Abstract
Introduction
Diabetes is a chronic metabolic disease caused by insulin resistance (IR) or/and insufficient insulin secretion, resulting in elevated blood glucose levels and macrovascular and microvascular complications, including cardiovascular disease, nephropathy, retinopathy, lipid disorders, neuropathy, and ketoacidosis.1,2 According to the latest data from the International Diabetes Federation, approximately 536.6 million adults (10.5%) worldwide had diabetes in 2021, and the total number is projected to increase to 643 million by 2030 and 783 million by 2045. 3 Type 2 diabetes (T2D), which is generally characterized by IR, is the most common type of diabetes, accounting for approximately 90% of all diabetes cases. 4 A healthy lifestyle, including a healthy diet, regular physical activity, not smoking, and maintaining a healthy body weight (BW), is the cornerstone of T2D management. 5 However, for many patients with T2D, pharmacological intervention is required to achieve and maintain glycemic control. Commonly used oral medications for T2D include metformin, sulfonylureas, thiazolidinediones, glinides, dipeptidyl peptidase-4 inhibitors, α-glycosidase inhibitors, glucagon-like peptide-1 receptor agonists, and sodium-glucose cotransporter-2 inhibitors. 6 Although these drugs are effective in lowering blood glucose and enhancing insulin sensitivity, they have adverse effects such as hypoglycemia, weight gain, allergic reactions, lactic acidosis, edema, gastrointestinal discomfort, anemia, cardiovascular risk, and liver and kidney dysfunction. 7 Therefore, there is an urgent need to develop milder and less toxic hypoglycemic drugs. Natural products are important resources for screening for bioactive agents against T2D. Natural products derived from medicinal plants, such as berberine, 8 andrographolide, 9 quercetin, 10 curcumin, 11 and naringin 12 have shown promising potential in the treatment of T2D.
Garcinia kola Heckel (family Guttiferae) seed, also known as bitter kola or false kola, is an edible nut originally found in lgbo, Nigeria. In folk medicine, bitter kola is recommended to control blood glucose levels and improve diabetes-related complications.13-16 Kolaviron, a biflavonoid complex from bitter kola containing Garcinia biflavonoid 1 (GB1), GB2, and kolaflavanone, has antidiabetic activity in alloxan-induced diabetic rabbits and streptozotocin-induced diabetic rats.17-19 GB1 (chemical structure shown in Figure 1A), one of the main components of kolaviron, exhibits antibacterial and antiplasmodial effects in vitro.20,21 Moreover, GB1 exhibits alpha-glucosidase and aromatase inhibitory activities. 21 We have previously reported that GB1 improves lipid metabolism in HepG2 human hepatoblastoma cell. 22 In the present study, db/db mice were used as a T2D model to investigate the effects of GB1 on blood glucose and lipid levels.
