Abstract
Objectives
The mechanism of tripterygium glycosides (TG) in the treatment of asthma was explored by utilizing network pharmacology, molecular docking, and in vitro preliminary pharmacological tests.
Methods
An analysis of the bioactive ingredients of TG was conducted using the Traditional Chinese Medicine Systems Pharmacology Database database or predicted by FAFdrugs4, with relevant targets gathered from HERB, Swiss Target Prediction, STITCH, and SEA databases, and the targets for asthma were collected in Drugbank, DisGeNET, and GeneCards databases. Compound-target-pathway interactions and the function of putative targets were annotated by Cytoscape 3.6.0 and Database Visualization and Integrated Discovery, respectively. The affinity of the key ingredient and hub targets was assessed by molecular docking using AutoDock Vina software. Real-time quantitative polymerase chain reaction, cell-secreted cytokine assay, and ELISA were used to explore the effect of TG and its key ingredient triptolide (TP) for asthma.
Results
There were 12 bioactive ingredients in TG with 335 corresponding targets, 2672 asthma-associated targets, and 204 TG anti-asthma targets. TP and Tripterine were identified as the two key ingredients. RELA and TP53 exhibited the best topological index in protein-protein interaction network analysis and were regarded as hub targets. TG might play a role in biological processes such as lipopolysaccharide-mediated signaling pathway, regulation of nitric oxide biosynthetic process and mainly related to toll-like receptor, TNF and apoptosis signaling pathways. Molecular docking revealed that TP effectively bound to hub targets. In vitro experiments demonstrated that TG and TP reversed the RNA expression of airway remodeling marker CDH1 and FN. Moreover, TP reduced the secretion of vascular endothelial growth factor, implicating its role in airway remodeling.
Conclusion
TG might act on target genes RELA and TP53, utilizing the active ingredient TP, to regulate signaling pathways such as toll-like receptor, TNF, and apoptosis, thereby playing a role in the improving airway remodeling in asthma.
Introduction
Asthma, characterized by chronic airway inflammation, bronchial hyper-reactivity and airway remodeling, manifests symptoms such as cough, wheezing, difficulty in breathing, and chest tightness. 1 However, asthma is still incurable and the goals of asthma treatment are to control symptoms and reduce the risk of disease progression. Inhaled corticosteroids are often used in asthma management but they have side effects and patients have poor responsiveness and compliance with them, 2 which prompted us to look for better drugs to supplement the treatment of asthma.
Tripterygium glycosides (TG), derived from the plant Tripterygium wilfordii Hook F, constitute a traditional Chinese medicine (TCM) proven effective in conditions like rheumatoid arthritis, systemic erythematosus lupus, and nephritis due to its immunosuppressive and anti-inflammatory properties. 3 Accordingly, TG has been utilized in asthma treatment for an extended period, 4 and a previous study demonstrated its efficacy in attenuating the established airway inflammation in asthmatic mice. 5 Currently, the mechanism underlying TG's actions remains incompletely elucidated, encompassing its targets, molecular mechanism, and related signaling pathways. A distinctive characteristic of TCM is its multi-components, multi-channels, and multi-target synergy. However, the investigation of a single target-pathway persists, posing an incompatibility with the features of Chinese medicine and resulting in inadequate explanations of action mechanisms. Network pharmacology integrating network biology and polypharmacology holds the promise to reveal the cooperative treatment of TCM against diseases. 6 Moreover, molecular docking, based on molecule structure, can elucidate the interactions between molecules and biological targets. 7 Consequently, a combined approach utilizing network pharmacology and molecular docking was employed in this study in order to identify TG's pharmacological targets and elucidate its mechanism of action against asthma (Figure 1). In addition, the elementary impact of TG and its bioactive ingredient triptolide (TP) on airway remodeling were investigated through in vitro experiments.

The flow chart of network pharmacology, molecular docking, and in vitro preliminary pharmacological evaluation for TG anti-asthma targets. TG, tripterygium glycosides.
