Abstract
Saponins have significant bioactivities in treating cardiovascular disease. Whereas there is a lack of in-depth knowledge about how saponins prevent cardiovascular disease. Tissue factor (TF) is the major initiator of the coagulation cascade and plays an important role in hemostasis and thrombosis. However structure–activity relationships (SARs) of saponins inhibiting TF activity have not been discussed in detail at present. To further clarify the relationships between saponins and TF, in this study, 10 representative saponins were selected to study the inhibitory effect on TF procoagulant activity of monocytes by an improved chromogenic substrate method, and the possible SARs were preliminarily analyzed. Furthermore, molecular docking analysis suggested that 4 representative saponins had a good affinity with TF/FVIIa. In addition, a representative saponin, ruscogenin, decreased both messenger ribonucleic acid and protein levels of TF in human monocytes partly due to its downregulation of nuclear factor kappa-light-chain-enhancer of activated B cells and c-Jun N-terminal kinase pathways. In conclusion, this study provides further explanation for the cardiovascular protection of saponins, and the analysis of SARs between inhibiting TF activity and saponins will be helpful to explore the therapeutic TF inhibitors.
Saponins are composed of sapogenins and sugars, uronic acids, or other organic acids, classified into two types according to the structure of sapogenin: (1) steroid saponin, whose sapogenin is derivative of spirosterane, mostly consisted of 27 carbon atoms (eg, diosgenin); (2) triterpenoid saponin, its sapogenin belongs to triterpene ramifications, mostly including 30 carbon atoms. Triterpene saponins are divided into tetracyclic triterpenes and pentacyclic triterpenes. Such saponins lie in plants like the genus Araliaceae and Umbelliferae. 1 -3 Individual saponins can engender special physiological activities. For example, panax notoginseng saponin R1, ginsenoside Rb1, and ginsenoside Rg1 have the characteristics of maintaining blood circulation, anti-arrhythmia, improving myocardial ischemia, 4 antidiabetes, 5 antioxidation, 6 and anticancer cell proliferation. 7 With the development of saponin chemistry, numerous new biological activities have been gradually discovered, especially the prevention and treatment of cardiovascular and cerebrovascular diseases have drawn extensive interest in the world. 8
Tissue factor (TF), or clotting factors III, also known as cytozyme or thromboplastin, is a single transmembrane glycoprotein containing 263 amino acid residues and holds structural homology with type II cytokine receptors. 9 The complex of TF–FVIIa gradually forms when TF is exposed to blood clotting factor VII/VIIa. Subsequently, TF–FVIIa activates coagulation factors X (FX) and IX (FIX) to initiate the coagulation cascade. 10 There are several inflammatory factors such as tumor necrosis factor-alpha (TNF-α), lipopolysaccharide (LPS), thrombin, histamine, which induce high expression of TF in monocytes/macrophages, vascular endothelial cells, smooth muscle cells, platelets, and atheromatous plaque. 11,12 Recent studies have discovered that increasing TF activity in vivo could cause atherosclerosis, disseminated intravascular coagulation, angina pectoris, thromboembolism, and so on. 13 Atherothrombosis occurs mainly due to the production of TF, rupture of atherosclerotic plaque with thrombosis will result in acute cardiovascular events exactly as myocardial infarction and stroke. 14 The research of TF/FVII inhibitors increasingly caught people’s attention and it was of great significance to search for TF/FVII inhibitors for anticardiovascular diseases.
Many traditional Chinese medicines such as Panax ginseng C.A.Mey, Panax notoginseng (Burk.) F.H.Chen, Anemarrhena asphodeloides Bunge, Polygala tenuifolia Willd., Glycyrrhiza uralensis Fisch., Platycodon grandiflorum (Jacq.)A.DC. contain plenty of saponins. 15,16 Some research reported that saponins have been treated as an important source of TF natural inhibitors and extensively used in clinical practice such as diaoxinxuekang capsule: representative of clinical application of total steroid saponins. 17 However, the SARs of various saponins relating to the inhibition of TF activity has not been discussed in detail until now.
In this present study, 10 representative saponins belonging to groups of steroid sapogenins, steroid saponins, triterpene saponin, and ecdysterone were investigated in order to determine their activity as inhibitors of TF in relation to the structure (the 10 chemical structures were shown in Figure 1). Besides, molecular docking simulations were implemented to probe the binding modes of the active ligands to the amino acids in the active site of TF/FVIIa protein. In addition, the effects of ruscogenin on the expression of TF messenger ribonucleic acid (mRNA) and protein were investigated by reverse transcriptase-polymerase chain reaction (RT-PCR) and Western blotting assay, respectively.

