Abstract
Cordyceps is a well-known and valuable fungal Chinese medicine and health food. The polysaccharides from
Liver is an important organ for biotransformation and detoxification of endogenous and exogenous harmful constituents. 1 The common causes of acute liver failure are the overdose of toxin ingestion, viral infection, metabolic disorder, immunological insult, and ischemic injury. 2 Furthermore, overdoses of free radicals are also considered as the cause of several chronic diseases. 3 Carbon tetrachloride (CCl4) is a well-known environmental biohazard and a common industrial solvent, which can particularly cause toxicity to the liver. The principle cause of CCl4-induced liver injury is lipid peroxidation which is induced by free radical derivatives of CCl4. Therefore, antioxidant processes play an important role in the protection of liver against CCl4-induced injury. 4
Cordyceps is a well-known and valuable fungal Chinese medicine and health food used as a tonic food for protection and improvement of kidney and lung functions and recuperation after illness.
5,6
There are over 400 described species distributed in 6 regions in various climatic areas and with a range of hosts (plants and insects).
7
The species are either endoparasitic on arthropods or parasitic on other fungi.
8
Cordyceps includes species assigned to genera like
Results and Discussions
ABTS and Hydroxyl Radical Scavenging Activities
Free radicals and reactive oxygen species (ROS) are considered as the direct or indirect causes of tissue damage and many human diseases, such as aging, cancer, atherosclerosis, and inflammation. The constituents of plants, fungi, and other sources have been reported to be useful for health protection and disease prevention. EPS from
As shown in Table 1, the free radical scavenging activities of EPS against ABTS• and hydroxyl radicals (∙OH) increased along with an increase in EPS concentration. The IC50 values of EPS for ABTS radical and in hydroxyl radical scavenging activities were 2.00 mg/mL and 3.78 mg/mL, respectively, whereas the respective IC50 values of vitamin C were about 0.03 mg/mL and 0.11 mg/mL (Table 1).
ABTS• and Hydroxyl Radical (∙OH) Scavenging Activity of EPS From

A study by Phuong-Tham Ho-Thi et al.
14
on antioxidant activities of polysaccharide extracts from several
Effects of EPS on Serum Activities of ALT and AST in CCl4-Intoxicated Mice
The hepatoprotective effects of EPS and the control on serum ALT and AST activities are shown in Figure 1. In the CCl4-induced liver damage groups, the activities of serum ALT and AST were 145.2 units/L and 57.5 units/L, respectively, while that of the control groups were 69.7 units/L and 40.8 units/L, respectively. These results indicate that CCl4 significantly increased the activities of both enzymes. It confirms thatCCL4 induced damage in rat liver, as previously reported by Jain et al. 17 and Campo et al., 18 who found that AST and ALT in serum of rats was significantly increased after administration of CCl4. The liver damage induced by CCl4 leads to changes in transport function and membrane permeability of hepatocytes, and thus enzymes in liver tissue, including ALT and AST, presented in cytoplasm are released into circulation. 19

Effects of EPS on serum AST (A) and ALT (B) activities in CCl4-intoxicated rats. Values are expressed as means ± SD(
The results in Figure 1 indicate that there were no significant differences in the activities of ALT and AST in serum between rats pretreated with EPS and the controls. However, the activities of these enzymes in serum of the model group were significantly different (
Effects of EPS on Levels of GSH and MDA in Liver Tissues
It has been reported that CCl4 is mainly metabolized to the highly reactive CCl3 and/or CCl3O2, which cause oxidative stress, weakening the antioxidant defense system and inducing lipid peroxidation, thereby leading to hepatotoxicity. 23,24 Therefore, to prevent liver damage caused by free radicals, one of the important pathways is to stimulate the antioxidant defense systems, including non-enzymatic and enzymatic antioxidants. 10 Hepatic MDA level represents the degree of membrane lipid peroxidation and oxidative damage in liver tissue and so a reduction in hepatic MDA formation can be used to identify the capability of the antioxidant defense system. 25
The effects of EPS on GSH and hepatic MDA levels in normal and model rats induced by CCl4 are shown in Figure 2. There were no significant differences in MDA levels among rats pretreated with EPS and silymarin in normal groups (Figure 2(A)). However, there was a remarkably increase in GSH contents (8.60 µM/g protein) in normal rats pretreated with EPS at 1.66 g/kg compared with that in rats pretreated with distilled water (7.17 µM/g protein) (Figure 2(B)). This suggests that EPS could stimulate the antioxidant system by non-enzymatic antioxidants in normal rats.

