Abstract
The aim of this study was to evaluate the hepatoprotective effects of the fruits of Polygonum orientale L. (POE) against fibrosis in carbon tetrachloride (CCl4)-induced liver injury. Bioactive components of POE were identified using liquid chromatography (LC)-mass spectrometry (MS)/MS by comparison with standards. Treatment with either silymarin (200 mg/kg) or POE (0.5 and 1.0 g/kg) caused significant decreases in the serum levels of enzymes and reduced the extent of liver lesions and fibrosis in histological analysis. POE (0.5 and 1.0 g/kg) decreased the levels of malondialdehyde, nitric oxide, proinflammatory cytokines (ie, tumor necrosis factor-α, interleukin [IL]-1β, and IL-6), an inflammatory cytokine (ie, cyclooxygenase-2), a profibrotic cytokine (ie, transforming growth factor-β), and fibrosis-related proteins (ie, connective tissue growth factor and α-smooth muscle actin) in the liver and enhanced the activities of the antioxidative enzymes superoxide dismutase, glutathione peroxidase, glutathione reductase, and catalase. Quantitative analysis of the active constituents in POE revealed an extract composition of 3.4 mg/g of protocatechuic acid, 20.8 mg/g of taxifolin, and 5.6 mg/g of quercetin. We have demonstrated that the hepatoprotective mechanisms of POE are likely to be associated with the decrease in inflammatory cytokines by increasing the activities of antioxidant enzymes. Our findings provide evidence that POE possesses a hepatoprotective activity to ameliorate chronic liver injury.
The liver is a crucial organ that plays an important role in the detoxification of diverse xenobiotics. Exposure of the general population to environmental pollutants reduces the protective mechanisms of the liver and causes hepatic failure. 1 Toxic chemicals, alcohol, drugs, and viral infections are also known to cause liver injury. Carbon tetrachloride (CCl4) is a chemical commonly used in animal models to study liver injury and fibrosis. 2 Cytochrome P-450 enzymes metabolize CCl4 to trichloromethyl radical and trichloromethyl peroxyl radical. These free radicals and related reactive oxygen species cause cellular damage through lipid peroxidation and the promotion of proinflammatory cytokine production from activated Kupffer cells (KCs). 3 This phenomenon indicates that CCl4 is a powerful hepatotoxic agent and may be used for the characterization of hepatic disease treatment medicines. 4 In addition, proinflammatory cytokines, including tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and IL-6, could be active in hepatic stellate cells (HSCs) and KCs. Those cytokines are believed to play central roles in the inflammatory reaction. TNF-α also stimulates the release of cytokines, such as cyclooxygenase-2 (COX-2), from KCs and induces nitric oxide (NO) production. 5 High levels of COX-2 and NO contribute to inflammatory response and oxidative stress in injured livers. 6
Liver fibrosis is an outcome of several chronic liver diseases, with scarring formation in acute or chronic injury. 7 During liver injury, HSCs change from the quiescent to the activated state and transfer into myofibroblasts in the formation of fibrosis with limited deposition of extracellular matrix (ECM). 8 In contrast to acute injury, chronic injury leads to the sustained production of oxidative stress, as well as inflammatory and fibrotic cytokines. These events culminate in the accumulation of ECM, which plays a major role in hepatic fibrosis and regeneration. 9 Previous studies have indicated that transforming growth factor-β (TGF-β) and connective tissue growth factor (CTGF) play critical roles in liver fibrosis. 10 TGF-β prompts fibroblasts to synthesize and contract ECM. 11
This cytokine has long been thought to be a central mediator of the fibrotic response. 12 α-Smooth muscle actin (α-SMA) can be measured in ECM; therefore, it could be an indicator of liver fibrosis. 13 Recent studies have employed the following methods of attenuating liver fibrosis: (1) suppressing the activation of HSCs, (2) reducing the formation of ECM, (3) inhibiting inflammatory response, and (4) reducing oxidative stress. 14
Polygonum orientale L. (POE), a member of the Polygonaceae family, has long been used as medicine and food in China. 15 Phytochemical studies have evidenced a high quantity of flavonoids in this plant, such as taxifolin and quercetin. 16 Gallic acid (GA) and protocatechuic acid, both phenolics, have also been identified. 17 GA reduced alanine aminotransferase (ALT), aspartate aminotransferase (AST), malondialdehyde (MDA), platelet-derived growth factor-BB levels, collagen deposition, and fibrosis score. 18 However, the antifibrotic and hepatoprotective effects of POE extract on chronic liver injury remain unknown.
The aim of the present study was to investigate the protective effects of POE against liver fibrosis and chronic injury induced by CCl4.
Materials and Methods
Plant Materials
The fruits of POE were collected from Toushe, Nantou County, Taiwan, in July 2013. The plant was identified by Chao-Lin Kuo, leader of the Department of Chinese Pharmaceutical Sciences and Chinese Medicine Resources, China Medical University (CMU-CMR). A voucher specimen (Number: CMU-CMR-PO-103001) was deposited at CMU-CMR for further reference.