Chemical structures of 10 saponins and sapogenins.
In order to evaluate the feasibility of saponins as TF inhibitor and explore the SARs, TF procoagulation activity was measured using improved chromogenic substrate methods.
18
As shown in Figure 2, 7 compounds including ruscogenin, diosgenin, pennogenin, parissaponin Pe, pm 13 (pennogenin-3-O-α-
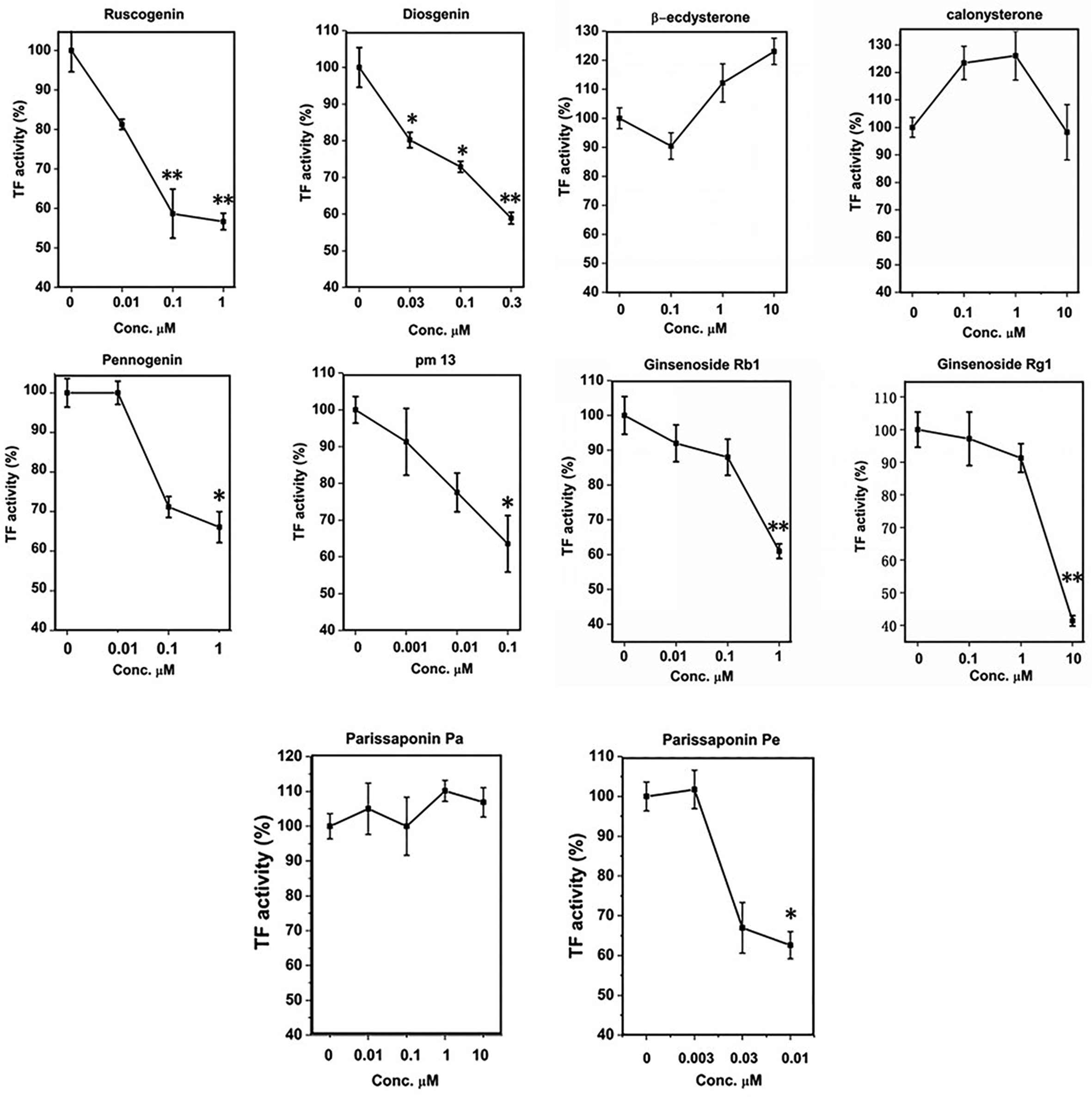
Concentration–response curves of 10 saponins on tissue factor (TF) procoagulation activity in human monocytes. All data presented are the mean ± standard deviation (n = 4) of 1 representative experiment from 3 independent experiments. *P < 0.05, **P < 0.01 compared with normal where the cells were treated with a control vehicle.
