Abstract
Cadmium (Cd), a highly toxic heavy metal, accumulates in the liver and kidneys in the human body by several routes. Tomatoes and related processed food possess multiple physiological functions such as antioxidation, antiinflammation, and anticancer. Herein, to investigate whether tomato extract (TE) can protect against cadmium chloride (CdCl2)-induced oxidative damage in the liver and kidneys, the oxidative damage models were established in vitro and in vivo, and the control group and TE intervention groups were also set up. A significant elevation was observed in Cd concentration and the level of alanine transaminase (ALT), aspartate aminotransferase (AST), blood urea nitrogen (BUN), creatinine (CR), and uric acid (UA) in the serum of CdCl2-treated rats compared with the control group, by contrast, these values were reduced following TE intervention. Moreover, it was found that exposure to CdCl2 decreased the superoxide dismutase (SOD) activity, glutathione (GSH) level, and catalase (CAT) activity and raised the malondialdehyde (MDA) level in serum, liver tissues, kidney tissues, and HepG2 cells, which can be reversed by TE to mitigate the oxidative damage. In addition, TE exerted an inhibitory effect on cell apoptosis in both liver and kidneys brought on by CdCl2 intoxication, as manifested by the downregulation of Caspase-3 protein and the up-regulation of B-cell lymphoma gene 2 (Bcl-2) protein after TE treatment. Altogether, our study reinforces previous findings that CdCl2 exposure disrupted the redox homeostasis, leading to oxidative damage in both liver and kidneys. More importantly, our findings suggest that the hepatorenal protective effect of TE against CdCl2 toxicity is implicated in the inhibition of oxidative stress and apoptosis.
Introduction
Cadmium (Cd) is a potent toxic heavy metal that is taken as one sort of ubiquitous environmental contaminant. 1 Occupational exposure to Cd and its compounds occurs mainly during mining, smelting, processing, and battery manufacturing, and there are a variety of means of environmental and nonoccupational exposures to the highly toxic metal, including contaminated water, polluted dust, and dietary and tobacco smoke sources. 2 Exposure to Cd results in its accumulation in several organs, contributing to nephrotoxicity, hepatotoxicity, genotoxicity, and apoptotic effects, which depend on the dose, route, and duration of exposure. 3 Additionally, it has been shown that Cd exposure can give rise to several malignancies including lung, prostate, kidney, and breast in humans. 4 Consequently, the widespread exposure to Cd in the environment poses a marked threat to human health as a public health concern, it is therefore of high importance to reduce the risk of Cd exposure in the environment and search for possible interventions.
With a biological half-life of approximately 20 years, Cd is a toxicant that is nonessential, accumulates over time, and is quite persistent. 5 About one-third of the body burden lies in the kidneys and about one-half in the liver and kidneys together. 5 The imbalanced antioxidant status is the most common and prominent toxic mechanism of Cd. 6 As one of the most severely compromised organs following Cd exposure, the liver is highly sensitive to acute and chronic Cd toxicity.5,7 Cd promoted the generation of reactive oxygen species (ROS), and as a result, subsequent oxidative damage can result in the antioxidant defense system being depleted, lipid peroxidation being accelerated, and protein and DNA being damaged followed by cell apoptosis and cell death in the liver. 8 The kidney is generally identified as the most critical organ influenced by chronic Cd exposure. 9 Long-term exposure to Cd by inhalation or ingestion may lead to kidney damage, from minor tubular dysfunctions to substantial impairment involving both tubuli and glomeruli. 10 Moreover, chronic kidney disease can be induced by Cd exposure, and the mechanism underlying Cd toxicity is cogently related to oxidative stress. 11 To lessen Cd-induced hepatotoxicity and nephrotoxicity, inhibition of oxidative damage may thus be crucial.
Tomatoes are common components of meals in daily life, and lycopene, one of the naturally occurring red carotenoid pigments in tomatoes, has been reported to possess exceptional antioxidant and antiinflammatory properties.12,13 In addition, lycopene exhibits a potent ability in quenching the singlet oxygen, which reduces the risk of numerous chronic diseases, such as cardiovascular diseases, neurodegenerative disorders, and malignant tumors. 12 Lycopene has been widely studied for more than 80 years along with various articles published in peer-reviewed journals. 14
A recent study, however, suggests that the intervention study of lycopene is limited since the beneficial findings cannot be merely attributed to lycopene which may have been affected by other nutrients. 15 Furthermore, the high cost of extraction of lycopene is arguably accountable for the limitation of wide application. In recent years, the study on the overall preventive function of natural food has become increasingly popular. In the present study, tomato extract (TE) was made by a simple method, and TE greatly retained the nutrients in tomatoes, which has more practical application value compared with the single compound used in previous studies.
Cd poisoning poses a severe health concern, yet there is still a lack of effective methods to protect against Cd-induced damage. Moreover, less research has focused on the protective effect of TE against Cd-induced oxidative damage in the liver and kidneys, and it is of practical significance to search for the interventions from the perspective of dietary intervention. Hence, a series of in vitro and in vivo experiments were carried out to investigate the role of TE exerted on Cd-induced oxidative damage and the possible mechanism, offering a basis for further exploration of interventions on Cd poisoning.
