Abstract
Introduction
Asparagi radix (AR) is the dried root of
Inflammation is an adaptive response triggered by harmful stimuli and conditions, such as infection and tissue damage. Inflammation is the basis of a variety of physiological and pathological processes. It is also the cause and complication of many diseases. For example, inflammation is involved in the study of cardiovascular system, nervous system, respiratory system, and digestive system diseases. 24 The pathological process of inflammation is complex, and its mechanisms are related to arachidonic acid metabolism, inflammatory cell function and autoimmune process. These processes are interrelated and influence each other. However, studies have found that arachidonic acid metabolism plays a major role in these processes. When various inflammatory factors and immunogens stimulate tissue cells, cell membranes are disrupted, and membrane phospholipase A2 (PLA2) is activated. It hydrolyzes intracellular phospholipids to release free arachidonic acid 25 and can be metabolized by the cycloxygenase (COX) biochemical pathway. Cyclooxygenase (COX), also known as prostaglandin synthase, is a key rate-limiting enzyme that catalyzes the synthesis of prostaglandin (PG) from arachidonic acid. COX has two isozymes, constitutive COX-1 and inducible COX-2. 26 COX-2 is induced by inflammation, and COX-2 is almost not expressed in various tissue cells under normal conditions. When the body is stimulated by inflammatory factors, some cells, such as endothelial cells, vascular smooth muscle cells, monocytes/macrophages, and fibroblasts, will be induced to express COX-2, so that the level of COX-2 increases sharply, causing the synthesis and accumulation of inflammatory PG in the damaged site, promoting an inflammatory response and tissue damage. 27 Hence, COX-2 has long been considered to play a key role in the pathogenesis of inflammation, as it can be rapidly expressed in response to a variety of pro-inflammatory mediators and cytokines. 28
With the deepening of research on the efficacy of active ingredients in AR, the research on their extraction, separation, and evaluation methods has been paid more and more attention. However, the traditional extraction, separation, and analysis methods are gradually replaced by some new extraction, separation, and analysis techniques due to the cumbersome steps, the loss of active ingredients, and the low preparation efficiency.29,30 Charged aerosol detector (CAD) is a new type of universal liquid chromatography detector. It has high sensitivity and good repeatability, and its response value does not depend on the optical properties or ionization efficiency of the measured substance. It can detect all non-volatile and many semi-volatile substances and can replace an ultraviolet detector (UV) to detect compounds with non-ultraviolet or weak ultraviolet activity.
31
It has become an important means of traditional Chinese medicine analysis.
32
Compared with high-performance liquid chromatography (HPLC), ultra-high-performance liquid chromatography (UPLC) is characterized by a good separation effect and relatively fast analysis speed, while quadrupole-time-of-flight tandem mass spectrometry (Q/TOF-MS) has strong selectivity and high sensitivity, which can accurately detect the relative molecular mass. The secondary mass spectrometry produced by it can prompt the information of characteristic ion fragments.
33
Therefore, it is urgent to establish a rapid, portable, and accurate method to screen and identify the bioactive components in
In this study, HPLC-CAD fractions technology is used to separate, collect, and enrich the components in AR. Then, the anti-inflammatory activity of the fractions collected at different times is determined, and the components in AR fractions is further analyzed and identified by UPLC-Q-TOF/MS (Figure 1). Finally, through data integration, the substances with potential anti-inflammatory activity in AR are obtained, hoping to provide some reference for the follow-up study of the anti-inflammatory activity of monomer compounds in AR.

