Abstract
Background
Zataria multiflora Boiss. (ZM) is known for its diverse bioactive components, including essential oils, phenolic compounds, and terpenoids, which possess significant antioxidant, antimicrobial, and anti-inflammatory properties. The production of inflammatory mediators and pro-inflammatory cytokines is linked to inflammatory pain, making the management of these mediators crucial in alleviating chronic conditions associated with oxidative damage and inflammation. This study aims to identify and isolate the antioxidant and anti-inflammatory compounds in the ethyl acetate (EtAc) fraction of ZM.
Methods
ZM leaves were sequentially extracted using petroleum ether, dichloromethane, and ethyl acetate via maceration method. The extract was standardized by determining total phenolic compounds using the Folin-Ciocalteau method. The EtAc extract was fractionated using column chromatography, and bioactive compounds were isolated then quantified by High-Performance Liquid Chromatography (HPLC). The in vitro antioxidant activities of the fractions were assessed using DPPH and Ferric Reducing Antioxidant Power (FRAP) assays. In vivo anti-inflammatory effects were evaluated in a carrageenan-induced paw inflammation model, supported by the Evans blue assay.
Results
Thymol, thymoquinone (TQ) and rosmarinic acid (RA) were identified as the main bioactive compounds in the EtAc fraction of ZM. Among these, TQ exhibited the strongest antioxidant activities and in vivo anti-inflammatory effects, with a lower toxicity profile. The concentrations in EtAc fraction were 5.38% for thymol, 0.0057% for TQ, and 0.0064% for RA (% w/w).
Conclusions
This study demonstrated that thymol, TQ and RA are the principal bioactive compounds in the ethyl acetate fraction of ZM. Although thymol was present in higher concentrations than the other two compounds, the predominant antioxidant and anti-inflammatory activities of the plant are attributed primarily to the monoterpenoid compound, TQ.
Introduction
Inflammation is a fundamental physiological response, crucial for protecting the body against a range of harmful stimuli, including pathogens, injuries, and irritants. This intricate process involves a cascade of events, beginning with vascular changes leading to edema and culminating in the migration of leukocytes to the injury site, establishing an inflammatory focus. This focus is essential for isolating and eliminating offending agents, thereby facilitating tissue regeneration and repair inflammation. 1 Pro-inflammatory cytokines, particularly interleukins such as IL-1β and interleukin-6 (IL-6), play a central role in orchestrating the inflammatory response. These cytokines regulate immune cell activation and behavior, inducing fever, and driving the recruitment of various inflammatory cells to the affected area. 2 Oxidative stress is closely related to inflammatory responses. The excess reactive oxygen species (ROS) produced can either oxidize biological molecules or can structurally alter proteins and genes to trigger signaling cascades that can lead to the initiation and progression of inflammatory diseases. Activation of transcription factors and pro-inflammatory genes caused by ROS leads to the initiation of inflammation. Inflammation causes immune cells to secrete various cytokines and chemokines to recruit various other immune cells to the site of oxidative stress/infection. Conversely, increased ROS production by immune cells at the site of inflammation causes oxidative stress and tissue damage. 3 Prolonged activation of these pathways can result in severe acute or chronic inflammatory conditions, necessitating the development of effective anti-inflammatory therapies. 4 While non-steroidal anti-inflammatory drugs (NSAIDs) are widely used to manage pain and inflammation, they often come with adverse effects on the gastrointestinal tract, kidneys, cardiovascular system, and liver. 5 Similarly, long-term use of corticosteroids is linked to a range of side effects that pose significant health risks. This highlights an urgent need for safer and more efficacious anti-inflammatory agents. 6
Traditional medicine has long recognized the potential of natural plant-based compounds as a source of therapeutic agents with anti-inflammatory properties. 7 Recent studies on anti-inflammatory effects have highlighted the role of phenolic compounds in alleviating oxidative stress at injury sites. These compounds help by reducing neutrophil infiltration and limiting the production of pro-inflammatory mediators such as nitric oxide (NO), tumor necrosis factor-alpha (TNF-α), and various interleukins. Generally, their anti-inflammatory properties are attributed to their ability to inhibit lipid peroxidation, downregulate the expression of inducible nitric oxide synthase (iNOS), cyclooxygenase-2 (COX-2), interleukin-1β (IL-1β), and IL-6, and disrupt oxidative stress-related signaling pathways. Additionally, phenolic compounds have been shown to suppress the release of pro-inflammatory cytokines, further contributing to their anti-inflammatory potential.8,9 Phenolic compounds, abundantly found in plants, are an essential part of the human diet due to their antioxidant and pro-oxidant properties for example, black tea contains phenolic compounds, which are responsible for the plant's antioxidant properties and other biological properties. 10 Numerous herbs contain bioactive compounds that can modulate inflammatory processes with minimal side effects, making them attractive alternatives for treatment.