Chemicals
Silymarin and quercetin were purchased from Sigma-Aldrich Chemical Co. (USA); CCl4 from Merck Co. (Germany); superoxide dismutase (SOD), glutathione peroxidase (GPx), and reduced glutathione (GRd) assay kits from Randox Laboratory Ltd; TNF-α, IL-1β, and IL-6 from eBioscience Inc. (San Diego, CA, USA); TGF-β from BioLegend (San Diego, CA, USA), and COX-2 from R&D Systems, Inc. (Minneapolis, USA). All other reagents used were of analytical grades.
Preparation of Plant Extract
The preparation of the crude extract was modified from the previous study. 19 Whole fruits were dried in a circulating air oven, crushed, and ground into a coarse powder (1.8 kg), which was extracted by maceration with 70% ethanol, 3 times. The filtered solution was collected and concentrated under reduced pressure to give 91.5 g extract with a yield of 5.08% (w/w). The remaining solution was lyophilized before the final POE was obtained. The extract was stored at −20 °C before the experiment.
Chromatographic Analyses
A high-performance liquid chromatography (HPLC) profile was established for the standards (protocatechuic acid, taxifolin, and quercetin) and POE; the method was modified from Zhou et al’s study. 20 The HPLC instruments used in this study consisted of a Shimadzu LC-20AT HPLC and SPD-20A UV-Vis detector system. An Ascentis C18 column (4.6 mm × 250 mm, 5 µm), purchased from Sigma-Aldrich, was used. The mobile phase consisted of 0.1% (v/v) aqueous phosphoric acid (A) and methanol (B) processing by a gradient elution program as minutes (%B): 0 minutes (10), 10 minutes (39), 20 minutes (47), 30 minutes (47), 40 minutes (58), 55 minutes (56), 65 minutes (10), and 75 minutes (10). All standards and samples were passed through a 0.45-µm Minipore filter before injection into the column with a sample size of 20 µL and detection at 270 nm. The quantitative data of protocatechuic acid, taxifolin, and quercetin in POE were calculated using their respective concentration versus peak area calibration curves.
The liquid chromatography-tandem mass spectrometry (LC-MS/MS) profile was established for the standards (protocatechuic acid, taxifolin, and quercetin) and POE. LC-MS was performed on a Dionex Ultimate 3000 HPLC system (Dionex, Germany) linked with electrospray ionization (ESI)-ion trap MS (HCT Ultra PTM Discovery, Bruker Daltonics, Germany). A linear gradient on a C18 LC column (Atlantis T3 C18, 5 µm; 2.1 mm × 150 mm) was used to separate the crude extracts with a flow rate of 0.25 mL/min. The mobile phase consisted of 5% (v/v) acetonitrile containing 0.1% (v/v) formic acid (A) and 99.9% (v/v) acetonitrile with 0.1% (v/v) formic acid (B). A gradient elution program as minutes (%B): 0 (1), 13 (60), and to 90% of B over 0.5 minutes. It was then held at 90% B for another 1.5 minutes at a flow rate of 0.25 mL/min followed by a return to the starting conditions and re-equilibration of the column for 1.5 minutes with 1% B prior to the next injection. The total run time was 18 minutes. The ESI source was operated in negative ion mode, and the scan range was 20 to 800 m/z. Nitrogen was used as the nebulizing (40 psi) and drying gas (10 L/min, 300 °C), and helium as the collision gas. For the MS/MS settings, the 6 most intense ions from each MS full scan spectrum were automatically selected as the precursor ion peaks for the following auto MS/MS experiments. The multiple reaction monitoring (MRM) collision amplifications were 0.5, 1, and 0.8 for protocatechuic acid, taxifolin, and quercetin, respectively. In MRM mode, 152.8→108.8, 300.8→178.8, and 302.8→284.8 were used as the transition ions (m/z) for the measurement of protocatechuic acid, taxifolin, and quercetin, respectively.
Animals and Experimental Design
Male Institute for Cancer Research (ICR) mice (22 ± 2 g) were obtained from BioLASCO Taiwan Co., Ltd. The mice were kept in the Animal Center of China Medical University at a controlled temperature of 22 ± 1 °C and relative humidity of 55 ± 5%, along with 12 hours light/12 hours dark cycles for 1 week before the experiment. Animals were provided with a rodent diet and clean water ad libitum. All studies were conducted in accordance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals and 3R (Replacement, Reduction, and Refinement). All tests were conducted under the guidelines of the International Association for the Study of Pain. The Committee on Animal Research, China Medical University, approved the experimental protocol. This study used cervical dislocation to sacrifice animals. Each animal experiment was performed in 6 groups of 10 mice each.
Acute Oral Toxicity
The acute oral toxicity test was performed according to the modified version in Liao’s study. 21 Male ICR mice (22 ± 2 g) were randomly divided into 2 groups with 10 mice each. One group was administered orally with the vehicle and the other with POE (5.0 g/kg). The experimental mice were given forage and water ad lbitum and were kept under regular observation for any mortality or behavioral changes within 14 days.