Results
Bioactive Ingredients in TG
A total of 12 bioactive ingredients were identified following the screening and prediction of the absorption, distribution, metabolism, and excretion (ADME)-Tox properties of compounds in TG. These included Celafurine, Celabenzine, Celacinnine, Triptolide, Triptonoditerpenic acid, Wilforlide A, Orthosphenic acid, Tripterine, and Triptotriterpenic acid C, which were retrieved from Traditional Chinese Medicine Systems Pharmacology Database (TCMSP) (Table S1). Additionally, ent-19-hydroxykaur-16-ene, Triptoquinone A, and Triptoquinone B were predicted by FAFdrugs4.
TG Targets, Asthma-Associated Targets, and TG Anti-Asthma Targets
A total of 335 targets were identified within 12 bioactive ingredients of TG. For the analysis of TG targets’ interactions, eight protein-protein interaction (PPI) network function clusters were found by Metascape platform (Figure 2). These clusters, numbered one through eight, featured seed genes EZH2, BCL2L11, PARP1, ESR1, CASP1, CD44, FH, and KCNMB4, respectively. A cluster, specifically Cluster 4, exhibited a highest MCODE_SCORE, consisting of 28 nodes and 275 edges. Its hub gene, ESR1, was associated with bronchial hyperresponsiveness and reduced lung function, especially in female subjects. 8 The seed gene of cluster 1, EZH2, was reported as an asthma-inducing factor. 9 A total of 2762 asthma-associated targets were collected, eliminating redundancies. The details are listed in Table S2. To reveal targets involved in the TG's treatment of asthma, 204 anti-asthma targets were determined through intersecting the 335 TG targets with 2762 asthma-associated targets (Figure 3, Table S3).

Eight clusters in 335 TG targets. Targets are represented by points, interactions by lines, and clusters by different colors. TG, tripterygium glycosides.

The Venn diagram was constructed between TG targets and asthma-associated targets. TG, tripterygium glycosides.
Gene Ontology and Pathway Enrichment
To identify the biological characteristics of putative targets of TG, the gene ontology (GO) and pathway enrichment analyses were conducted. Three aspects were considered in the GO analysis of gene functions: biological processes (BP), cellular components (CCs), and molecular functions (MFs). In terms of BP, the TG targets were involved in apoptotic processes, drug response, and cell proliferation (Figure 4a). As Figure 4b showed that the apoptosis, toll-like receptor (TLR) signaling