Abstract
The aim of this study was to investigate the antitumor effect of lycorine hydrochloride (LH) and discuss the correlation between LH-induced apoptosis and autophagy in the human colorectal cancer cell line HCT116. Here the results by the Cell Counting Kit-8 and colony formation assays showed that LH concentration-dependently decreased cell viability and colony formation in HCT116 cells, suggesting inhibition of cell proliferation by LH. By flow cytometry, LH was found to increase apoptotic rate in HCT116 cells. Mechanistically, Western blot results revealed that LH increased the expression of the protein of Bax and Caspase-3, and decreased Bcl-2 proteins expression. Moreover, the reverse transcriptase quantitative polymerase chain reaction and Western blot analysis also showed that LH increased the expression of Beclin-1 and LC3B-II/LC3B-I ratio, indicating that autophagy was induced by LH. LH induced autophagy via downregulating phospho-mammalian target of rapamycin and upregulating phospho-AMPK (5′ adenosine monophosphate-activated protein kinase). Furthermore, to understand the role of LH-induced autophagy and its association with apoptosis, cells were analyzed after Beclin-1 small interfering RNA transfection. The results indicated that the proapoptotic ability of LH was increased by inhibition of autophagy. In conclusion, the present investigation suggested that LH induced apoptosis and autophagy in HCT116 cells via the mitochondrial and AMPK/mTOR pathways. The suppression of autophagy promoted LH-induced apoptosis by modulating Beclin-1 and Bcl-2.
Colorectal cancer (CRC) is the third and second leading cancer in men and women respectively, with 1.2 million new cases and 600 000 deaths per year. 1 Existing treatment options for CRC include surgery, radiation therapy, chemotherapy, and immunotherapy. 2 Each treatment option typically involves tradeoffs between patient survival and quality of life. As Chemotherapy drugs display toxic side effects, natural products with anticancer effects are desirable.
Lycorine is the major bioactive component of the
Autophagy is another primary type of cell death, which also has been considered to be associated with cell survival and tumor maintenance. 9 Interestingly, lycorine as a potential anticancer agent has been demonstrated to have opposite effects on autophagy. On one hand, lycorine reduced HMGB1 to inhibit autophagy in multiple myeloma. 10 On the other hand, lycorine has been shown to induce autophagy in hepatocellular carcinoma cells 11 and A549 cells. 12 However, it remains unknown whether lycorine also affects autophagy in human CRC cells.
The interplay between autophagy and apoptosis is complex in the pathological process. 13 Autophagy in apoptosis has a dual role, apoptosis induction and inhibition. 14,15 Few studies explored the correlation between lycorine-induced apoptosis and autophagy. The previous research showed that lower levels of lycorine-induced autophagy promoted apoptosis in A549 cells. 12 But there is lack of adequate evidence to elucidate the interplay between lycorine-induced apoptosis and autophagy in human CRC cells.
Therefore, this study aimed to explore the effect of lycorine hydrochloride (LH) on proliferation, autophagy, and apoptosis in HCT116 cells. Meanwhile, we also analyzed the interaction mechanism between the autophagy and apoptosis induced by LH. The results of our study will provide a novel and effective treatment way for CRC.
Effects on Cell Viability in HCT116 Cells
The effect of LH on HCT116 cell viability was first investigated by the Cell Counting Kit-8 (CCK-8) assay. Figure 1 reveals that the exposure of HCT116 cells to increasing concentrations of LH (0, 2.5, 5, and 10 μmol/L) resulted in a dose-dependent reduction in cell viability. At a concentration of 10 μmol/L, LH exhibited maximal inhibition of approximately 61.3% of HCT116 cell proliferation compared with the control group.