Experimental Design of CCl4-Induced Liver Fibrosis
The experimental animals were randomly divided into 6 groups (10 mice in each group). Mice in the control group (Con) and CCl4 group (CCl4) were orally administered distilled water. The silymarin group (S) was orally administered silymarin (200 mg/kg in 1% carboxymethylcellulose), and the POE group was orally administered POE (0.1, 0.5, and 1.0 g/kg) every day for 8 weeks. The doses given for POE were based on traditional use and translated from humans to mice. 22 One hour after the last administration of the experimental drugs, CCl4 (10 mL/kg body weight, 20% in olive oil) was injected intraperitoneally (ip) into each group, except for the control group, 3 times a week for 8 weeks. The control mice received an equivalent volume of olive oil (ip).
All mice were sacrificed under anesthesia on the 8th weekend. Blood samples were centrifuged at 3000 rpm for 30 minutes at 4 °C, and the serum was kept at −80 °C for measurement of biochemical parameters. The livers were weighed after rinsing with saline and blotted on filter paper. One part of the liver was cut for histological analysis, and the remaining liver tissues were stored at −80 °C for later analysis.
Body Weight and Liver Index Analysis
Body weight was recorded once every 2 days. Livers were removed after sacrifice. The liver index was calculated as liver weight/body weight, mg/g.
Serum Biochemistry
The blood was centrifuged at 3000 rpm (Beckman GS-6R, Germany) at 4 °C for 30 minutes to separate the serum. ALT, AST, and alkaline phosphatase (ALP) were measured using spectrophotometric diagnostic kits in a ChemWell-T Automated Chemistry Analyzer.
Histopathological Examination
Liver tissues were collected from the same lobes and trimmed to approximately 2 mm thickness. The tissues were then fixed in a 10% buffered formaldehyde solution and embedded in paraffin. They were further cut into 2 µm sections, stained with hematoxylin and eosin (H&E) and Sirius red, and then examined by light microscopy.
Proinflammatory, Inflammatory, and Profibrotic Cytokines
Proinflammatory cytokines (TNF-α, IL-1β, IL-6), inflammatory cytokines (COX-2), and profibrotic cytokines (TGF-β) were measured by enzyme-linked immunosorbent assay kits according to the manufacturer’s instructions. The captured antibodies of TNF-α, IL-1β, IL-6, COX-2, and TGF-β were seeded to each well of a 96-well plate overnight. The next day, a second set of the biotinylated antibody was incubated with either sample tissues or standard antigens in the plate before streptavidin-horseradish peroxidase was finally added. TNF-α, IL-1β, IL-6, COX-2, and TGF-β were measured at 450 nm to determine their amount represented as pg/mg protein.
Hepatic Antioxidant Enzymes
The liver was removed, washed thoroughly in ice-cold saline, and a homogenate (10%) prepared in phosphate-buffered saline (50 mM, pH 7). Liver tissue homogenates were collected for the estimation of SOD, GPx, and GRd enzymes to detect the antioxidant activities of POE using a ChemWell-T Automated Chemistry Analyzer. Part of the homogenate was used for the estimation of GRd. 23 The remaining homogenate was centrifuged at 5000×g for 10 minutes in a centrifuge at 4 °C, and after removal of the cell debris, the supernatant was used for the assay of SOD and GPx. 24
Assessment of Lipid Peroxidation
The level of lipid peroxidation in the liver was defined by measuring MDA. The MDA levels were evaluated by the thiobarbituric acid reacting substance (TBARS) method. 25 Briefly, MDA, when reacted with thiobarbituric acid in acidic conditions at high temperature, forms a red-complex, TBARS, the absorbance of which was determined at 532 nm.
NO Assay
NO was measured based on the method reported by Moshage et al. 26 Nitrate was converted to nitrite utilizing nitrate reductase. Nitrite was subsequently reacted with sulfanilic acid to produce the diazonium ion and reacted with N-(1-naphthyl) ethylenediamine to give a chromophoric azo derivative (purplish red), which could be recorded at 540 nm. Values obtained by this procedure represented the sum of nitrite and nitrate and are represented as μM.
Hydroxyproline Measurement
Hydroxyproline is the most important constituent of collagen, which forms hepatic ECM. The analysis of hydroxyproline in the liver was based on the chloramine-T method. 27 Liver tissue was homogenized and hydrolyzed in 6N hydrochloric acid at 120 °C, then centrifuged at 10,000 rpm for 15 minutes. To the hydrolyzed supernatant, chloramine T reagent was added, and the mixture was maintained at room temperature for 25 minutes. The final step was the addition of Ehrlich’s reagent. Absorbance at 560 nm was recorded with a spectrophotometer.
Western Blotting Assay
The expression of metabolic regulatory signals in the liver was examined using Western blot analysis. In brief, ice-cold radioimmunoprecipitation assay buffer containing phosphatase and protease inhibitors (50 mM sodium vanadate, 0.5 mM phenylmethylsulfonyl fluoride, 2 mg/mL aprotinin, and 0.5 mg/mL leupeptin) was used for protein extraction. The protein concentrations were then measured by DC protein assay (Bio-Rad Laboratories Inc., USA). Total protein samples (60 µg) were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (10% acrylamide gel) using the Bio-Rad Trans-Blot system and then transferred to membranes. The membranes were blocked with 5% bovine serum albumin in Tris-buffered saline containing 0.1% Tween 20 (TBS-T), incubated for 60 minutes, and then hybridized with primary antibodies specific for CTGF (Novus Biologicals, USA), catalase (CAT; Abcam, UK), α-SMA (Abcam, UK), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH; Novus Biologicals, USA) at suitable concentrations in TBS-T for 16 hours. The membranes were then incubated with a secondary antibody for an additional 3 hours. Detection of the antigen-antibody complex was performed using an enhanced chemiluminescence kit (Merck Millipore, Germany) with GAPDH used as the internal control. After comparing with the marker for specificity, the immunoblots of CTGF (38 KDa), CAT (60 KDa), α-SMA (42 KDa), and GAPDH (38 KDa) were quantified with a laser densitometer.