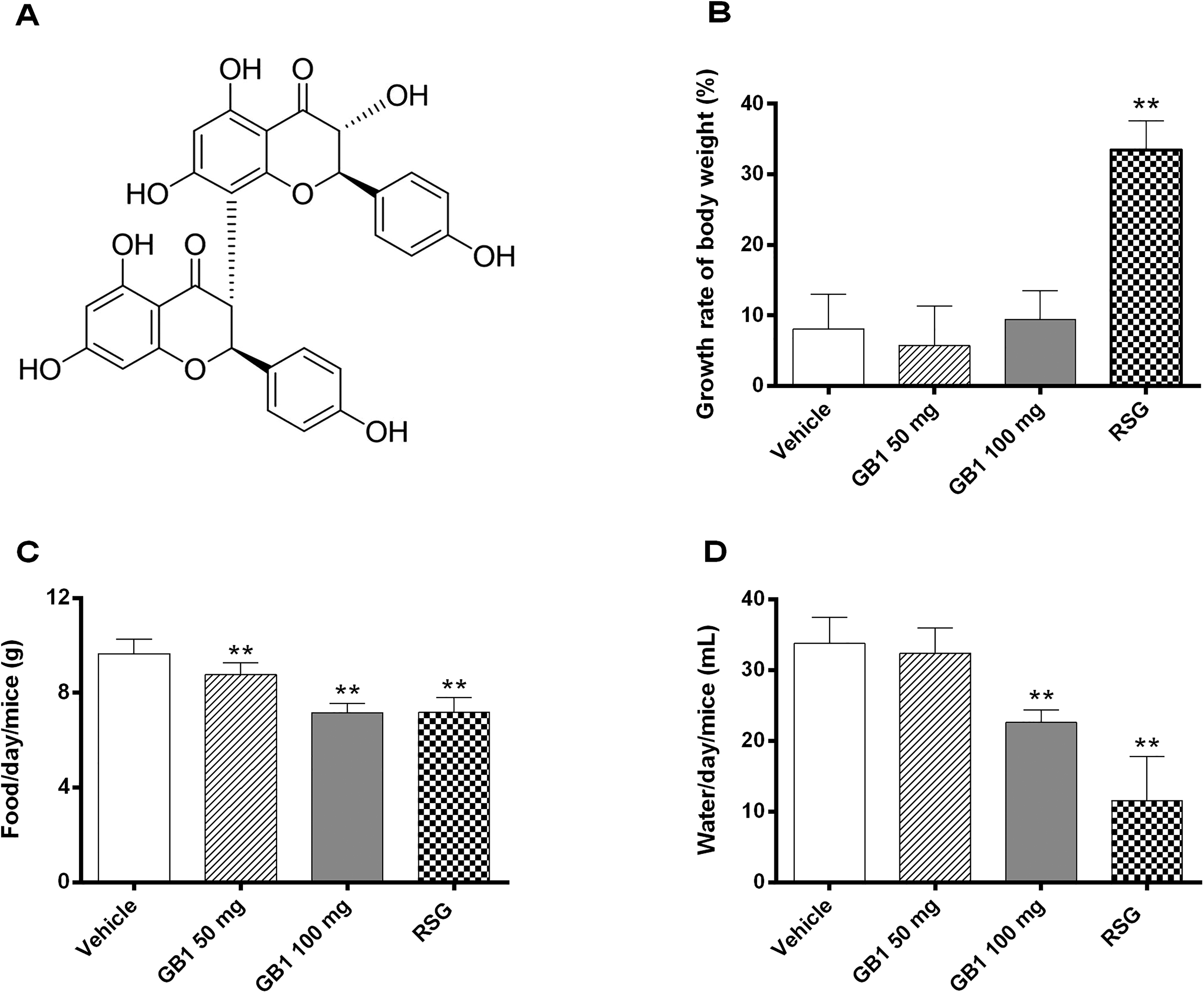
Effects of Garcinia biflavonoid 1 (GB1) on body weight, food, and water intake. (A) Structure of GB1. (B) Body weight changes. (C) Average daily food intake. (D) Average daily water intake. All data are presented as the mean ± standard deviation. *P < .05, **P < .01, significantly different from the vehicle group.
Results
Effects of GB1 on BW, food and water intake
There was no significant difference in the BW between the GB1 and vehicle groups. In contrast, the positive control drug, rosiglitazone (RSG), significantly induced weight gain in mice. Mice in the RSG group gained significant weight after 12 d of administration and were 33.5% heavier at the end of the experiment than at the start (Figure 1B). Compared to the vehicle group, mice administered with RSG and GB1 showed a marked decrease in their consumption of food and water (Figure 1C and D).
