Abstract
Keywords
Introduction
Pneumonia accounted for nearly 2.5 million fatalities globally in 2019, ranking as the fourth leading cause of death worldwide. 1 A total of 544 000 hospitalizations from the emergency department are attributed to community-acquired pneumonia (CAP) per year. 2 Relevant reports have received much attention in recent years, attracting scientists to explore new and effective methods to treat pneumonia. Pneumonia is a prevalent acute respiratory illness that is characterized by inflammation of the distal lung regions, specifically the alveoli and bronchial tree. 3 Pneumonia is generally caused by infection with microorganisms, and current antibiotic regimens for the empiric treatment of CAP include monotherapy with respiratory fluoroquinolone or combination therapy with β-lactam and macrolide antibiotics. 4 Although many microbiological infectious diseases can be treated with these regimens, they may also contribute to the emergence of antibiotic resistance. 5 With the widespread implementation of medicines and vaccines, pneumonia continues to have high death and disability rates, placing a huge burden on global health.6,7 The failure to incorporate immunocompromised people in clinical guidelines and treatment trials exacerbates the situation. 8 Researchers are gradually concentrating increasingly on traditional Chinese medicine (TCM) to find a cure that will work and prevent the emergence of drug resistance.
The Huangqin Qingfei decoction (HQFD), which originated from Health Treasure (Weisheng Baojian in China) in the Yuan Dynasty, was formulated by Luo Tianyi, a follower of Li Dong-Yuan (the founder of the theory of the spleen and stomach). The herbal medicines of the HQFD include 2 TCMs, namely Scutellaria baicalensis Georgi and Gardenia jasminoides J.Ellis. The HQFD invigorates the lungs, detoxifies the body, and mitigates dryness and heat. Pharmacological investigations have demonstrated that the HQFD has anti-inflammatory, antiviral, antibacterial, anticancer, antioxidant, and antiapoptotic properties.9–11 The prescription is extensively used clinically to manage pulmonary inflammation, neurological disorders, and neuronal dysfunction, as well as various cardiovascular and cerebrovascular conditions.12,13 However, the precise mechanisms underlying the synergistic effects of these 2 TCMs remain incompletely understood. As such, further investigation is required to elucidate their anti-inflammatory and antibacterial properties.
Network pharmacology can be used to study the components of TCMs, and it has been increasingly used in recent years. 14 With the advancement of systems biology research, the concept of network pharmacology has revolutionized the conventional approach of “one target, one drug,” instead favoring a “network-target, multiple-component-therapeutics” strategy. 15 The intricate chemical compounds and multifaceted compositions of TCMs present a challenge in comprehending their molecular mechanisms, given the lack of effective research methods available for investigation. Network pharmacology can be used to address scientific obstacles in a systematic manner and at multiple levels, while the holistic perspective of TCM aligns with the contemporary medical research paradigm that emphasizes network and systems analyses. 16
In this study, we used network pharmacology to identify the main targets and signaling pathways affected by the components of the HQFD in the treatment of pneumonia. We also aimed to elucidate the molecular biological mechanisms of the HQFD to provide pertinent references for subsequent research. Moreover, we verified the mechanism of lung protection with the HQFD in animal experiments and described the effective ingredients of the HQFD and their mechanisms in the treatment of pneumonia.
Network Pharmacology Analysis Materials and Methods
Identification of the Active Ingredients and Potential Targets of the HQFD
The active ingredients of the HQFD and the corresponding targets of Scutellaria baicalensis Georgi and Gardenia jasminoides J.Ellis were retrieved from the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP; https://old.tcmsp-e.com/tcmsp.php). 17 Oral bioavailability (OB) was defined by quantifying the absorption and systemic distribution of the active ingredients. Drug-likeness (DL) was determined by evaluating the chemical resemblance between the drug and known pharmaceutical compounds. Oral bioavailability and DL were considered as the most important constraints to evaluate drug development; therefore, we selected an OB of ≥30% and a DL of ≥0.18 as pharmacokinetic data-retrieving information according to absorption, distribution, metabolism, excretion criteria. 18 Subsequently, the UniProt website (https://www.UniProt.org) was utilized to standardize gene identification and get target abbreviations for the targets. The main constituents of the HQFD were identified along with their respective target proteins.
Identification of Targets in Pneumonia Treatment
The study systematically identified potential therapeutic targets for pneumonia through comprehensive searches across multiple databases, including GeneCards (https://www.genecards.org/), the Therapeutic Target Database (https://idrblab.net), and Online Mendelian Inheritance in Man (https://omim.org/).19,20 The resulting targets were then integrated, and duplicates were removed to prepare for the construction of the protein–protein interaction (PPI) network in the subsequent step.
Establishment of the Active Ingredients and Their Corresponding Targets and PPI Network Construction
Active ingredient targets in the HQFD and pneumonia targets were imported into the Venn website (http://www.bioinformatics.com.cn/static/others/jvenn/example.html) to obtain overlapping targets for drugs and diseases. The STRING database (https://string-db.org/) was used to access protein interaction network data by importing the targets identified in the intersection. The filters were configured to retrieve organism-specific information for Homo sapiens. 21
Cytoscape is a network data integration, analysis, and visualization software that can integrate biomolecular interaction networks, especially PPIs, protein–DNA interactions, and genetic interactions. 22 Cytoscape software is generally used for network pharmacology prediction.
For visualization, Cytoscape 3.9.1 was used to import the active ingredients and corresponding drug targets (joint drug targets) of the HQFD, enabling the construction of a network depicting the interconnections between the active ingredients of the HQFD and their targets. The PPI data were entered into Cytoscape 3.9.1 to generate a visual representation, and the PPI network was constructed. From the PPI network graph, Cytoscape 3.9.1 software was used to calculate the parameters of each node in the network, including degree centrality (DC), closeness centrality (CC), and betweenness centrality (BC). According to the DC values, the nodes in the PPI network were ranked, and 4 core targets with high scores were selected for further investigation. 23
Meta-analysis of Clustered Biological Terms
With the aim of exploring PPI the biological functions of pneumonia targets, Metascape (http://metascape.org/) was used to conduct the Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses. Gene Ontology functions with P values of <.01 were screened. 24 The KEGG enrichment analysis facilitated the identification of biologically relevant processes that encompass significant signaling pathways. Subsequently, a bioinformatics platform was used to visualize the GO and KEGG pathway analyses.
Molecular Docking Analysis
As a computational approach that facilitates the investigation of how small-molecule ligands interact with receptor biomacromolecules, molecular docking enables the prediction of affinity strength and binding mode, which is of great importance in drug research. 25 Using standard molecular docking methods, we verified whether the core proteins identified by network pharmacology exhibited associations with the active ingredients of the HQFD. 26
Network Pharmacology Analysis Result
Compound Composition and Target Analysis of the HQFD
Thirty-six highly active components from Scutellaria baicalensis Georgi and 15 from Gardenia jasminoides J.Ellis were identified after previous screening. Table 1 displays the target details for part ingredients.
Part of the Active Ingredient in HQFD.