Effects of EPS on MDA (A) and GSH (B) contents in CCl4-intoxicated rats. Values are expressed as means ± SD (n = 6). Different letters (a–f) above the bars indicate significant differences at
In comparison with the normal group, the GSH content of the model group was significantly decreased after being induced by CCl4 (
Regarding hepatic MDA, the content of this compound was remarkably increased after rats were induced by CCl4 (
Effects of EPS on Histopathological Changes in Liver Tissue
The results of histopathological examination used for evaluating the hepatoprotective effects of EPS and the positive control on CCl4-induced acute liver damage are shown in Figure 3. As shown in Figure 3(A), the control slides show hepatic cells with well-preserved cytoplasm, prominent nucleus, and nucleolus and visible central veins. In contrast, the liver of the CCl4-intoxicated rats present extensive liver injuries, characterized by strong fatty changes in hepatocytes around the central vein, loss of cellular boundaries, focal necrosis, fibrosis and cytoplasmic vacuolization, as well as inflammatory cell infiltration (Figure 3(B)). However, the pretreatments with EPS and silymarin (positive control) significantly reduced the damage induced by CCl4(Figure 3(C, D, E)). This is in agreement with the decrease in the activities of serum aminotransferases (ALT and AST) (Figure 1) and the reduction in hepatic oxidative product levels (Figure 2(A)). The results further confirm the hepatoprotective effect of EPS from
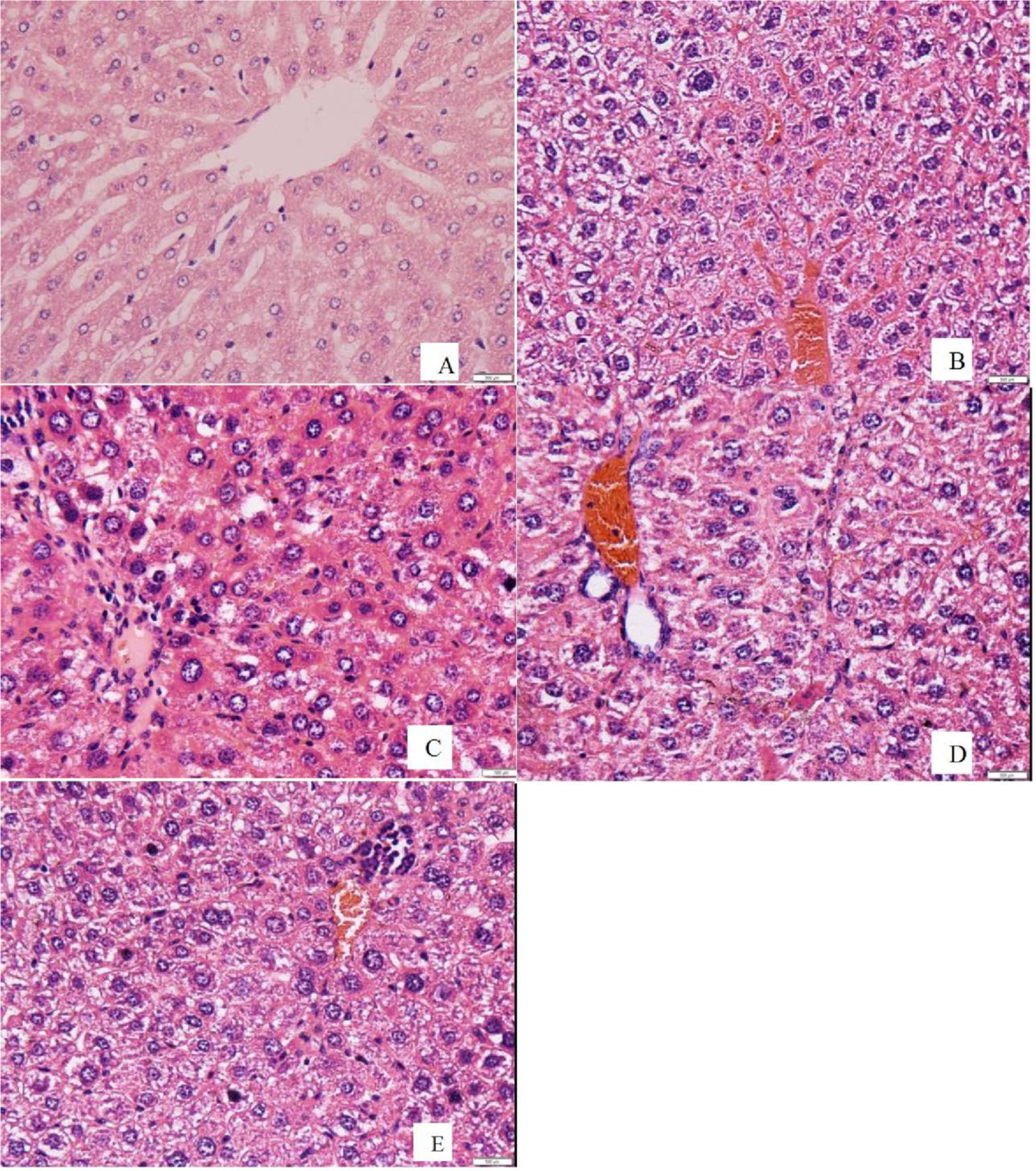