Materials and Methods
The Preparation of Tomato Extract
The 1 kg fresh tomatoes were washed, followed by being crushed with a crusher, and samples were then dried at 98 °C to achieve a constant weight. Next, the dried sample (0.1 g) was thoroughly mixed with 100 mL of sunflower seed oil. Subsequently, extraction was conducted at 37 °C in a constant-temperature water bath. The stock solution of TE was made by collecting the filtrate after vigorous squeezing with two layers of gauze and stored at −20 °C for analysis. The density of TE was measured to be 0.931 g/mL and the density of sunflower seed oil was 0.916 g/mL. That is to say, the stock solution of TE contained approximately 15 mg/mL of active ingredients. Additionally, samples were determined and identified by high-performance liquid chromatography (HPLC, Shimadzu LC-15C, Japan) and lycopene was detected at 475 nm.
Animals and Experimental Design
Thirty male Sprague-Dawley rats, weighing 180-220 g, of specific pathogen-free grade, were purchased from North Sichuan Medical College Laboratory Animal Center. The animals were kept in a temperature-controlled (18 °C-25 °C) and humidity-controlled (60%-70%) barrier system. Rats were randomly assigned to five groups according to body weight (six rats per group), namely, the control group, the model group, the TE low-dose group, the TE medium-dose group, and the TE high-dose group. Throughout the experiment, the rats in TE low, medium, and high-dose groups were, respectively, treated with TE at 37.5 mg/kg, 75 mg/kg, and 150 mg/kg by intragastric administration once a day for 37 consecutive days. Meantime, for rats in the control group and model groups, intragastric administration of 10 mL/kg solvent control was carried out. Besides, on day 7, animals in the model, TE low-dose, TE medium-dose, and TE high-dose groups received an intraperitoneal injection of 2 mg/kg CdCl216,17 once daily for the following 7 days, and control rats were intraperitoneally injected with 1 mL/kg normal saline once per day for a week (Figure 1A). At the end of the experiment, rats were sacrificed to harvest blood samples and organs, including the liver and kidneys. This experiment conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health and acquired approval from the North Sichuan Medical College Ethics Committee (Appl. No. 04/2019).

Animal experiment design and preparation of tomato extract. (A) Animal experiment design in this research. (B) Body weight changes in each group during the study period. (C) Identification of active component lycopene in the TE. (D) The serum cadmium concentration. Values are presented as mean ± standard deviation (n = 3). **P < .01, compared with the control group; #P < .05, compared with the model group.
Determination of Cd Concentration
To collect the serum, the non-anticoagulated blood was allowed to stand for 30 minutes at room temperature, followed by centrifugation at 1000 rpm for 10 min at 4 °C. Subsequently, the supernatant fluid, namely serum, was separated and stored at −80 °C for further analysis. The serum Cd concentration was measured using a PinAAcle 900T Atomic Absorption Spectrometer (PerkinElmer).
Measurement of Activities of ALT, AST, BUN, CR, and UA
To delineate the effect of CdCl2 exposure and TE intervention on liver function in rats, we collected the serum to examine the enzymatic activities of alanine transaminase (ALT) and aspartate aminotransferase (AST) using an ALT assay kit (Nanjing Jiancheng Bioengineering Institute) and AST assay kit (Nanjing Jiancheng Bioengineering Institute) according to the manufacturer's protocol.
Next, we used a fully automatic biochemical analyzer (Hitachi) to measure BUN, CR, and UA in rat serum.
Cell Culture
The human hepatocellular carcinoma HepG2 cells, purchased from MINGJING BIOLOGY (Shanghai), were used in this experiment. The HepG2 cells were cultured in RBMI-1640 medium (Gibico) supplemented with 10% fetal bovine serum (Sijiqing) and 1% (v/v) penicillin/streptomycin (Beyotime). Cells were maintained in a humidified incubator at 37 °C under 5% CO2 atmosphere. When cells occupied 80%-90% of the bottom of the dish, they were digested with 0.25% trypsin (Beyotime) and subcultured.
Cell Viability Assay
To verify the toxic effect of cadmium chloride (CdCl2) (analytically pure, Chengdu Kelong Chemical Co., Ltd) on HepG2 cells, the cell viability of HepG2 cells was assessed by CCK-8 assay (Beyotime) after the administration of CdCl2 alone at different concentrations (0, 1, 2, 4, 8, 16, 32, 64 μmol/L). Following this, the TE stock solution was diluted to an intervention dose of 7.5 μg/mL, 15 μg/mL, and 30 μg/mL, respectively. The cell grouping was as follows: the blank control group, the DMSO control group, the TE control group, the CdCl2 control group, the TE low-dose group (7.5 μg/mL TE + 16 μmol/L CdCl2), the TE medium-dose group (15 μg/mL TE + 16 μmol/L CdCl2), and the TE high-dose group (30 μg/mL TE + 16 μmol/L CdCl2). The cell viability of cells was detected again by CCK-8 assay. The absorbance values were measured at 450 nm using a microplate reader (SpectraMax M2, Molecular Devices).