Effects of LH on the viability of HCT116 cells. (a) Chemical structure of LH. (b) HCT116 cells were treated with indicated concentrations of LH (0, 2.5, 5, and 10 µM) for 48 hours. Cell viability was determined by Cell Counting Kit-8 assay. Data represent the mean values ± standard error of the mean (
Effects of LH on Clonogenic Ability
Colony formation assays were performed to investigate the long-term effects of LH on the growth of HCT116 cells. Our results revealed that colony formation abilities of HCT116 cells in the LH groups were significantly lower than those of the corresponding control group (

Effects of LH on colony forming. (a) HCT116 cells were treated with vehicle or LH for 2 weeks. Colony formation was fixed in methanol, stained with crystal violet, and counted. (b) The number of colonies was analyzed. Data are represented as mean ±standard error of the mean (
Effects of LH on Apoptosis
In the former study, LH was proven to be effective against several cancers by inhibiting proliferation and inducing apoptosis of cancer cells. 5 -7 Next, we examined if exposure to LH could activate apoptosis in HCT116 cells. In this experiment, apoptotic cell death was observed to be activated following LH (0, 2.5, 5, and 10 μmol/L) treatments. Ten micromoles per liter LH treatment was observed to lead to 35% propidium iodide (PI)-positive cells (Figure 3).

Effects of LH on apoptosis of HCT116 cells. HCT116 cells were treated with different concentrations of LH for 48 hours. Additionally, cells were transfected with NC siRNA or Beclin-1 siRNA, followed by 10 µM LH treatment. (a) The collected cells were stained with Annexin V-fluorescein isothiocyanate and propidium iodide and analyzed by flow cytometry. (b) The apoptosis rate was calculated. Data represent the mean values ± standard error of the mean (
To investigate the mechanism of LH affecting HCT116 cell apoptosis, major cell apoptosis regulatory proteins, including Bcl-2, Bax, and Caspase-3, were detected by Western blot. HCT116 cells were treated with different concentrations of LH (0, 2.5, 5, and 10 μmol/L). The results showed that the expression of Bax tended to a positive increase, but the expression of Bcl-2 was reduced by LH. Moreover, cleaved caspases-3, the active type of the caspases, were observed to significantly increase expression as the dose of LH increased (Figure 4).