Effects of EPS on the liver histological damage after CCl4 treatment in rats. Animals were given orally either EPS (0.83 g/kg and 1.66 g/kg) or silymarin (0.1 g/kg) once daily for 8 consecutive days prior to the single administration of CCl4, and then a portion of the left lobe of the liver tissues, stained with hematoxylin and eosin (H&E), was used for histological assessment under a microscope. Representative photographs of liver sections stained with HE showing the pathological changes in hepatic tissues (400×): (A) control group, (B) CCl4-intoxicated group, (C) EPS (0.83 g/kg) and CCl4 group, (D) EPS (1.66 g/kg) and CCl4 group, (E) Silymarin (0.1 g/kg) and CCl4 group.
Conclusions
The findings of this study indicate that EPS from cultivated
Materials and Methods
Materials
ABTS (2,2'-azino-bis 3-ethylbenzothiazoline-6-sulfonic acid), hydrogen peroxide, TCA (trichloroacetic acid), and silymarin were purchased from Sigma Aldrich, Inc., USA; Elman [5,5’-dithiobis-(2-nitrobenzoic acid)], thiobarbituric acid (TBA), Coomassine Brilliant Blue, and Tris- HCL 0.2M, pH 7.4 from Merck Co., Germany; carbon tetrachloride- CCl4 from Chongqing Chuandong Chemical Co., Ltd.; and GPT (ALT) IFCC mod. liquid UV kit and GOT (AST) IFCC mod. liquid UV kit from Human GmbH, Germany. All other chemicals and solvents were of analytical grade.
ABTS Radical Cation Scavenging Activity
The ABTS radical cation scavenging activity was determined based on the method described by Shirwaikar et al., 31 with slight modifications. The ABTS+ radical solution was prepared by mixing 7 mM ABTS with 2.45 mM potassium persulfate and the mixture was stored in the dark place for 12 hours before use. Prior to use, the solution was diluted with ethanol to get an absorbance of 0.700 ± 0.025 at 734 nm. ABTS free radical scavenging activity was measured by mixing 10 µl of EPS with 1.0 ml of ABTS working standard solution in a microcuvette. The decrease in absorbance of the solution was measured exactly after 6 minutes at 734 nm. Ascorbic acid (Vitamin C) was used as the positive control.