Assessment of Indicators of Oxidative Stress
By assaying antioxidant defense systems: superoxide dismutase (SOD), glutathione (GSH), catalase (CAT), and malondialdehyde (MDA), we explored the oxidative stress status of serum, liver tissues, and kidney tissues in rats and HepG2 cells after CdCl2 poisoning and TE treatment. An appropriate amount of precooled sterile saline was added to liver and kidney tissues, respectively, according to the ratio of weight (g): volume (mL) = 1:9, followed by homogenization on ice using a glass homogenizer. In the next step, centrifugation at 2500 rpm for 10 min at 4 °C was performed to obtain the supernatant, stored at −80 °C. The activity of SOD and the levels of GSH and MDA were measured using the SOD assay kit (A001-3, Nanjing Jiancheng Bioengineering Institute), GSH assay kit (A006-2, Nanjing Jiancheng Bioengineering Institute), CAT assay kit (A007-1-1, Nanjing Jiancheng Bioengineering Institute), and MDA assay kit (A003-2, Nanjing Jiancheng Bioengineering Institute).
Histopathological Observation
Hematoxylin and eosin (H&E) staining was applied to observe histopathological changes in liver and kidney tissues after the administration of CdCl2 and TE intervention. The liver and kidney tissues were collected and fixed in 4% paraformaldehyde and then processed routinely with graded alcohol and xylene, followed by paraffin embedding and sectioning. Next, the slides were subjected to H&E staining and examined under a light microscope for histopathological analysis. Image acquisition was conducted using a Pannoramic 250 digital scanner (3DHISTECH).
Immunohistochemical Study
The slides were deparaffinized and dehydrated with xylene and a graded series of ethanol solutions, respectively. Antigen retrieval was carried out in citrate buffer solution (pH 6.0) using a microwave oven for 20 min. After being washed with PBS, the sections were incubated with 3% hydrogen peroxide solution for 10 min at ambient temperature to quench endogenous peroxidase activity. Subsequently, the tissues were treated at room temperature for 20 min with goat serum as a blocking buffer. In the next step, these slides were probed with primary antibodies against B-cell lymphoma gene 2 (Bcl-2) (M00040-3; Boster) and Caspase-3 (M00334-5; Boster) at 4 °C overnight followed by secondary antibody at 37 °C for 30 min. Next, the sections underwent color development with DAB substrate and hematoxylin counterstaining. Finally, all sections were imaged by microscope (Olympus) and analyzed with Image-Pro Plus 6.0 software (Media Cybernetics).
Statistical Analysis
The results were expressed as mean ± standard deviation. Data were analyzed using one-way analysis of variance (ANOVA) by SPSS 22.0 (IBM). P < .05 was considered statistically significant.
Results
Animal Experiment Design and Preparation of TE
The rats in the control group had a good mental state, a good appetite, many activities, shiny hair, and increasing body weight. The rats in the model group exhibited a poor mental state, decreased food intake and activity, dried and messy fur, and a slow weight gain. Moreover, the general condition of the TE group rats was better than that of the model group rats. From day 13, the weight of rats following CdCl2 administration alone was significantly decreased when compared to the control group, by contrast, the body weight of the TE groups had higher body weight in comparison with the model group (Figure 1B).
Lycopene is an important antioxidant compound abundantly present in tomatoes. 12 In this research, HPLC analysis was carried out and the major active component lycopene in TE was identified by comparison with an authentic standard. From Figure 1C, we can see the representative chromatograms for the lycopene control and TE samples at 475 nm. The chromatogram does suggest that the tomato extract used in this research contains lycopene.
As displayed in Figure 1D, as opposed to the control group, the model group presented a considerable rise in serum Cd concentration of rats (P < .01), whereas the TE groups exhibited a reduction in serum Cd concentration, which was particularly noticeable in the TE high-dose group (P < .05).
TE Mitigated CdCl2-Induced Liver Injury and Oxidative Stress in Serum
The activities of ALT and AST are reliable parameters for the evaluation of liver function. 18 A significant increase in the serum activities of both ALT and AST was observed in the CdCl2 group in contrast to the control group (P < .01), as can be shown in Table 1. Moreover, it has been noted that the serum ALT and AST levels decreased after TE intervention in comparison with the model group, notably in the TE high-dose group (P < .01). As for the indicators of renal injury, we selected the measurement of serum BUN, CR, and UA and found that the three indicators were significantly higher in the CdCl2 group than in the control group, and these indicators were decreased after giving different doses of TE intervention (Table 1).
