Abstract
Objective
Our study aimed to investigate the underlying molecular mechanisms of selin-11-en-4α-ol purified from Artemisia vulgaris against anti-inflammation through network pharmacology, molecular docking, and in vitro experiments.
Methods
Potential targets for selin-11-en-4α-ol and inflammation were obtained using Swiss Target Prediction, Pharm Mapper, and GeneCards databases. Venn diagrams were used to obtain the targets of selin-11-en-4α-ol for the treatment of inflammation, and the intersecting targets were uploaded to the STRING database to construct a protein–protein interaction (PPI) network of selin-11-en-4α-ol anti-inflammatory target proteins for further screening of the core targets. The Gene Ontology and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses were conducted using the DAVID database. In addition, molecular docking validation of core target binding to selin-11-en-4α-ol was performed. The anti-inflammatory effects were verified by in vitro experiments of lipopolysaccharide-induced RAW264.7 cells.
Results
PPI network analysis showed that mitogen-activated protein kinase 14 (MAPK14), SRC, and HSP90AA1 were predicted as potential anti-inflammatory targets, and KEGG enrichment analysis revealed that the MAPK signaling pathway presented the highest gene enrichment counts. Molecular docking and western blot results suggested that selin-11-en-4α-ol regulated MAPK and nuclear transcription factor-κB (NF-κB) signaling pathways to alleviate inflammation. In addition, selin-11-en-4α-ol inhibited the production of inflammatory factors, chemokines, and reactive oxygen species and exhibited anti-inflammatory and antioxidant effects.
Conclusion
Selin-11-en-4α-ol treats inflammation by targeting the MAPK and NF-κB signaling pathway, demonstrating that network pharmacology and molecular docking are effective tools for studying traditional Chinese medicine and providing a theoretical basis for developing anti-inflammatory drugs.
Keywords
Introduction
Inflammation is a comprehensive series of physiological responses of the organism to foreign pathogens and viruses, including acute and chronic inflammation. 1 A growing number of studies have shown that excessive inflammation is a predisposing factor for many diseases, such as asthma, cancer, cardiovascular disease, and rheumatoid arthritis, causing considerable health risks and economic burdens and seriously affecting people's quality of life.2-4 Macrophages are the main coordinators of the inflammatory response, and infiltration and aggregation of immune effector cells, such as macrophages, can be observed at the site of inflammation. 5 Lipopolysaccharide (LPS), a gram-negative endotoxin, can polarize macrophages into the M1 type by activating the downstream inhibitory kappa B kinase (IKK) kinase complex and MAP kinase family through binding to TLR4, releasing inflammatory factors including IL-1β, IL-6, TNF-α, NO, and other inflammatory factors to exert pro-inflammatory functions, which can aggravate tissue damage if the M1 phase is not maintained continuously.6,7
Traditional Chinese medicine has been used for thousands of years to treat inflammatory diseases and has been widely used worldwide as a preventive or therapeutic strategy in modern medicine due to its low side effects and significant efficacy. 8 Artemisia vulgaris, a genus of Artemisia in the family Asteraceae, contains volatile oils, terpenoids, organic acids, and flavonoids as active ingredients, which have hepatoprotective, antitussive, analgesic, antibacterial, warming, and hemostatic effects and are widely used in the treatment of various diseases, among which sesquiterpenoids are the most abundant in Avulgaris ingredients. 9 The molecular formula of selin-11-en-4α-ol is C15H26O and the chemical name is (1R,4aR,7R,8aR)-1,4a-dimethyl-7-prop-1-en-2-yl-2,3,4,5,6,7,8,8a-octahydronaphthalen-1-ol. In our research, selin-11-en-4α-ol is a sesquiterpene, which is a metabolite of A vulgaris. Selin-11-en-4α-ol has been found in the essential oils of Chrysanthemum officinale, Guava leaf, Cornus officinalis, and Artemisia annua.10-12 Previous studies found that selin-11-en-4α-ol was not significantly toxic to MCF-7, X-17, and other cells, 13 had strong antifungal activity, 14 and inhibited nitric oxide (NO) production in LPS-activated mouse peritoneal macrophages and the release of β-hexosaminidase from RBL-2H3 cells. 15 Anti-inflammatory studies of selin-11-en-4α-ol are limited, and the underlying molecular mechanisms remain unclear.
Network pharmacology is based on systems biology, genomics, proteomics, and other multidisciplinary theories, combined with high-throughput genomics and bioinformatics technologies to screen drug and disease target information to reveal the molecular mechanism of Chinese medicine treatment of disease.16,17 Nowadays, more and more reports about the application of network pharmacology in monomeric compounds are appearing. Xue et al explored the molecular mechanism of berberine in improving premature ovarian failure by network pharmacology approach and performed experimental validation, and Liang et al elucidated the mechanism of lignocaine in treating osteoporosis by network pharmacology approach, which provided scientific support for rational clinical use.18,19 Molecular docking is an effective tool to study the degree of affinity between small molecule ligands and protein receptors, which can effectively predict the binding activity and model between them, and has been widely used in the study of pharmacological mechanisms of drug treatment diseases. 20 The combined application of network pharmacology and molecular docking techniques could better elucidate the anti-inflammatory effects and potential molecular mechanisms of selin-11-en-4α-ol, for which, to our knowledge, no relevant reports are available.
Our study predicted the potential anti-inflammatory targets of selin-11-en-4α-ol, cellular signaling pathways that may be involved in regulation, and potential pharmacological mechanisms through network pharmacological analysis. The binding mode and activity of selin-11-en-4α-ol and key targets were predicted by molecular docking, and in vitro experiments were conducted by LPS-induced inflammation model of RAW264.7 cells to verify the potential anti-inflammatory mechanism of selin-11-en-4α-ol, providing a scientific basis for drug development and clinical treatment. The workflow diagram is shown in Figure 1.

Workflow of selin-11-en-4α-ol anti-inflammatory.