Statistical Analyses
All data were expressed as mean ± SEM. Statistical analyses were performed using a one-way analysis of variance followed by Scheffe’s multiple range tests by SPSS software (IBM Corp. Released 2011. IBM SPSS Statistics for Windows, Version 20.0. Armonk, NY: IBM Corp). Histological analyses were carried out using the nonparametric Kruskal-Wallis test followed by the Mann-Whitney U-test. The criterion for statistical significance was P < 0.05.
Results
Chromatographic Analyses of POE
An LC-MS fingerprint profile was established as the base peak chromatogram (BPC) for the standards (ie, protocatechuic acid, taxifolin, and quercetin), identified with retention times (RTs) of 5.5, 11.4, and 15.6 minutes, respectively. The BPC for POE was consistent with RTs, as well as molecular and fragment ions. RTs of 11.3, 19.6, and 39.4 minutes were detected, representing protocatechuic acid, taxifolin, and quercetin, respectively. These peaks were also found in the POE chromatogram indicating the relative amounts of each at 270 nm. Chromatograms were baseline separated according to the time program, and the identification was achieved through spiking each standard in the sample. Quantitative analysis of POE gave contents of 3.4 mg/g of protocatechuic acid, 20.8 mg/g of taxifolin, and 5.6 mg/g of quercetin.
Acute Oral Toxicity Test
An acute toxicity test indicated no mortality or unusual behavior among the mice orally administered POE 5.0 g/kg for 14 days. In addition, no significant difference existed between the control and POE groups in liver or kidney function assays examining AST, ALT, creatinine (CRE), blood urea nitrogen (BUN), and albumin (ALB) in serum. Therefore, we concluded that the weight of the median lethal dose (LD50) in POE is greater than 5.0 g/kg and that POE is a safe extract (Table 1).
Serum Biochemical Parameters From POE and Control at 5.0 g/kg Body Weight in Mice.

Abbreviations: ALB, albumin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nirtogen; CRE, creatinine; POE, Polygonum orientale L.
Effects of serum biochemical parameters on (A) control and (B) POE. Values are mean ± SEM (n = 10). “*” indicates significant difference from the control group (*P < 0.05).
Body Weight and Liver Index
The mean body weight of the surviving mice in all groups is shown in Figure 1(A). CCl4 caused a significant reduction in body weight in the CCl4 group compared with that in the control group from the fourth week. After administration of either silymarin (200 mg/kg) or POE (1.0 g/kg) for 5 weeks, the body weight of the treated mice significantly recovered from the effect of CCl4. As shown in Figure 1(B), in the CCl4 group, hepatomegaly was observed, and the liver index was significantly higher than that of the control group. The liver indices of the silymarin (200 mg/kg) and POE (0.5 and 1.0 g/kg) groups were significantly lower than that of the CCl4 group.

Effects of POE and silymarin on body weight (A) and liver index (B) induced by carbon tetrachloride (CCl4) during 8 weeks. Values are mean ± SEM (n = 10). “#” Indicates significant difference from the control group (### P < 0.001). “*” Indicates significant difference from the CCl4 group (*P < 0.05, **P < 0.01, ***P < 0.001). POE, Polygonum orientale L.
Hepatic Serum Levels
As shown in Figure 2(A-C), the CCl4 group exhibited a significant increase in AST, ALT, and ALP serum levels. Following treatment with either silymarin (200 mg/kg) or POE (0.5 and 1.0 g/kg), AST, ALT, and ALP levels significantly decreased compared with those of the control group. These findings suggest that POE possesses protective properties against CCl4-induced liver injury.

Effect of POE and silymarin exerted on activities of serum (A) aspartate aminotransferase, (B) alanine aminotransferase, and (C) alkaline phosphatase in mice treated with carbon tetrachloride (CCl4). Values are mean ± SEM (n = 10). “#” Indicates significant difference from the control group (### P < 0.001). “*” Indicates significant difference from the CCl4 group (*P < 0.01, **P < 0.005, ***P < 0.001). POE, Polygonum orientale L.
Histological Analyses
The results of H&E staining showed obvious changes in the histological analyses in the CCl4 group, including increased vacuolization, inflammation, and hepatocellular necrosis (Figure 3(B-F)) compared with the control group (Figure 3(A)). Liver damage was reduced through treatment with silymarin (200 mg/kg) and POE (0.5 g/kg and 1.0 g/kg) (Figure 3(C, E, F)), and the injury scores for vacuolization, inflammation, and hepatocellular necrosis were significantly decreased (Table 2). As shown in Figure 4(B) and Table 2, prolonged administration of CCl4 led to an obvious accumulation of collagen in the liver, indicated by Sirius red staining in the CCl4 group. Treatment with silymarin (200 mg/kg) and POE (0.5 g/kg and 1.0 g/kg) significantly inhibited the progression of liver fibrosis (Figure 3(C, E, F) and Table 2). The histological analyses revealed that treatment with POE significantly inhibited CCl4-induced liver fibrosis.