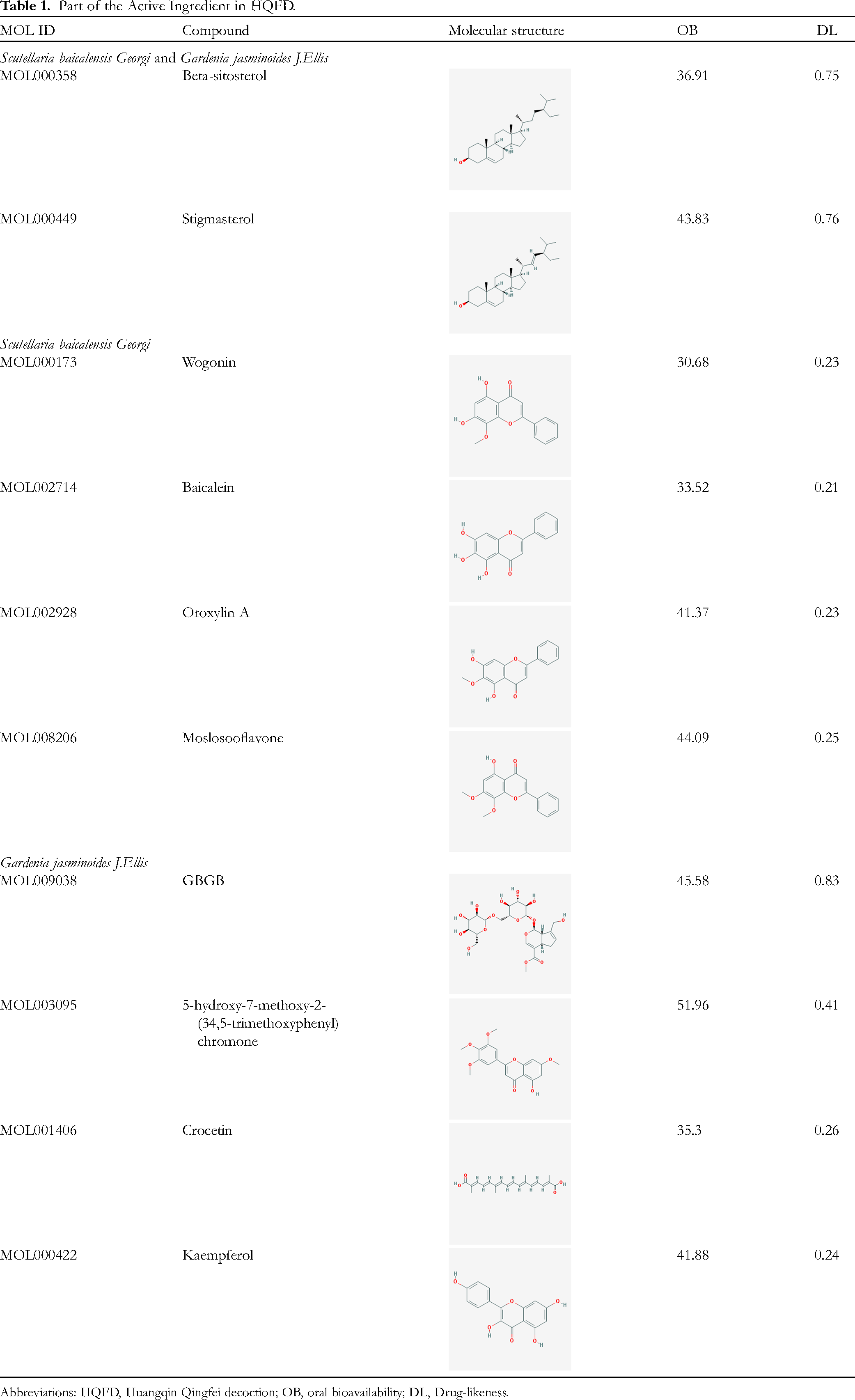
Abbreviations: HQFD, Huangqin Qingfei decoction; OB, oral bioavailability; DL, Drug-likeness.
In the compound–target–disease network analysis, 42 compounds and 89 intersection targets were selected from the compounds with a large number of intersections. We found that beta-sitosterol and Stigmasterol both in Scutellaria baicalensis Georgi and Gardenia jasminoides J.Ellis, wogonin, baicalein, and oroxylin A in Scutellaria baicalensis Georgi, quercetin, and kaempferol in Gardenia jasminoides J.Ellis regulated multiple targets. The characteristics of the components regulating the same target, which indicate the multicomponent and multitarget functions of the HQFD, are shown in Figure 1A.

(a) Compounds-targets-disease network the 28 yellow ellipses represent the components of Scutellaria baicalensis Georgi, and the 10 green ellipses represent the components of Gardenia jasminoides J.Ellis. The 2 red ellipses represent the components that are common between Scutellaria baicalensis Georgi and Gardenia jasminoides J.Ellis. The pink rectangles represent the 89 targets of the HQFD (Intersection of Scutellaria baicalensis Georgi and Gardenia jasminoides J.Ellis). Blue rectangles represent pneumonia and are associated with targets. The component with the highest correlation was beta-sitosterol, and the target with the highest correlation was PTGS2. (b and c) Venn diagram of the targets of the HQFD and the targets of pneumonia. There were 117 targets unique to HQFD, 1901 targets unique to pneumonia, and 96 targets shared by HQFD and pneumonia. (d) Depiction of the PPI network involving the HQFD and pneumonia targets. The ellipses represent proteins targeted by both pneumonia and HQFD. The target protein with the highest correlation was AKT1. (e) Target–pathway interaction network. The yellow triangles represent the targets, and the blue ellipses represent the pathways. The target protein with the highest correlation was RELA. The pathway with the highest correlation was pathways in cancer. HQFD, Huangqin Qingfei decoction; PPI, protein–protein interaction.
Protein–Protein Interaction Network Construction and Analysis
A total of 1997 pneumonia targets were identified by searching GeneCards and other databases. The common targets of the ingredients in the HQFD were mapped with pneumonia targets using bioinformatics to draw a Venn diagram (Figure 1B-C), with 32 common targets obtained after mapping. The results indicate that the HQFD has the potential to treat pneumonia through multiple targets. The PPI network was created by visualizing the common targets, as shown in Figure 1D. The DC values of the nodes in the graph determine whether the targets are core targets. We identified part of the important targets based on the DC, BC, and CC values (Table 2).
Some Important Targets and Information From the PPI Network.
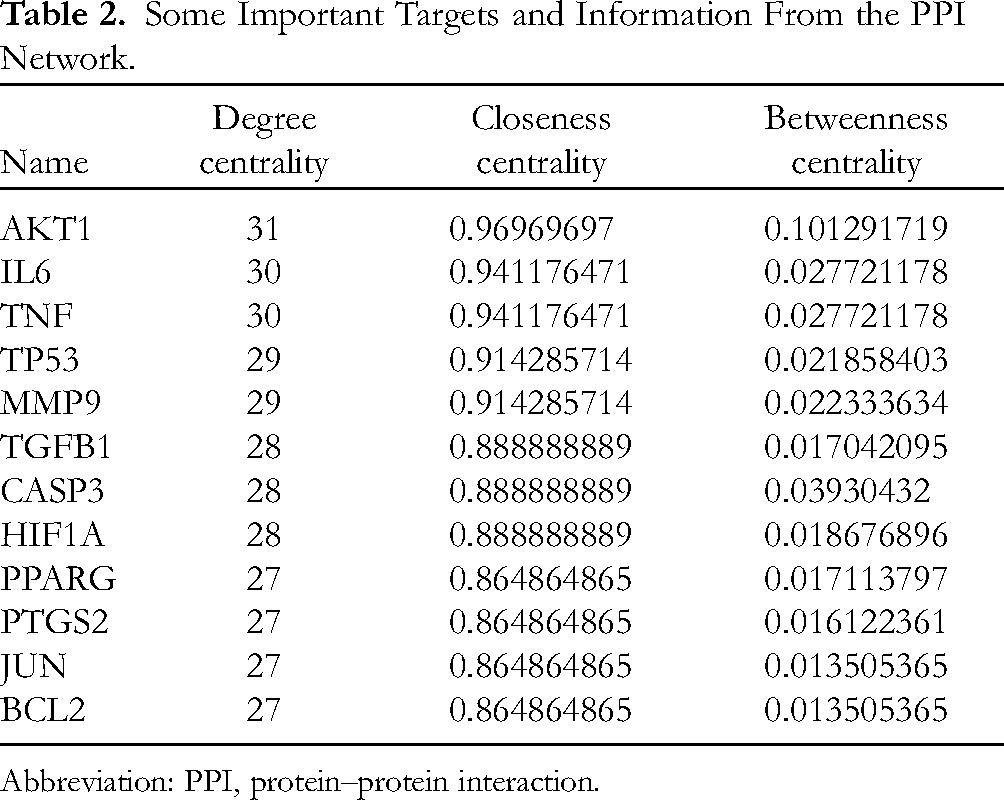
Abbreviation: PPI, protein–protein interaction.
Gene Ontology and KEGG Enrichment Analyses
As shown in Figure 2A, we screened 20 signaling pathways in the KEGG enrichment analysis to explore the signaling mechanisms of the HQFD in pneumonia treatment. We identified the pathways in cancer, lipid and atherosclerosis, and the advanced glycosylation end products and receptor of advanced glycosylation end products (AGE-RAGE) signaling pathway in diabetic complications and so on. As shown in Figure 2B, the GO analysis showed that the HQFD plays an important role in pneumonia treatment, which is related to the response to lipopolysaccharide (LPS), the response to molecule of bacterial origin, the tube morphogenesis, and the response to decreased oxygen levels and other biological processes.