pathway and TNF signaling pathway were closely related to the treatment of diseases with TG. Likewise, the GO and pathway enrichment analyses of 204 TG anti-asthma targets were also performed. For GO analysis, 49 genes were enriched in the BP of negative regulation of apoptosis, 54 genes were enriched in the CC of extracellular space, and the MF of enzyme binding had an enrichment of 39 genes, the top 15 terms are displayed in Figure 5a-c. Figure 5d illustrated that among the enriched pathways, TLR signaling pathway, TNF signaling pathway, and apoptosis were closely related to the mechanism of TG in anti-asthma.

Analysis of TG targets enriched by GO and pathways (a) the top 20 terms of GO analyses and (b) pathway enrichment analyses. GO, gene ontology. TG, tripterygium glycosides.
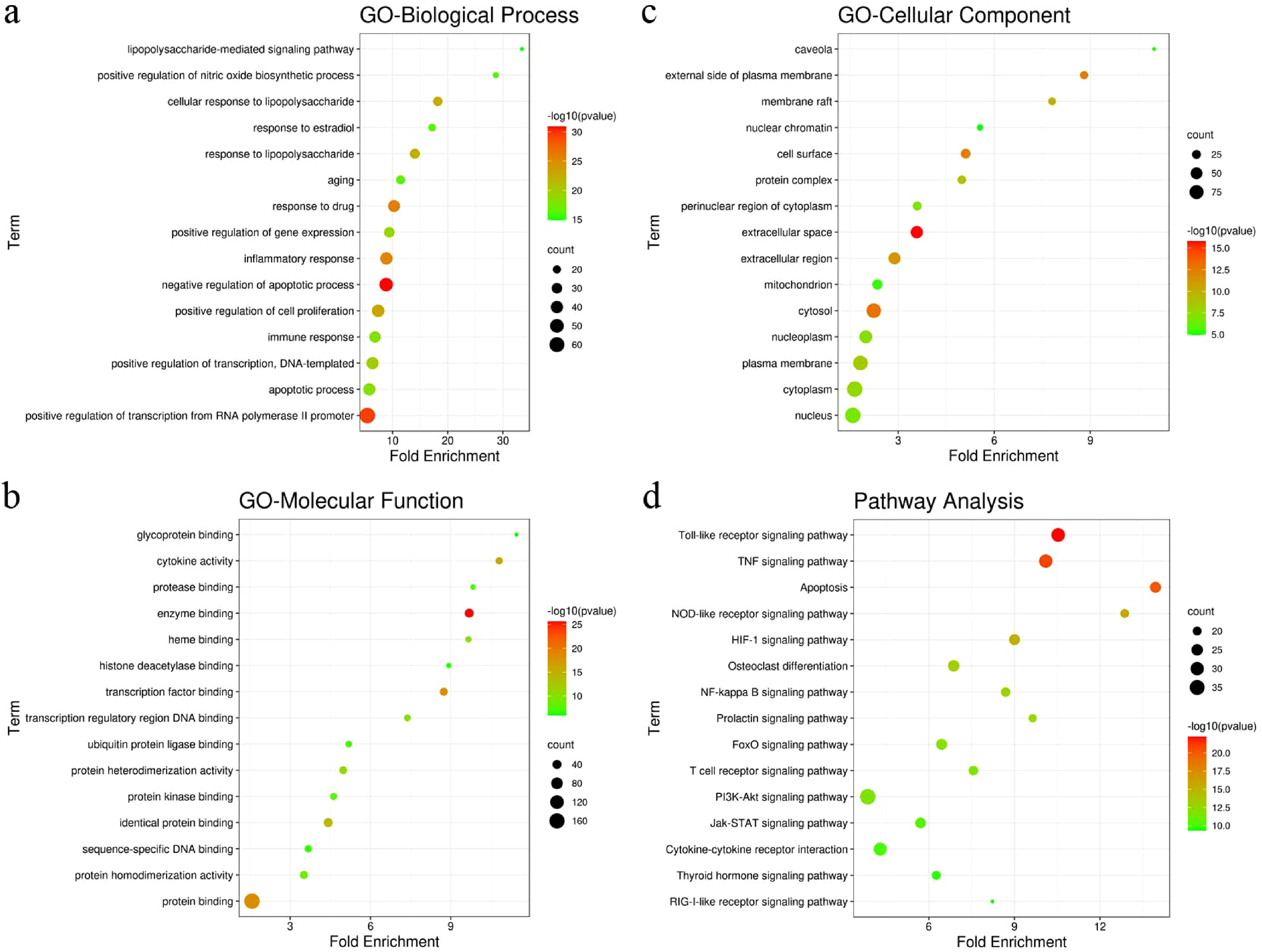
The results of GO and pathway enrichment for TG anti-asthma targets. The top 15 terms of biological processes (a), molecular functions (b), cellular components (c), and pathway enrichment analyses (d). GO, gene ontology. TG, tripterygium glycosides.
Construction of PPI Network
To analyze the PPI among 204 TG anti-asthma targets, a PPI network was constructed using Cytoscape (Figure 6a). RELA and TP53 were identified as hub targets by the analysis of the topological index of degree. The detailed topological properties of top 10 targets are presented in Table 1.

Interaction network. The interaction network of 204 TG anti-asthma targets. Node size of targets is proportional to the number of degrees and the green nodes represent the top2 targets (a). Compound-target network. The yellow nodes are bioactive compounds. The green nodes represent key targets and the black lines represent the relationship between the targets and the compounds (b). The target-top 5 pathway network. Purple octagons represent KEGG terms and green nodes represent pathway targets and the blue ones are the key targets in this study. The black lines reflect how the target participates in the pathway (c). TG, tripterygium glycosides.
The Topological Properties of top 10 TG Anti-Asthma Targets.

Abbreviation: RELA, transcription factor p65; TP53, cellular tumor antigen p53; AKT1, protein kinase B; EGFR, epidermal growth factor receptor; HDAC1, histone deacetylase 1; CTNNB1, catenin beta-1; HIF1A, hypoxia-inducible factor 1-alpha; MYC, Myc proto-oncogene protein; JUN, transcription factor AP-1; AR, androgen receptor; TG, tripterygium glycosides.