Effects of GB1 on Glycometabolism
Figure 2A shows dynamic changes in fasting blood glucose (FBG) levels at different points during the study period. In untreated db/db mice, FBG increased from 17.6 ± 4.0 mmol/L at the baseline to 34.1 ± 6.3 mmol/L at the end of the experiment. Compared with untreated mice, GB1 (100 mg/kg) significantly reduced FBG levels after 10 days of treatment but increased the levels of liver glycogen (Figure 2A and B). The area under the curve (AUC) values of the oral glucose tolerance test (OGTT) and insulin tolerance test (ITT) were significantly lower in the two GB1 groups than in the vehicle group, indicating that GB1 alleviated glucose intolerance and IR (Figure 2C-E). A slight, non-significant decline in fasting insulin (FINS) levels was observed in GB1-treated mice (Figure 2F). However, compared with the untreated group, the homeostasis model assessment of insulin resistance (HOMA-IR) was significantly lower, whereas the homeostasis model assessment of β-cell function (HOMA-β) and quantitative insulin sensitivity check index (QUICKI) were significantly higher in the high-dose GB1 group (Figure 2G and H).
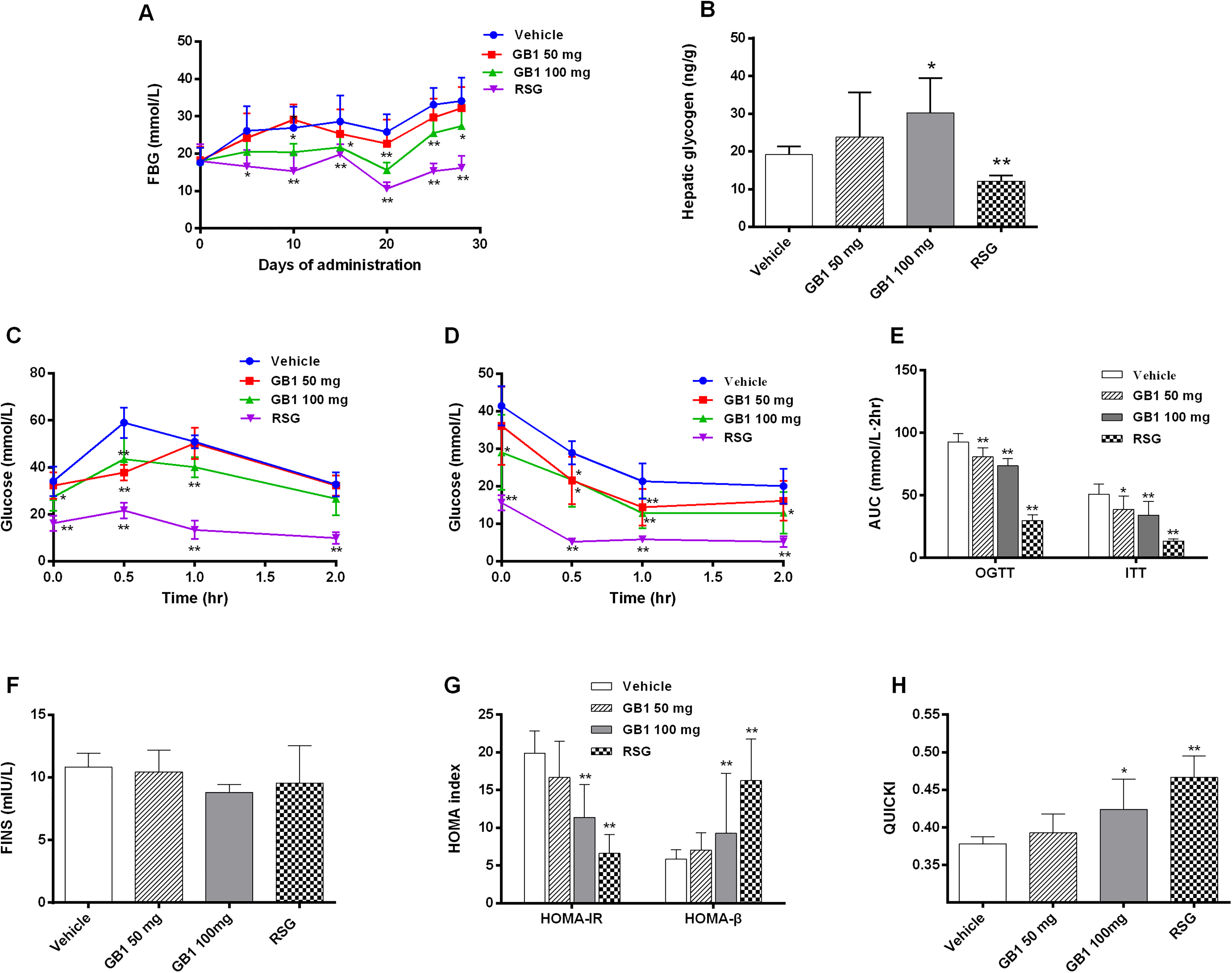