(a) Top 20 KEGG pathways of the hub genes. The pathway with the lowest P value was the pathway in cancer. (b) GO terms of hub genes. The GO biological process with the highest P value was the response to lipopolysaccharide. The GO biological process with the lowest P value was the response to lipopolysaccharide. The GO cellular component with the lowest P value was the membrane raft. The GO molecular function with the lowest P value was cytokine receptor binding. GO, Gene Ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes.
Target–Pathway Interaction Network Construction
We imported the enriched pathways and their corresponding genes into Cytoscape software for analysis. The target–pathway interaction network is constructed in Figure 1E. The analysis result showed that AKT1, AR, BAX, and CCND1 play an essential part in the signaling pathway.
Molecular Docking
According to the network pharmacology results, we screened the top 3 compounds (beta-sitosterol, wogonin, and quercetin). Then, the top 3 compounds were conjugated with the 4 important proteins (AKT1 [PDB ID: 5AAR], PTGS2 [PDB ID: 4COX], MAPK14 [PDB ID: 1KV1], and MMP9 [PDB ID: 5TH6]). The docking fraction between the protein and the compound is presented in Table 3. The docking cases with the lowest binding energy were chosen as the best docking images of the receptor and ligand after visualization (Figure 3A-I).

(a-i) Schematic of docking between the active compounds of the HQFD and AKT, PTGS2, MAPK14, and MMP9. (a) Beta-sitosterol—AKT1 (b) Beta-sitosterol—MAPK14 (c) Beta-sitosterol—MMP9 (d) Beta-sitosterol—PTGS2 (e) Wogonin—AKT1 (f) Wogonin—MAPK14 (g) Wogonin—MMP9 (h) Wogonin—PTGS2 (i) Quercetin - AKT1 (j) Quercetin—MAPK14 (k) Quercetin—MMP9 (l) Quercetin—PTGS2. The orange structural formula is the chemical composition of small molecules, the blue 3-dimensional structure is the protein, the red structural formula is the amino acid residues connected by hydrogen bonds in the protein molecules, and the name of the amino acid residues is shown. The yellow bonds are the hydrogen bonds corresponding to the protein of the small molecule, and the numbers above are the lengths of the hydrogen bonds. The 3 pairs with the lowest binding energy were PTGS2 and beta-sitosterol, MMP9 and beta-sitosterol, and MAPK14 and quercetin. (M) Heatmap of binding energies. HQFD, Huangqin Qingfei decoction.
The results show that beta-sitosterol interacts with AKT1, PTGS2, MAPK14, and MMP9 (Figure 3A-D). Figure 4A shows that beta-sitosterol interacts with Glu699, through the establishment of a solitary hydrogen bond in AKT1. Similarly, Figure 4B illustrates that beta-sitosterol forms one hydrogen bond with Tyr342 in MAPK14. In Figure 3C, beta-sitosterol does not form hydrogen bonds with MMP9 in the selected molecular conformation. As depicted in Figure 4D, beta-sitosterol establishes one hydrogen bond with Lys468 in PTGS2.
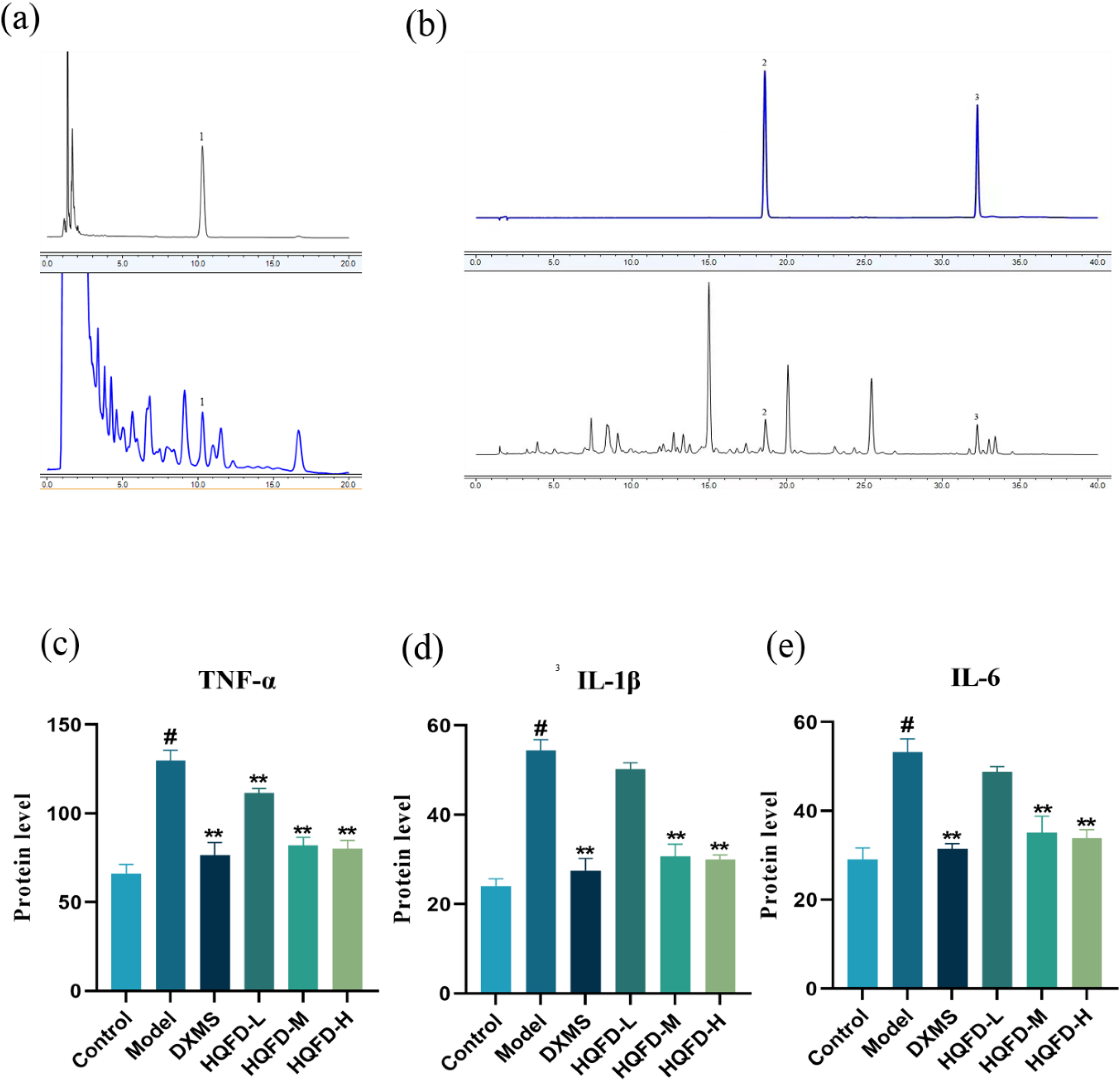
(a) Chromatogram of HQFD in 206 nm mode. 1 denotes β-sitosterol; 2 denotes quercetin; 3 denotes wogonin. (b) Chromatogram of HQFD in 350 nm mode. (c-E) Effect of the HQFD on TNF-α, IL-1β andreceptor–ligand IL-6 cytokines expression by ELISA. Compared with the control group, the cytokines expression levels of TNF-α, IL-1β, and IL-6 in the model group were significantly increased, while those in the medium- and high-dose HQFD groups were significantly decreased compared to the model group. # P < .05 versus control group, *P < .05 versus model group, **P < .01 versus model group. HQFD, Huangqin Qingfei decoction; ELISA, enzyme-linked immunosorbent assay.
Wogonin interacts with AKT1, PTGS2, MAPK14, and MMP9 (Figure 3E-I). Figure 4E shows that wogonin forms one hydrogen bond with Cys523 and Ser547 in AKT1 while interacting with Leu522 through 2 hydrogen bonds. Similarly, in Figure 4F, wogonin interacts with Ser293 via one hydrogen bond, as well as Asp292 and Leu291 through 2 hydrogen bonds, in MAPK14. Figure 4G shows that wogonin forms a hydrogen bond with Pro415 and His432 in MMP9. Figure 4H shows that wogonin interacts with Trp323, Gln327, and Tyr130 along with Glu46 via one hydrogen bond, while also interacting with Ser49 through 2 hydrogen bonds in PTGS2.