Compound-target and compound-pathway networks were also constructed based on target identification and pathway analysis. The compound-target network consists of 215 nodes and 282 edges, highlighting TP and Tripterine as the most significant compounds in TG anti-asthma treatment (Figure 6b). The androgen receptor target was influenced by a variety of bioingredients derived from TG, such as Triptonoditerpenic acid C, Triptotriterpenic acid C, TP, Tripterine, Wilforlide A, and Orthosphenic acid. Notably, both TP and Tripterine were associated with hub targets of RELA and TP53. And the target-pathway network showed that TP53 gene was exclusively mapped to the apoptosis pathway, while the genes of RELA were inclusively mapped to TLR signaling pathway, TNF signaling pathway, and apoptosis and HIF-1 signaling pathway. This suggests that TG exerted anti-asthma effects through multiple pathways and multiple targets (Figure 6c).
Molecular Docking
The results suggested that TP played a significant role in compound-target network, with RELA and TP53 listed as the top 2 TG anti-asthma targets. Therefore, the docking between TP and these two proteins was performed (Figure 7). The binding energy between TP and the two targets was less than −6 kcal/mol, which indicated that TP could form stable conformations with these targets for asthma treatment.
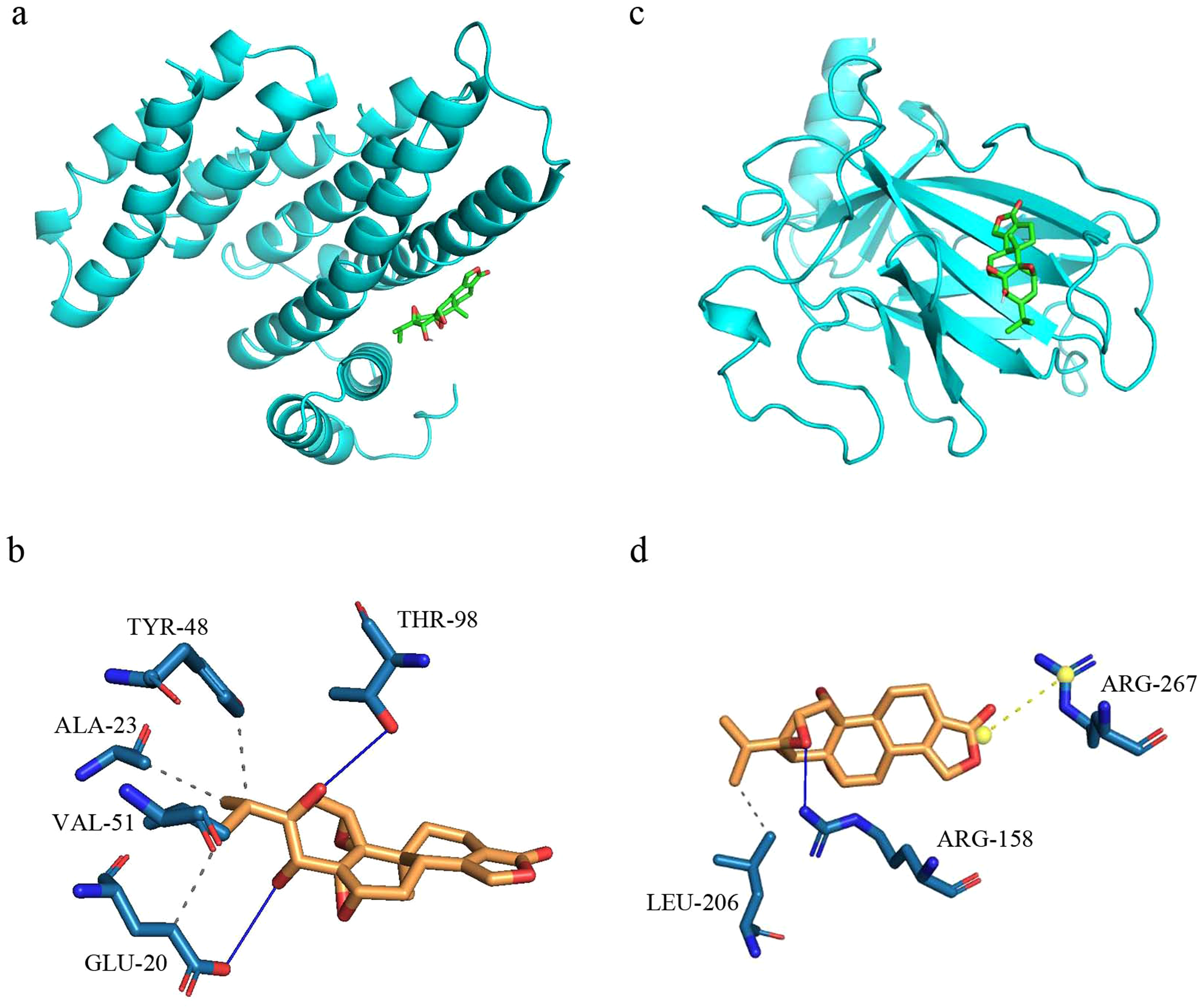
Molecular docking of TP and anti-asthma targets. TP with RELA (a, b), and TP53 (c, d). There is a ball-and-stick representation for molecules, dotted lines for hydrogen bonds, and distances in Angstroms. TP, triptolide.