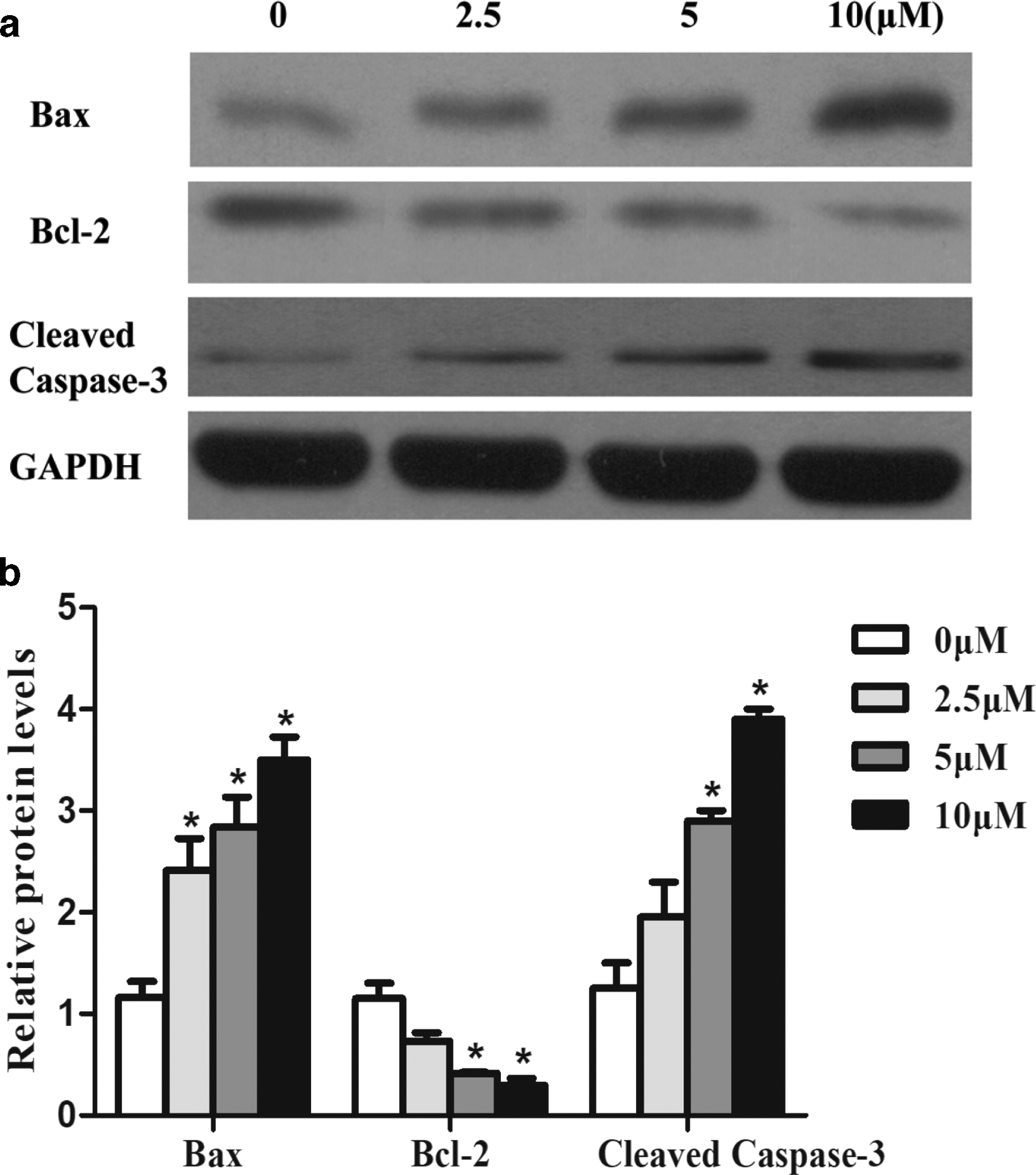
Effects of LH on the mitochondrial apoptotic pathway in HCT116 cells. HCT116 cells were treated with indicated concentrations of LH for 48 hours. (a) Western blotting analysis indicating the expressions of P53, Bax, Bcl-2, and cleaved caspase-3, 9 proteins in HCT116 cells. (b) Analysis of P53, Bax, Bcl-2, and cleaved caspase-3, 9. Data represent the mean values ± standard error of the mean (
Effects of LH on Autophagy
Autophagy is a process for recycling cellular constituents, and it is associated with cell survival and tumor maintenance. 15 We investigated if LH could induce autophagy in HCT116 cells. Autophagy is characterized by the conversion of LC3B-I (cytosolic form) into a lipidized LC3B-II (autophagosome membrane-bound form). 16 Accordingly, we analyzed the expression of LC3B-II/I protein by Western blot and LC3B-II mRNA level by reverse transcriptase quantitative polymerase chain reaction (RT-qPCR). We found that LH could significantly downregulate the ratio of LC3B-II/I protein and reduced LC3B-II mRNA level, which confirmed the role of LH in the induction of autophagy in HCT116 cells. Next, we assessed the expression of Beclin-1, which is an autophagy effector responsible for autophagosome formation. Surprisingly, we observed that the expression of Beclin-1 protein and mRNA increased in a dose-dependent manner following treatment of HCT116 cells with LH (Figure 5).

Effects of LH on autophagy in HCT116 cells. (a) After 48 hours treatment with vehicle or LH, expressions of LC3B and Beclin-1 protein were assessed by Western blot. (b) Analysis of LC3B and Beclin-1 proteins. (c) The expression of LC3B and Beclin-1 mRNA was detected by reverse transcriptase quantitative polymerase chain reaction. Data represent the mean values ± standard error of the mean (
Then, we examined the mechanism through which LH can exert its effects on autophagy. AMPK/mTOR is a classic autophagic pathway. The AMPK system is considered a sensor of cellular energy status and a modulator of metabolism. 17 We observed that the levels of AMPK and mTOR were not decreased by LH treatment. However, LH increased the expression of phosphorylated AMPK and significantly suppressed phosphorylated mTOR protein expression in a dose-dependent manner (Figure 6).