The results also show that quercetin interacts with AKT1, PTGS2, MAPK14, and MMP9 (Figure 3I-L). The configuration of quercetin in Figure 3I suggests its potential to engage in a single hydrogen bond interaction with Ala627, while simultaneously having 2 hydrogen bonds with Glu669, Leu699, and Phe700 in ATK1. In Figure 3J, the presence of one hydrogen bond is demonstrated between quercetin and Leu195, Ser252, as well as Ser251, while simultaneously 2 hydrogen bonds are demonstrated between quercetin and Leu291, Lys249 in MAPK14. MMP9 forms one hydrogen bond with Pro429 and Tyr420, while also establishing 2 hydrogen bonds with His432 and Pro430 (Figure 3K). Finally, PTGS2 establishes one hydrogen bond with Ser471, Cys41, and Asn39, and 2 hydrogen bonds with Gln461 and Asn43 during its interaction with quercetin (Figure 3L).
A common approach used to evaluate the affinity between ingredients and protein targets is binding energy computation, which can also serve as an indicator of the probability of a receptor–ligand interaction. It is generally accepted that when the binding energy between the ligand and the receptor is less than 0, the 2 can bind spontaneously. The lower the binding energy between the ligand and the receptor, the higher the stability of the conformation and the higher the affinity between the ligand and the receptor. The binding energy results obtained by molecular docking are shown in Table 3. To visualize the binding affinity between the ligand and the receptor, a heatmap was generated (Figure 3M).
The Energy Required for Binding Between the HQFD Compounds and the Core Targets (kcal/mol).

Abbreviations: HQFD, Huangqin Qingfei decoction; COX, cyclooxygenase.
Among the tested compounds, beta-sitosterol exhibited the strongest binding affinity with PTGS2 (−11.44 kcal/mol). Quercetin exhibited the weakest binding affinity with AKT1 (−5.71 kcal/mol). The 3 compounds (beta-sitosterol, wogonin, and quercetin) exhibited good binding affinity with the 4 core targets (AKT1, PTGS2, MAKP14, and MMP9). Based on these results, it is speculated that all of these compounds play a significant role in mediating the therapeutic effects of the HQFD in pneumonia.
Animal Experiments Validation Materials and Methods
Materials
Experiment Animals
Sixty ICR mice weighing 32.5 ± 2.5 g were procured from Zhejiang Chinese Medical University and maintained in a strictly controlled pathogen-free environment. The mice were housed in a breeding environment at a consistent temperature of 22 °C to 26 °C and a relative humidity of between 48% and 56%. The mice were subjected to a light/dark cycle of equal duration, with each period lasting for 12 h. Before the commencement of the experiment, an acclimatization period of 1 week was designated for the mice to adjust to the standard laboratory conditions. During the acclimatization period, the mice were provided with ad libitum access to a regular diet and water supply.
Reagents and Equipment
The medicinal ingredients of Scutellaria baicalensis Georgi and Gardenia jasminoides J.Ellis were procured from Hangzhou Hospital of Traditional Chinese Medicine and authenticated by Associate Professor Chen Jin from the College of Biology Science at Zhejiang Chinese Medicine University. Scutellaria baicalensis Georgi 30 g and Gardenia jasminoides J.Ellis 6 g were soaked in tap water for 1 h. Then, the condensation reflux method was used to boil the Chinese medicine twice for 2 h each time. The liquid was merged twice and concentrated with a rotary evaporator to 90 mL. Based on the ratio of human to mouse surface area, the prescription was converted into 3 concentrations (high, medium, and low) for human clinical use. The high dose was 3.12 g/kg, the medium dose was 1.56 g/kg, and the low dose was 0.78 g/kg.
The RNase-Free DNase Set (Qiagen), TRIzol Plus RNA Purification Kit (Thermo Fisher Scientific), Power SYBR Green PCR Master Mix (Applied Biosystems), SuperScript III First-Strand Synthesis SuperMix (Thermo Fisher Scientific), 30% acrylamide solution (Bio-Rad), Halt Protease and Phosphatase Inhibitor Cocktail (100X) (Thermo Pierce), T-PER Tissue Protein Extraction Reagent (Thermo Pierce), bicinchoninic acid protein concentration determination kit (Beyotime Biotechnology), SuperSignal West Dura Extended Duration Substrate (Thermo Pierce), ECL DualVue WB Marker (GE Healthcare), polyvinylidene fluoride (PVDF) transfer printing film (Millipore), x-ray film (Huadong Medicine Co. Ltd), and bovine serum albumin (Sangon Biotech) were used in the experiments.
Methods
Animal Grouping and Intervention
Female and male ICR mice (30 females, 30 males) at 4 to 6 weeks of age and weighing 30 to 34 g were obtained from Shanghai BK Company. The mice were randomly divided into 6 groups, including the control group, model group, positive control group (namely DXMS group), and Chinese medicine group (namely HQFD-high, -medium, and -low group). Each group had 5 female and 5 male mice. The mice were administered by gavage once daily for 7 days. The dose of the positive drug was obtained by converting the body surface area of humans and mice. The positive control group was administered 2 mg/kg dexamethasone, the Chinese medicine groups were administered the doses specified above, and the control and model groups were administered equal amounts of saline.
In the study, the mice were anesthetized by intraperitoneal injection of atropine solution at 0.02 mL/10 g, and then LPS solution was inhaled at 20 g/kg using a 12-gauge mouse gavage needle. 27 The head of the gavage needle was inserted approximately 1 cm into the tracheal orifice, which is the epiglottis cartilage in mice. The required dose of LPS suspension was slowly injected, and the mice were kept upright until the liquid infusion was completed. The cannula was withdrawn immediately after completion of the installation. If moist rales could be heard during breathing, this indicated that the installation was successful, and the mice were resuscitated and returned to the original cage for 24 h. Twelve hours before the induction of pneumonia, the mice were fasted. After inducing 12 h, we collected the lung tissues from the mice.
The Analysis of Active Components of HQFD by High-Performance Liquid Chromatography
To qualitatively analyze the effective components of HQFD in network pharmacology, Scutellaria baicalensis Georgi and Gardenia jasminoides J.Ellis were pulverized (through a sieve with a mesh size of 2). Subsequently, 1 g of each powder was accurately weighed and dissolved in methanol. The resulting mixture was subjected to extraction at 50 °C using a 200 W ultrasonic bath for 30 min. After filtration, the obtained filtrate was dried using a water bath and further filtered with 10 mL of methanol through a 0.22-μm filter membrane. The collected filtrate was then used for high-performance liquid chromatography (HPLC) analysis.
High-performance liquid chromatography analysis was performed by Thermo U3000 UHPLC system (Thermo) and the column employed for the analysis was an Agilent Technologies E clipse XDB-C18 column (4.6 × 150 mm, 5 μm). The β-sitosterol was subjected to isocratic elution for 20 min using a mobile phase composed of methanol. The column oven temperature was maintained at 30 °C, with an injection volume of 10 μL and a flow rate of 1.0 mL/min. Chromatograms were recorded at a wavelength of 206 nm. The gradient elution of quercetin and wogonin was performed using a mobile phase consisting of 0.1% aqueous phosphoric acid (mobile phase A) and acetonitrile (mobile phase B). The programmed gradient was as follows: 0 to 25 min, 15% to 35% B; 25 to 35 min, 35% to 55% B; 35 to 35.1 min, 55% to 15% B; 35.1 to 40 min, with a final concentration of 15% B. The column oven temperature was maintained at 30 °C, while the injection volume and flow rate were set at 10 μL and 1.0 mL/min, respectively. Chromatograms were recorded at a wavelength of 350 nm.
The Quantification of Inflammatory Cytokines IL-1β, IL-6, and TNF-α by Using Enzyme-Linked Immunosorbent Assay Technique
The lung tissue was homogenized and centrifuged to extract the supernatant, followed by quantification of TNF-α, IL-1β, and IL-6 levels using enzyme-linked immunosorbent assay (ELISA) kits specifically for TNF-α, IL-1β, and IL-6 in the lung tissue.
The Observation of Pathological Damage of the Mice by Hematoxylin and Eosin Staining
The right lower lung was fixed for paraffin sections and stained with hematoxylin and eosin (H&E). The degree of lung tissue damage and inflammatory cell infiltration was compared between the groups.