Materials and Methods
Extraction and Isolation of Selin-11-en-4α-ol
In May 2018, fresh leaves of A vulgaris were collected and air-dried in Tangyin, Henan, China. 2 kg air-dried leaves were extracted 3 times (18 L each time) with 95% ethanol, combined and concentrated to obtain 210 g of crude extracts. The crude extract was suspended in 1.3 L of warm water and sequentially extracted 3 times with petroleum ether (1.3 L) and ethyl acetate (1.3 L), obtained petroleum ether fraction (45 g) and the ethyl acetate fraction (47 g). The petroleum ether fraction (45 g) was subjected to an MCI column (50 × 4.5 cm) and sequentially eluted with 40%-100% aqueous methanol solution to obtain 7 subfractions (Fr. A-G). The Fr.D (5.1 g) was subjected to Sephadex LH-20 (110 × 2 cm) eluted with methanol to obtain 5 subfractions (Fr.D.1-Fr.D.5). The fraction of Fr.D.3 (3.8 g) was subjected to ODS column chromatography (10 × 4 cm) eluted with 40%-60% aqueous methanol solution to give 7 subfractions (Fr.D.3.1∼Fr. D.3.7). The Fr.D.3.6 (1.9 g) was subjected to silica gel column (6 × 4 cm 200-300 mesh) eluted with PE/EtOAc 95:5 to 0:100 and obtained 9 subfractions (Fr. D.3.6.1-Fr. D.3.6.9), and the Fr.D.3.6.3 (900 mg) was recrystallized to obtain the compound (385 mg), accounted for 0.193 ‰ of the total dry weight. Subsequently, the compound was dried and dissolved in deuterated chloroform, and the 1H NMR(600 MHz), 13C NMR (125 MHz), and two-dimensional (2D) NMR spectra were obtained on a Bruker AVANCE-600.
Prediction of Potential Targets for Selin-11-en-4α-ol and Inflammation
The chemical structure of selin-11-en-4α-ol (PubChem CID: 15560330) was taken from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/), the Swiss Target Prediction database (http://www.swisstargetprediction.ch/) and the Pharm Mapper database (http://www.lilab-ec.ust.cn/pharmmapper/) were used to predict the potential targets of selin-11-en-4α-ol. The UniProt database (http://www.uniprot.org/) was used to normalize the protein names. The species was selected as “homo sapiens” to obtain the predicted target of selin-11-en-4α-ol. Inflammation-related targets were screened by the GeneCards database (https://www.genecards.org/), and selin-11-en-4α-ol targets were intersected with inflammatory targets using “Venny” to obtain selin-11-en-4α-ol anti-inflammatory target, which was used in the follow-up study.
Protein–Protein Interaction Network
Anti-inflammatory targets of selin-11-en-4α-ol were imported into the STRING database (https://cn.string-db.org/) with the protein species “homo sapiens,” and a minimum confidence level of 0.700 and a single node in the network was hidden to obtain a protein-protein interaction (PPI) network. Optimization and analysis of PPI networks using Cytoscape v3.7.1 software.
Gene Ontology and Kyoto Encyclopedia of Genes and Genomes Pathway Enrichment Analysis
To explain the role of the above targets in gene function and the signaling pathways enriched, the above target proteins were imported into the DAVID database (https://david.ncifcrf.gov/) for gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis, and functional terms and pathways with P < .05 were considered significant.
Molecular Docking
The target proteins mitogen-activated protein kinase 14 (MAPK14), SRC, and HSP90AA1 with high degree values in the PPI network were selected for molecular docking, and the target protein structures with relatively small Å values were selected from the UniProt database. Combine AutoDock tools 1.5.6 with Vina for molecular docking. The docking box parameters were taken from AutoDock tools 1.5.6, and the docking was conducted by Vina. 2D model presentation of the docking model via Pymol and Discovery StudioVisualizer 2021 software.
Cell Culture
The murine macrophage cell line, RAW264.7, was purchased from the Typical Culture Collection, added to DMEM medium containing 10% by volume of Australian fetal bovine serum and 1% penicillin-streptomycin solution, and incubated at 37 °C in a 5% CO2 incubator.
Cell Viability Assay
Cell viability was determined by the CCK8 method, and the cell suspension was diluted to a density of 2 × 105 cells/mL after cell passaging and inoculated on 96-well plates at 100 μL per well. The selin-11-en-4α-ol was diluted two-fold to 7 different concentrations (200-3.125 μM), and 100 μL per well was added to 96-well plates while blank control and normal cell groups were set up. After 24 h of treatment, the old culture medium was discarded, and DMEM medium containing 10% CCK8 was added to each well and incubated for 1 h away from light. The absorbance was measured at 450 nm using a spectrophotometer (BioTek Epoch 2).
NO Measurement
Griess reagent (Beyotime) was used to determine nitrite (NO2−) levels in RAW264.7 cell culture supernatant, and cells were inoculated at 5 × 105 cells/mL in 6-well culture plates. After overnight incubation, cells were treated with 10 μM of dexamethasone (DEX; Tianjin KingYork Group Hubei Tianyao Pharmaceutical Co., Ltd) as well as different concentrations of selin-11-en-4α-ol for 4 h and then in the presence or absence of 200 ng/mL LPS (Sigma-Aldrich) for 20 h. The supernatant was mixed with an equal volume of Griess reagent, and the absorbance of selin-11-en-4α-ol was measured at 570 nm using a spectrophotometer (BioTek Epoch 2). Three replicate wells were set up for each sample group to ensure the experimental data's validity.
Enzyme-linked Immunosorbent Assay
The production of IL-6 and TNF-α in RAW264.7 cell culture supernatant was measured by enzyme-linked immunosorbent assay (ELISA), and the cells were inoculated at 5 × 105 cells/mL in 6-well culture plates. After overnight incubation, cells were treated with 10 μM of DEX as well as different concentrations of selin-11-en-4α-ol for 4 h and then in the presence or absence of LPS (200 ng/mL) for 20 h. The levels of IL-6 and TNF-α in cell supernatants were determined using an ELISA kit (Multisciences) and the manufacturer's instructions. Three replicate wells were set up for each sample group to ensure the experimental data's validity.