Technology roadmap.
Experimental Methods and Materials
Reagents and Chemicals
The fresh AR is collected from Guizhou Fengtai Agricultural Development Co., Ltd (Qianxinan Prefecture, China) (Batch number: 20211005), harvested in autumn. AR is identified as the root tuber of
Preparation of AR Aqueous Extract
A total of 500 g of AR fresh product was taken, and the fibrous roots were removed. After washing, it was boiled in boiling water for 30 min. After cooling, it was peeled and dried in an oven at 60°C for 24 h to obtain AR medicinal materials. After the powder was beaten, 5 g of crude powder of
Preparation of HPLC-CAD Fractions
Distillates were prepared using a fraction collector (BSZ-100, Shanghai Jiapeng Scientific Instruments, Shanghai, China). The samples were injected into the HPLC system for separation, and then the fractions were collected by setting the fraction collector FC every 60 s, evaporated to dryness with nitrogen, redissolved with 100 μL methanol, and analyzed by UPLC-Q/TOF-MS.
Data Statistical Analysis
By consulting Pubchem, CNKI, ChemSpider, and other databases and websites, the names, structural formulas, molecular formulas, relative molecular weight, characteristic fragment ions, and mass spectrometry fragmentation rules of the chemical constituents of Asparagi radix were summarized. The simulated molecular formula function of SCIEX OS software was used, and then the structural formula was imported to match the characteristic fragment ions to determine the compound.
The anti-inflammatory activity of the water extract was measured, the fluorescence value (RFU) of each well was recorded, and the inhibition rate of each sample was calculated according to the following formula. The dose–effect curve was analyzed according to the analysis results of SPSS software. The dose–effect curve was analyzed by GraphPad Prism, and the IC50 value of the sample was calculated.
Finally, the anti-inflammatory activity of different fractions was determined by the IC50 value, and the results were statistically analyzed by SPSS, GraphPad Prism, Origin, and other software to determine the anti-inflammatory activity of each fraction. Finally, through the mass spectrometry information of different fractions and their anti-inflammatory activity results, compounds with potential anti-inflammatory activity were screened out.。
Identification of AR Chemical Constituents
HPLC-CAD Analysis
Chromatographic column: Waters chromatographic column (XBridge BEH Amide, 130 Å, 5 μm, 4.6 mm × 250 mm, Waters, USA); mobile phase: 10 mmol/L ammonium formate solution (pH4.0, A)-acetonitrile (B), gradient elution, see Table 1; flow rate: 0.7 mL/min; column temperature: 30°C; injection volume: 3 μL; atomization temperature: 50°C.
HPLC-CAD Gradient Elution Table.

UPLC-Q/TOF-MS/MS Analysis
Chromatographic column: ChromCore amide column (HILIC-Amide, 3 μm, 2.1 mm × 150 mm, nanoanalysis, China). Mobile phase: 0.1% formic acid water (A)-0.1% formic acid acetonitrile (B), gradient elution, as shown in Table 2. Flow rate: 0.35 mL/min. Column temperature: 35°C. Injection volume: 2.0 μL.
AR Water Extract UHPLC Gradient Elution.