The Analysis of mRNA Expression Levels of Anti-Inflammatory Pathway Targets of HQFD by Real-Time Polymerase Chain Reaction
The right upper lung was homogenized to measure the mRNA levels of MMP9, PTGS2, AKT1, and MAPK14 were detected by polymerase chain reaction (PCR) in lung tissue homogenates to observe the effect of HQFD on the mRNA expression of anti-inflammatory pathway targets. The method is as follows: The standard RNA extraction method using TRIzol (Invitrogen Life Technologies) was used to isolate total RNA from the tissue samples. The content, purity, and quality of the RNA were determined by ultraviolet spectrophotometry and electrophoresis. Reverse transcription was performed on all extracted RNA. The reaction system was supplemented with 10 μL 2 × RT Reaction Mix and 2 μL RT Enzyme Mix, followed by the addition of RNase-free water to a final volume of 20 μL. The mixture was incubated at 25 °C for 10 min, subjected to thermal cycling at 50 °C for 30 min, and heated to 85 °C for 5 min to synthesize the first cDNA strand. Quantitative PCR primers were designed using Primer Premier 6.0 and Beacon Designer 7.8 software. The primers are detailed in Table 4. The mRNA expression of related genes was normalized to GAPDH and analyzed using the 2ΔCt method.
Real-Time PCR Primers.

Abbreviation: PCR, Polymerase chain reaction.
The Analysis of Expression Levels of Anti-Inflammatory Pathway Target Proteins of HQFD by Western Blotting
The remaining lung tissue homogenate was used to detect the expression of target proteins of MMP9, PTGS2, AKT1, and MAPK14 by Western blotting to determine the impact of HQFD on the expression level of anti-inflammatory pathway target proteins. The methodology was as follows: Initially, the proteins in the lung homogenate were separated. The protein isolates were subsequently resolved using sodium dodecyl sulfate–polyacrylamide gel electrophoresis with a loading amount of 60 μg per well. The proteins were transferred to PVDF membranes, which were subsequently blocked in T-TBS for 1 h. The membranes were incubated overnight at 4 °C with primary antibody (1:1000 dilution), followed by a 1-h incubation with secondary antibody. After washing the membrane 5 times with T-TBS, detection was performed by enhanced chemiluminescence. Image J software was used to quantify the optical density of the replicated bands, with each band being replicated 3 times. The relative expression of the target protein was calculated using the following formula, and the results are expressed as the mean ± standard deviation:
Statistical Analysis
All data were analyzed and visualized with SPSS 25.0 statistical software and GraphPad Prism 9.5. One-way ANOVA analyzed the data between groups, and, P < .05; P < .01 are considered significant.
Animal Experiments Validation Result
The Active Components in HQFD
As shown in Figure 4A and B, the presence of 3 active ingredients, namely β-sitosterol, wogonin, and quercetin, in HQFD was confirmed.
Impact of HQFD on Inflammatory Cytokines
The anti-inflammatory effect of HQFD was verified by performing ELISA to measure the expression levels of inflammatory cytokines, including IL-1β, IL-6, and TNF-α, and the results are shown in Figure 4C to E. As shown in Figure 4C, TNF-α in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the levels of TNF-α in the HQFD-treated groups and the positive control group were significantly decreased (P < .01). As shown in Figure 4D, IL-1β in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the IL-1β of the groups treated with medium and high doses of HQFD and the positive control group were significantly decreased (P < .01). As shown in Figure 4E, the expression of IL-6 in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the expression of IL-6 was significantly decreased in the medium, and high doses of HQFD-treated groups and the positive control group (P < .01).
Morphological Results of the Lungs in Mice
The results are shown in Figure 5A to F. In the control group, the structure of alveoli and bronchi at all levels was clear and complete, and the morphology was normal, without any signs of inflammatory cell infiltration. In the model group, the alveolar septum was thickened, the alveolar structure was severely damaged, and there were coagulated cavities and extensive inflammatory cell infiltration (mainly lymphocytes) accompanied by serous exudation. In the dexamethasone-positive control group, the lung tissue still had inflammatory cell infiltration, alveolar septum thickening, and alveolar septum inflammatory cell infiltration, which was speculated to be related to the adverse reactions of the drug. Compared with the model group, the low-dose HQFD group had better improvement of inflammatory damage in lung tissue, thinner alveolar septum, reduced inflammatory cell infiltration (mainly lymphocytes), and improved alveolar and perivascular cell damage. In HQFD, the inflammatory damage of lung tissue in the HQFD-H group was significantly improved compared with that in the model group, with almost no inflammatory cells and clear and complete alveolar structure.
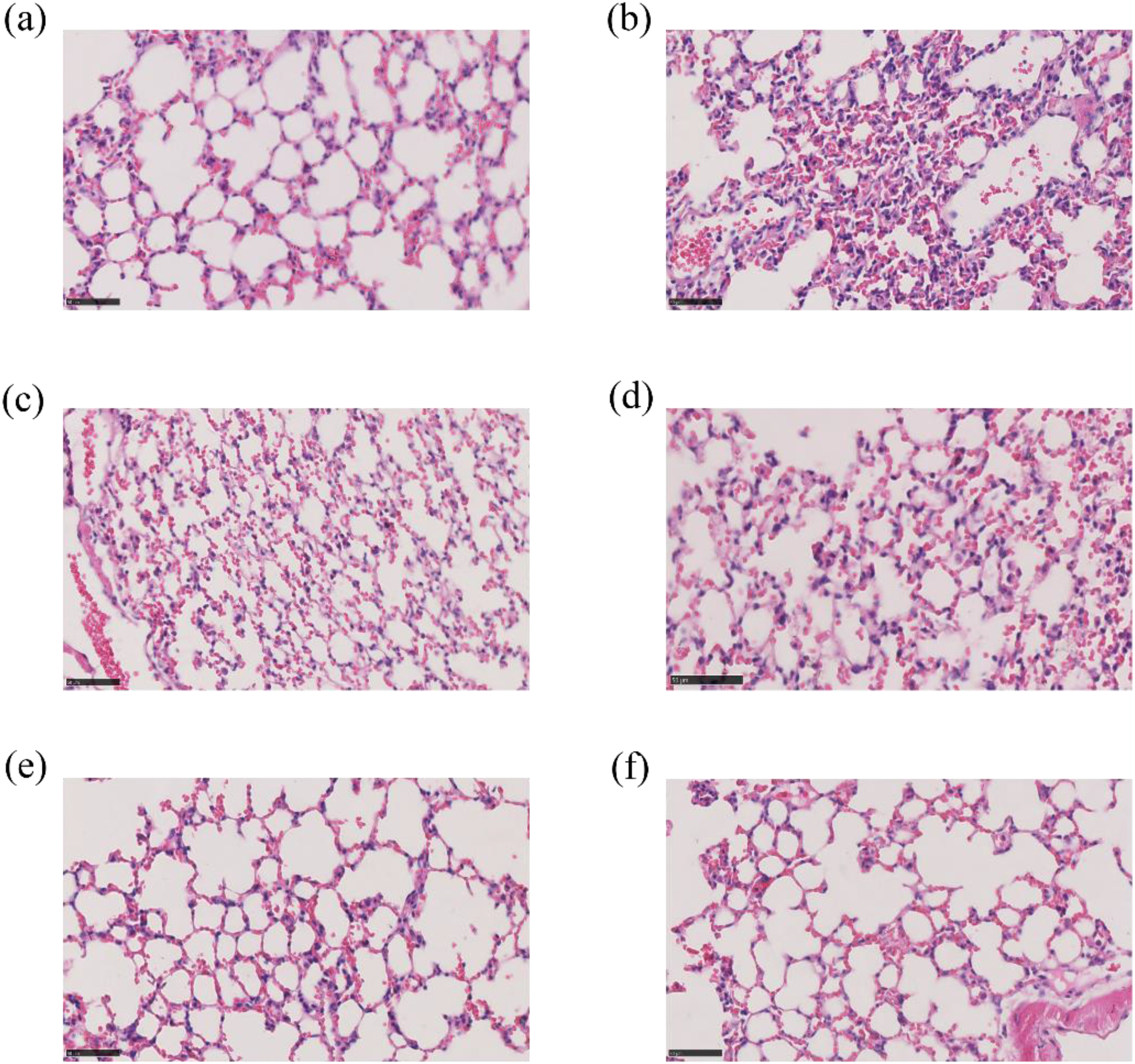
Schematic diagram of pathological sections of lung tissue in each group. (a) Pathological section of lung tissue in the control group; the lung tissue structure was intact, the morphology was normal, and there was no inflammatory cell infiltration. (b) The pathological section of lung tissue in the model group; Lung tissue was severely damaged in the model group, with coagulated cavities and extensive inflammatory cell infiltration accompanied by serous outflow. (c) Pathological sections of lung tissue in the positive group; Alveolar structural integrity and alveolar morphology were slightly improved, and inflammatory cell infiltration was reduced. (d) Pathological sections of lung tissue in the HQFD-L group; the alveolar structural integrity and alveolar morphology were improved, and there was still inflammatory cell infiltration. (e) Pathological sections of lung tissue HQFD-M group; Alveolar structural integrity and alveolar morphology were significantly improved, and inflammatory cell infiltration was significantly reduced. (f) Pathological sections of lung tissue in the HQFD-H group. Both alveolar structural integrity and alveolar morphology were significantly improved, and there was almost no inflammatory cell infiltration. HQFD, Huangqin Qingfei decoction.