Real-Time Quantitative PCR
To examine the effect of selin-11-en-4α-ol on the expression of inflammatory and chemokines in LPS-stimulated RAW264.7 cells, RAW264.7 cells were inoculated on 6-well plates (8 × 105cells/mL) preincubated overnight and treated with DEX (10 μM) as well as different concentrations of selin-11-en-4α-ol for 4 h, followed by treatment of cells in the presence or absence of LPS (200 ng/mL) for 6 h. Total RNA was extracted using TRIzol reagent (Thermo Fisher Scientific) according to the manufacturer's protocol and reverse transcribed with oligo-dT using PrimeScript™ RT kit (Takara Bio Inc.). RT-qPCR was performed by monitoring the increase in SYBR Green fluorescence using the ViiA 7 Real-Time PCR System (Thermo Fisher Scientific). The expression of GAPDH was used as an internal control to normalize all target genes. The data were analyzed using the ΔΔCT method. The primer sequences used in this work are shown in Table 1, and 3 replicate wells were set up for each set of samples to ensure the validity of the experimental data.
Primer Sequences.

Detection of Reactive Oxygen Species
Intracellular reactive oxygen species (ROS) levels in RAW264.7 cells were determined by the ROS Assay Kit (Beyotime), and RAW264.7 cells were inoculated on 6-well plates (5 × 105 cells/mL) and preincubated overnight with DEX (10 μM) as well as various concentrations of selin-11-en-4α- ol for 4 h, followed by treatment for 20 h in the presence or absence of LPS (200 ng/mL). After washing with PBS, 2', 7'-dichloro fluoro resin diacetate (DCFH-DA) was added and incubated for 50 min at 37 °C protected from light. Cells were washed 3 times with PBS and visualized and photographed by an ECLIPSE Ti-DH inverted fluorescence microscope (Nikon). ImageJ 1.53c software (National Institutes of Health) was used to analyze the fluorescence intensity.
Western Blot
RAW264.7 cells were inoculated on 6-well plates (8 × 105 cells/mL) and preincubated overnight, treated with DEX (10 μM) as well as different concentrations of selin-11-en-4α-ol for 4 h, and then treated for 20 h in the presence or absence of LPS (200 ng/mL). The cell extracts were washed twice with PBS, RIPA lysate containing protease inhibitor and phosphatase inhibitor (1:1:50) was added, incubated on ice for 15 min, and the supernatant was collected by centrifugation (12 000 × g) at 4 °C for 10 min to obtain cellular proteins. Protein concentration was determined by BCA Protein Assay Kit (Beyotime), and the protein concentration of each group was made uniform by adding the corresponding volume of RIPA, 5 × loading buffer, and denatured by heating for 15 min at 100 °C in a metal bath heater. The sodium dodecyl sulfate-polyacrylamide gel electrophoresis loading buffer separated equal amounts of protein samples and transferred them to nitrocellulose membranes (PVDF) at 70 V for 90 min at 4 °C. After blocking with 5% bovine serum albumin (BSA, Amresco) for 2 h at room temperature, the membranes were washed 3 times with tris-buffered saline tween-20 (TBST) to remove the blocking solution, and then the membranes were treated with 1:1000 primary antibodies against inducible nitric oxide synthase (iNOS), COX-2, p-ERK1/2, ERK1/2, p-JNK1/2, JNK1/2, p-p38, p38, p-IKKβ, IKKβ, p-IκBα, IκBα, p-p65, and p65 primary antibodies (Cell Signaling Technology) and incubated the membranes overnight at 4 °C. The corresponding primary antibodies were recovered, and the membranes were washed 3 times with TBST to remove residual primary antibodies and then blotted with the respective horseradish peroxidase-conjugated secondary antimouse or antirabbit antibodies. Specific proteins were detected using an enhanced chemiluminescence assay kit (Thermo Fisher Scientific) according to the manufacturer's instructions, and the membranes were scanned and visualized using a chemiluminescence imaging system (Tanon).
Statistical Analysis
All data are expressed as mean ± standard deviation (SD, n = 3). Graphs were created using GraphPad Prism 8 (San Diego) to analyze all the data statistically. One-way analysis of variance was used to analyze differences between experimental groups, and the P-values < .05 were considered statistically significant.
Results
Structural Identification of Selin-11-en-4α-ol
The compound (Figure 2) was obtained as a white powder, and structural identification using spectroscopy techniques, 1H NMR (δH, CDCl3, 600 MHz): 4.71 (1H, br.s, H-12), 4.69 (1H, br.s, H-12), 1.95 (1H, m, H-7), 1.85 (1H, m, H-6), 1.78 (1H, m, H-1), 1.75 (3H, s, H-15),1.25 (1H, m, H-5),1.12 (3H, s, H-13), 0.89 (3H, s, H-14). 13C NMR (δC, 150 MHz, CDCl3):150.8 (C-11), 108.3 (C-12),72.4 (C-4), 55.0 (C-5), 46.5 (C-7), 44.8 (C-3), 43.5 (C-1), 41.2 (C-9), 34.7 (C-10), 27.0 (C-8), 26.2 (C-6), 22.8 (C-13), 21.2 (C-15), 20.3 (C-2), 18.8 (C-14).