Electrospray ionization (ESI) source; information-dependent acquisition (IDA) is performed under positive or negative ion detection. Ion spray voltage:5500 and −4500 V; ion source temperature: 550°C; atomizing gas (N2) pressure: 55 psi; auxiliary gas (N2) pressure: 55 psi; air curtain gas (N2) pressure: 35 psi; In TOF-MS mode, the scanning range is m/z 100-1500 Da. Declustering voltage (DP): 80 V (positive ion)/−80 V (negative ion); collision energy (CE): 10 V (positive ion)/−10 V (negative ion); the TOF-MS/MS mode scanning range: m/z 50-1500 Da. Declustering voltage (DP): 80 V (positive ion)/−80 V (negative ion); collision energy (CE): 30 V (positive ion), − 30 V or − 50 V (negative ion).
Data Analysis
By consulting Pubchem, CNKI, ChemSpider, and other databases; and the name, structural formula, molecular formula, relative molecular weight, characteristic fragment ions, and mass spectrometry fragmentation rules of AR chemical components were summarized. The molecular formula function of SCIEX OS software was used to simulate the molecular formula function, and then the structural formula was introduced to match the characteristic fragment ions to determine the compounds, and then the anti-inflammatory active substances in AR were determined by combining the anti-inflammatory activity determination results.
Screening of AR Anti-Inflammatory Active Ingredients
Determination of Total Anti-Inflammatory Activity of Water Extract
The aqueous extract liquor (5 mL) of AR was diluted into 44.93, 22.46, 11.23, 7.49, 5.16, 4.49, 3.00, and 2.25 mg/mL sample solutions by adding purified water. The COX-2 inhibitory activity was screened according to the kit method. The 96-well plate was used to set the control hole and the sample hole. After the sample and each solution were added in turn, they were mixed and incubated at 37°C for 10 min. Each sample was set up with three repeat holes (n = 3). The inhibition rate was calculated according to the following formula.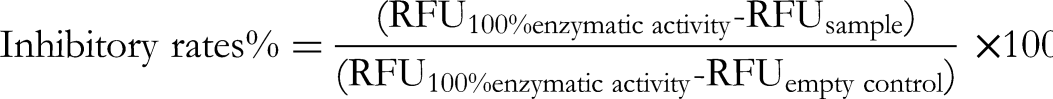
Determination of Anti-Inflammatory Activity of HPLC-CAD Fraction in Vitro
Water extracts (2 mL) of AR were centrifuged at 12 000 rpm for 10 min, and then filtered with 0.45 μm microporous membrane. UPLC-CAD was used to analyze the samples according to the chromatographic conditions of “Preparation of AR Aqueous Extract”. The fractions collected by FC were used, and some collectors were set to collect every 60 s. A total of 60 fractions were collected each time, and CAD was collected 6 times. The collected fractions were dried with nitrogen and redissolved with 80% methanol and water in 100 μL. Then, according to the method under Determination of total anti-inflammatory activity of water extract Section, three replicates were set for each sample (n = 3), and the anti-inflammatory activity of 60 fractions in different time periods was determined, and the COX-2 inhibition rate was calculated.
Results and Analysis
Identification of 50 Components in Asparagus cochinchinensis
Through the UPLC-Q/TOF-MS/MS analysis of the water extract of AR, the positive and negative ion extraction ion current chromatograms (XIC) of the identified compounds were obtained, as shown in Figure 2. As shown in the figure, the compounds of AR respond better in the negative ion mode; the excimer ion peaks are [M−H]− and [M + COOH]−, and in the positive ion mode, the main excimer ion peak is [M + H]+. Through the database information, the retention time, accurate molecular weight, fragment ions, and mass spectrometry fragmentation rules of the compounds were compared, and a total of 50 compounds were identified. The identified chemical constituents and mass spectrometry information are shown in Table 3.
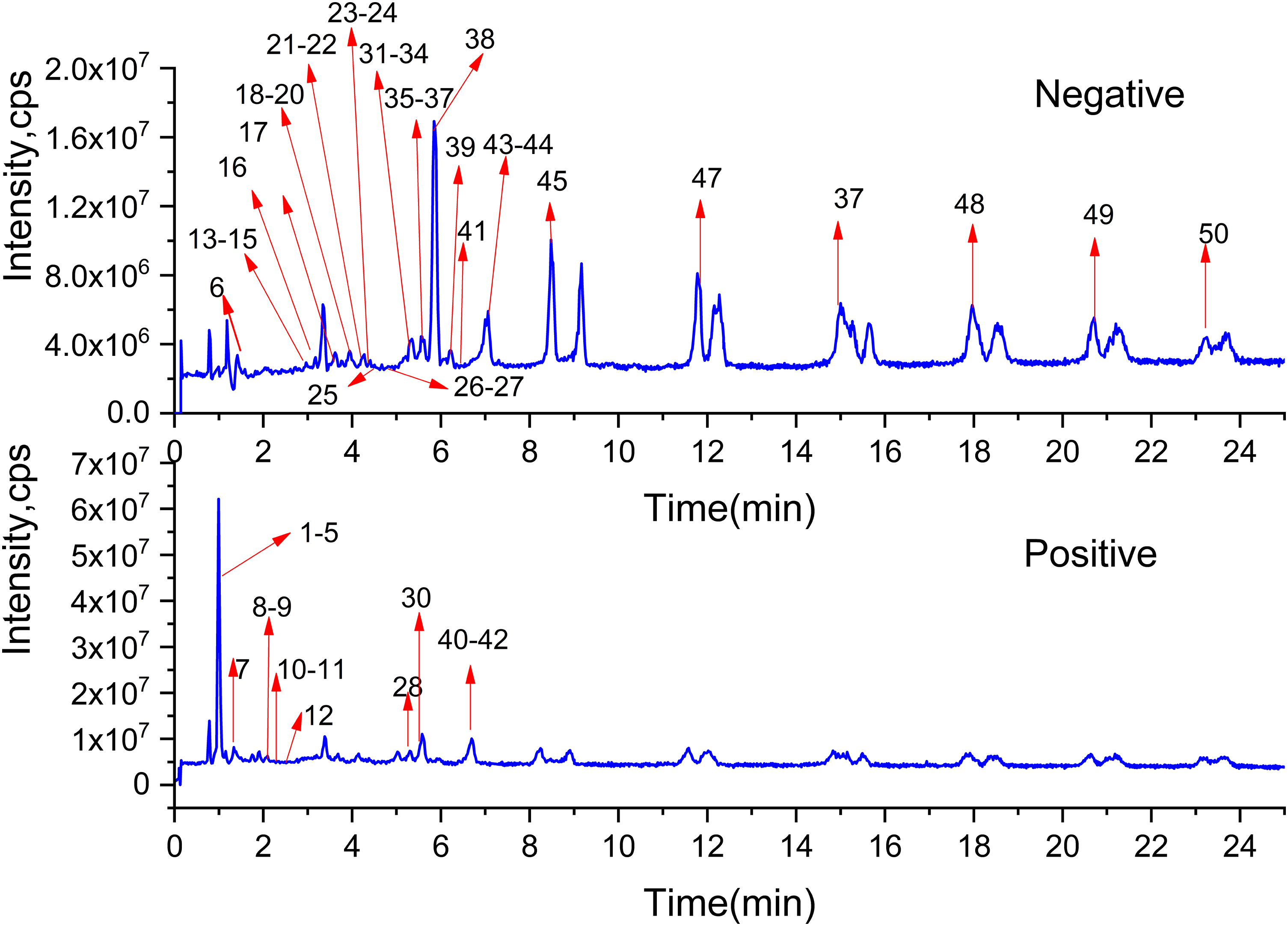
Total ion current chromatogram.
UHPLC-Q-TOF/MS identification of 50 chemical constituents in