Impact of HQFD on Anti-Inflammatory Pathways
To verify the mechanism of the HQFD in ICR mice, Western blotting was performed to measure the expression of p-AKT, AKT, P38, p-P38, MMP9, PTGS2 (COX2), and β-actin. As shown in Figure 6E, the results demonstrate that compared with the control group, the LPS-induced pneumonia model group heightened the phosphorylation of AKT and P38, as well as the expression of MMP9 and PTGS2. Compared with the model group, the HQFD significantly attenuated p-Akt and p-P38 expression. Moreover, the protein expression of MMP9 and PTGS2 was markedly reduced in the HQFD-treated groups. These results are consistent with the network pharmacology predictions. Quantitative analysis was performed using optical density values, which are presented in Figure 6A to D. As shown in Figure 6A, MMP9 in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the levels of MMP9 in the HQFD-treated groups and the positive control group were significantly decreased (P < .01). As shown in Figure 6B, PTGS2 in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the PTGS2 of the groups treated with low, medium, and high doses of HQFD and the positive control group were significantly decreased (P < .01). As shown in Figure 6C, the expression of p-Akt in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the expression of p-Akt was significantly decreased in the HQFD-treated groups and the positive control group (P < .01). As shown in Figure 6D, the expression of p-P38 in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the expression of p-P38 was significantly decreased in the HQFD-treated groups and the positive control group (P < .01).

(a-d) Effect of the HQFD on p-AKT, p-P38, MMP9, and PTGS2 (COX2) protein expression by Western blot. (e) Western blot strip diagram (f-i) Effect of the HQFD on the mRNA expression of AKT, PTGS2, MAPK14, and MMP9 in lung tissues graph. Compared with the control group, the mRNA and protein expression levels of AKT1, MAPK14, MMP9, and PTGS2 in the model group were significantly increased, while those in the medium- and high-dose HQFD groups were significantly decreased compared to the model group. # P < .05 versus control group, *P < .05 versus model group, **P < .01 versus model group. HQFD, Huangqin Qingfei decoction; COX, cyclooxygenase.
Real-time PCR was used to validate the mRNA expression of pivotal targets, including AKT1, MAPK14, MMP9, and PTGS2. HQFD treatment resulted in significantly decreased expression of AKT1, MAPK14, MMP9, and PTGS2. The detailed results are presented in Figure 6E to I. As shown in Figure 6E, the expression of MMP9 mRNA in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the MMP9 mRNA expression was significantly decreased in the HQFD-M and HQFD-H groups and the positive control group (P < .01). Notably, MMP9 mRNA expression in the HQFD-H group was close to that in the control group (P > .05). As shown in Figure 6F, the expression of PTGS2 mRNA in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the PTGS2 mRNA in the group treated with the medium dose of HQFD was significantly decreased (P < .05). PTGS2 mRNA was significantly decreased in the HQFD-H group and positive control group (P < .01). As shown in Figure 6G, the expression of AKT1 mRNA in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the expression of AKT1 mRNA in the groups treated with low, medium, and high doses of HQFD and the positive control group decreased significantly (P < .01). As shown in Figure 6H, the expression of MAPK14 mRNA in the lung tissue of the model group was significantly higher than that of the control group (P < .01). Compared with the model group, the expression levels of MAPK14 mRNA in the HQFD-treated groups and the positive control group were significantly decreased (P < .01).
Discussion
The objective of this study was to elucidate the mechanisms underlying the therapeutic effects of HQFD in pneumonia. Utilizing the TCMSP, a total of 51 chemical compounds were identified, among which the main chemical components included beta-sitosterol, wogonin, and quercetin, which modulate various signaling pathways. These findings suggest that the HQFD exerts its therapeutic effects on pneumonia through multiple active ingredients.
Commonly obtained from medicinal plants, beta-sitosterol exhibits pharmacokinetic characteristics similar to those of dexamethasone, showing potential utility in the management of inflammatory disorders. The secretion of inflammatory factors, including TNF-α, IL-8, IL-1β, and IL-6 by keratinocytes, was significantly reduced in the presence of beta-sitosterol, beta-sitosterol effectively inhibited the generation of reactive oxygen species and macrophages induced by PGN, TNF-α, or LPS. The β-sitosterol has also been reported that it has the potential of antioxidant and antimicrobial in another study. 28 Additionally, in a lung chronic infection mouse model, β-sitosterol dramatically decreased leukocyte recruitment and bacteria recovery, exhibiting anti-inflammatory and anti-infection capabilities and enhancing health status. 29 Wogonin has previously demonstrated significant therapeutic effects in pneumonia.30,31 Wogonin, a naturally occurring flavonoid from the root extract of Scutellaria baicalensis Georgi, exhibits diverse pharmacological properties, such as anti-inflammatory, anticancer, antibacterial, and antiviral properties. 32 In in vitro and in vivo experiments, wogonin has been observed to modulate the expression of proteins associated with inflammation and inhibit enzymes associated with inflammation, particularly cyclooxygenases (COX) and lipoxygenases. Systematic reviews have found that wogonin exerts direct effects on immune cells, including macrophages, lymphocytes, dendritic cells, mast cells, monocytes, and neutrophils. 33 Wogonin impedes the generation of various pro-inflammatory cytokines, chemokines, and inflammatory mediators, while simultaneously stimulating the production of specific anti-inflammatory cytokines. Their regulation of immune activation and anti-inflammatory activity is related to the suppression of binding to the arachidonic acid metabolism and Toll-like receptor (TLR) expression. It has been demonstrated that wogonin induces PPAR-γ expression, which in turn promotes the suppression of NF-kappaB-p65 activation and drastically lowers TNF-α and IL-6 expression. Additionally, it is linked to decreased activation of important molecular pathways (JAK-STAT, NF-kappa B, and MAPK), which has the impact of downregulating COX-2 and inducible nitric oxide synthase (iNOS). Quercetin, a natural flavonoid found in various plants, possesses numerous medicinal properties such as antioxidant, anticancer, antimicrobial, neuroprotective, and anti-inflammatory activities. Quercetin has demonstrated protective effects against acute lung injury by reducing lung edema and improving lung histological changes. Importantly, the beneficial effects of quercin can be attributed to its ability to inhibit inflammation. Mechanistically, quercetin downregulates the NF-kappa B signaling pathway and upregulates the cAMP-Epac and HO-1 pathways. 34 Furthermore, quercetin also displayed inhibitory activity against pneumolysin, suggesting its potential as a candidate for treating pneumococcal infections. 35 Taken together, the identified components may play a crucial role in the therapeutic efficacy of the HQFD in pneumonia, warranting further investigation.
The common key targets of the HQFD ingredients and pneumonia targets include AKT1, PTGS2, MAPK14, and MMP9. During the molecular docking process, beta-sitosterol, wogonin, and quercetin all demonstrated strong binding to these key targets, among which the 3 pairs with the lowest binding energy were PTGS2 and beta-sitosterol, MMP9 and beta-sitosterol, and MAPK14 and quercetin, suggesting that MMP9 may be the key target mediating the therapeutic effects of the HQFD.