Hepatic histological analyses of the effect of POE and silymarin on CCl4-induced liver fibrosis in mice based on hematoxylin and eosin staining (100×). (A) Control group, (B) animals treated with 20% CCl4 (10 mL/kg of body weight), (C) animals treated with silymarin (200 mg/kg) and then treated with CCl4, and (D-F) animal treated with POE (0.1, 0.5, and 1.0 g/kg) and then treated with CCl4. CCl4, carbon tetrachloride; POE, Polygonum orientale L.

Hepatic histological analyses of the effect of POE and silymarin on CCl4-induced liver fibrosis in mice based on Sirius red staining (100 ×). (A) Control group, (B) animals treated with 20% CCl4 (10 mL/kg of body weight), (C) animals treated with silymarin (200 mg/kg) and CCl4, and (D-F) animals treated with POE (0.1, 0.5, and 1.0 g/kg) and CCl4. CCl4, carbon tetrachloride; POE, Polygonum orientale L.
Quantitative Summary of the Protective Effects of POE and silymarin on CCl4-Induced Liver Fibrosis and Damage Based on Histological Observations.

Abbreviations: CCl4, carbon tetrachloride; POE, Polygonum orientale L.
Values are mean ± SEM (n = 10). “#” Indicates significant difference from the control group (### P < 0.001). “*” Indicates significant difference with respect to the CCl4 group (*P < 0.05, **P < 0.01, ***P < 0.001).
aThe histological indices of hepatic inflammation and necrosis were quantified based on the method of Knodell’s study. 28 The liver damage was graded 0-4 as following: none (0); slight (1); mild (2); moderate (3); and remarkable (4).
bHepatic fibrosis was graded 0-4 according to the method of Ruwart’s study 29 as the following: none (0), normal liver; slight (1), increase of collagen without formation of septa; mild (2), formation of incomplete septa from portal tract to central vein (septa that do not interconnect with each other); moderate (3), complete, but thin septa interconnecting with each other (incomplete cirrhosis); and remarkable (4), with thick septa (complete cirrhosis).
Effects of POE on TNF-α, IL-1β, IL-6, COX-2, and TGF-Β in the Liver
As shown in Tables 3 and 4, the TNF-α, IL-1β, IL-6, COX-2, and TGF-α levels in the CCl4-induced liver fibrosis group were markedly increased. Treatment with either silymarin (200 mg/kg) or POE at 0.5 and 1.0 g/kg significantly suppressed the concentration of TNF-α. Similarly, IL-1β, IL-6, COX-2, and TGF-α levels were significantly reduced by silymarin (200 mg/kg) and POE (0.5 and 1.0 g/kg).
Effect of POE and silymarin on Proinflammatory Cytokines.

Abbreviations: CCl4, carbon tetrachloride; IL, interleukin; POE, Polygonum orientale L.; TNF-α, tumor necrosis factor-alpha.
Values are mean ± SEM (n = 10). “#” Indicates significant difference from the control group (### P < 0.001). “*” Indicates significant difference with respect to the CCl4 group (*P < 0.05, **P < 0.01, ***P < 0.001).
Effect of POE and silymarin on Inflammatory Cytokine and Profibrotic Cytokine.

Abbreviations: CCl4, carbon tetrachloride; COX-2, cyclooxygenase-2; POE, Polygonum orientale L.; TGF-β, transforming growth factor-β.
Values are mean ± SEM (n = 10). “#” Indicates significant difference from the control group (### P < 0.001). “*” Indicates significant difference with respect to the CCl4 group (**P < 0.01, ***P < 0.001).
Effects of POE on Hepatic Antioxidant Enzymes
SOD, GPx, GRd, and CAT activities were measured to evaluate the antioxidant effects of POE in the liver. Compared with the activities of hepatic enzymes in the control group, those in the CCl4 group were significantly decreased, as shown in Table 5 and Figure 5. Treatment with either POE (0.5 g/kg and 1.0 g/kg) or silymarin (200 mg/kg) significantly increased the levels of SOD, GPx, GRd, and CAT activity.
Effect of POE and silymarin on the Activities of Different Antioxidant Enzymes in CCl4-Treated Mice.