Effects of 10 Saponins and Sapogenins on Lipopolysaccharide-Induced Tissue Factor Activity in Human Monocytes.

IC50, half-maximal inhibitory concentration.
aThe IC50 are mean values from at least two replicates.
bEffects can be neglected at the tested concentrations.
The results of this study indicated that for steroid saponins or sapogenins, the intensity of the activity of the 3 steroid sapogenins was: ruscogenin (IC50 = 20 nM) > diosgenin (IC50 = 40 nM) > pennogenin (IC50 = 480 nM). There were the only difference in the number and position of hydroxy group among the 3 steroid sapogenins: ruscogenin showed 2 hydroxy groups at C-1 and C-3 position, diosgenin with 1 hydroxy group situated at C-3 position, and pennogenin had hydroxy groups at C-3 and C-17 position. The results suggested that the hydroxyl group at C-1 position could increase the inhibitory activity of TF, and probably hydroxyl group at C-3 position was the necessary group of sapogenin relating to the inhibition of TF activity. The explanation might be the enhanced hydrophily afforded by the hydroxyl groups. Nevertheless, the activities of steroid saponin were higher than the corresponding sapogenin: parissaponin Pe (IC50 = 6.0 nM) > diosgenin (IC50 = 40 nM); pm 13 (IC50 = 50 nM) > pennogenin (IC50 = 480 nM), which indicated that C-3 glycosyl group played an important role in TF inhibitory activity of these compounds. The intensity of the activity of three steroid saponins was: parissaponin Pe > pm 13 > parissaponin Pa. The number and type of glycosyl groups linked at the C-3 position of steroid saponins were different, and the activities of disaccharide glycosides in spirosterane compounds evaluated in this article were stronger than that of trisaccharide glycosides. Calonysterone and β-ecdysterone belonged to the ecdysterone compound, which had no inhibitory effect on TF activity. It suggested that if E and F ring of the steroid mother nucleus opened, the activity of inhibition of TF would disappear.
Ginsenoside Rb1 and Rg1 were attributed to triterpene saponin, according to its structure, which can be segmented into 20 (S)-protopanaxadiol (PPD) and 20 (S)-peotopanaxatriol (PPT). PPD or PPT ginsenosides combined with glycosyl at C-3 and/or C-20 positions, while PPT ginsenosides connected with hydroxyl at C-3 position. Among them, Rb1 was a part of PPD type and Rg1 pertained to PPT type. 8,19 Their IC50 values were 340 nm and 1030 nm, respectively, which illustrated that tetracyclic triterpenoid saponins of damane type could generate some inhibitory effect on TF activity, but their activity was not as strong as steroid saponins. However, the activity of ginsenoside Rg1 was quite different from that of ginsenoside Rb1. The reason might be that some of the glycosides on ginsenoside Rg1 were hydrolyzed.
In recent years, molecular docking has played an important role in obtaining new drug candidates and reducing costs via computational tools in a short time. The purpose of docking is to combine the ligand into the binding pocket of the receptor and further predict the interaction and energy between the receptor and the ligand. 20 For the sake of further evaluating saponins’ biological activities, we selected 4 representative saponins including parissaponin Pe, ruscogenin, diosgenin, and ginsenoside Rb1. Among them, parissaponin Pe was steroid saponin, ruscogenin and diosgenin belonged to steroid sapogenins, and ginsenoside Rb1 was triterpenoid saponin. They were representative saponins and had strong bioactivities. In order to observe their binding posture with TF/FVIIa protein, the program of Autodock-vina was used, which utilized a semiempirical free-energy force field to predict the binding energy of receptor and ligand, 21 as well as determined which residues from its active site were involved in the interaction with these compounds. As shown in Table 2, the 4 ligands showed high binding energy ranging from −6.2 to −8.5 kcal/mol. Figure 3 represented the interaction mode between ligands and TF/FVIIa protein. From the results, we came to know that all 4 ligands got into the active pocket of the protein and matched with the active pocket well. In the hydrophobic pocket of TF/FVIIa protein, 4 ligands could form the hydrogen bonds with relevant amino acids such as Tyr94, Asp60, Gly193, Leu40, Arg147, Asp146, which could exert an important role in maintaining the supramolecular structure of the complex. To sum up, the results of molecular docking studies revealed the interaction and precise binding sites between receptor protein TF/FVIIa and 4 saponins, which further supported the data of bioactivity determination and pointed out the potential direction of future chemical modification. These molecular simulations provided a reasonable explanation for the interaction between the 4 saponins and TF/FVIIa and supplied valuable information for further discovery of TF/FVII inhibitors.