MMPs are a group of enzymes that participate in the modification of the extracellular matrix (ECM) under both physiological and pathological conditions through proteolytic activity. 36 In particular, MMPs play a crucial role in mediating wound healing, airway remodeling, and cell transport during pulmonary inflammation. However, excessive MMP expression has been implicated in tissue degradation associated with pulmonary inflammation and lung disorders. 37 MMP9 is the largest MMP, and it acts as a gelatinase. 38 MMP9 causes the degradation of gelatine and types IV, V, XI, and XVI collagen during tissue remodeling. Moreover, it degrades non-ECM molecules, such as interleukin-1β and TNF-α, which are essential for inflammation. MMP9 is typically absent in healthy lungs, but it can be released by lymphocytes, neutrophils, mast cells, and macrophages during inflammation.37,39 The association between MMP9 and neutrophil migration toward the lungs in response to infection has been well-established for a long period. In the pathological progression of pneumonia, the synergistic action of MMP9 and integrin facilitates the inflammatory response and enhances permeability in pulmonary microvascular endothelial cells, leading to endothelial dysfunction, edema, and severe hypoxemia. This cascade effect may potentially trigger or exacerbate pneumonia.40,41
In this study, AKT1, PTGS2, and MAPK14 showed a strong affinity with beta-sitosterol, wogonin, and quercetin. AKT1, one of the serine/threonine protein kinases, regulates the growth and function of innate immune cells, such as dendritic cells, macrophages, and neutrophils. 42 AKT1 deletion promotes the upregulation of iNOS and interleukin-12β. This results in enhanced bacterial eradication, increased sensitivity to LPS, and abolished tolerance to endotoxin.43,44 Moreover, AKT is closely related to the cellular immune response and B cells. 45 It is noteworthy that activation of PI3K-dependent p38 kinase (p38-MAPK) modulates the activity of AKT through phosphorylation of Ser473 in human neutrophils.46,47 MAPKs include p38 kinases, 48 and p38α (also known as MAPK14) is the most extensively studied member of the p38 family and exhibits ubiquitous expression across various cell types. 49 It has been demonstrated that multiple extracellular factors can activate P38α, such as cytokines, chemokines, chemoattractants, oxidative stress, ischemia, bacterial LPS, interleukin-1, and TNF-α. 50 The activation of p38α is an essential event in the generation of pro-inflammatory cytokines, such as TNF-α, interleukin-6, and interleukin-1β. Additionally, it triggers the expression of iNOS, which is responsible for regulating oxidation. It also initiates adherent proteins and induces the COX-2 enzyme, which plays a pivotal role in regulating connective tissue remodeling under pathological conditions. 51 Cyclooxygenase enzymes, also known as prostaglandin-endoperoxide synthases, are the rate-limiting enzymes in the conversion of arachidonic acid (derived from damaged cell membranes) into prostaglandins. These enzymes exist in at least 2 isoforms: PTGS1 (COX-1) and PTGS2 (COX-2). Unlike PTGS1, which is constitutively expressed, PTGS2 expression is induced to high levels during inflammation by growth factors and cytokines. 52 PTGS2 activation leads to prostaglandin E2 (PGE2) production, which regulates vascular tone, thrombosis, inflammation, and pain. 53 The significance of IL6 and TNF as activators of P38 in the context of common targets for diseases and drugs cannot be overlooked in this analysis. As a pivotal mediator of inflammation, TNF-α is synthesized and secreted by activated macrophages, promoting lung fibroblast proliferation and collagen synthesis, facilitating the recruitment of inflammatory cells, and modulating the inflammatory cascade in alveolar epithelial cells. 54 Several studies have demonstrated that by inhibiting the transcriptional activity of NF-kappa B induced by TNF-α, it is possible to disrupt the signaling pathway of NF-kappa B and ultimately prevent the expression of IL8 and IL6 messages, thereby protecting cell. 55 The pro-inflammatory cytokine IL-6 is implicated in a diverse range of physiological and pathological processes, exerting a potent immunomodulatory effect. 56 Previous studies have demonstrated that IL6 serves as a pivotal pro-inflammatory factor responsible for initiating the inflammatory cascade in patients with COVID-19. 57 Following infection, pathogenic T cells undergo rapid activation, subsequently stimulating CD14+ and CD16+ inflammatory monocytes to secrete substantial quantities of IL6, thereby culminating in the development of an exaggerated inflammatory response. Its elevated levels have been correlated with the severity of infection. 58 It is evident that common key targets of the HQFD and pneumonia targets mutually regulate, indicating their involvement in the occurrence and pathogenic mechanisms of pneumonia, such as oxidative stress and inflammation. This finding supports the GO and KEGG enrichment analyses.
Based on the results of the GO analysis, we identified predominant enrichment in biological processes among the target genes, including the response to LPS, the response to molecule of bacterial origin, the tube morphogenesis, and the response to decreased oxygen levels. Among them, LPS plays a pivotal role in the pathogenesis of pneumonia. Toll-like receptors serve as prototypical ligands for LPS, and the TLR signaling pathway represents a fundamental component of the immune response. Lipopolysaccharide binds to the TLR4, which activates signaling pathways. The binding of LPS to TLR4 triggers the activation of signaling cascades. This activation mechanism primarily involves MyD88 and/or other associated proteins, ultimately promoting the production of pro-inflammatory cytokines.59,60 Oxidative stress, inflammation, and endothelial cell damage are facilitated by macrophages, neutrophils, and various other activated cell types in response to LPS. Several studies have demonstrated the advantageous role of modulating MAPK and NF-kappa B, in preserving endothelial barrier integrity and ultimately suppressing TLR4 transduction and raft protein activation in LPS-induced pro-inflammatory signaling. 61 The results of the KEGG enrichment analysis indicate that the HQFD for pneumonia treatment activated multiple signaling pathways, including the pathways in cancer, the lipid and atherosclerosis, as well as the AGE-RAGE signaling pathway in diabetic complications. Based on the enrichment analysis results, the PI3K-AKT signaling pathway, MAPK signaling pathway, and NF-kappa B signaling pathway have great potential as one of the main signaling cascades of HQFD in the treatment of pneumonia. The PI3 K/Akt pathway plays a central role in regulating inflammatory response, cell activation, and apoptosis. Studies have reported that the PI3 K/Akt signaling pathway is activated in a variety of inflammatory and autoimmune diseases. 62 In addition, the PI3 K/Akt pathway can be the upstream activator of the NF-kappa B signaling cascade, and the NF-kappa B pathway has long been considered as a canonical pro-inflammatory pathway. The MAPK pathway serves diverse cellular functions, including the regulation of gene expression, cell cycle progression, and cell survival. 63 Inhibitors targeting 2 members of the p38 family have demonstrated anti-inflammatory effects primarily by suppressing inflammatory mediators. 64 These physiological processes are crucial in infectious airway diseases.
In summary, this study employed pharmacology and molecular docking to identify active components (beta-sitosterol, wogonin, and quercetin) and 4 representative targets (AKT1, PTGS2, MAPK14, and MMP9). Molecular docking results demonstrated favorable binding of all 3 compounds to the respective proteins. Furthermore, PTGS2 exhibited the strongest affinity with beta-sitosterol showing the lowest binding energy indicative of a highly stable interaction. Enrichment revealed that HQFD exerts its therapeutic effects on pneumonia through modulation of the PI3K-AKT signaling pathway, MAPK signaling pathway, and NF-kappa B signaling pathway.
The prediction results of network pharmacology were further validated through subsequent animal experiments in this study. In animal experiments, H&E staining results revealed that when compared to the control group, the model group exhibited severe alveolar damage with prominent infiltration of inflammatory cells in the alveolar space, indicating pneumonia can lead to pulmonary injury characterized by thick alveolar septum, cavities of solidification and extensive infiltration of inflammatory cells. Conversely, treatment with HQFD resulted in an improvement in alveolar structure and a reduction in inflammatory cell infiltration compared to the model. These findings suggest that HQFD effectively mitigated lung tissue damage and suppressed inflammatory cell infiltration within the alveolar space. The ELISA results concurrently demonstrated that HQFD effectively attenuated the levels of inflammatory cytokines IL-6, IL-1β, and TNF-α in lung tissue, thereby mitigating the detrimental impact of the inflammatory response on pulmonary tissue.
Polymerase chain reaction results revealed significant upregulation of AKT1, PTGS2, MAPK14, and MMP9 mRNA in lung tissues from the model group compared with the control group. Meanwhile, the mRNA expression of AKT1, PTGS2, MAPK14, and MMP9 was significantly lower in the HQFD group than in the model group. Furthermore, it was observed that the mRNA expression of AKT1, PTGS2, MAPK14, and MMP9 was even lower in the high-dose HQFD group than in the positive control group. The Western blot analysis confirmed that the expression levels of PTGS2 and MMP9 were significantly reduced in the HQFD group compared to the model group, indicating that HQFD exerts its anti-inflammatory effect by suppressing gene expression of PTGS2 MMP9.