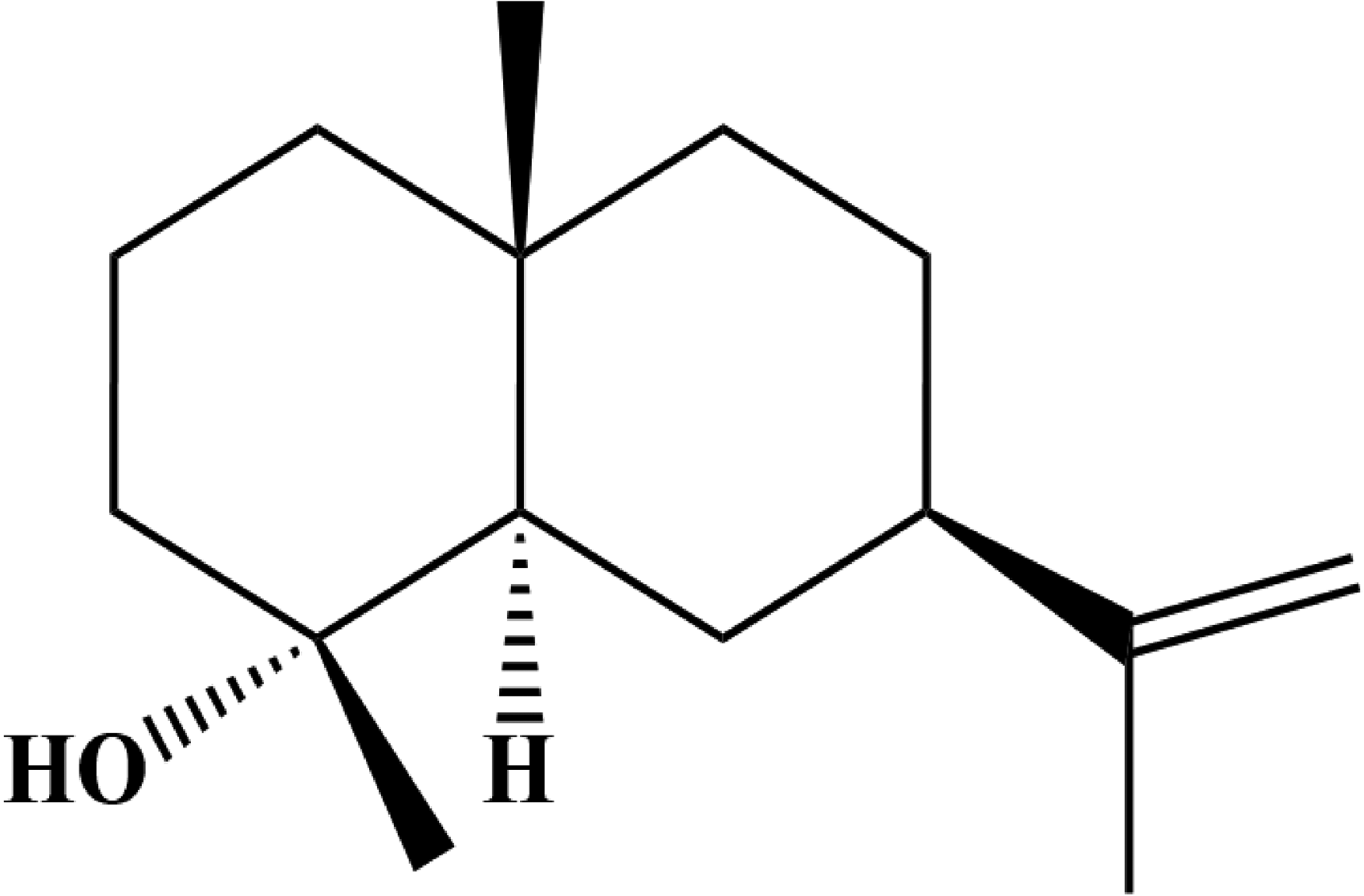
Chemical structure of selin-11-en-4α-ol.
Target Prediction and Drug-Disease Target Composite Network Construction
In total, 16 and 99 targets of selin-11-en-4α-ol were obtained from the Swiss Target Prediction database and Pharm Mapper database, respectively, and 112 targets associated with selin-11-en-4α-ol were obtained after removing duplicates (Figure 3A). Based on the correlation score and using the median method, 1364 inflammatory targets were screened from the GeneCards database (Figure 3B). Fifty-one targets, including MAPK14, are considered potential targets for selin-11-en-4α-ol in treating inflammation (Figure 3C). The above targets were imported into Cytoscape_v3.7.1 software to construct the selin-11-en-4α-ol-target-inflammation network, which consisted of 53 nodes and 102 edges (Figure 3D).
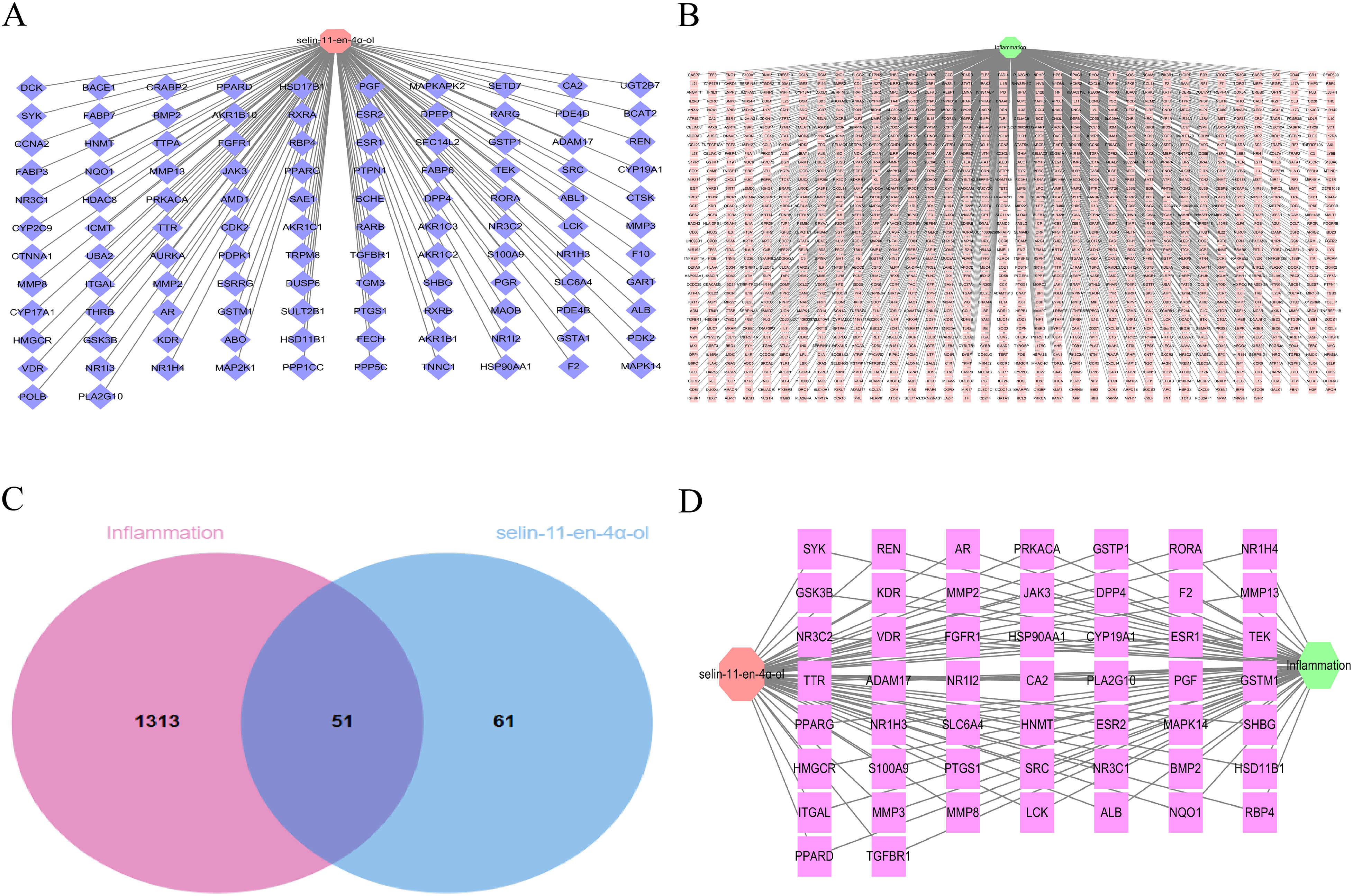
Selin-11-en-4α-ol and the targets of inflammation. (A) Target network of selin-11-en-4α-ol. The orange-red octagonal node represents selin-11-en-4α-ol, and the dark blue diamond nodes represent the predicted targets of selin-11-en-4α-ol. (B) Target network of inflammation. The light green hexagonal node represents inflammation, and the pink square nodes represent the predicted inflammation targets. (C) Venn diagram of selin-11-en-4α-ol and inflammation. The red circle represents the predicted target of inflammation, and the light blue circle represents the target of selin-11-en-4α-ol. (D) selin-11-en-4α-ol-target-inflammatory network. The orange-red octagonal node represents selin-11-en-4α-ol, the light green hexagonal node represents inflammation, and the red squares represent potential targets of selin-11-en-4α-ol for treating inflammation.
PPI Network Analysis
The obtained 51 common targets were uploaded to the online STRING 11.0 database to obtain a PPI network consisting of 40 targets (Figure 4A), and Cytoscape was used to optimize the PPI network, with darker node colors representing higher degree values and higher importance of nodes in the network (Figure 4B). Targets with high, moderate values in the PPI network included SRC, HSP90AA1, MAPK14, AR, and ESR1, suggesting that these could be effective targets of selin-11-en-4α-ol for treating inflammation.

Protein–protein interaction network. (A) PPI network derived from the STRING database (high confidence: >0.700). (B) Cytoscape optimized PPI network. The shade of the circle represents the size of the corresponding degree value, with the redder the color representing the more important the target in selin-11-en-4α-ol for the treatment of inflammation.
GO and KEGG Pathway Enrichment Analysis