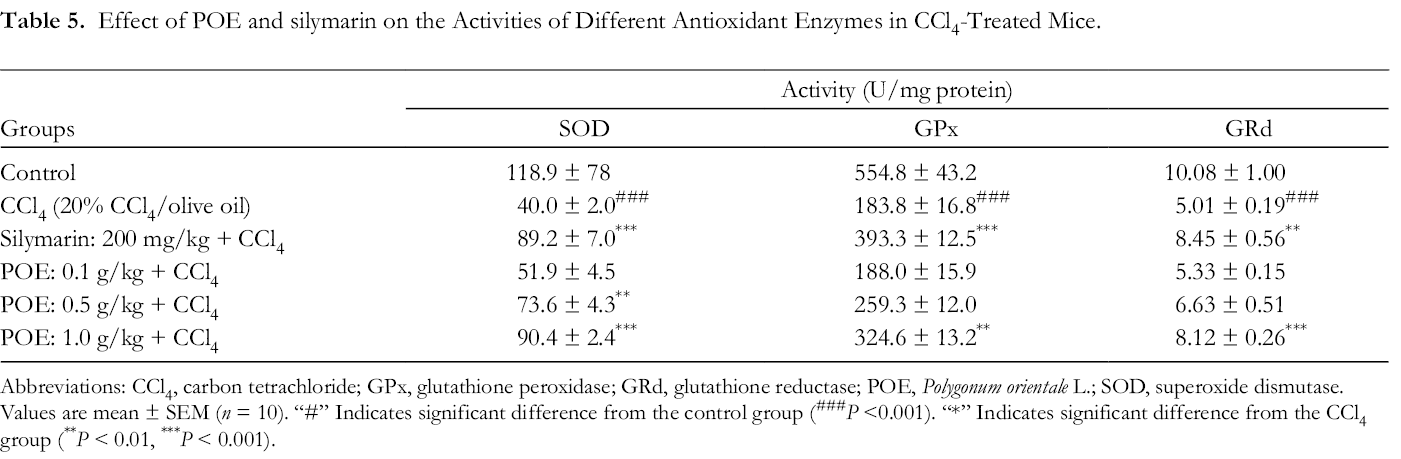
Abbreviations: CCl4, carbon tetrachloride; GPx, glutathione peroxidase; GRd, glutathione reductase; POE, Polygonum orientale L.; SOD, superoxide dismutase.
Values are mean ± SEM (n = 10). “#” Indicates significant difference from the control group (### P <0.001). “*” Indicates significant difference from the CCl4 group (** P < 0.01, *** P < 0.001).

Effect of POE and silymarin on the protein expression of CAT in the liver treated with CCl4. Values are mean ± SEM (n = 10). “#” Indicates significant difference from the control group (### P < 0.001). “*” Indicates significant difference from the CCl4 group (**P < 0.01, ***P < 0.001). CAT, catalase; CCl4, carbon tetrachloride; POE, Polygonum orientale L.
Effects of POE on MDA Level in the Liver
MDA level was used to express the degree of hepatic lipid peroxidation. The effects of POE on CCl4-induced lipid peroxidation are shown in Figure 6. The level of MDA in the CCl4 group significantly increased (### P < 0.01). However, MDA levels were reduced through treatment with either POE (0.5 and 1.0 g/kg) or silymarin (200 mg/kg). The results showed that POE could reduce the lipid peroxidation induced by CCl4.

Effect of POE and silymarin on CCl4-induced MDA contents in mice liver. Values are mean ± SEM (n = 10). “#” Indicates significant difference from the control group (### P < 0.001). “*” Indicates significant difference from the CCl4 group (**P < 0.01, ***P < 0.001). CCl4, carbon tetrachloride; MDA, malondialdehyde; POE, Polygonum orientale L.
Effects of POE on NO Level in the Liver
Compared with the NO level of the control group, the liver was significantly higher in the CCl4 group. Treatment with either silymarin (200 mg/kg) or POE (0.5 and 1.0 g/kg) yielded significant inhibition of the increased NO level in the mouse livers of the mice compared with that of the CCl4 group (Figure 7).

Effect of POE and silymarin on the level of nitric oxide in the liver treated with CCl4. Values are mean ± SEM (n = 10). “#” Indicates significant difference from the control group (### P < 0.001). “*” Indicates significant difference from the CCl4 group (***P < 0.001). CCl4, carbon tetrachloride; POE, Polygonum orientale L.
Effects of POE on Liver Fibrosis Caused by CCl4 in Mice
Hydroxyproline content was used as an effective indicator of liver fibrosis, as shown in Figure 8(A). Compared with the control group, the CCl4-treated group exhibited significantly increased hydroxyproline in the liver. However, the silymarin (200 mg/kg) and POE (0.5 and 1.0 g/kg) groups showed decreased hydroxyproline content compared with that of the CCl4 group. The effects of POE on α-SMA expression were investigated (Figure 8(B)). The CCl4 group displayed a significant increase in α-SMA expression compared with that of the control group. However, the expressions of α-SMA in the silymarin (200 mg/kg) and POE (0.5 and 1.0 g/kg) groups were clearly lower than that of the CCl4 group. As indicated in Figure 8(C), the expression of CTGF protein was upregulated in the CCl4 group. By contrast, after silymarin (200 mg/kg) and POE (0.5, 1.0 g/kg) treatment, the expression of CTGF protein was downregulated compared with that of the CCl4 group.