Western blot results revealed that compared with the control group, the expression of PTGS2 and MMP9 protein in lung tissues from the model control group was significantly upregulated. Compared to the model group, the high-dose HQFD group exhibited reduced p-P38, p-AKT, MMP9, and PTGS2 expression. However, there was no significant correlation between the protein expression and the mRNA expression of AKT1 and MAPK14, suggesting that posttranslational modifications may play a role in regulating these genes under inflammatory conditions. Based on the characteristics of pathological tissue sections and the Western blot results, the study proposed a hypothesis for the mechanism of high expression in pulmonary inflammation (Figure 7). During the pathological development of pulmonary inflammation, external factors, such as LPS and oxidative stress, promote the expression of the AKT and MAPK enzymes. In the PI3 K/Akt signaling pathway, Akt, a protein downstream, is activated, to promote immune cell generation and pro-inflammatory cytokine release for effective bacterial clearance. In the NF-kappa B signaling pathway and MAPK signaling pathway, the proteins of downstream PTGS2 and MMP9 are activated by NF-kappa B and MAPK, regulating pulmonary vascular tension, mediating wound healing under pathological conditions, and restoring homeostasis through airway remodeling. However, low immunity in some populations may lead to excessive expression of AKT, PTGS2, MAPK14, and MMP9. The alveolar structure is damaged, leading to immune cell infiltration into the lung tissue due to excessive release of pro-inflammatory cytokines, causing irreversible degradation of lung tissue and further promoting their expression, resulting in significant life-threatening damage from pneumonia. The HQFD can reduce the excessive expression of AKT, PTGS2, MAPK14, and MMP9, as well as inhibit pro-inflammatory factor phosphorylation to mitigate the inflammatory response caused by LPS. The core mechanism of polydatin is similar to that which has been reported to mitigate the inflammatory response induced by cytokine storm in COVID-19, through the inhibition of nitric oxide and PGE2 production, as well as the phosphorylation of NF-kappa B and MAPK in LPS-induced RAW 264.7 macrophages. 65 This aligns with the anti-inflammatory therapeutic effects observed with HQFD treatment. This hypothesis is supported by our experimental results, which indicate elevated expression of AKT, PTGS2, MAPK14, and MMP9 during inflammation.

Mechanistic map of anti-inflammatory pathways. Pulmonary inflammation is mainly caused by infection of the lungs with foreign pathogenic microorganisms (such as bacteria and pathogens), which stimulate lung cells. Firstly, TNFα on the surface of the lung cell membrane is activated by pathogen stimulation. TNF signals through 2 distinct cell surface receptors: TNF-R1 and TNF-R2. TNF-R1 recruits TNF receptor type 1-associated DEATH domain protein to establish the “death-inducing signaling complex” to initiate signal transduction. TNF-R1 signaling activates numerous genes that are primarily controlled by 2 distinct pathways, namely the MAPK cascade and the NF-kappa B pathway, resulting in necroptosis and apoptosis. Among them, c/EBPβ downstream of the MAPK cascade and NF-kappa B downstream of the NF-kappa B pathway enter the nucleus and activate the expression of pro-inflammatory genes. PTGS2 and MMP9 are transcription and translation products. PTGS2 mediates the reconstruction of the extracellular matrix and regulates vascular tone. MMP9 promoted the expression of inflammatory mediators. In contrast, TNF-R2 signaling induces activation of the NF-kappa B pathway, including the PI3K-dependent NF-kappa B pathway, leading to survival. In pathophysiological conditions, TNF overproduction leads to the emergence of inflammatory reactions that are implicated in various lung diseases characterized by inflammation. HQFD mainly regulates the PI3K-AKT signaling pathway, MAPK signaling pathway, and NF-kappa B signaling pathway to inhibit the overexpression of pro-inflammatory factors, thereby achieving anti-inflammatory effects. By Figdraw. HQFD, Huangqin Qingfei decoction.
Among them, MMP9 and PTGS are targets of the NF-kappa B signaling pathway, MAPK14 is a target of the MAPK cascade signaling, and Akt is a target of the PI3K-AKT signaling pathway. These pathways play pivotal roles in inflammation and other biological processes. In the NF-kappa B pathway, upstream molecules are activated by external stimuli to promote the expression of multiple genes. MMP9 and PTGS2, as crucial gene transcription products regulated by NF-kappa B, serve as key components of this signaling pathway that governs ECM remodeling and inflammatory mediators.66,67 The PI3K-AKT signaling pathway represents a classical signal transduction cascade involved in cell proliferation, apoptosis, and various cellular processes. Akt functions as an important downstream effector molecule of PI3 K and participates in regulating cell survival and apoptosis. Within the MAPK signaling pathway, MAPK acts as a regulatory signal modulating diverse physiological/path effects such as cell growth, differentiation, stress response, and inflammatory reactions.
The results of in vitro experiments were consistent with the network pharmacology analysis. It was observed that beta-sitosterol, wogonin, and quercetin emerged as the primary active constituents of HQFD exhibiting significant efficacy in reducing lung tissue suppressing inflammatory cell infiltration within the alveolar space and inhibiting the activation of PI3K-AKT signaling pathway, MAPK signaling pathway, and NF-kappa B signaling pathway. Consequently, HQFD holds promising potential as an ideal therapeutic agent for pneumonia treatment.
This study has some limitations that should be noted. First, the present study was limited to lung tissue analysis without considering inflammatory markers in blood samples. Second, the establishment of the experimental animal model as a single strain neglected the fact that CAP is predominantly caused by mixed infection. Finally, there are notable differences in the mode of infection between experimental animal lung infections and clinical patient infections. These limitations impeded the advancement of theories elucidating the mechanisms of the HQFD in pneumonia treatment and the regulation of immune function by the HQFD. Addressing these limitations will be prioritized in future research.
Conclusion
In this study, we successfully identified the active constituents, prospective targets, and pathways underpinning the therapeutic effects of HQFD in pneumonia. Animal experiments were conducted to verify the results. The therapeutic mechanism of the HQFD in pneumonia may be attributed to its active ingredients, including beta-sitosterol, wogonin, and quercetin. This mechanism involves multiple targets and pathways, including the PI3K-AKT signaling pathway, MAPK signaling pathway, and NF-kappa B signaling pathway, as well as the expression of AKT, PTGS2, MAPK14, and MMP9. By reducing inflammatory damage and alleviating pneumonia symptoms through these mechanisms, the HQFD demonstrates promising potential as a treatment for pneumonia.
Overall, the findings contribute to the understanding of the mechanism underlying the therapeutic effects of the HQFD in pneumonia, furnish experimental evidence for the use of the HQFD as a therapeutic approach for pneumonia, and ultimately offer strategies for the development of novel antipneumonia drugs.
Supplemental Material
sj-tif-1-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-1-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-2-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-2-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-3-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-3-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-4-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-4-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-5-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-5-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-6-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-6-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-7-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-7-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-8-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-8-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-9-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-9-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-10-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-10-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-11-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-11-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-12-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-12-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-13-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-13-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-14-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-14-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-15-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-15-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-16-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-16-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-17-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-17-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-18-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-18-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-19-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-19-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-20-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-20-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Supplemental Material
sj-tif-21-npx-10.1177_1934578X241239843 - Supplemental material for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation
Supplemental material, sj-tif-21-npx-10.1177_1934578X241239843 for Exploring the Mechanisms of the Huangqin Qingfei Decoction in Pneumonia Treatment for Lipopolysaccharide-Induced Mouse Model: A Network Pharmacology Analysis and Experimental Validation by Zengle Zheng, Jingyun Lei, Jia Chang, Xiuxiu Yi, Guoying Zhou and Jing Chen in Natural Product Communications
Footnotes
Acknowledgments
This research was funded by the National Natural Science Foundation of China (No. 82174272) and the Key Research and Development Program of Zhejiang Province (No. 2020C03052).
Data Availability Statement
The data used to support the findings of this study are included within the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 82174272), Key Research and Development Program of Zhejiang Province (grant number National Natural Science Foundation of China, 2020C03052).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the NIH Guidelines for the Care and Use of Laboratory Animals and were approved by the Animal Ethics Committee of Zhejiang Chinese Medicine University (The ethical approval code: IACUC-20211206-12. The ethical approval date: December 06, 2021).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