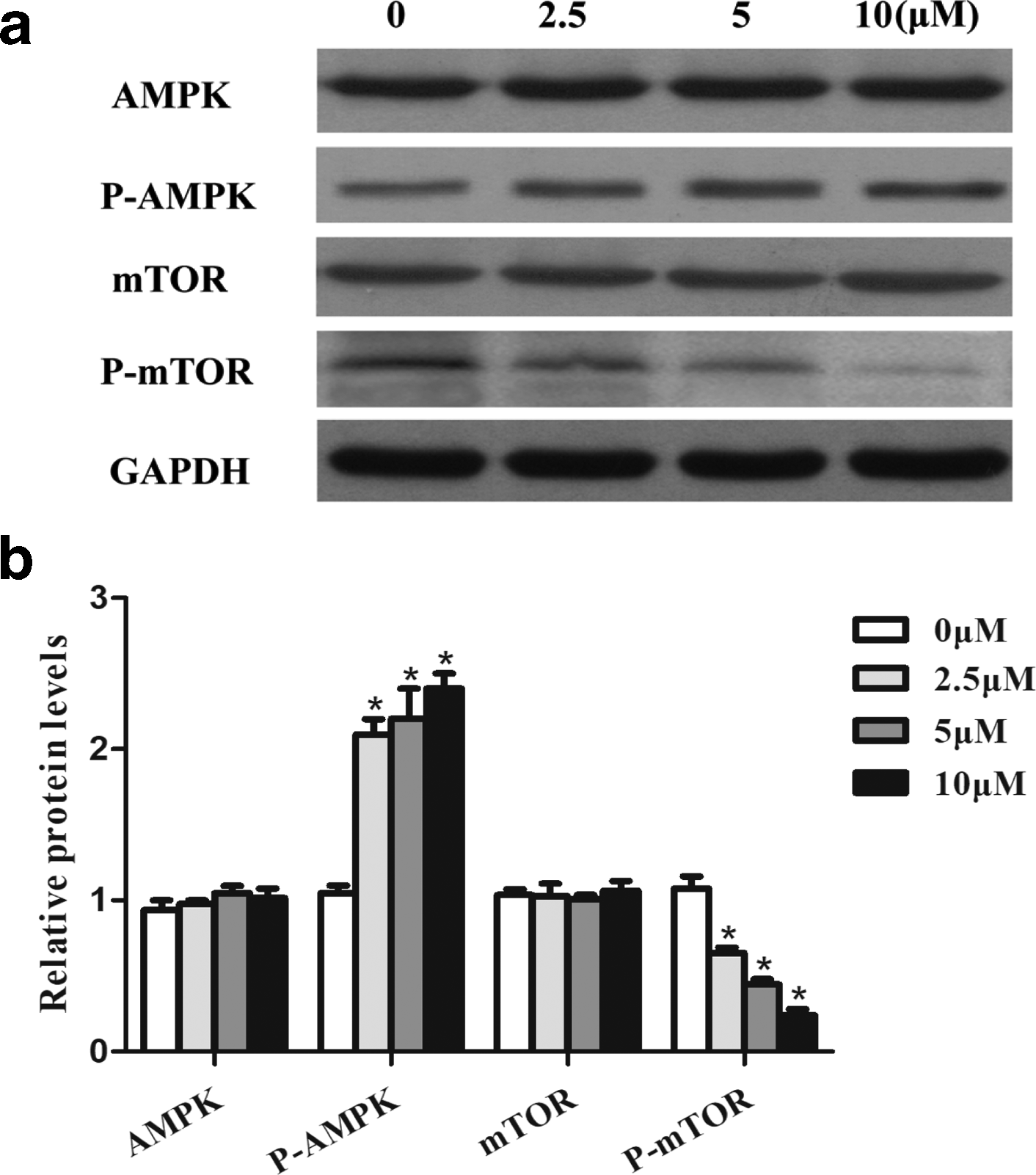
Effects of LH on the AMPK/mTOR autophagic pathway in HCT116 cells. (a) HCT116 cells were treated with vehicle or LH for 48 hours to assess the protein levels of AMPK, P-AMPK, mTOR, and p-mTOR by Western blot. (b) Quantitative analysis of AMPK, p-AMPK, mTOR, and p-mTOR. Data represent the mean values ± standard error of the mean (
Inhibition of Autophagy Promotes LH-Induced Apoptosis
Although apoptosis and autophagy are two distinct modes of programmed cell death, they have complex interconnections in order to maintain cellular homeostasis. 18 The relationship between the 2 types of programmed cell death, autophagy and apoptosis, can be positive or negative. 19 Many reports suggested that inhibition of autophagy may enhance cancer drug curative effects. 20,21 Furthermore, to confirm the role of autophagy induced by LH, we performed an autophagy gene knockdown assay by silencing Beclin-1 gene. First, Beclin-1 levels in Beclin-1 small interfering RNA (siRNA)-transfected cells were significantly reduced