TG/TP Reversed the mRNA Expression of Epithelial-Mesothelial Transdifferentiation Airway Remodeling Markers CDH1 and FN1
The fundamental impact of TG and TP in asthma was assessed in vitro. No survival inhibition for 16HBE was observed when the concentration of TG lower than 0.3 μg/ml or TP was lower than 5 ng/ml (Figure 8a,b).Therefore, for the subsequent treatments of 16HBE, the TG and TP dosages were adjusted to 0.3 μg/ml and 5 ng/ml, respectively. In this study, real-time quantitative polymerase chain reaction (RT-PCR) was employed to confirm the effects of TG/TP on airway modeling. The results illustrated that TG or TP reversed the expression levels of epithelial-mesothelial transdifferentiation airway remodeling markers CDH1 and FN1 in 16HBE cells stimulated by TGF-β1 (Figure 8c) in a dose-dependent manner(data were not shown).

The effect of TG/TP on 16HBE cell viability (a, b) and the mRNA expression of airway remodeling markers FN1 and CDH1 (c). *<0.05; **<0.01; ***<0.001. TG, tripterygium glycosides; TP, triptolide.
TP Reduced Vascular Endothelial Growth Factor Secretion of 16HBE Cells
To further explore the potential impact of TP for asthma, a liquid suspension chip was used to analyze the secreted cytokines of 16HBE cells treated with TP. The heatmaps described the changes of all 27 secreted cytokines and four specific secreted cytokines (Figure 9a,b), respectively. Figure 9b suggested TGF-β1 significantly promoted the secretion of vascular endothelial growth factor (VEGF) in 16HBE cells, an effect significantly attenuated by TP as further confirmed by ELISA (Figure 9c).

Differential expression of cytokines detected by liquid suspension chip and the expression of VEGF was confirmed by ELISA. (a) Heatmap representation of liquid suspension chip analysis. (b) Heatmap representation and bar charts, (c) the VEGF expression detection by ELISA. VEGF, vascular endothelial growth factor
Discussion
There are numerous phenotypes and endotypes associated with asthma, and a complex network of molecules is involved in the treatment of asthma. 10 TCM, characterized by multi-components, multi-channels, and multi-targets synergistic effect, holds promise for asthma treatment. 11 In the current study, the target and possible mechanisms of TG against asthma were explored by network pharmacology. After identifying 12 bioactive ingredients in TG, we identified 335 targets for these ingredients and 2762 asthma-associated targets. Subsequently, 204 anti-asthma targets of TG were obtained. Among them, RELA (NF-κB p65) and TP53 were shown as key targets. RELA, a subunit of the transcription factor for NF-κB, could induce various pro-inflammatory chemokines, cytokines, adhesion molecules, and respiratory mucins. 12 Higher levels of NF-κB p65 protein were observed in the peripheral blood mononuclear cells from asthmatic patients compared to normal individuals, 13 demonstrating that RELA might act as a crucial role in asthma. Regarding TP53, asthma symptoms were statistically significantly associated with polymorphism in codon 72 of p53, 14 and the DNA methylation of TP53 was associated with the severity of late-onset asthma, 15 which showed that TP53 also participates in the development of asthma. Therefore, TG could be employed to treat asthma by targeting RELA and TP53. KEGG pathway analysis and the target-pathway network shed light on the mechanism of TG in anti-asthma. It was evident that apoptosis and TLR signaling pathway showed great potential in anti-asthma (Figure 5). Many existing anti-asthma drugs have been found to exert a pro-apoptotic effect. For example, Benralizumab, an antagonist of the IL-5 receptor, significantly increased eosinophil apoptosis. 16 Additionally, Ketamine administration induced the apoptosis of neutrophil and eosinophil in BALF, potentially contributing to the resolution of inflammation in asthma. 