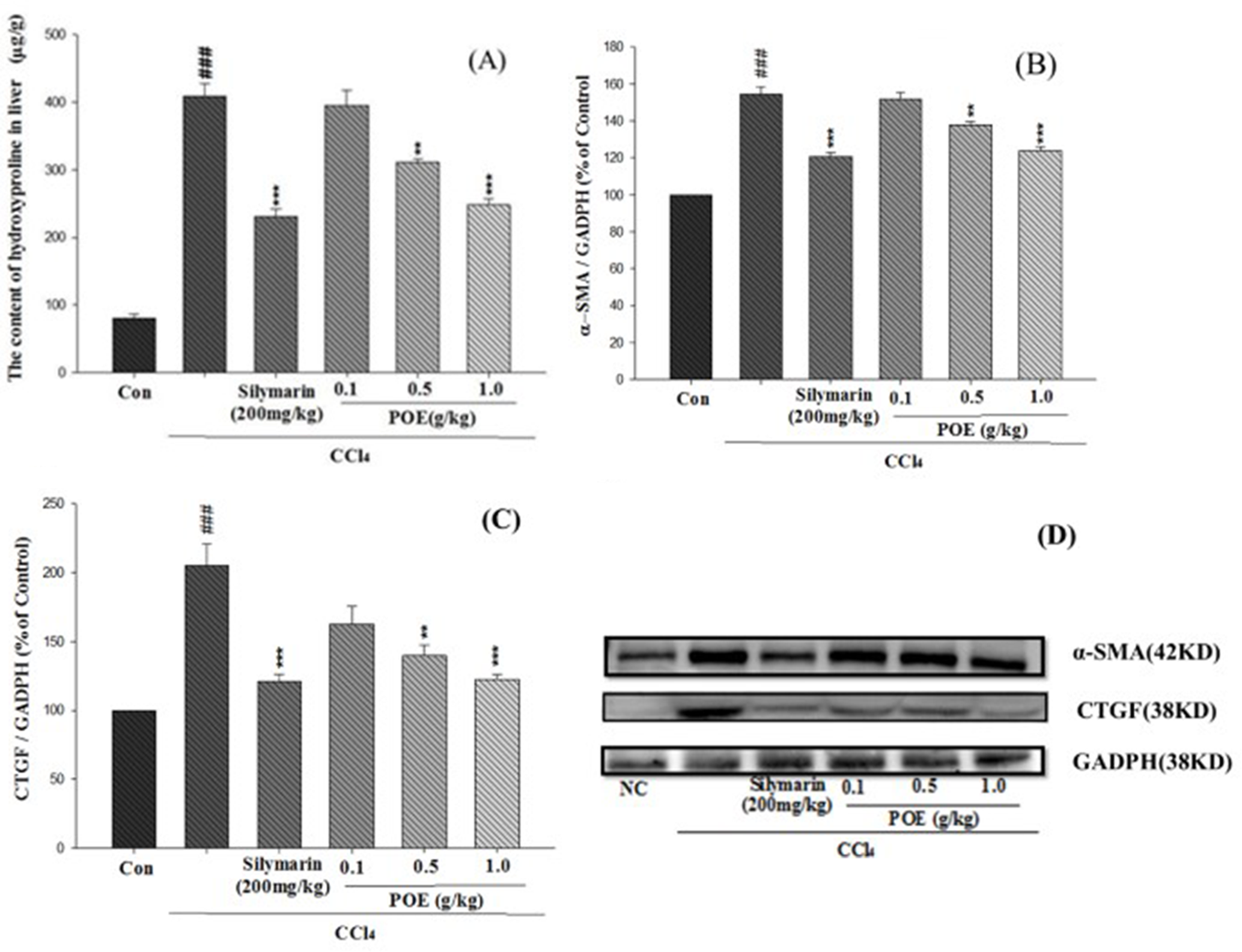
Effects of POE and silymarin on fibrosis of liver tissue induced by CCl4. (A) Content of hydroxyproline; (B) protein expression of α-SMA; (C) CTGF; and (D) Western blot of α-SMA and CTGF. Values are mean ± SEM (n = 10). “#” Indicates significant difference from the control group (### P < 0.001). “*” Indicates significant difference from the CCl4 group (**P < 0.005, ***P < 0.001). CCl4, carbon tetrachloride; CTGF, connective tissue growth factor; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; POE, Polygonum orientale L. α-SMA, α-smooth muscle actin.
