QZJFD were searched (restricted to Homo sapiens) in the UniProt database (https://www.uniprot.org/).
Acquisition of IPF-Related Targets
The target genes related to IPF were collected by GeneCards (https://www.genecards.org/) and GeneMap (https://omim.org/search/advanced/geneMap) in The Online Mendelian Inheritance in Man (OMIM, https://omim.org/) using “Idiopathic pulmonary fibrosis.” After collection, the disease-related target genes were obtained by merging and de-duplication.
Construction of the QZJFD-Compound-Target-IPF Network
The intersection of the target genes related to the active ingredients of QZJFD and IPF was screened using the Venny2.1 online software mapping tool platform. The QZJFD-compound-target-IPF network was constructed by importing Chinese herbal medicines, active ingredients, key targets, and diseases into Cytoscape 3.9.0 based on the correspondence and properties.
Construction and Analysis of PPI Networks for Key Targets
The intersecting drug targets and IPF targets were uploaded to the STRING database (https://cn.string-db.org/) to construct the PPI network, with the species restricted to Homo sapiens. The PPI network TSV file was then imported into Cytoscape 3.9.0 to visualize the PPI network.
Analysis of Key Target Pathways and Functional Enrichment
The key targets (P < .05) identified in the screening process were imported into The Database for Annotation, Visualization, and Integrated Discovery (DAVID, https://david.ncifcrf.gov) for Gene Ontology (GO http://www.geneontology.org) function and the Kyoto Encyclopedia of Genes and Genomes (KEGG, https://www.genome.jp/kegg/) enrichment analyses.
Molecular Docking Simulation
The top 10 key active ingredients in the drug-component-target-disease regulatory network and the top 5 target genes in terms of the degree value corresponding to the active ingredient in the PPI network analysis were selected for validation by molecular docking simulation. The 3D structures of key target proteins were downloaded from the Protein Data Bank database (PDB, https://www.rcsb.org/), and the SDF files of key active ingredients were downloaded from the PubChem database and converted to the PDBQT format using Open Babel 3.1.1. The key target proteins were dehydrated and hydrogenated using PyMOL Version 2.0 (https://www.pymol.org/). Using the protein as the receptor and the small molecule as the ligand, the active pocket with the highest drug score was selected as the active site for molecular docking simulation according to the Proteins Plus prediction results. The binding ability was predicted using AutoDock Vina software, with lower binding energy reflecting greater stability of the interaction. 79 The top 3 groups of proteins with the best binding activity were selected and the interaction with the active ingredient was visualized using PyMOL Version 2.0.
Animal Experiments
Animal Grouping, Modeling, and Drug Administration
Forty-eight male Sprague–Dawley rats were randomly divided into blank, model, pirfenidone, QZJFD low-dose, QZJFD medium-dose, and QZJFD high-dose groups (n = 8 rats per group). The pulmonary fibrosis model was established as previously described. 80 Briefly, animals were anesthetized by intraperitoneal injection of 2% sodium pentobarbital (3 mL/kg), the neck was disinfected with iodophor and the trachea was exposed via a neck incision. Rats were then placed in the vertical position and saline was injected into the trachea of the rats in the blank group, while the remaining 40 rats were injected with bleomycin/saline solution (5 mg/kg). The skin was sutured and disinfected. After 3 days, rats in the blank and model groups received saline by oral gavage, while rats in the pirfenidone group received pirfenidone (100 mg/kg), 81 and rats in the QZJFD high-, medium-, and low-dose groups received QZJFD at 12.257, 6.137, and 3.069 g·kg−1 of the drug solution, respectively, for 21 days. 82 At 12 h after the final administration, the rats were anesthetized with 3% sodium pentobarbital intraperitoneally. Blood (2 mL) was sampled from the abdominal aorta and then centrifuged (3000 rpm for 10 min at 4 °C). The serum was collected and stored at −80 °C prior to analysis. Rats were then sacrificed by intraperitoneal injection of 3% sodium pentobarbital. For each rat, the right lung tissue was excised into a freezing tube and stored at −80 °C. The left lung tissue was fixed in 4% paraformaldehyde.
Serum Inflammatory Factor Levels
Serum levels of IL-6, TNF-α, and IL-1β of rats were measured using ELISA kits according to the manufacturer's instructions.
Histomorphological Observation of Lung Tissue
Paraformaldehyde fixed lung tissues were routinely dehydrated, and embedded in paraffin, and sections (4 μm thickness) were prepared. The sections were stained with hematoxylin and eosin (HE) and Masson's trichrome. Histomorphological changes were evaluated under a light microscope (200 × magnification).