The percentage inhibition was calculated according to the formula:

where A0 was the absorbance of the control, and A1 was the absorbance of the sample at 734 nm.
Hydroxyl Radical Scavenging Assay
The hydroxyl radical scavenging activity of the extract was assayed by the method described by Akashi et al. 32 The reaction mixture (3.0 ml) contained 1.0 ml of 1.5 mM FeSO4, 0.7 ml of 6 mM hydrogen peroxide, 0.3 ml of 20 mM sodium salicylate, and varied concentrations of EPS (1 ml). After incubation for 1 hours at 37 °C, the absorbance of the hydroxylated salicylate complex was measured at 562 nm. Ascorbic acid (Vitamin C) was used as the positive control. The scavenging activity of the hydroxyl radical effect was calculated as follows:

where A0 is the absorbance of the control (without extract), A1 is the absorbance of the solution in the presence of the sample, and A2 is the absorbance of the solution without sodium salicylate.
Animal
Adult male Wistar albino rats (27 ± 2 g) used for this study were supplied by Nha Trang Institute of Vaccine and Medical Biologicals, Khanh Hoa Province, Vietnam. They were kept in plastic cages housed in a room with a 12 hours light/12 hours dark cycle at 25 ± 2°C and fed with a standard rodent diet and water
Experimental Design
Adult male Wistar albino rats (27 ± 2 g) were fed for 1 week. After that, they were divided into 8 groups (6 rats in each group) including a normal control group (distilled water), model groups (EPS, 0.83 g/kg bw or 1.66 g/kg bw) and positive control groups, including normal rats and rats with CCl4-induced liver damage (silymarin, 0.1 g/kg bw). Rats in the normal and model groups were intragastrically administrated with distilled water, and those in the other 6 groups were administrated with silymarin and different concentrations of EPS, respectively, applying once a day for 8 successive days. On the first, third, and fifth days, acute liver injury was induced experimentally by injection with CCl4 diluted in olive oil (1:3 v/v) at a dose of 1.0 mL/kg bw, while the mice in the normal control group were injected with an equal amount of olive oil without CCl4.
29
All the rats were fasted for 16 hours, but water was supplied
Biochemical Testes in Serum
ALT and AST assays to measure the function of liver after CCl4-induced liver damage were determined by the method described in the technical manual of the commercially available diagnostic kits (Human, Germany).
Determination of MDA and GSH Levels
Levels of MDA and GSH in the liver homogenate were estimated using commercially available diagnostic kits. The level of MDA, a determination of lipid peroxidation, was measured by the thiobarbituric acid (TBA) reaction method. The non-protein liver GSH level was measured according to the instructions given with the detection kits.
Histopathological Examinations
A portion of the left lobe of the liver was preserved in 10% formalin solution for histopathological sections. The fixed tissues were embedded in paraffin; sections 3‐5 μm thick were obtained, deparaffinized, dehydrated in ethanol (50% to 100%), and cleared with xylene. The extent of CCl4-induced damage, including cell necrosis, steatosis, hyaline regeneration, and ballooning degeneration, was evaluated by assessing the morphological changes in liver sections stained with hematoxylin and eosin (H&E) under an Olympus BX50 light microscope. The data were analyzed by the image analysis system MetaMorph Offline (UIC/ OLYMPUS).
Footnotes
Statement of Human and Animal Rights
All experiment procedures involving animals were conducted in accordance with the Research Center of Ginseng and Medical Materials, Ho Chi Minh City; National Institute of medical materials, Ha Noi, Vietnam.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by The Department of Science and Technology and Center of Science and Technology Development in Ho Chi Minh City.