when compared to negative control (NC) siRNA-transfected cells confirming effective transfection. Secondly, in cells transfected with Beclin-1 siRNA, followed by LH treatment, the levels of Beclin-1 and LC3B-II were found to be significantly reduced when compared to NC group (transfected with NC siRNA and then treated with 10 µM LH) indicating the induction of autophagy by LH (Figure 7). Finally, since apoptosis rates increased significantly in Beclin-1 siRNA transfected cells followed by LH treatment compared to the NC group, this indicated that LH-induced autophagy inhibited apoptotic cell death in HCT116 cells. Protein expression levels of cleaved Caspase-3 and Bax were significantly reduced in Beclin-1 siRNA transfected cells followed by LH treatment compared to the NC group, which was consistent with the results of flow cytometry, further confirmed autophagy induced by LH inhibited apoptosis in CRC cells (Figure 8).
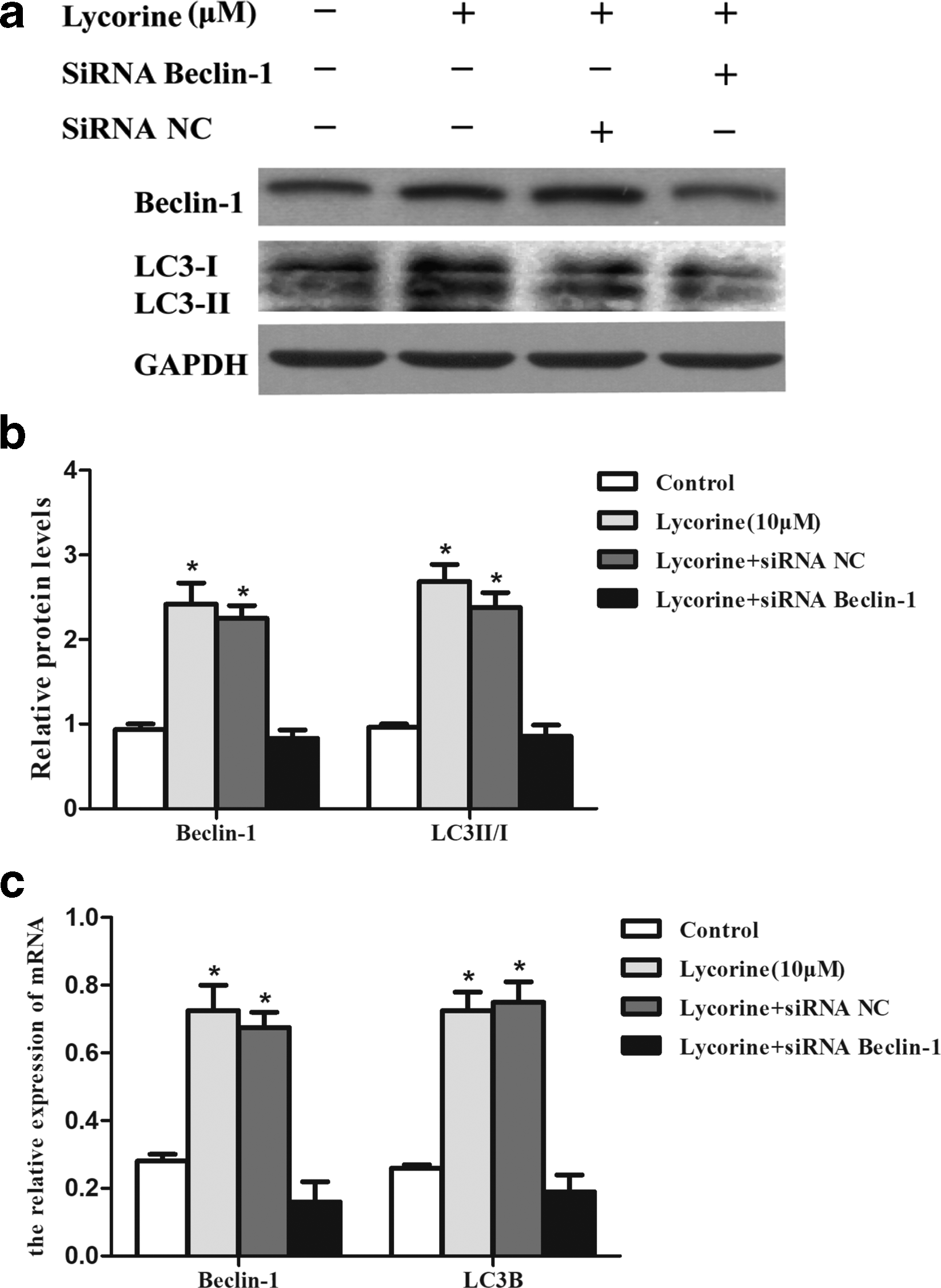