Results of Serum ALT, AST, BUN, CR, and UA Levels in Rats (n = 6,
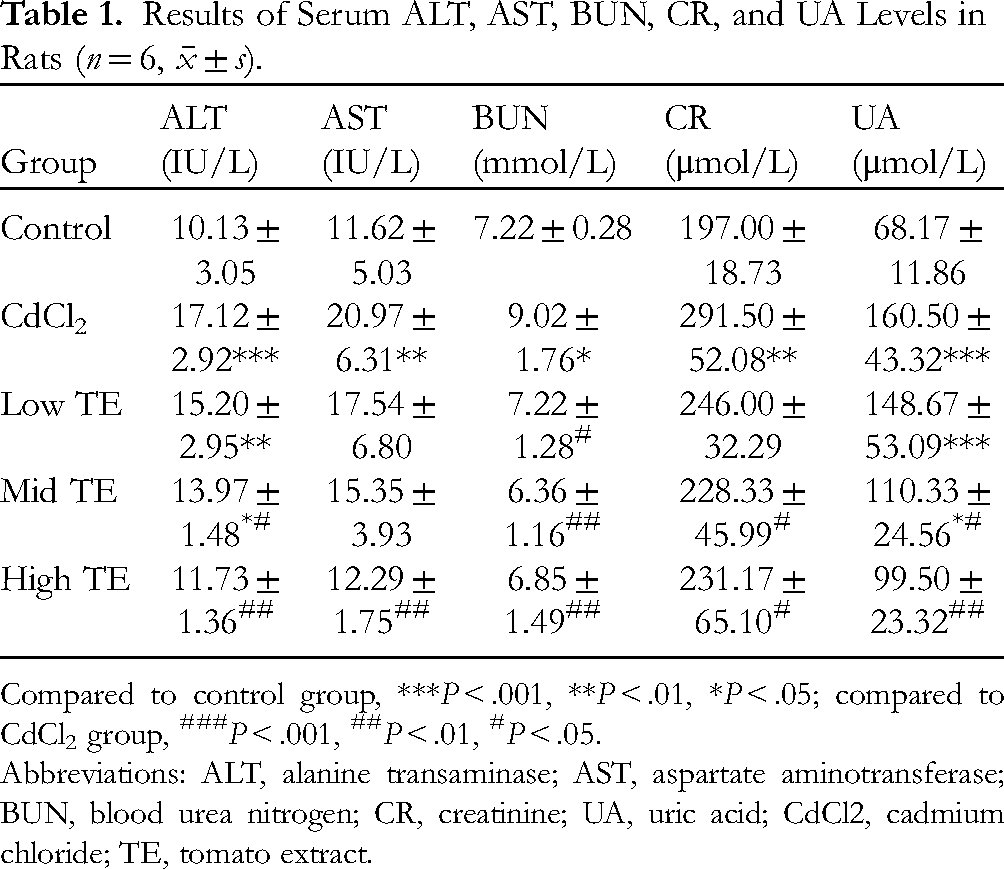
Compared to control group, ***P < .001, **P < .01, *P < .05; compared to CdCl2 group, ###P < .001, ##P < .01, #P < .05.
Abbreviations: ALT, alanine transaminase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; CR, creatinine; UA, uric acid; CdCl2, cadmium chloride; TE, tomato extract.
In terms of oxidative stress, SOD functions as an essential antioxidant enzyme that may scavenge superoxide anion radicals, and MDA is a stable end product of lipid peroxidation.19,20 GSH is the main intracellular thiol compound involved in the defense against oxidative damage.19,20 The SOD activity, as well as the levels of MDA and GSH, can therefore be used to determine the status of oxidative stress. In the present study, CdCl2 exposure significantly reduced the activity of SOD and the level of GSH and markedly enhanced the level of MDA in serum (P < .01), whereas treatment with TE elevated the SOD activity and GSH level and decreased the MDA level, which was particularly obvious in the TE high-dose group (P < .05, P < .01) (Figure 2).

Effects of tomato extract on serum oxidative stress in the CdCl2-exposed rats. The activity of SOD and the levels of GSH and MDA in serum after CdCl2 exposure and TE intervention. Values are presented as mean ± standard deviation (n = 3). *P < .05, **P < .01, compared with the control group; #P < .05, ##P < .01, compared with the model group.
TE Attenuated CdCl2-Induced Oxidative Stress in Hepatic and Renal Tissues of Rats
As seen in Figure 3, the hepatic and renal tissues in the model group had lower levels of GSH and lower levels of CAT and SOD activity than the control group did, but greater levels of MDA (P < .05, P < .001). Both SOD activity and GSH and CAT levels raised according to increasing TE dose, contrary to MDA level, which declined in hepatic and renal tissues as the TE dose increased, especially in the TE high-dose group (P < .01, P < .001).

Effects of tomato extract on oxidative stress in hepatic and renal tissues in the CdCl2-exposed rats. (A-D) The activity of SOD and the levels of GSH, CAT, and MDA in hepatic tissues after CdCl2 exposure and TE intervention. (E-H) The activity of SOD and the levels of GSH, CAT, and MDA in renal tissues after CdCl2 exposure and TE intervention. Values are presented as mean ± standard deviation (n = 3). *P < .05, ***P < .001, compared with the control group; #P < .05, ##P < .01, ###P < .001, compared with the model group.