Determination of Total Anti-Inflammatory Activity of AR
The COX-2 inhibition rate of different concentrations of AR extract under “Determination of total anti-inflammatory activity of water extract” section is shown in Table 4, and the IC50 value was calculated by GraphPad Prism at 11.77 mg/mL (Figure 3). The results showed that water extract of AR had strong inhibition of COX-2 enzyme activity, and played an anti-inflammatory activity by inhibiting the COX-2 enzyme to reduce the production of prostaglandin G2 and prostaglandin H2 catalyzed by arachidonic acid.

COX-2 enzyme inhibition rate of AR total extract(n = 3).
Different Concentrations of AR Water Extract COX-2 Enzyme Inhibition Rate (n = 3).

Anti-inflammatory Activity Analysis of HPLC-CAD Fractions
The results of anti-inflammatory activity of 60 HPLC-CAD fractions under “Determination of anti-inflammatory activity of HPLC-CAD fraction in vitro” section are shown in Table 5, Figure 4, 29, 34, 42, and 43 fractions had strong inhibition of COX-2 enzyme activity, as shown in Figure 5.

HPLC-CAD liquid chromatogram.

HPLC-CAD fraction anti-inflammatory activity chromatogram (n = 3) (the upper layer is liquid chromatography, the lower layer is the COX-2 enzyme inhibition rate histogram).
HPLC-CAD Fraction COX-2 Enzyme Inhibition Rate. (Mean ± SD,n = 3).

Integration of AR Active Ingredients
In this study, an HPLC-CAD/FC system was used to establish an anti-inflammatory activity integration strategy to screen potential bioactive components in AR. The fraction samples enriched by HPLC-CAD/FC system were used for the screening of anti-inflammatory active ingredients in vitro. The anti-inflammatory activity test results of the fractions integrated with HPLC-CAD/FC showed that the anti-inflammatory activity of peaks 9, 12, 14, and 15 of the constructed CAD chromatographic method was more significant than that of other chromatographic peaks, and the activity of peaks 14 and 15 was greater than 60%. Then, UPLC-Q-TOF/MS was used to analyze the data to determine the chemical structure information of the active compounds.
After comparing with the literature, the results were: The peak 9: [M-H]−m/z 1035.5380 (C50H84O22), fragment ions: 903.4960 [M-H-Xyl]−, 757.4830 [M-H-Xyl-Rha]−, was identified as (25S)-26-O-β-D-glucopyranosyl-5β-furanostan-3β, 22α,26-triol-3-O-[α-L-rhamnopyranosyl-(1→4)]-[β-D-xylopyranosyl-(1→2)]-β-D-glucopyranoside (Figure 6A). Peak 12, quasi-molecular ion [M-H]− m/z 1047.5234 (C51H84O22), fragment ion: 901.5099 [M-H-Rha]−, 755.4472 [M-H-2Rha]−, was identified as protodioscin(Figure 6B). Peak 14:quasi-molecular ion [M-H]− m/z 1065.5493 (C51H86O23), fragment ions: 903.4978 [M-H-Glc]−, 757.4387 [-M-H-Glc-Rha-]−, 595.3868 [-M-H-2Glc-Rha-]−, identified as (25R)-26-O-β-D-glucopyranosyl-5β-furostan-3β, 22α. 26-triol-3-O-[α-L-rhamnopyranosyl-(1→4)]-[β-D-glucopyranosyl-(1→2)]-β-D-glucopyranoside(Figure 6C). Chromatographic peak 15, quasi-molecular ion [M-H]− m/z 919.4895 (C45H76O19), fragment ion: 757.4372 [M-H-Glc]−, 595.3853 [M-H-2Glc]−, 433.321 [M-H-3Glc]−, was identified as (25S)-26-O-β-D-glucopyranosyl-5β-furostan-3β,22α,26-triol-3-O-[β-D-glucopyranosyl-(1→2)]-β-D-glucopyranoside(Figure 6D). 34