The 51 intersecting targets were entered into the DAVID database for further analysis. The GO enrichment analysis included 3 aspects of biological processes (BPs), cellular components (CCs), and molecular functions (MFs), and a total of 220 GO entries were obtained (P < .05), of which 147, 22, and 51 entries were retrieved for BP, CC, and MF, accounting for 67%, 10%, and 23%, respectively (Figure 5A). The top 5 BP terms were: Intracellular steroid hormone receptor signaling pathway, intracellular receptor signaling pathway, positive regulation of gene expression, negative regulation of inflammatory response, and response to estrogen (Figure 5B). The top 5 CC terms were: Extracellular region, receptor complex, membrane raft, chromatin, and extracellular space (Figure 5C). The top 5 MF terms were: RNA polymerase II transcription factor activity, ligand-activated sequence-specific DNA binding, zinc ion binding, steroid binding, sequence-specific DNA binding, and protein tyrosine kinase activity (Figure 5D). KEGG signaling pathway analysis identified 16 related signaling pathways, including MAPK signaling pathway, lipid and atherosclerosis, Ras signaling pathway, Bile secretion, and hepatocellular carcinoma (Figure 5E). Among them, the MAPK signaling pathway may be the most effective in the anti-inflammatory process of selin-11-en-4α-ol. In addition, Figure 5F shows a selin-11-en-4α-ol-target-pathway network consisting of 47 nodes and 73 edges.

Go and KEGG enrichment analysis of selin-11-en-4α-ol therapeutic targets for inflammation. (A) Values of BP, CC, and MF entries. (B) GO-enriched entries for biological processes (top 16). (C) The molecular function of GO-enriched entries (top 16). (D) GO-enriched entries of cellular components (top 16). (E) KEGG pathway analysis for enrichment and gene counting. (F) selin-11-en-4α-ol-target-pathway network. The red V-nodes represent pathways, and the green rectangular nodes represent predicted targets of selin-11-en-4α-ol in inflammation therapy. Abbreviations: GO, gene ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes; BP, biological process; CC, cellular component; MF, molecular function.
Molecular Docking
The binding energy calculated by molecular docking can reflect the degree of affinity between the ligand and the receptor. It is usually considered that if the binding energy is less than −5.0 kcal/mol, it indicates that the active ingredient has good binding activity with the target protein, and if the binding energy is less than −7.0 kcal/mol, it indicates that both have strong binding activity. The binding energies of selin-11-en-4α-ol to SRC (PDB ID 1A1B), HSP90AA1 (PDB ID 1BYQ), and MAPK14 (PDB ID 1A9U) were −5.3, −5.0, and −6.3 kcal/mol, respectively, as calculated by AutoDock and Vina software, while the binding energies of DEX to SRC, HSP90AA1, and MAPK14 were −7.2, −7.1, and −7.5 kcal/mol, indicating that DEX and the corresponding target proteins showed stronger binding activity. The interactions were visualized using Discovery Studio Visualizer 2021 and Pymol (three-dimensional [3D]) (Figure 6A-F). Among them, the binding affinity of selin-11-en-4α-ol to MAPK14 was contributed by: Hydrophobic interactions with residues VAL38, LYS53, ALA51, LEU167, LEU104, ILE84, and LEU75; and interactions with residues ASP168, MET109, GLU71, and THR106 via van der Waals forces. The results showed that selin-11-en-4α-ol had good binding activity to MAPK14, consistent with the prediction of PPI interaction analysis, suggesting that MAPK14 may play an important role in treating inflammation by selin-11-en-4α-ol.