Effects of GB1 on glycometabolism. (A) FBG. (B) Hepatic glycogen. (C) Glucose curves of OGTT. (D) Glucose curves of ITT. (E) AUC of OGTT and ITT. (F) FINS. (G) HOMA index. (I) QUICKI. All data are presented as the mean ± standard deviation. *P < .05, **P < .01, significantly different from the vehicle group. Abbreviations: GB1, Garcinia biflavonoid 1; FBG, fasting blood glucose; OGTT, oral glucose tolerance test; ITT, insulin tolerance test; AUC: area under the curve; FINS, fasting insulin; HOMA, homeostasis model assessment; QUICKI, quantitative insulin sensitivity check index.
Effects of GB1 on Blood Lipids
Serum lipid parameters showed that GB1 reduced the triglyceride (TG) and free fatty acid (FFA) levels without affecting the total cholesterol (TC) levels. RSG decreased TG and FFA concentrations and increased TC levels (Figure 3A-C). The triglyceride-glucose index (TyG) index was reduced in both GB1 and RSG groups (Figure 3D).

Effects of GB1 on lipids levels. (A) TC. (B) TG. (C) FFA. (D) TyG index. All data are presented as the mean ± standard deviation. *P < .05, **P < .01, significantly different from the vehicle group. Abbreviations: GB1, Garcinia biflavonoid 1; FFA, free fatty acid; TC, total cholesterol; TGs, triglycerides; TyG, triglyceride-glucose index.