17 Therefore, our results indicated that TG could play an anti-asthmatic effect by regulating apoptosis pathway. Moreover, it was previously reported that TLRs played a crucial role in allergic diseases by recognizing environmental antigens and initiating pro- or anti-allergy responses. 18 Furthermore, asthma susceptibility among children was demonstrated to be influenced by variant alleles of TLR2 and TLR4. 19 Lim et al suggested in the lungs of TLR4 knockout mice exposed to aluminum oxide nanoparticles and in a human airway epithelial cell line with downregulated TLR4, asthma-related allergic responses were reduced. 20 Therefore, asthma treatment and prevention may be achievable by targeting TLRs. 21 For instance, inflammatory monocytes can produce cytokines associated with Th1 when the TLR signaling pathway is activated, potentially alleviating allergy-induced asthma triggered by OVA. 22 In this study, the results also suggested that TG might be used to treat asthma by TLR signaling. Collectively, TG probably plays a role in preventing and treating asthma through apoptosis and TLR signaling pathways.
In this study, we randomly choose one of the primary TG components TP to verify. The compound-target network showed the compound TP from TG targeting RELA and TP53 was the most significant in anti-asthma (Figure 6b). Molecular docking also showed that TP formed stable conformations with RELA as well as TP53, supporting the notion TP might play an anti-asthma effect by targeting RELA or TP53. Previous studies have demonstrated that TP can suppress ASMC proliferation through G0/G1 cell cycle arrest and inhibition of the AKT/NF-κB/cyclinD1 signaling pathway, contributing to asthma airway remodeling. 23 This finding aligns with our result that TP might play a role in improving airway remodeling by targeting RELA. Regarding TP53, although it has been implicated in asthma, 15 there was no report on the effect of TP53 regulation by TP on asthma, which promoted us to further investigate.
Through preliminary in vitro experiments, we found the effectiveness of TG and TP in improving airway remodeling by reversing the mRNA expression of epithelial-mesothelial transdifferentiation markers CDH1 and FN1. Moreover, using the Luminex liquid suspension array, we observed that the compound TP reduced VEGF secretion of 16HBE cells. VEGF, known as a pro-inflammatory factor, is primarily secreted by bronchial epithelial cells and related to airway remodeling by modulating MUC5AC or matrix metalloproteinase-9 in asthma. 24 Sputum supernatants from asthmatic patients exhibited higher levels of VEGF than those from normal subjects, positively correlated with airway vascular permeability index. 25 Plasma VEGF could serve as a serological marker relevant to recurrent wheezing in allergic patients. 26 VEGF enhanced the activity of peripheral eosinophils (P < 0.05), but it was independent of the VEGF concentration. 27 In addition, Shen et al found that sevoflurane inhibited the OVA-induced VEGF expression and effectively suppressed airway remodeling. 28 Therefore, targeting VEGF is a promising therapeutic approach for asthma. Our results illustrated that TP inhibited the secretion of VEGF, which provided a new potential mechanism by which TP improved airway remodeling and implied that TP would be a promising method to treat asthma.
Conclusions
In summary, TP and Tripterine emerge as key compounds of TG for asthma treatment. TG exhibits potential effective against asthma by targeting RELA and TP53 and influencing pathways such as TLR, TNF, and apoptosis. TP is identified as capable of improving airway remodeling, likely through the reduction of VEGF expression. Accelerating asthma drug development requires further in vivo research to verify the efficacy of TG and TP.