Molecular modeling results pointed out the binding modes and interactions of 4 ligands with tissue factor (TF)/FVIIa protein. Protein is rendered in light yellow ribbons and compounds and relevant amino acids in sticks. H-bonds are indicated as yellow dashed lines. Results for parissaponin Pe (a), ruscogenin (b), diosgenin (c), and ginsenoside Rb1 (d) are illustrated.
Binding Energy and H-Bond Interaction of Compounds with Tissue Factor/FVIIa.

On the other hand, monocytes did not express or express a small amount of TF in physiological cases. However, when some inflammation medium (interleukin (IL)-1, IL-6, TNF-α) and certain factors (thrombin, LPS, interferon-γ, C-reactive protein) stimulated, monocytes could induce expression of TF, the most studied of which was the induced expression of TF by bacterial endotoxin LPS. 22,23 Modern studies have shown that the induced expression of TF was mainly regulated at the transcriptional level and then adjusted the expression of TF protein. 24 The expression of TF mRNA in PBMC induced by LPS and TNF-α was investigated by RT-PCR (Figure 4). Considering its supernal inhibitory effect on TF activity and its typical structure, we observed the effect of ruscogenin on TF mRNA expression. The results revealed that ruscogenin could inhibit the expression of TF mRNA in monocytes induced by LPS or TNF-α, suggesting that ruscogenin also regulated the expression of TF at the transcriptional level. Meanwhile, to further evaluate whether ruscogenin suppressed TF protein expression, TF protein expressions were detected in PBMCs and THP-1 cells induced by TNF-α by western blotting assay. As shown in Figure 5(a), TF protein was almost not detectable in unstimulated monocytes, but TNF-α (10 ng/mL, 5 hours) markedly induced TF expression by about fourfold. Ruscogenin (0.01-1 μM) diminished TF expression induced by TNF-α in a concentration-dependent manner in human monocytes. Furthermore, similar results were obtained with THP-1 cells. TNF-α (10 ng/mL, 5 hours) markedly induced TF protein expression, and ruscogenin (0.01-1 μM) also suppressed TNF-α induced TF expression dose-dependently in THP-1 cells (Figure 5(b)). Therefore, ruscogenin decreased both mRNA and protein levels of TF stimulated with LPS or TNF-α in human monocytes. This might be one of the anti-inflammatory and antithrombotic mechanisms of ruscogenin.

Effect of ruscogenin on tissue factor (TF) messenger ribonucleic acid expression in human monocytes stimulated by tumor necrosis factor-alpha (TNF-α) (a) or lipopolysaccharide (LPS) (b). All data presented are the mean ± standard deviation of 3 independent experiments. ## P < 0.01 compared with control; *P < 0.05, **P < 0.01 compared with model where the cells were treated with TNF-α or LPS.

Effects of ruscogenin on tissue factor (TF) protein expression in human peripheral blood monocytes (a) and THP-1 cells (b) stimulated by tumor necrosis factor-alpha (TNF-α). All data presented are the mean ± standard deviation of 3 independent experiments. ## P < 0.01 compared with control; *P < 0.05 compared with TNF-α alone.
To further determine the mechanism by which ruscogenin inhibited TF expression and TF activity, we investigated the expression and phosphorylation of p38MAPK, c-Jun N-terminal kinase (JNK), and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)/p65 induced by TNF-α in THP-1 cells. TNF-α plays a key role in chronic inflammatory conditions and cardiovascular risk. 25 Therefore, we chose TNF-α as the main stimulator in the study for molecular mechanisms. First, we investigated the effect of ruscogenin on TNF-α induced NF-κB activation. NF-κB/p65 phosphorylation was induced by TNF-α (10 ng/mL) after stimulation for 5, 15, and 30 minutes, and pretreatment with ruscogenin at a concentration of 1 µM decreased p65 phosphorylation by about 38% at 5 minutes and 56% at 15 minutes (Figure 6(a)). Next, we determined whether ruscogenin affects TNF-α-induced p38MAPK and JNK activation in THP-1 monocytic cells. In unstimulated THP-1 monocytic cells, p38MAPK and JNK were minimally phosphorylated. JNK was phosphorylated peaking at 5 minutes after TNF-α (10 ng/mL) stimulation and pretreatment with ruscogenin at a concentration of 1 µM decreased the phosphorylation of JNK by about 68% at 5 minutes (Figure 6(b)). Phosphorylation of p38MAPK was gradually increased from 5 to 30 minutes after TNF-α (10 ng/mL) stimulation. However, no significant effect on p38MAPK activation was observed with ruscogenin at a concentration of 1 µM (Figure 6(c)). These results indicated that ruscogenin might downregulate the phosphorylation of NF-κB/p65 and JNK induced by TNF-α, but not the phosphorylation of p38MAPK in monocytes.