Histopathological and Immunohistochemical Examinations
H&E staining of liver tissues after CdCl2 administration showed cloudy swelling of hepatocytes, granular and vacuolar degeneration of hepatocytes, brown–yellow granules in the cytoplasm, punctate necrosis of hepatocytes, hepatic central vein dilation, hepatic sinusoid dilation, and inflammatory cell infiltration, as represented in Figure 4. Liver sections from rats treated with TE showed that the overall liver lesion was milder in the TE groups than in the model group. In addition, the kidneys from CdCl2-exposed rats exhibited histopathological changes: renal tubular epithelial cell swelling, vacuolar degeneration of renal tubular epithelium, structureless filaments or floc in the lumen, dilatation and congestion of interstitial blood vessels and glomerular capillary tuft. Overall, the degree of lesion was lighter in the TE intervention groups than in the model group.

Histopathological and immunohistochemical examinations in hepatic and renal tissues. Histopathological changes in hepatic and renal tissues after CdCl2 exposure and TE intervention (magnification 200× and scale bar 100 μm). Immunohistochemical staining of Bcl-2 and Caspase-3 protein among five groups. The positive staining of Bcl-2 and Caspase-3 was indicated by a brown color (magnification 200× and scale bar 100 μm).
The antiapoptotic Bcl-2 protein and pro-apoptotic Caspase-3 protein played a pivotal role in the regulation of apoptosis.21,22 As displayed in Figure 4, immunostaining for Bcl-2 was significantly decreased in response to CdCl2 administration when compared to control animals in hepatic and renal tissues. However, the immunopositive areas in the TE medium and high-dose groups were apparently higher than those of the model group. Additionally, a rise in Caspase-3 expression was also seen in the liver and kidneys after individual administration of CdCl2. The protective effects of TE were revealed in rats treated with TE where the expression of Caspase-3 was reduced in the liver and kidneys.
CdCl2 Exposure Induced Oxidative Stress in HepG2 Cells
From Figure 5A, we can see that the cell viability of HepG2 cells changed in varying degrees with the prolongation of CdCl2 exposure time and increase of exposure concentration. The cell viability of HepG2 cells exhibited a downward trend after being exposed to more than 8 μmol/L of CdCl2, in which the lowest viability was observed under the condition of 64 μmol/L CdCl2 for 24 h. The activity of SOD and the level of GSH were downregulated with the increasing concentration of CdCl2 exposure, and MDA production was augmented in a dose-dependent manner, with the high-dose CdCl2 group (32 μmol/L) being the most pronounced (P < .01) (Figure 5B-G).
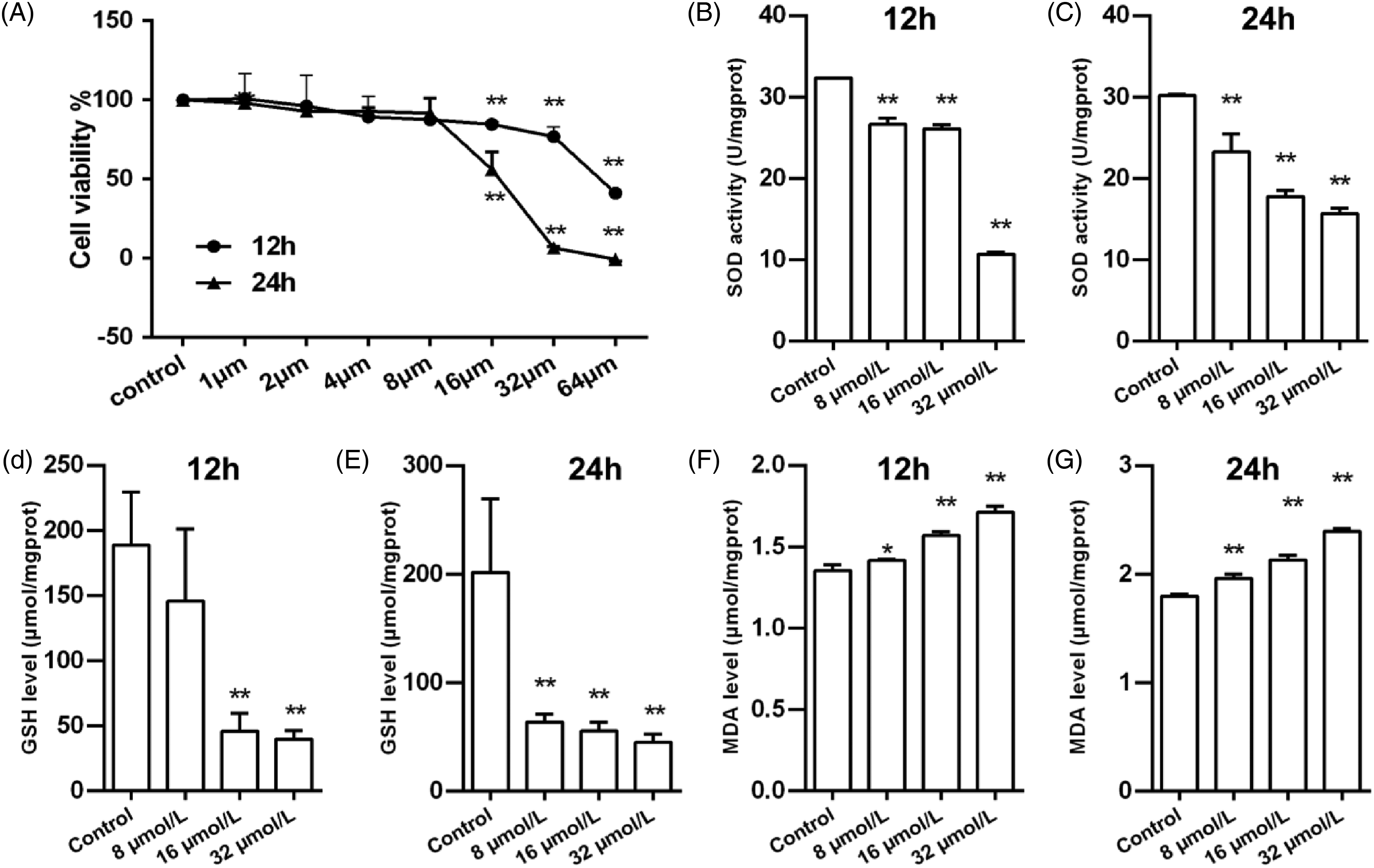
Effects of CdCl2 poisoning at different concentrations on cell viability and oxidative stress in HepG2 cells. (A) The cell viability of HepG2 cells after CdCl2 poisoning at different concentrations. (B, D, and F) The activity of SOD and the levels of GSH and MDA in HepG2 cells after CdCl2 poisoning at different concentrations for 12 h. (C, E, and G) The activity of SOD and the levels of GSH and MDA in HepG2 cells after CdCl2 poisoning at different concentrations for 24 h. Values are presented as mean ± standard deviation (n = 3). *P < .05, **P < .01, compared with the control group.
TE Antagonized CdCl2-Induced Oxidative Stress in HepG2 Cells