Beclin-1 knockdown inhibited LH-induced autophagy. HCT116 cells were transfected with NC siRNA or Beclin-1 siRNA, and then treated with 10 µM LH for 48 hours. (a) Whole cell lysates were analyzed for protein levels of LC3B and Beclin-1 by Western blot. (b) Quantitative analysis of LC3B and Beclin-1. (c) LC3B and Beclin-1 mRNA levels were determined by RT-qPCR. Data represent the mean values ± standard error of the mean (
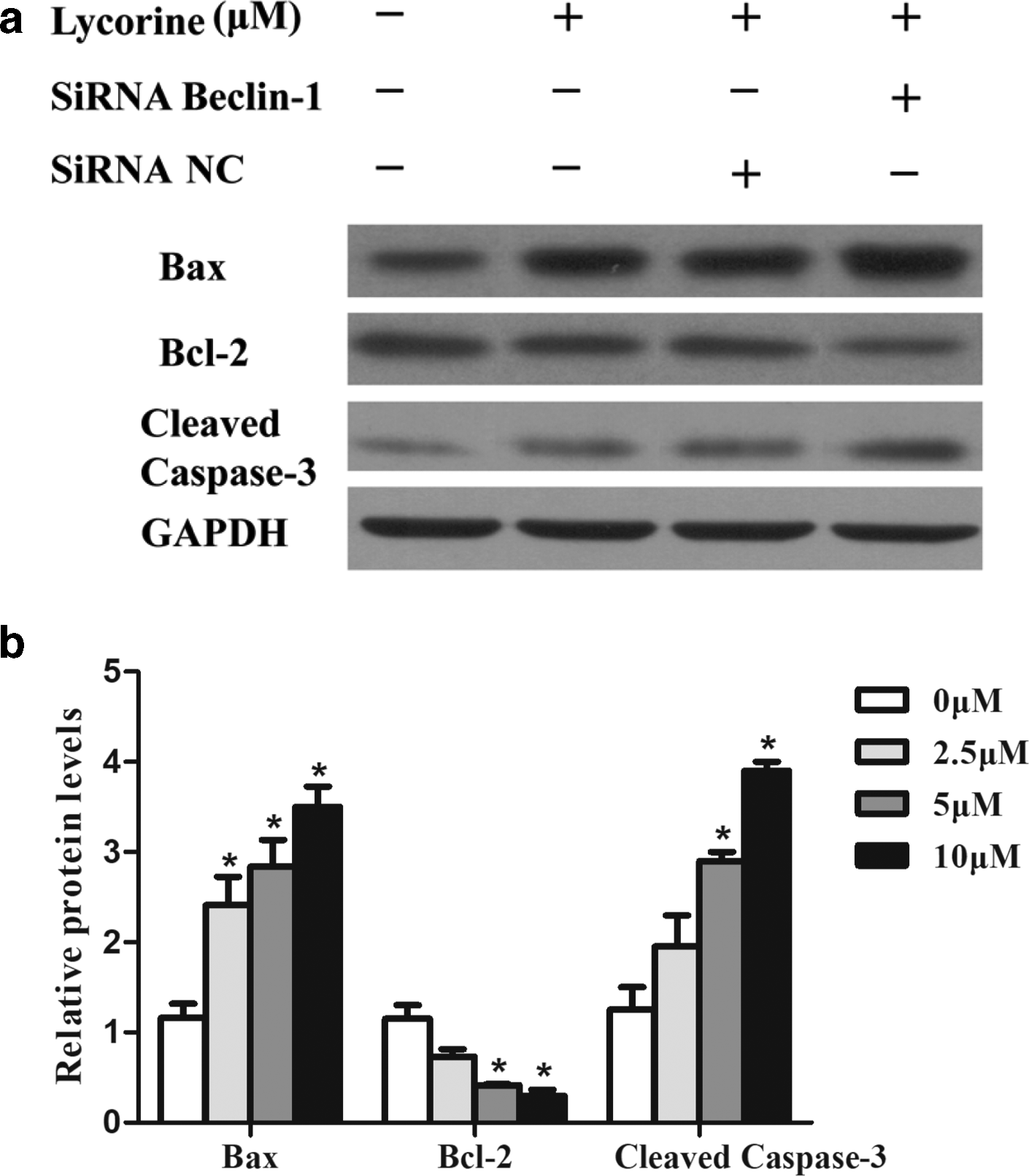
Beclin-1 knockdown promoted LH-induced apoptosis. HCT116 cells were subjected to Beclin-1 knockdown, followed by 10 µM LH treatment for 48 hours. (a) Proteins were then extracted for analysis of Bax, Bcl-2, and caspase-3 by Western blot. (b) Quantitative analysis of Bax, Bcl-2, and caspase-3. Data represent the mean values ± standard error of the mean (
In conclusion, we demonstrated that LH has potential antitumor activity in human CRC HCT116 cells by inhibiting proliferation and increasing cell apoptosis. LH induced autophagy of HCT116 cells via AMPK/mTOR autophagic pathway. Additionally, the inhibition of autophagy promoted apoptosis induced by LH via modulating Beclin-1 and Bcl-2. Therefore, inhibiting autophagy in human CRC may be a meaningful way to increase the efficacy of anticancer treatment by LH.
Experimental
Compound
LH was obtained from Dalian Meilun Biological Technology Co., Ltd. (C16H17NO4.HCl, molecular weight: 323.77, purity >98%, Dalian, China).
Cell Culture
Human CRC cell line HCT116 (purchased from Cell Culture Centre, Institute of Basic Medical Sciences, Chinese Academy of Medical Sciences) were cultured in Dulbecco’s modified Eagle medium (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) containing 10% fetal bovine serum (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA). The cell line was usually passaged 2 to 3 times per week.
Cell Viability Assay
The HCT116 cells were treated by the various concentrations of LH for 48 hours. Thereafter, 10 µL of CCK-8 solution was added to each well and the cells were incubated for an additional 2 hours at 37°C. Finally, optical density at 450 nm was measured by a SpetraMax M5 microplate reader (Molecular Devices, Sunnyvale, CA, USA).