Discussion
Chronic liver disease is one of the most prevalent causes of human sufferings and death, and liver fibrosis is considered to be the most important pathological feature of chronic liver disease. 30 Numerous hepatoprotective activities derived from traditional Chinese medicines and herbs have been reported in recent years. Flavonoids and phenolics have exhibited various pharmacological effects, particularly relating to anti-inflammatory and hepatoprotective activities. 31 Phytochemical studies have identified abundant flavonoids in the fruit of POE. 32 In addition, our previous research showed that POE has a protective effect against acute liver inflammation caused by CCl4. 33 Because of this, we wanted to continue to explore whether the role of POE in long-term liver injury can also protect the liver. CCl4 is a well-known toxic substance used to induce liver injury in animal models evaluating hepatoprotective effects. It is also one of the most reported substances employed in response to physical, biochemical, and histological changes associated with fibrosis and inflammation of the liver. 34 Intraperitoneal injection of CCl4 (10 mL/kg body weight, 20% in olive oil) twice weekly for 8 weeks induced hepatocellular apoptosis and necrosis associated with HSC activation, leading to fibrosis, inflammation, and cirrhosis. 35 The liver index in the CCl4 group was clearly higher than that of the control group; however, the liver indices of the silymarin (200 mg/kg) and POE (0.5 and 1.0 g/kg) groups were clearly lower than that of the CCl4 group (Figure 1). The hepatotoxic effects of CCl4 were evaluated according to the serum levels of AST, ALT, and ALP. 36 Treatment with either silymarin (200 mg/kg) or POE (0.5 and 1.0 g/kg) clearly attenuated the hepatotoxic effect of CCl4, as indicated by the significant decrease in the serum levels of AST, ALT, and ALP in the silymarin and POE groups compared with that of the CCl4 group (Figure 2). Excessive NO is related to several acute and chronic diseases such as hepatic injuries and diabetes. 37 Leukocytes and KCs produce high quantities of NO and cytokines, particularly TGF-β and TNF-α, which are a potent profibrogenic cytokine modulator and an inflammation modulator, respectively. 12 In liver tissue, hepatocellular necrosis and inflammation can be observed in a model of CCl4-induced mice, attributable to the activation of cytochrome P450 isozymes, including CYP2E1, involved in CCl4 bioactivation. 38 In the present study, treatment with either silymarin (200 mg/kg) or POE (0.5 and 1.0 g/kg) clearly reduced the TNF-α, IL-1β, IL-6, COX-2, and NO concentrations in liver tissue compared with that of the CCl4 group (Table 3 and Figure 7). Moreover, the histopathological examination of H&E staining revealed significant differences among the silymarin (200 mg/kg), POE (0.5, 1.0 g/kg), and CCl4 groups (Table 2). Our data suggest that the hepatoprotective effect of silymarin (200 mg/kg) and POE (0.5 and 1.0 g/kg) results from their ability to reduce the levels of proinflammatory and profibrotic cytokines. CCl4 treatment reduces the antioxidant enzymes SOD, CAT, GPx, and GRd in the liver because of their rapid depletion during the reduction of oxidative stress. 37 Lipid peroxidation caused by free radicals from CCl4 was found to lead to liver injury in an experimental model. 5 Furthermore, the MDA level in the liver tissue is an indicator of lipid peroxidation. 39 The results of the present study demonstrated that treatment with either silymarin (200 mg/kg) or POE (0.5, 1.0 g/kg) significantly reduced the MDA level and increased the levels of SOD, CAT, GPx, and GRd in the silymarin and POE groups compared with those of the CCl4 group (Figures 5 and 6). Lipid peroxidation was suppressed through upregulated antioxidants. This result suggested that treatment with either silymarin (200 mg/kg) or POE (0.5 and 1.0 g/kg) prevented liver injury caused by free radicals and consecutive pathological changes in CCl4-induced liver injury.
Continuous injury and chronic wound healing response, including inflammatory response and cellular proliferation, produce cytokines and ECM components. 40 The excessive accumulation of ECM by KCs releases proinflammatory cytokines and free radicals to cause oxidative stress and liver fibrogenesis. 41 TNF-α is mainly derived from KCs after liver injury and during the wound-healing response. 42 TGF-β is induced and activated in various fibrotic diseases; suppressing TGF-β-induced macrophages attenuate the progression of hepatic fibrosis. 43 Hydroxyproline and α-SMA are both indicators for ECM in fibrotic tissue according to Lee and Friedman (2011). 44 We observed significantly higher hydroxyproline content and higher expression of α-SMA in the CCl4 group than in the control group. However, lower hydroxyproline content and lower expression of α-SMA were observed in the silymarin (200 mg/kg) and POE (0.5 and 1.0 g/kg) groups (Figure 8(A, B)). A crucial factor in liver fibrosis, CTGF is synthesized from hepatocytes and HSCs. 45 During liver fibrosis, CTGF acts as a hepatic fibrotic facilitator in the epithelial-to-mesenchymal transition, plays a key role in the increase of ECM synthesis, and is a critical downstream mediator of TGF-β on fibroblasts. In the present study, CTGF expression was upregulated in the CCl4 group, whereas it was significantly downregulated with silymarin (200 mg/kg) and POE (0.5 and 1.0 g/kg) treatment (Figure 8(C)). We also found that hepatic fibrosis in the CCl4 group was more severe than that in the silymarin (200 mg/kg) and POE (0.5 and 1.0 g/kg) groups according to the histological observation of Sirius red staining (Figure 4 and Table 2). We suggest that the antifibrotic effects of silymarin (200 mg/kg) and POE (0.5 and 1.0 g/kg) result from their capacity to reduce the formation of ECM.
Conclusions
The findings of the present study demonstrate that the ethanolic extract from the fruits of POE is effective in reducing liver inflammation and fibrosis induced by CCl4 in mice. Possible mechanisms are as follows: (1) treatment of hepatic injury through reduced serum levels of AST, ALT, and ALP; (2) inhibition of inflammation by decreasing the levels of TNF-α, IL-1β, IL-6, and COX-2; (3) upregulation of SOD, CAT, GPx, and GRd levels against lipid peroxidation; and (4) reduction of ECM through downregulation of TGF-β and CTGF. This study provides scientific evidence for the clinical use of POE extract for liver inflammation and fibrosis.
Footnotes
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human. Animal study was approved by the Committee on Animal Research, China Medical University (Number: 104–248).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Department of Chinese Medicine and Pharmacy, Ministry of Health and Welfare (MOHW104-CMAP-M-114-000423).