Methods
Screening Bioactive Ingredients from TG
The compounds of TG were compiled from Chinese literature reports available on China National Knowledge Infrastructure (https://www.cnki.net/). Subsequently, each compound underwent drug-likely evaluation using the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform database (TCMSP) (http://lsp.nwu.edu.cn/tcmsp.php) that contains the ADME information. Bioactive ingredients were identified with oral bioavailability ≥ 20% and drug likeness ≥ 0.18. For compounds not registered in TCMSP, the canonical SMILES from PubChem (https://pubchem.ncbi.nlm.nih.gov/) were downloaded and subjected to the FAFdrugs4 (http://fafdrugs4.mti.univ-paris-diderot.fr) for computational prediction of ADME-Tox properties, assisting hit selection before chemical synthesis or ordering.
Identification of TG Anti-Asthma Targets
TG targets were identified through high-throughput experiment and reference-guided database of TCM (HERB, http://herb.ac.cn/). For ingredients lacking targets in HERB, predictions were obtained using Swiss Target Prediction(http://www.swisstargetprediction.ch/), STITCH (http://stitch.embl.de/), and SEA (http://sea.bkslab.org) to gather the predicted targets. String (https://string-db.org/) was employed to standardize target names. To identify the asthma-associated targets, the Drugbank (https://go.drugbank.com/), DisGeNET (http://www.disgenet.org/), and GeneCards (https://www.genecards.org/) databases were searched using the keywords “asthma.” Finally, a Venn diagram was used to pinpoint TG anti-asthma targets, elucidating the mechanism of TG's anti-asthma activity.
Enrichment Analysis
To annotate the functions associated with putative targets, pathway enrichment analysis and GO terms were performed using the Database Visualization and Integrated Discovery software (DAVID, http://david.abcc.ncifcrf.gov/home) with a significance threshold set at P < 0.05. The top GO terms and pathways were selected and visualized on the bioinformatics websites (http://www.bioinformatics.com.cn/).
Relationship map Construction
By integrating TG targets into Metascape(https://metascape.org/gp/index.html), we obtained the clustered PPI network using, using MCODE, and this network was further analyzed and modified using Cytoscape 3.6.0 (http://cytoscape.org/). The TG anti-asthma targets were then imported into Cytoscape 3.6.0 and the background network was downloaded from High-quality INTeractomes (HINT, http://hint.yulab.org). Afterwards, potential targets were screened using topological properties. Finally, an interaction network involving ingredients, targets, and pathways was also established by Cytoscape 3.6.0.
Molecular Docking
We downloaded the 2D structure of TP from Pubchem, minimized its energy, and saved it as a mol2 file by ChemOffice software. Then, the mol2 file was imported into AutoDock Tools 1.5.6 as a ligand. After detecting the root, selecting the twisted key and exporting in pdpqt format, the file was prepared for docking. The 3D structure of RELA and TP53 targets, 6XNU and 3D08 were retrieved from the protein data bank database (https://www.rcsb.org/), respectively. Pymol software was utilized to eliminate the solvent and organic components from these targets, and AutoDock Tools 1.5.6 was used to add hydrogen atoms. AutoDock Vina facilitated the docking process, and critical interactions were visualized.
Cell Culture and Drug Treatment
The bronchial epithelial cell 16HBE was obtained from WheLab (Shanghai, China). The cells were cultured in Dulbecco's modified Eagle's medium supplemented with 10% FBS (Gibco, USA) and 1× penicillin-streptomycin (Gibco, USA). Subsequently, cells were sub-cultured in 6-well plates at a density of 2 × 105 cells per well or in 96-well plate at a density of 5 × 103 cells per well 24 h prior to experimentation. Following a 16-24 h period of serum-free medium starvation, cells were subjected to stimulation with 10 ng/ml TGF-β1 (PeproTech, USA), either alone or in combination with TG (Xi’an Haoxuan Biotechnology Co. Ltd, Shanxi, China) or TP (Xi’an Haoxuan Biotechnology Co. Ltd, Shanxi, China), for 48 h. The experiment involved four groups: TGF-β1 group, TGF-β1 + TG group, TGF-β1 + TP group, and Control group.