Molecular docking model. (A) Binding mode of selin-11-en-4α-ol to SRC. (B) Binding mode of selin-11-en-4α-ol to HSP90AA1. (C) Binding mode of selin-11-en-4α-ol to MAPK14. (D) Binding mode of DEX to SRC. (E) Binding mode of DEX to HSP90AA1 . (F) Binding mode of DEX to MAPK14.
Selin-11-en-4α-ol Inhibits LPS-Induced Secretion of TNF-α and IL-6 in RAW264.7 Cells
Selin-11-en-4α-ol was assayed for cytotoxicity to RAW264.7 using the CCK8 assay. No significant cytotoxicity was observed in the series of concentrations from 3.125 to 100 μM, and 25 μM (low dose), 50 μM (medium dose), and 100 μM (high dose) were subsequently used as the therapeutic doses for our study(Figure 7A). To investigate the effect of selin-11-en-4α-ol on the secretion of TNF-α and IL-6 in LPS-induced RAW264.7 cells, TNF-α and IL-6 in RAW264.7 cell culture supernatants were quantified. As shown in Figure 7B-C, the levels of TNF-α and IL-6 were significantly higher in the LPS group compared with the control group (P < .001), and pretreatment with DEX (10 μM) and different doses of selin-11-en-4α-ol inhibited the secretion of TNF-α and IL-6. 50 μM and 100 μM of selin-11-en-4α-ol significantly inhibited the secretion of TNF-α, and 25, 50, and 100 μM of selin-11-en-4α-ol significantly inhibited the secretion of IL-6.
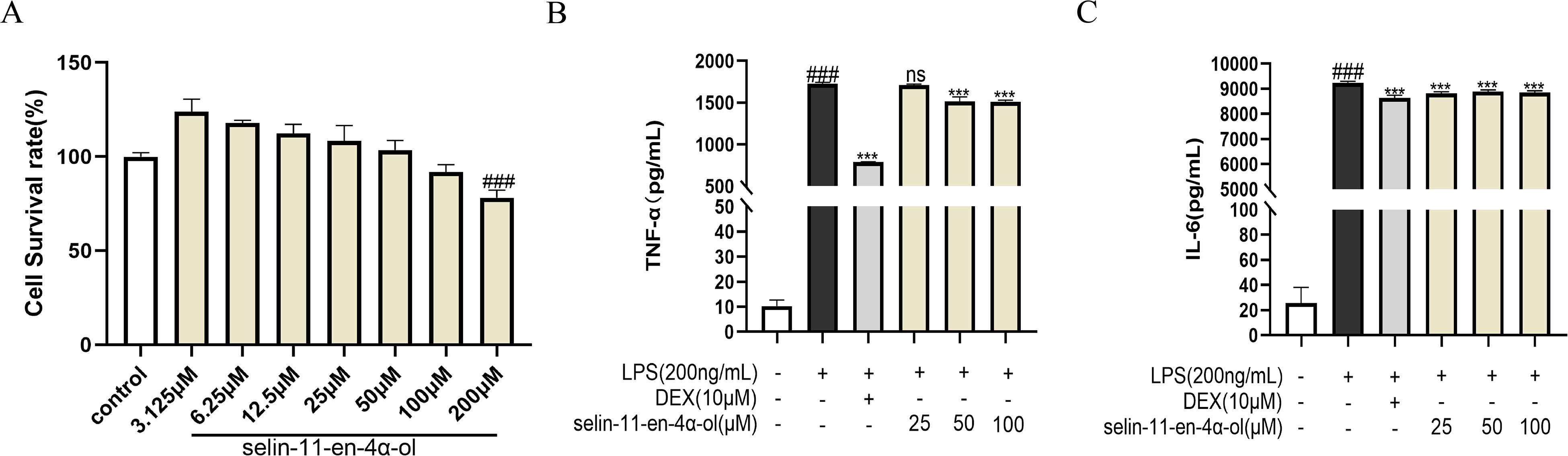
The effect of selin-11-en-4α-ol on TNF-α and IL-6 secretion in lipopolysaccharide (LPS)-stimulated RAW264.7 cells. (A) Cell viability as determined by the CCK8. (B) Effect of TNF-α expression. (C) Effect of IL-6 expression. Results are expressed as mean ± SD (n = 3). ###P < .001 versus control group; *P < .05, **P < .01, ***P < .001 versus LPS group.
Selin-11-en-4α-ol Inhibits Inflammatory Cytokine and Chemokine Expression
The effects of selin-11-en-4α-ol on LPS-induced pro-inflammatory cytokines and chemokines were examined by RT-qPCR at the mRNA level. LPS stimulation upregulated the mRNA levels of TNF-α, IL-6, IL-1β, MIP-1α, and MCP-1, and pretreatment with DEX (10 μM) and different concentrations of selin-11-en-4α-ol significantly inhibited the mRNA expression levels of inflammatory factors and chemokines compared with the model group (Figure 8A-E), tentatively indicating that selin-11-en-4α-ol could inhibit the LPS-induced inflammatory response.

Effect of selin-11-en-4α-ol on inflammatory and chemokine expression in lipopolysaccharide (LPS)-stimulated RAW264.7 cells. (A) TNF-α, (B) IL-6, (C) IL-1β, (D) MIP-1α, (E) MCP-1. Results are expressed as mean ± SD (n = 3). ###P < .001 versus control group; *P < .05, **P < .01, ***P < .001 versus LPS group.