Colony Forming
The cells were then exposed to LH for 24 hours. The medium was then replaced with complete medium and cultured at 37°C for 2 weeks. Colony formation was photographed and counted after staining with 0.1% crystal violet. The stained colonies (consisting of 50 cells or more) were scored.
SiRNA Transfection
The siRNA-targeting Beclin-1 and NC were purchased from Shanghai GenePharma Co., Ltd. (Shanghai, China). The siRNA sequences were as follows: Beclin-1 siRNA, 5′-GCTGCCGTTATACTGTTCT-3′, NC siRNA 5′-GTTCTCCGAACG TGTCACGT-3′. 15 Cells were grown in a 6-well plate until 60% confluence and transfected with 100 nM NC siRNA and 100 nM Beclin-1 siRNA using Lipofectamine 2000 transfection reagent (Thermo Fisher Scientific, MA, USA) following manufacturer protocol. After 48 hours of transfection, the indicated experiments were conducted.
Apoptosis Assay
The cells were subjected to Beclin-1 knockdown, followed by 10 µM LH treatment. After 24 hours of incubation, cells were stained with Annexin V-fluorescein isothiocyanate and PI, followed by flow cytometry analysis according to the manufacturer instructions (BD Biosciences, San Jose, CA, USA).
Quantitative RT-PCR for mRNA Expression
The expressions of Beclin-1 and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) mRNA were detected by RT-PCR. Total RNA was isolated from HCT116 cells by RNAiso Plus (Takara, Shiga, Japan) method. cDNA was synthesized by using PrimeScript RT reagent Kit with gDNA Eraser (Takara, Shiga, Japan). Quantitative analysis of the expression of Beclin-1 and GAPDH mRNA was performed with SYBR Premix Ex Taq (Takara, Shiga, Japan). PCR was performed using the following conditions: 30 seconds at 95°C, followed by 40 cycles of 5 seconds at 95°C, and extension at 60°C for 30 seconds. The data were analyzed using the relative gene expression (2-∆∆Ct) method. The primers were used as follows: beclin-1 5′-GACCGAGTGACCATTCAGGAAC-3′ (forward) and 5′-GGTTCTCCATGGTGCCACCATCAG-3′ (reverse) 15 ; LC3B-II 5′-GAGAAGCAGCTTCCTGTTCTGG-3′ (forward) and 5′-GTGTCCGTTCACCAACAGGAAG-3′ (reverse); GAPDH 5′-GGACTGACCTGCCGTCTAG-3′ (forward) and 5′-TAGCCCAGG ATGCCCTTGAG-3′ (reverse). 22
Western Blot Analysis
The HCT116 cells were treated with LH for 48 hours. Meanwhile, the cells were transfected with 100 nM Beclin-1 siRNA, followed by 10 µM LH treatment. The treated cells were washed with cold PBS followed by lysis in a cell lysis buffer (Beyotime Biotech. Co., Ltd, Shanghai, China). Protein concentration was measured by the BCA protein assay (Beyotime Biotech. Co., Ltd, Shanghai, China). Equal amounts of protein were separated on sodium dodecyl sulfate polyacrylamide gel electrophoresis gels and subsequently transferred to polyvinylidene fluoride membranes. Then, the membranes were blocked with 5% bovine serum albumin at room temperature for 1 hour. Thereafter, the membranes were incubated overnight at 4°C with different primary antibodies (anti-Caspase-3, anti-Bax, anti-Bcl-2, anti-Beclin-1, and anti-LC3B, 1: 1000, Cell Signaling Technology, Inc., MA, USA). The membranes were then incubated with secondary antibodies at room temperature and washed 3 times with TBST (1× TBS containing 0.1% Tween 20). After being rinsed, the immunoreactive bands were visualized with Western blotting luminol reagent and gel images were analyzed with Image J (NIH image, Bethesda, MD, USA).
Statistical Analysis
Data were expressed as mean ± standard error of mean and all analyses were performed in triplicate. Data were analyzed by one-way analysis of variance conducted by GraphPad Prism Software (San Diego, CA, USA). Statistical significance was considered for
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared no financial support for the research, authorship, and/or publication of this article.