Effects of GB1 on Pancreas, Liver, Kidney, and Heart
Compared to untreated mice, there were no significant changes in alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels or in the weight of the pancreas, liver, kidneys, and heart in GB1-treated mice (Figure 4A to C). However, RSG increased the AST and ALT levels and liver weight (Figure 4A and B). Hematoxylin and eosin (H&E) staining showed no significant histopathological alterations in the pancreas, kidney, or heart tissues in the GB1 and RSG groups compared to the vehicle group (Figure 4D-F).
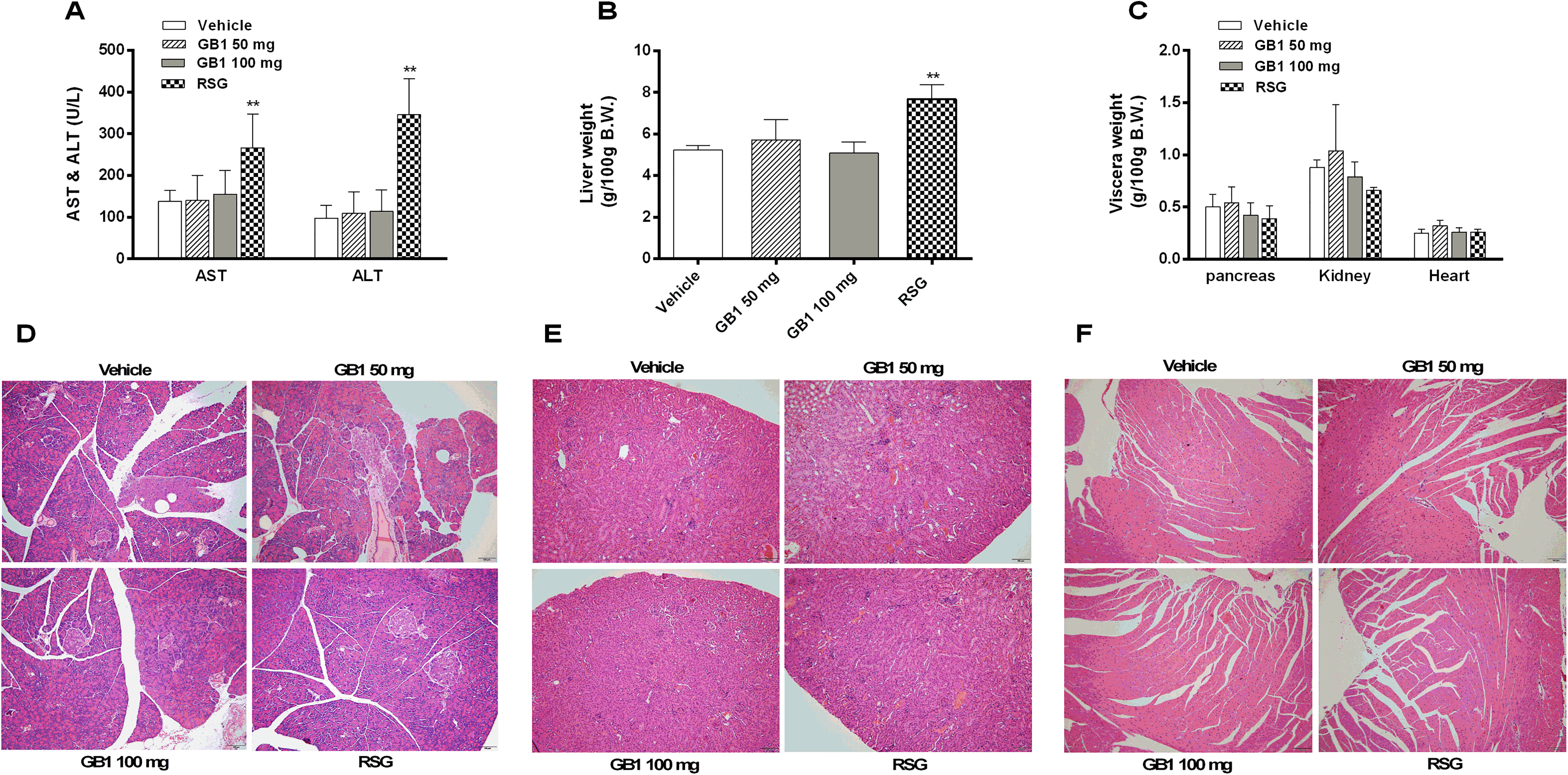
Effects of GB1 on pancreas, liver, kidney, and heart. (A) The levels of ALT and AST. (B) Liver weight. (C) Pancreas, kidney, and heart weight. (D) Representative H&E staining of pancreas sections. Scale bars: 100 μm. (E) Representative H&E staining of kidney sections. Scale bars: 100 μm. (F) Representative H&E staining of heart sections. Scale bars: 100 μm. All data are presented as the mean ± standard deviation. *P < .05, **P < .01, significantly different from the vehicle group. Abbreviations: GB1, Garcinia biflavonoid 1; ALT, alanine aminotransferase; AST, aspartate aminotransferase; H&E, hematoxylin and eosin.