Selin-11-en-4α-ol Inhibits NO, iNOS, and COX-2 Expression
The effects of selin-11-en-4α-ol on the expression of NO, iNOS, and COX-2 were investigated by RT-qPCR, ELISA, and Western blot at the level of gene transcription and protein secretion. Western blot results revealed that preadministration of DEX and high doses of selin-11-en-4α-ol inhibited the protein expression of iNOS, COX-2 (Figure 9A). Nitrite (NO2−) levels in cell supernatants were measured by Griess reagent, and pretreatment with DEX and selin-11-en-4α-ol at 50 and 100 μM significantly inhibited NO secretion compared to the LPS group, while low doses of the drug selin-11-en-4α-ol did not significantly differ on NO secretion (Figure 9B). At the transcriptional level, 25, 50 and 100 μM of selin-11-en-4α-ol significantly inhibited the mRNA expression of iNOS, COX-2 compared with the model group (Figure 9C-D).

Effect of selin-11-en-4α-ol on the expression of NO, iNOS, and COX-2 inflammatory mediators. (A) Representative immunoblot images of iNOS and COX-2. (B) NO secretion. (C) mRNA levels of iNOS. (D) mRNA levels of COX-2. Results are expressed as mean ± SD (n = 3). ###P < .001 versus control group; *P < .05, **P < .01, ***P < .001 versus LPS group.
Selin-11-en-4α-ol Inhibits LPS-Induced ROS Production in RAW264.7 Cells
DCFH-DA was performed to evaluate the intracellular level of ROS. As shown in Figure 10, LPS treatment increased ROS production, and pretreatment with selin-11-en-4α-ol (25, 50, 100 μM) and DEX (10 μM) inhibited the accumulation of intracellular ROS, indicating that selin-11-en-4α-ol had a mitigating effect on oxidative stress.

Selin-11-en-4α-ol pretreatment reduced the production of ROS. (A) Fluorescence microscopy showing intracellular ROS production (scale bar: 50 μm). (B) Relative fluorescence intensity was analyzed by ImageJ software to assess the level of ROS. Values indicate mean ± standard deviation (n = 3), ###P < .001 versus control group; ***P < .001 versus LPS group.

Effect of selin-11-en-4α-ol on LPS-induced MAPK/NF-κB pathway protein expression in RAW264.7. (A) Representative immunoblot images of p-ERK1/2, ERK1/2, p-JNK1/2, JNK1/2, p-p38, and p38. (B) Representative immunoblot images of p-IKKβ, IKKβ, p-IκBα, IκBα, p-p65, and p65.

Schematic diagram of the anti-inflammatory mechanism of selin-11-en-4α-ol.
Selin-11-en-4α-ol Inhibits LPS-Induced Protein Expression of MAPK/Nuclear Transcription Factor-κB Signaling Pathway in RAW264.7 Cells
The PPI network interaction analysis and KEGG pathway enrichment analysis revealed that p38 was an important target and MAPK-like receptor signaling pathway may be an effective pathway in selin-11-en-4α-ol anti-inflammation. Therefore, the potential anti-inflammatory mechanism of selin-11-en-4α-ol was further investigated by Western blot to verify the network pharmacology predictions. As shown in Figure 11A, the phosphorylation levels of ERK1/2, JNK1/2, and p38 were increased in LPS-stimulated RAW264.7 cells, and selin-11-en-4α-ol decreased the phosphorylation levels of the above proteins, indicating that it could exert anti-inflammatory effects by regulating the production of inflammatory factors and chemokines through inhibiting the protein phosphorylation expression of MAPK signaling pathway. The above results were consistent with the predictions of PPI network analysis, KEGG enrichment analysis, and molecular docking. In addition, selin-11-en-4α-ol could also inhibit LPS-induced elevated phosphorylation of nuclear transcription factor-κB (NF-κB) signaling pathway-related proteins IKKβ, IκBα, and p65 (Figure 11B).
Discussion
Inflammation is a common and prevalent disease that threatens human health, and excessive inflammation can be dangerous, disabling, or even life-threatening. DEX is widely used to treat inflammatory diseases but has side effects such as increased infections and gastrointestinal reactions. 21 Selin-11-en-4α-ol was a monomeric compound obtained by extraction and isolation from A vulgaris. Previous studies have shown an inhibitory effect on LPS-induced NO secretion from RAW264.7 cells. 15 However, a more comprehensive anti-inflammatory characterization and potential anti-inflammatory molecular mechanisms have not been reported. This study explored the potential anti-inflammatory targets and pathways of selin-11-en-4α-ol through network pharmacology, molecular docking, and in vitro cellular assays to provide a scientific basis for developing new anti-inflammatory drugs based on natural products.
In our study, 51 potential anti-inflammatory targets of selin-11-en-4α-ol were predicted. Based on the PPI network, selin-11-en-4α-ol could interfere with inflammation by regulating SRC, HSP90AA1, MAPK14, and other targets. SRC is a proto-oncogene tyrosine-protein kinase that is activated by binding to various cellular receptors such as immune response receptors, G protein-coupled receptors, and cytokines and controls various biological activities such as gene transcription, immune response, and apoptosis. 22 SRC can mediate IL-6 signaling through activation of the YAP1-NOTCH pathway to induce inflammation during regeneration of damaged tissues. 23 HSP90AA1 is a heat shock protein, HSP 90-α, which can be involved in cell cycle control and signal transduction as a molecular chaperone. 24 HSP90AA1 can bind bacterial LPS to induce TNF secretion by monocytes, thereby mediating the inflammatory response. 25 MAPK14 (p38) is mitogen-activated protein kinase 14, which plays an important role in the pro-inflammatory cytokine-induced cellular response cascade as an essential component of the MAP kinase signaling pathway. 26 It has been reported that phosphorylation of MAPK14 is significantly increased in LPS-stimulated myeloid cells, which promotes the expression of the inflammatory factors IL6, IL8, and IL12B, and also enhances the recruitment of NF-kappa-B, which further mediated inflammation. 27 The molecular docking results showed that selin-11-en-4α-ol showed good binding activity to MAPK14.