HepG2 cells exposed to CdCl2 had a severe decline in viability (P < .001) that was alleviated by TE treatment (Figure 6A). After CdCl2 exposure and TE intervention in HepG2 cells for 12 h, decreased SOD ability and GSH and CAT levels, and raised MDA production occurred with CdCl2 toxicity (P < .05, P < .01), by contrast, the TE groups have a higher level of SOD, GSH and CAT, and a lower level of MDA when compared to the CdCl2 control group (Figure 6B, D, F and H). After CdCl2 exposure and TE intervention in HepG2 cells for 24 h, a substantial reduction in the activity of SOD and the level of GSH and CAT was observed in the model group, whereas CdCl2 poisoning evidently increased the level of MDA (P < .05, P < .001) (Figure 6C, E, G and I). Moreover, the high-dose TE caused a significant rise in SOD activity (P < .001), and the GSH and CAT levels were found to be elevated in the TE groups, while TE dose-dependently made the MDA level downregulated (Figure 6C, E, G and I).

Effects of tomato extract on cell viability and oxidative stress in CdCl2-exposed HepG2 cells. (A) The cell viability of HepG2 cells after CdCl2 exposure and TE intervention. (B, D, F, and H) The activity of SOD and the levels of GSH, CAT, and MDA in HepG2 cells after CdCl2 exposure and TE intervention for 12 h. (C, E, G, and I) The activity of SOD and the levels of GSH, CAT, and MDA in HepG2 cells after CdCl2 exposure and TE intervention for 24 h. Values are presented as mean ± standard deviation (n = 3). *P < .05, **P < .01, ***P < .001, compared with the blank control group; #P < .05, ##P < .01, ###P < .001, compared with the CdCl2 control group.
Discussion
Cadmium is recognized as a dangerous environmental and occupational pollutant that, even at low exposure levels, represents a potential hazard to human and animal health. 23 The heavy metal Cd was categorized as a human carcinogen (group Ⅰ) by the International Agency for Research on Cancer (IARC, 1993) based on sufficient evidence of carcinogenicity in both human beings and experimental animals. 24 Many studies have found that some plant or fruit extracts have the ability to counteract the harmful effects of Cd.25–27 Tomatoes and tomato products have high nutritional value with antioxidant capacity and antiinflammatory and anticancerous activities.28,29 In this study, changes in experimental animals’ body weight can be taken as an important index to evaluate their health condition. 30 We found that TE treatment attenuated the reduction in body weight of CdCl2-exposed rats, especially in the TE medium and high-dose groups. Similar findings have been described in Cd-related studies, wherein the body weight of rats dramatically dropped after exposure to Cd for 2-4 weeks, while weight loss in mice brought on by Cd administration was efficiently reversed by betulinic acid at different doses.25,31
The dose of tomato extract we used in this study was very close to the daily dietary intake of tomatoes. We assessed the lycopene content of tomato extracts using lycopene as an indication, keeping the safety of the extracts in account. Even in the high-dose group of tomato extract, our results showed no harm to animals or cells.

Total experimental design. The tomato extract samples were determined and identified by high-performance liquid chromatography and lycopene was detected at 475 nm. By assaying antioxidant defense systems: superoxide dismutase (SOD), glutathione (GSH), catalase (CAT), and malondialdehyde (MDA), we explored the oxidative stress status of serum, liver tissues, and kidney tissues in rats and HepG2 cells after CdCl2 poisoning and TE intervention. Moreover, the levels of ALT, AST, BUN, CR, and UA were measured to assess the liver function of rats, and the immunohistochemical staining of B-cell lymphoma gene 2 (Bcl-2) and Caspase-3 protein was analyzed to evaluate the apoptosis status in the liver and kidneys.