Secondary fragment ion peak and structural formula (A, B, C, and D correspond to Peak 9, Peak 12, Peak 14, and Peak 15, respectively).
In summary, the four components of 3-O-
Conclusion
This experiment is based on UPLC with high resolution, narrow chromatographic peaks, a good separation effect, and relatively fast analysis speed, while quadrupole-time-of-flight tandem mass spectrometry (Q/TOF-MS) has strong selectivity and high sensitivity, which can accurately detect the relative molecular mass. The secondary mass spectrometry can prompt the information of characteristic ion fragments. The chemical components of Asparagi radix is identified by UPLC-Q-TOF/MS. According to the properties of Asparagi radix compounds, an HPLC method is established to separate the water extract of AR, and the CAD detector is connected. It has the characteristics of high sensitivity, and good repeatability, and its response value does not depend on the optical properties and ionization efficiency of the measured substance. It can detect all non-volatile and many semi-volatile substances. It can replace the UV detector to detect non-UV or weak UV active compounds as well. It is used to detect steroidal saponins, amino acids and sugars with weak or no UV absorption in AR. The water extract of Asparagi radix is flowed out of the tandem part collector FC by HPLC, and the fractions is collected at an interval of 1 min. A single group is used for in vitro anti-inflammatory activity determination and mass spectrometry identification. After integrating the activity screening results, the qualitative analysis data of mass spectrometry is combined with the activity determination results to obtain the anti-inflammatory active components in Asparagi radix.
In this experiment, UPLC-Q-TOF/MS is used to qualitatively study the main chemical components with molecular weight in the range of 100-1500 in the water extract of Asparagi radix. A total of 50 compounds is identified, including 21 steroidal saponins, 16 amino acids, 8 small molecule sugars, and 5 other types of small molecule compounds. The experimental results show that the compounds in Asparagi radix mainly include saponins, amino acids, and sugars, which are highly consistent with the research direction of Asparagi radix. The established mass spectrometry method can quickly identify the main chemical components in the water extract of AR and preliminarily analyze the main chemical composition information of AR, which lays a material foundation for the screening and quality evaluation of its active substances.
Currently, COX-2 is an important target in the treatment of chronic inflammation, and many drugs targeting this target have been marketed. However, there are serious side effects in these drugs, such as strong cardiovascular side effects leading to the delisting of rofecoxib and etoricoxib. 59 Therefore, it is very meaningful to study new COX-2 inhibitors. In this experiment, when screening the anti-inflammatory active components of Asparagi radix, the cyclooxygenase-2 inhibitor screening kit is used to determine the anti-inflammatory activity of the fractions at different time periods in vitro. Four steroidal saponins is obtained with potential anti-inflammatory activity. It is concluded that the saponins of Asparagi radix can significantly reduce the expression of COX-2, which is of great significance for the development of new COX-2 selective inhibitors. However, this experiment only tested its anti-inflammatory activity in vitro, and its anti-inflammatory activity in vivo and its mechanism of action is not studied in depth. As mentioned above, Asparagi radix plants have broad research prospects, and the study of their anti-inflammatory activity will provide new ideas and directions for the treatment of inflammation-related diseases.
Footnotes
Acknowledgments
The authors thank Shenzhen Institute for Drug Control (Shenzhen, China) for providing technical support for the research.
Authors’ Contribution
YL and BW contributed to design, data curation, investigation, and writing—original draft. WL, CH, QQ, HS, and WW contributed to investigation and validation. GZ, XS, and ZH contributed to formal analysis and conceptualization. GF and XY contributed to resources, supervision, funding acquisition, and writing—review and editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guizhou Province Science and Technology Foundation Project, the National training program for innovative backbone talents for TCM, the Guiyang College of TCM Doctor Startup Fund Project, the “Thousand” level Innovative Talents Project in Guizhou Province (grant no. Qkhzc [2021]417, grant no. Zjjh [2019] 128, grant no. Guizhongyi Doctor Fund [2017] 1, grant no. Qrlf [2020] 4).
Informed Consent
The authors have read the final manuscript and approved of the submission.