Immunofluorescence Labeling of α-SMA, TGF-β1, JNK, p-JNK, P38 MAPK, p-P38 MAPK, Collagen I, and FN1 in Lung Tissue
Paraformaldehyde-fixed lung tissues were routinely dehydrated and embedded in paraffin. Following the dewaxing wash, high-temperature antigen repair was performed with a 3% citric acid antigen repair solution, followed by incubation in 5% bovine serum albumin (BSA) for 30 min at room temperature. Sections were then stained overnight at 4 °C with primary antibodies (P38, 1:100; α-SMA, 1:150; p-P38, 1:100; p-JNK, 1:90; JNK, 1:100; TGF-β, 1:100; collagen I, 1:150; and FN1, 1:200). Sections were then washed 3 times with phosphate buffered saline (PBS) (5 min/wash) and incubated for 30 min at 37 °C with the corresponding fluorescently-labeled secondary antibody. After washing 3 times with PBS, the sections were washed with tap water, sealed with a cover slip, and observed under a fluorescence microscope. The nucleus was defined by blue fluorescence and the target protein was defined by red/green fluorescence.83,84
Western Blot Analysis of α-SMA, TGF-β1, JNK, p-JNK, P38 MAPK, p-P38 MAPK, Collagen I, and FN1 Expression in Lung Tissue
Tissue (50 g) was ground to a fine powder in liquid nitrogen and incubated overnight at 4 °C in radioimmunoprecipitation (RIPA) lysis buffer to extract the total proteins. After centrifugation (12 000 rpm for 10 min at 4 °C), the supernatant was collected and proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) before transfer to a polyvinylidene difluoride (PVDF) membrane at room temperature with slow shaking for 2 h. The membrane was then incubated overnight at 4 °C with primary antibodies for the detection of P38 (1:1200), p-P38 (1:800), α-SMA (1:2000), JNK (1:1000), p-JNK (1:1300), TGF-β (1:1000), collagen I (1:1000), and FN1 (1:1000). The membrane was then washed 3 times with TBS + Tween (TBST) before incubation with the corresponding secondary antibody at room temperature. After washing 3 times with TBST, protein bands were visualized using an enhanced chemiluminescent (ECL) kit and quantified by measurement of grayscale values using Image-J software.
Statistical Analysis
SPSS 22.0 software was used for statistical analysis. Data were expressed as mean ± standard deviation (SD). Differences between groups were evaluated by one-way ANOVA and P < .05 was considered to indicate statistical significance.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X241242274 - Supplemental material for Investigation of the Mechanism of Action of Qingzaojiufei Decoction on Idiopathic Pulmonary Fibrosis Based on Network Pharmacology and Experimental Validation
Supplemental material, sj-doc-1-npx-10.1177_1934578X241242274 for Investigation of the Mechanism of Action of Qingzaojiufei Decoction on Idiopathic Pulmonary Fibrosis Based on Network Pharmacology and Experimental Validation by Chunyang Mao, Yan Du, Kun Niu, Fan Yang and Changcai Bai, Bo Zhou, Yang Niu in Natural Product Communications
Footnotes
Abbreviations
Acknowledgments
Thank you to the Experimental Animal Center of Ningxia Medical University and Ningxia Regional Key Laboratory of Integrated Traditional Chinese and Western Medicine for the Prevention and Treatment of Regional High Incidence Disease for providing platforms and experimental sites.
Authors’ Contributions
Yang Niu and Chunyang Mao: conceptualization; Yan Du: data curation; Kun Niu: formal analysis; Chunyang Mao, Yang Niu, and Bo Zhou: funding acquisition; Kun Niu and Fan Yang: methodology; Bo Zhou and Yang Niu: supervision; Changcai Bai: validation; Yan Du: writing–original draft preparation; Chunyang Mao: writing–review and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ningxia Medical University Animal Ethics Committee. Ethical approval to report this case was obtained from Ningxia Medical University of Ethics Committee (IACUC-NYLAC-2022-151).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Ningxia Natural Science Foundation [2023AAC03212]; Ningxia Medical University 2021 University-level Key Research Project [XZ2021006]; Ningxia Minority Medicine Modernization Key Laboratory of the Ministry of Education/Ningxia Regional High Morbidity Combined Chinese and Western Medicine Prevention and Treatment Research Key Laboratory 2022 [20023302]; National Natural Science Foundation of China Joint Fund for Regional Innovation Development [U20A20404].
Statement of Human and Animal Rights
There are no human subjects in this article and human and animal rights are not applicable.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