Oxidative stress has been substantiated to be one of the primary mechanisms behind Cd toxicity. 20 Cadmium causes oxidative stress and inflammation by increasing reactive oxygen species (ROS), which can cause liver and kidney damage. Cadmium also activates the extracellular signal-regulated kinase (ERK) and p38 mitogen-activated protein kinase (MAPK) pathways, resulting in mitochondria-mediated intrinsic apoptosis. 32 Cadmium also decreases the body's antioxidant capacity, interferes with mitochondrial function, inhibits enzymes, and alters hepatocyte metabolism, according to research.
On the other hand, cadmium has the ability to reduce the activities of superoxide dismutase (SOD), glutathione peroxidase (GPx), and catalase (CAT), thus enhancing their damaging effects. Oxidative stress plays a key role in the destructive effects of cadmium. Cadmium increases the production of superoxide anion, leading to the conversion of ferrous ions to ferrous ions and the generation of hydroxyl radicals via the Fenton reaction, which exacerbates oxidative damage, especially lipid damage. 33 Studies have shown that under pathological conditions, nitric oxide (NO) reacts with superoxide anion to generate peroxynitrite, which can lead to lipid peroxidation and inflammatory responses that directly trigger renal dysfunction.
Cadmium increases inflammatory cytokines in different tissues, leading to various tissue damage and oxidative reactions. Among them, TNF-α is associated with downregulation of antioxidant enzymes and free radical production. 34 There is evidence that cadmium-induced oxidative stress is mediated through activation of the nuclear factor erythrocyte 2-associated factor 2 (Nrf2) signaling pathway. In addition, cadmium binds to cysteine groups in the structure of glutathione, thereby weakening the intracellular antioxidant system.
Oxidative stress is distinguished by the generation of free radicals which induces lipid peroxidation and cellular membrane rupture, and oxidative status could be inferred by evaluating the levels of indicators such as SOD, MDA, and GSH.20,35 SOD, a classical antioxidant enzyme, is recognized as the first line of defense against free radical formation and plays a vital role in circumventing oxidative stress. 19 GSH is required for many crucial cellular processes and performs a nonnegligible role in maintaining and regulating the intracellular thiol-redox status. 19 MDA, generated by the peroxidation of polyunsaturated fatty acids, represents a final and stable product of lipid peroxidation and also serves as an index of lipid peroxidation. 19 SOD, GSH, and MDA were therefore employed to assess the extent of Cd-induced oxidative damage. In addition to inducing direct cellular damage, oxidative stress acts as a key mediator in apoptosis and/or necrosis. 9
The primary detoxifying process in plants that is impacted by heavy metals is phytochelatins, which work by forming complexes with the metals to lessen their toxic effects. This system can shield plant cells from heavy metals, and it can lessen the buildup of heavy metals in tissues and organs in mammals by preventing their absorption.36,37 Lycopene, which is derived from tomatoes and tomato-based products, is the primary active component of tomatoes. According to linked studies, lycopene possesses antiinflammatory, antioxidant, antiproliferation, and other properties that help prevent cancer and tumors as well as lower the risk of cardiovascular disease. 38 Lycopene is a sort of particularly important phytochemical present in tomatoes, and antioxidation and antiapoptosis were regarded as the main mechanisms by which lycopene exerted its protective effect.39–42 Tomato extract is a mixture with preventive properties that can enhance cadmium metabolism in the body and lower serum cadmium levels. However, due to the complicated composition of tomato extracts, a representative lycopene was chosen, and its quality and principal components were investigated and verified by HPLC after fat-soluble extraction.
Moreover, it was revealed that the antioxidant role of lycopene may indirectly affect the apoptotic effect in cells. 43 Furthermore, in the mitochondrial apoptotic pathway, it is well known that Bcl-2 displayed an antiapoptotic effect via inhibition of mitochondrial cytochrome c release, while the release of cytochrome c can activate the effector caspases, eventually executing apoptosis.27,28
ALT and AST, used as sensitive indicators for estimating liver function, are typical biomarkers of liver injury. 18 It was demonstrated that hepatic function was impaired after exposure to CdCl2, as evidenced by the raised ALT and AST activities in serum. 44 In the present study, we found that the activities of ALT and AST were increased in the serum of CdCl2-intoxicated rats, which tied in well with other findings reporting the alterations of ALT and AST activities brought on by Cd exposure.44,45 The results described above suggested that the administration of CdCl2 resulted in the destruction of cell membrane structure and an increase in cell membrane permeability which subsequently disrupted the cellular enzymatic system and released enzymes into the circulation. 46 The histopathological alteration is thought of as a direct parameter of how damaged the organ is. 44 H&E staining of liver sections after CdCl2 exposure showed brown–yellow granules in the cytoplasm, punctate necrosis of hepatocytes, hepatic central vein dilation and hepatic sinusoid dilation, inflammatory cell infiltration, and so on.