Cell Viability Assay
The cells were cultured in 96-well plates prior to experimentation. After starvation, cells were treated with varying concentrations of TG or TP for 48 h. Subsequently, 10 μl cell counting Kit-8 reagent (Solarbio, China) was added to medium and incubated for 2 h. The optical density (OD) was detected at 450 nm and the cell viability was calculated using the following formula:
Real-Time Quantitative Polymerase Chain Reaction
Total RNA was extracted from cells using RNAiso Reagent (Takara, Janpan). An equal amount of 1 μg RNA was taken from each group for removing genomic DNA and reverse transcribed into cDNA using a PrimeScript RT Reagent Kit (Takara, Japan). PCR was performed using SGExcel FastSYBR Master (Sangon Biotech, Shanghai, China). Internal reference, actin, was used to standardize the data, which were analyzed using the 2−ΔΔCT method. Primers used in the study are detailed in Table 2 and were synthesized by Sangon Biotech (Shanghai, China).
The List of Specific Primer Sequences Used in RT-PCR.

RT-PCR, real-time quantitative polymerase chain reaction.
Cell-Secreted Cytokine Assay
After the cells were stimulated by TGF-β1 or treated with TP, cell supernatants were collected following centrifugation at 1000 rpm,10 min with four duplicate samples for each group. The liquid suspension array was performed using the Bio-Plex Pro Human Cytokine Grp I Panel 27-plex (Wayen Biotechnologies, Shanghai, China) in accordance with the manufacturer's instructions. The values were read using the Bio-Plex MAGPIX System (Bio-Rad). The target cytokines were further valuated in cell supernatants using commercial enzyme linked immunosorbent assay kit (MEIMIAN, Jiangsu, China) according to the manufacturer's instructions.
Statistical Analysis
Data were analyzed using graphpad prism (version 7, GraphPad Software, San Diego, CA). For normally distributed data, between-group comparisons were made by using Student's t test and comparisons of three or more sets of data were conducted by one-way ANOVA or two-way ANOVA. For data that were not normally distributed, Kruskal-Wallis or Friedman ANOVA was used. All comparisons were two-tailed, and P-values of less than 0.05 were considered significant.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241238904 - Supplemental material for Tripterygium Glycosides in the Treatment of Asthma: Integrating Network Pharmacology, Molecular Docking and In Vitro Preliminary Pharmacological Evaluation
Supplemental material, sj-docx-1-npx-10.1177_1934578X241238904 for Tripterygium Glycosides in the Treatment of Asthma: Integrating Network Pharmacology, Molecular Docking and In Vitro Preliminary Pharmacological Evaluation by Shuhui Gao, Min Yan, Tianci Jiang, Jingyao Lian, Zhe Cheng, Huahui Zeng and Junwei Zhao in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X241238904 - Supplemental material for Tripterygium Glycosides in the Treatment of Asthma: Integrating Network Pharmacology, Molecular Docking and In Vitro Preliminary Pharmacological Evaluation
Supplemental material, sj-docx-2-npx-10.1177_1934578X241238904 for Tripterygium Glycosides in the Treatment of Asthma: Integrating Network Pharmacology, Molecular Docking and In Vitro Preliminary Pharmacological Evaluation by Shuhui Gao, Min Yan, Tianci Jiang, Jingyao Lian, Zhe Cheng, Huahui Zeng and Junwei Zhao in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X241238904 - Supplemental material for Tripterygium Glycosides in the Treatment of Asthma: Integrating Network Pharmacology, Molecular Docking and In Vitro Preliminary Pharmacological Evaluation
Supplemental material, sj-docx-3-npx-10.1177_1934578X241238904 for Tripterygium Glycosides in the Treatment of Asthma: Integrating Network Pharmacology, Molecular Docking and In Vitro Preliminary Pharmacological Evaluation by Shuhui Gao, Min Yan, Tianci Jiang, Jingyao Lian, Zhe Cheng, Huahui Zeng and Junwei Zhao in Natural Product Communications
Footnotes
Author Contributions
Methodology, investigation, formal analysis, visualization, and writing-original draft: Shuhui Gao and Min Yan. Validation and writing-original draft: Tianci Jiang and Jingyao Lian. Conceptualization, writing-review & editing, supervision, and project administration: Zhe Cheng, Huahui Zeng and Junwei Zhao.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation for Young Teachers of Zhengzhou University, (grant number JC21854032).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