Discussion
In our study, leptin receptor-deficient db/db mice developed hyperglycemia at 9 weeks of age with an average FBG of 17.6 mmol/L, reaching 34.1 mmol/L after 4 weeks without treatment. The FBG levels in the low-dose GB1 (50 mg/kg) group were similar to those in the untreated group on days 5 and 10 of administration and decreased slightly, but not significantly, after 15 days. After 10 days of administration until the end of the experiment, FBG levels in the high-dose GB1 (100 mg/kg) group were significantly lower than those in the control group. This indicates that GB1, particularly at high doses, alleviated hyperglycemia in db/db mice. We also observed a remarkable increase in the hepatic glycogen concentration after GB1 treatment. Hepatic glycogen synthesis plays an important role in the autoregulation of blood glucose. 23 GB1 promotes liver glycogen synthesis and may be conducive to blood glucose reduction. Furthermore, OGTT and ITT results showed that GB1 improved glucose tolerance and insulin response. Subsequently, we assessed HOMA-IR, HOMA-β, and QUICKI, the indirect parameters most commonly used in the treatment of T2D,24,25 and found that GB1 (100 mg/kg) significantly reduced HOMA-IR and increased HOMA-β and QUICKI, indicating that GB1 effectively improved insulin sensitivity and β-cell function. In summary, GB1 alleviated glucose metabolism disorders in db/db mice.
Dyslipidemia is another condition often associated with T2D. Our previous study revealed that GB1 ameliorated oleic acid- and palmitic acid-induced lipid deposition in HepG2 cells by reducing TG levels. 22 In the present study, GB1 significantly decreased the TG and FFA levels in db/db mice. Excess FFA levels are a risk factor for the development of T2D. First, FFA loading may suppress glucose metabolism according to the Randle cycle or the glucose-fatty acid cycle hypothesis.26,27 Moreover, FFAs induce IR via a variety of signaling mechanisms, such as disruption of phosphorylation, oxidative stress, and inflammatory signaling cascades.28-31 Therefore, a reduction in FFA levels contributes to insulin sensitization, glucose uptake, and metabolism.32,33 High TG levels significantly increase the risk of cardiovascular disease in patients with T2D. 34 Mounting evidence indicates that the TyG index, derived from the product of TG and FBG levels, is a valuable predictor of glycemic control and cardiovascular status.35,36 Our data showed that the TyG index was reduced after GB1 treatment, suggesting that GB1 not only affected glycemic control but also decreased the risk of cardiovascular disease by lowering blood lipid levels.
RSG, a peroxisome proliferator-activated receptor gamma agonist, acts as an insulin sensitizer in the treatment of T2D by activating the insulin pathway and regulating fatty acid metabolism.37,38 However, RSG is clinically associated with edema, weight gain, anemia, liver dysfunction (mildly to moderately reversible increase in aminotransferase), high TC and low-density lipoprotein cholesterol levels, osteoporosis, and an increased risk of heart failure.39-42 In our study, although RSG provided good glycemic control in db/db mice, it also led to weight gain and elevated ASL, ATL, and TC levels. GB1 had a modest hypoglycemic effect without the side effects caused by RSG.
In conclusion, this study demonstrated that GB1, a natural biflavonoid isolated from Garcinia kola seeds, exerts both hypoglycemic and hypolipidemic effects in db/db mice. Moreover, GB1 improved glucose tolerance and IR without any apparent adverse reactions. Thus, GB1 may serve as a potential dietary supplement or phytomedicine for the treatment of T2D through the regulation of glycolipid metabolism. However, further experiments using other T2D models are required to confirm their efficacy and safety.
Materials and Methods
GB1 Preparation
Garcinia kola nuts were obtained from Nigeria (Africa) in 2017 and identified by Professor Gang Hao of South China Agricultural University (Guangzhou, China). The voucher specimens were deposited at the Artemisinin Research Center, Guangzhou University of Chinese Medicine. GB1 was isolated as previously described 22 and identified using HPLC, 1H-NMR, and 13C-NMR. The yield and purity were determined to be approximately 1.0 and 99.6%, respectively.