Effects of ruscogenin on activation of p38, c-Jun N-terminal kinase (JNK), and NF-κB/p65 in THP-1 cells stimulated by tumor necrosis factor-alpha (TNF-α). (a–c) THP-1 cells were pretreated with or without ruscogenin (1 μM) for 1 hour and then exposed to TNF-α (10 ng/mL) for 0-30 min; then p-p38, p-JNK, and p-NF-κB/p65 were determined by Western blotting analysis (upper panel) and quantitation of protein phosphorylation were performed by densitometric analysis (lower panel). All data presented are the mean ± standard deviation (n = 3) of 1 representative experiment from 3 independent experiments.
In conclusion, preliminary SAR analysis stated that 3-hydroxyl group was the possible necessary group of sapogenin for inhibiting TF procoagulant activity, the 1-hydroxyl group increased the inhibitory activity of TF, and it was also equally important that the presence of the glycosylation groups greatly affected the inhibitory action of spirostane compounds. Steroid saponin showed higher activity than the corresponding sapogenin. Ecdysterone had no inhibitory effect on TF activity, suggesting that the inhibitory activity of TF disappeared when the E ring and F ring were unfolded in the steroid mother nucleus. The activity of dammarane-type tetracyclic triterpenoid saponin was not as strong as steroid saponin. Meanwhile, the results of autodock/Vina correlated well with the results of inhibiting TF activity in vitro for these saponins and it further provided a reasonable explanation for the SARs of saponins inhibition of TF. In addition, a representative saponin, ruscogenin, decreased both mRNA and protein levels of TF in human monocytes partly due to its downregulation of NF-κB and JNK pathways. To the best of our knowledge, this was the first time that studies have been conducted on the SARs of several saponins related to inhibition of TF activity. These results will really help to explore therapeutic TF inhibitors of saponins and further understand the protective mechanisms of saponins against cardiovascular disease.
Experimental
Reagents
Compounds
PBMC Isolation and Culture
PBMC were from the blood of healthy volunteers. As described in previous studies, 26 PBMC were isolated by density gradient centrifugation using Ficoll–Hypaque solution (density: 1.077). Heparin was added to 20-50 mL of blood as an anticoagulant at a final concentration of 10 U/mL. After diluted with phosphate-buffered saline (PBS) at 1:1 ratio, the whole blood was superinduced to the upper layer of the human Ficoll–Hypaque solution along the tube wall in the same proportion, and centrifuged at room temperature for 15 minutes to gain 1-2 mL of white membrane cells at the interface between plasma and solution. Then washing cells twice with Hank’s balanced salt solution by centrifugation for 10 minutes, white cell precipitation was visualized through centrifugation, PBS was discarded, and the cell pellet was suspended in RPMI 1640 medium containing 10% heat-inactivated calf serum.
THP-1 Cell Culture
We also used monoblastic leukemia cell line THP-1 cells in this study, which were kindly provided by Stem Cell Bank, Chinese Academy of Sciences. Cells were cultured in RPMI-1640 medium supplemented with 10% fetal calf serum, 0.2% sodium carbonate, 50 U/mL penicillin–streptomycin, and 2-mercaptoethanol to a final concentration of 0.05 mM.
Induction of TF and Drug Treatment
PBMC were suspended in 10% 1640 medium, inoculated in 96-well culture plates at a cell density of 3-5 × 106 cells/mL, and then incubated at 37°C in 5% carbondioxide. Primary cultured PBMC were incubated with control vehicle (90 µL 0.1% dimethyl sulfoxide [DMSO], 10 µL PBS) or experimental vehicle (90 µL 0.1% DMSO, 10 µL LPS or TNF-α) in 96-well plates for 1 hour, then 10μL LPS (100 ng/mL) or TNF-α (20 ng/mL) stimulated for 5 hours in order to induce TF activity. 27 After the completion of the culture, the cells were pelleted by centrifugation and resuscitated in RPMI medium. The cell suspension was frozen to TF activity assay at −20°C.