GO enrichment analysis showed that the target genes were mainly enriched in extracellular regions, receptor complexes, and other loci and were associated with BPs such as intracellular receptor signaling, positive gene expression regulation, and negative inflammatory response regulation. KEGG pathway enrichment analysis was performed to explore the anti-inflammatory signaling pathway of selin-11-en-4α-ol. KEGG enrichment analysis identified 16 relevant signaling pathways, suggesting that selin-11-en-4α-ol may exert anti-inflammatory effects through multiple pathways, with the MAPK signaling pathway exhibiting the highest gene count enrichment, indicating that selin-11-en-4-ol may interfere with inflammation primarily through regulation of the MAPK signaling pathway. MAPKs are a family of serine/threonine protein kinases that function as signaling agents in various cells, mediating fundamental BPs such as cellular proliferation, differentiation, and apoptosis. 28 The MAPK signaling pathway includes ERK, p38MAPK, BMK1, and JNK. 29 Although these 4 pathways are distinct, the same stimulus can activate multiple signaling pathways to generate interactions and participate in the organism's BPs. In mononuclear macrophages, LPS can activate ERK, p38MAPK, and JNK pathways to increase their corresponding protein phosphorylation levels, which further mediates the synthesis of inflammatory cytokines such as TNF-α, IL-6, and IL-1.30-32 Many natural products can inhibit MAPK signaling pathway-mediated inflammation as potential targets for anti-inflammatory therapy.
TLRs/NF-κB signaling pathway activity is mainly affected by IKK, a complex composed of IKKα, IKKβ, and IKKγ. IKKβ regulates the activation of the classical NF-kB pathway through the phosphorylation of IkBs, first catalyzing the phosphorylation of IκB proteins leading to the degradation of IκB proteins during normal cellular. The degradation of the IκB protein during the normal state of the cell in interaction with the transcription factor NF-κB further promotes NF-κB nuclear translocation and binding to the promoter sequence of the corresponding target gene to induce the expression of inflammatory factors, chemokines, and other inflammation-related mediators, exerting a pro-inflammatory effect.33,34 It has been reported that the MAPK signaling pathway and the NF-κB signaling pathway are closely related, and the phosphorylation of p38 promotes the nuclear translocation of p65, while the NF-κB signaling pathway is inhibited after treatment of cells with p38 inhibitors. 35 In summary, we speculated that selin-11-en-4α-ol may interfere with the onset and development of inflammation by regulating the MAPK/NF-κB signaling pathway. To test this hypothesis, we found that selin-11-en-4α-ol down-regulated the phosphorylation levels of ERK1/2, JNK1/2, p38, IKKβ, IκBα, and p65 by protein blotting experiments, confirming the prediction of network pharmacology and molecular docking. It was shown that selin-11-en-4α-ol exerted anti-inflammatory effects by regulating the expression of target proteins related to the MAPK/NF-κB signaling pathway to inhibit inflammatory signaling (Figure 12).
The anti-inflammatory effect of selin-11-en-4α-ol was investigated by LPS-induced RAW264.7 cells in an in vitro assay. It has been reported that the inflammatory factors TNF-α, IL-1β, IL-6, COX-2, iNOS, and the chemokines MCP-1 and MIP-1α are abundantly expressed in LPS-activated macrophages.36-38 TNF-α and IL-1β are the main inflammatory cytokines involved in inflammation and can induce the expression of other inflammatory factors to maintain and amplify the inflammatory response.39,40 When the body is infected, monocytes and macrophages can release IL-6, and excessive IL-6 production plays an important pathological role in developing inflammatory diseases. 41 The expression of iNOS further regulates the production of the inflammatory mediator NO, which is thus involved in the inflammatory response. 42 COX-2 is a key enzyme in the synthesis of prostaglandins, which can be significantly expressed during infection and injury in the body, inducing the synthesis of prostaglandin E2 and playing an important role in the inflammatory response, and many clinical nonsteroidal anti-inflammatory drugs target COX-2 to exert anti-inflammatory effects. 43 Chemokines (MCP-1 and MIP-1α), a family of chemotactic molecules, play a crucial role in various inflammatory diseases such as neuroinflammatory diseases, rheumatoid arthritis, and acute pancreatitis by recruiting monocytes and macrophages to sites of inflammation to enhance the expression of inflammatory factors.44,45 In addition, the expression of MIP-1α and MCP-1 is associated with activating the JNK/p38 MAPK signaling pathway and is considered one of the targets to inhibit the LPS-induced inflammatory response. 46 Therefore, inflammation can be alleviated by inhibiting the expression of the above-mentioned inflammatory factors and chemokines. We found that selin-11-en-4α-ol pretreatment significantly inhibited the secretion of NO, TNF-α, and IL-6 and had a significant inhibitory effect on the mRNA levels of TNF-α, IL-1β, COX-2, iNOS, MIP-1α, and MCP-1, confirming that selin-11-en-4α-ol could exert anti-inflammatory effects by down-regulating the expression of inflammatory factors and chemokines. ROS plays an important role in macrophage-mediated immune responses, and it has been reported that the expression of ROS is significantly elevated in LPS-induced macrophages, which further activates MAPK and NF-κB to drive a series of pro-inflammatory factors and reducing or blocking ROS production has also become a strategy for the treatment of related inflammatory diseases.47,48 The effect of selin-11-en-4α-ol on intracellular ROS levels was evaluated using the ROS probe DCFH-DA. We found that 3 concentrations of selin-11-en-4α-ol significantly inhibited ROS production and exhibited strong antioxidant damage, but more in-depth mechanistic studies are still unclear and need further exploration.
Conclusions
Our study investigated the anti-inflammatory effects and potential molecular mechanisms of selin-11-en-4α-ol by combining network pharmacological analysis, molecular docking techniques, and experimental validation. The results showed that selin-11-en-4α-ol exerted anti-inflammatory effects by inhibiting the phosphorylation levels of MAPK/NF-κB signaling pathway-related target proteins to downregulate the production of TNF-α, IL-1β, IL-6, COX-2, iNOS inflammatory factors and chemokines (MCP-1, MIP-1α). The combination of network pharmacology and pharmacological experiments effectively elucidates natural products to treat inflammation through multiple targets and pathways, providing new ideas and strategies for future clinical drug treatment of inflammatory diseases.
Footnotes
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Guangdong Province ordinary universities characteristic innovation project (2020KTSCX060), Undergraduate Innovation and Entrepreneurship Training Program of Guangdong Pharmaceutical University (202210573062), and Guangdong Modern Agricultural Industrial Technology System Innovation Team Project (2022KJ142), and Guangzhou key research and development plan (202206010008).