Animals and Administration
Leptin receptor-deficient db/db mice with congenital obesity, hyperglycemia, hyperinsulinemia, and hyperlipidemia were used as T2D models. 43 Male db/db mice (8-week-old) were purchased from GemPharmatech Co., Ltd (Jiangsu province, China; license number: SCXK 2018-0008) and kept at 20 to 25 °C and 40% to 70% relative humidity with a 12-h light-dark cycle. After 1 week of adaptive feeding, db/db mice with FBG > 11.1 mmol/L were randomly divided into 4 groups (n = 9 per group): Vehicle (0.5% sodium carboxymethyl cellulose (CMC-Na) solution), low-dose GB1 (50 mg/kg), high-dose GB1 (100 mg/kg) and RSG (4 mg/kg). GB1 was dissolved in a 0.5% CMC-Na solution. RSG, an insulin sensitizer used for the treatment of T2D, was used as a positive control. 44 RSG sodium tablets were purchased from Taiji Group (Chongqing Fuling Pharmaceutical Factory Co., Ltd.). All drugs were administered by oral gavage once daily for 4 weeks. Animal experiments were conducted with the approval and supervision of the Experimental Animal Ethics Committee of Guangzhou University of Chinese Medicine (No. 20190902003). During the experiment, all the mice were in good health with normal food intake.
Detection of FBG and Hepatic Glycogen
BW was measured every 2 days, and food and water intake was recorded every 3 days. Blood samples were collected every 5 days from the tail vein to monitor blood glucose levels following a 12-h overnight fast. At the end of the experiment, liver tissue was separated and homogenized, and hepatic glycogen was detected using a liver/muscle glycogen assay kit (Cat No. A043-1-1; Nanjing Jiancheng Bioengineering Institute).
OGTT and ITT
The OGTT and ITT were performed on the day 1 and 2 after the last administration. After fasting for 12 h, the mice were administered 2 g/kg of glucose orally (OGTT) or 2 IU/kg insulin intraperitoneally (ITT). 45 Blood glucose levels were measured before (0 h) and after (0.5, 1, and 2 h) glucose/insulin administration, using an Accu-Chek Active Glucometer (Roche Diagnostics GmbH). The AUC of the blood glucose levels between baseline and 2 h was calculated using GraphPad Prism 6.01 (GraphPad Software).
Quantification of Serum Parameters
At the end of the experiment, the mice were fasted overnight, and anesthetized with 1% isoflurane, 46 and blood was collected from the aortaventralis. After centrifugation at 3000 r/min for 10 min, the supernatant was collected as serum and stored at −20 °C until analysis. The serum concentrations of TC, TG, ALT, and AST were measured using a HITACHI 7080 Automatic Biochemical Analyzer (Hitachi Ltd.). FFA and FINS levels were detected using enzyme-linked immunoassay kits (Cat No. MM0326M2 and MM0579M2; Jiangsu Meimian Co., Ltd.). The HOMA-IR and HOMA-β, QUICKI, and TyG index were calculated to evaluate insulin sensitivity and β-cell function, using the following formulas: HOMA-IR = FBG × FINS/22.5; HOMA-β = 20 × FINS/(FBG-3.5); QUICKI = 1/(lgFINS + lgFBG); TyG index = ln [fasting TG (mg/dL) × FBG (mg/dL)/2].35,45,47
Histopathological Examination
Mice were anesthetized with 5% isoflurane. Tissues, including the pancreas, liver, heart, and kidneys, were removed from the mice and weighed. Then the pancreas, heart and kidneys were fixed in 4% paraformaldehyde for 24 h, embedded in paraffin, and sliced into 4-μm sections as previously described. 45 After H&E staining, all sections were randomly evaluated under a light microscope (Olympus).
Statistical Analysis
SPSS software (version 24.0; SPSS, Inc.) was used for the statistical analysis. Results are expressed as the mean ± standard deviation. Comparisons between groups were analyzed via one-way analysis of variance with post hoc Tukey's test, and P < .05 was considered statistically significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Experimental Animal Ethics Committee of Guangzhou University of Chinese Medicine.
Funding
The author(s) disclosed receipt of the following financial support for the research: Natural Science Foundation of Guangdong Province (2018A030313731) and Traditional Chinese Medicine Bureau of Guangdong Province (2019 (43)).
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
This study does not contain any studies on human subjects. The standard Guidelines of the Ethical Committee for animals were followed.