Measurement of Monocytes TF Activity
TF activity was measured by an improved chromogenic substrate method. When the drug was incubated with the cells for 5 hours, aspirated the drug-containing medium in the cell plate. One hundred microliters of serum-free 1640 culture solution were added to each well, and the cells were repeatedly frozen and thawed at −70°C to room temperature 3 times, the cells were lysed and the lysate was evenly blown; 45 µL was absorbed and inoculated it in another new 96-well plates, which was incubated for 5 minutes, the freshly prepared human prothrombin complex (PPSB) solution containing 5 mmol/L CaCl2 (0.01 g/mL, 5 µL/well) was superinduced to the culture medium, incubated for 15 minutes, and the reader microplate meteraged absorbance at 405 nm, at length the purified recombinant human TF was used to generate the standard curve. The relative IC50 values of natural compounds on TF inhibition were calculated using origin 8.0 software according to the fitted dose response curves, and the formula of TF inhibition was shown below.

Molecular Docking
Molecular docking using Autodock Vina 1.1.2 was performed to study the binding mode of compounds with TF/FVIIa. 28 The 3-dimensional structures of parissaponin Pe, ruscogenin, diosgenin, and ginsenoside Rb1 compounds were plotted by ChemBioDraw Ultra 14.0 and ChemBio3D Ultra 14.0 software. We chose the complex (TF–FVIIa) as protein model. Construction of molecular docking protein model—setting up the keyword “Coagulation Factor VII” to retrieve the conformation in RCSB PDB database (http://www.rcsb.org/pdb/home/home.do), then qualified conditions for screening, and the final PDB number was 4YLQ. 29 The protein was first treated with autotools 1.5.7 to remove water molecules, and the energy lattice was calculated with autogrid.The grid box coordinates were set at 1.588, −6.319, −52.186; the box size was 20 × 20 × 20 mesh points; the distance of each small mesh point was 1 nm. Autodock vina 1.1.2 was used for batch docking of small molecules with proteins. The results were set to return to 9 conformations. The optimum scoring posture judged by Vina docking score was selected. Visual analysis was performed using Schrodinger Maestro 12.0.
RT-PCR for Detection of TF mRNA
The total RNA was extracted by 1-step method with Trizol reagent; the concentration and purity were detected by ultraviolet spectrophotometry, and further by reverse transcription synthesized complementary deoxyribonucleic acid (cDNA). RT-PCR reaction was performed as described in the previous study. 30 The reverse transcripted cDNA was then amplified by PCR with the following primers (Genebase, Shanghai, China): TF, 5′-GAACCCAAACCCGTCAAT-3′ (sense) and 5′-TCTCATACAGAGGCTCCC-3′ (antisense); β-actin, 5′-AGCGGGAAATCGTGCGTGAC-3′ (sense) and 5′-AGTTTC GTGGATGCCACAGGAC-3′ (antisense). The reaction conditions were as follows: 5 minutes at 94°C, then 32 cycles of 72°C for 30 seconds, 55°C for 45 seconds, and 72°C for 45 seconds. After amplification, under the conditions of voltage 90 V, 1.5%-1.8% agarose gel electrophoresis comprising 5 g/mL ethidium bromide electrophoresed, electrophoresis for 30 minutes, followed by ultraviolet scanning, ultimately the ratio of TF/β-actin represented the expression abundance of TF mRNA.
Western Blotting Assay
Human PBMCs and THP-1 cells were harvested in lysis buffer for Western blotting. The samples (20-40 μg) were treated with 5× sodium dodecyl sulfate–polyacrylamide gel electrophoresis sample buffer, followed by heating at 95°C for 5 minutes and were then subjected to 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis. The proteins were then transferred to polyvinylidene difluoride membranes. The membranes were blocked with 3% bovine serum albumin and incubated with the primary antibodies. Immunoreactive bands were detected by a chemiluminescence system (ECL plus, Amersham).
Statistical Analysis
All data were expressed as mean ± standard deviation. Data were analyzed by one-way analysis of variance and analyzed by Student’s 2-tailed t-test for comparison between 2 groups, in succession Dunnett’s test when the data involved 3 or more groups.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Graduate Innovation Fund Project of Guizhou Province of China (No. YJSCXJH2019036), the Talent Introduction Project of Guizhou University of China (No. 2010039) and the Science and Technology Project of Guizhou Province of China (No.[2019] 1120).