It is plausible to assume that liver damage can arise from CdCl2 poisoning based on the aforementioned experimental findings. Besides, TE counteracted CdCl2-induced liver injury, as manifested by the decrease of ALT and AST activities and the mitigation of histopathological damage in the TE groups. Moreover, the reduction of SOD activity and GSH level and the elevation of MDA level were found in serum and hepatic tissues of rats after exposure to CdCl2 which were compatible with the previous study. 47 From these results, it might be speculated that increased ALT and AST in serum are implicated in the rupture of cellular membranes resulting from lipid peroxidation. 46 TE presented a significant blunting of oxidative stress in serum and hepatic tissues by increasing SOD activity and GSH level and lowering MDA level, particularly in the TE high-dose group.
A previous study suggested that compared with the control group, the SOD activity and GSH level were obviously reduced, while the MDA level was significantly enhanced in renal tissues of Cd-administered rats, indicating that Cd was capable of inducing severe oxidative stress. 9 Besides, physalis extract restored the activities of antioxidant enzymes including SOD, CAT, and GPx, improved GSH homeostasis and/or its rate of synthesis, and reduced the level of MDA, all of which conferred enhanced protection against Cd-induced oxidative damage in the kidneys. 9 In this experiment, TE recovered the redox homeostasis disrupted by CdCl2 poisoning in renal tissues, presenting as a higher level of SOD activity and GSH level and a lower level of MDA in the TE groups. Similar results were also observed in HepG2 cells. Besides, exposure to CdCl2 distinctly downregulated the expression of Bcl-2 protein and considerably up-regulated Caspase-3 protein expression in the liver and kidneys, which could be reversed by TE intervention. The underlying mechanism through which TE exhibited its protective role may entail the suppression of mitochondrial oxidative stress, thus defending against cell apoptosis caused by CdCl2, which was at least in part, in line with those reported by Yin et al. 35 Nevertheless, as hepatorenal injury occurrence is a complex and dynamic process, it should be pointed out that, in this research, the interactions between oxidative stress and apoptosis in CdCl2-induced kidney injury have yet to be fully elucidated and require further explorations.
There are, of course, other mechanisms in addition to defending against oxidative damage when it comes to the potential mechanisms behind the protective action of TE. It has been reported that TE, mixed with the rat diet, prevents the accumulation of heavy metals in the liver, percentage protection by tomatoes to accumulation was Cd > Hg > Pb in the liver, which the protective mechanism may be related to the phytochelatin contained in tomatoes.48–50 Herein, since tomatoes contain abundant fat-soluble antioxidants, we used oil extraction and heating methods in the preparation of TE, which significantly preserved the fat-soluble substances in tomatoes and is more accordant with the way tomatoes are processed in daily life. Our study may assist in better understanding the mechanism involved with Cd toxicity and the part that TE played in Cd-induced hepatotoxicity and nephrotoxicity (Figure 7).
Our research, however, is still in its early phases. We only examined simple oxidation damage indicators, but the results revealed that TE has a protective effect against cadmium-induced liver and kidney toxicity damage. In this investigation, tomato extract was a combination, and no functional components in tomato extract were identified. The specific mechanism has not been completely investigated and will be tested in the subsequent tests.
Conclusions
In conclusion, TE exhibited antagonistic roles against the reduction in HepG2 cell viability and weight loss of rats brought on by CdCl2 exposure. TE intervention also effectively mitigated lipid peroxidative damage and enhanced antioxidant capabilities, and also lessened the histopathological alteration induced by CdCl2 toxicity in the liver and kidneys. In addition, TE exerted a protective activity in CdCl2-induced injury by suppressing cell apoptosis. The present study deepens our understanding concerning the toxic effect of CdCl2 acting on the liver and kidneys and indicates that TE may be an effective option for the prevention of Cd-induced diseases.
Footnotes
Authors’ Contributions
Z.Z.L. and H.Q.W. obtained funding for the study. Z.Z.L. and L.C. wrote the manuscript. Z.Z.L., L.C., L.X.Z., Q.H.L., C.T.P., Z.Y.Z., Q.F., Y.L., R.J.S., S.M.L., B.D., and H.Q.W. designed the experiments. Z.Z.L. analyzed the experimental data. L.C., L.X.Z., Q.H.L., C.T.P., Z.Y.Z., Q.F., Y.L., R.J.S., S.M.L., and B.D. performed the experiments. All authors contributed to the discussion and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the North Sichuan Medical College Ethics Committee, Sichuan Province, China.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Project of the Education Department of Sichuan Province [grant number 17ZA0177]; Sichuan Science and Technology Program [grant number 2020YJ0382]; Scientific research project of health commission of Sichuan [grant number 19PJ199]; and Government University specific Cooperative Scientific Research Project of Nanchong [grant number 19SXHZ0188].
Statement of Human and Animal Rights
The animal experimental procedure was conducted in accordance with the Institutional Animal Care guidelines of North Sichuan Medical College, Sichuan Province, China and approved by the Administration Committee of Experimental Animals, Sichuan Province, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