One such plant, Zataria multiflora Boiss.(ZM), a member of the Lamiaceae family native to Iran, Pakistan, and Afghanistan, 7 has attracted significant attention for its rich pharmacological profile. Traditionally used for its anti-inflammatory, antimicrobial, and analgesic properties, this herb has also been employed as a culinary spice. Recent studies have demonstrated the potential of ZM essential oil and its constituents to exhibit antioxidant and anti-inflammatory effects, both in vitro and in vivo. The essential oil has been shown to enhance the levels of key antioxidant enzymes while effectively scavenging free radicals, thereby mitigating oxidative stress and inflammation. 11 The plant and its terpenoid may hold therapeutic effect in certain inflammatory diseases characterized by a crucial role of free radical scavenging. 12 Toxicity and safety studies conducted in cellular and animal models have not revealed significant adverse effects at therapeutic doses. 13 Furthermore, no significant complications have been documented when administered in therapeutic dosages. The biological and medicinal importance of the family Lamiaceae, genus Zataria and ZM makes it important to investigate the phytochemical composition of ZM. In a study, two para-cymene derivatives: multiflutriol (1), multiflurol (2), a new aromatic ester of para-hydroxybenzoic acid (3), and three compounds: dihydroxyaromadendrane, luteolin, and α-tocopherolquinone were identified in ZM. 14 The aerial parts of this plant contain essential oil. The essential oil content (EO) of ZM varies from 2.75% to 5.89% and the essential oil of this plant is rich in phenolic oxygenated monoterpenes. Carvacrol (73.31-10.56%), thymol (48.12-3.51%), linalool (55.38-0.90%) and p-cymene (1.66-13.96%) are the main components of the essential oil. Carvacrol (CAR) with the chemical name (2-methyl-5-(1-methylethyl)-phenol) is a potent monoterpenic phenol found in several essential oils of the Lamiaceae family. CAR has a beneficial effect on many diseases such as asthma and chronic obstructive pulmonary disease (COPD). 15 In more detail, it can be said that the essential oil of ZM contains phenolic (thymol, carvacrol, and linalool) and non-phenolic (p-cymene, γ-terpinene, and α-pinene) compounds. 16 Despite the promising pharmacological properties of ZM, the understanding of its bioactive constituents and their mechanisms of action remains limited. Our previous findings suggest that the ethyl acetate (EtAc) fraction of this plant displays robust antioxidant and anti-inflammatory capacities. Therefore, this study aims to identify and isolate the active compounds within the EtAc fraction of ZM using bioassay-guided fractionation, while evaluating their potential antioxidant and anti-inflammatory effects.
Materials and Methods
Reagents
Analytical-grade solvents and reagents were sourced from E-Merck (Darmstadt, Germany). λ-carrageenan and Evans blue dye were obtained from Sigma-Aldrich (USA). All reagents used conformed to analytical grade standards. Standard compounds thymol, thymoquinone (TQ) and rosmarinic acid (RA) (purity > 93.0%) were purchased from Sigma (St. Louis, MO, USA). High-Performance Liquid Chromatography (HPLC) grade methanol (MeOH) and trichloroacetic acid (TCA) were supplied by Merck (Darmstadt, Germany), respectively. Deionized water was prepared using a Milli-Q water purification system (Millipore, Bedford, MA, USA).
Plant Materials, Extraction of the Plant and Phytochemical Analysis
The plant was purchased from a local market, authenticated by a botanist and a voucher specimen was deposited in Herbarium of the Department of Pharmacognosy, Kerman University of Medical Sciences, Iran (KF1241-2).
One thousand five hundred (1500) g of plant leaves was sequentially extracted using petroleum ether, dichloromethane, ethyl acetate by maceration method. The solvent to sample ratio was 4:1. The extraction with each solvent continued for 72 h and the extract was replaced with a fresh solvent every 24 h. The extracts were concentrated and dry in vacuum oven at 40 °C and then stored at −20 °C until further experiments. Phytochemical screening for flavonoids, terpenoids, tannins, alkaloids and phenolic compounds was performed using standard methods. 17 Total phenolic content was quantified using the Folin-Ciocalteu method, with Gallic acid as a standard. 18 The Folin-Ciocalteu method measures poly(phenol) intake and may serve as an anti-inflammatory biomarker. Phenolic compounds in plants are important for human health, offering antioxidant and pro-oxidant benefits. 19
Chromatographic Purification
Bioactive phenolic compounds were identified in ZM EtAc extract via TLC. 30 g of EtAc extract was subjected to stepwise column chromatography, involving a 1 m × 4 cm silica column with an activated silica gel bed (300 g). The initial elution with petroleum ether was gradually switched to petroleum ether/EtAc gradient, resulting in 62 fractions. TLC analysis of the fractions led to the pooling of 13 homogeneous fractions. Fraction 6 (F6) showed superior antioxidant and anti-inflammatory activity (IC50: 62.95 µg/mL), prompting further purification to sub-fraction 1 (SF1; IC50: 7.13 µg/mL). SF1 purity was confirmed by TLC and HPLC, with structural elucidation achieved using Nuclear Magnetic Resonance (NMR) and IR spectroscopy.
Antioxidant Assays
The antioxidant activity of the samples was evaluated using two well-established methods: DPPH (2,2-diphenyl-1-picrylhydrazyl) and FRAP (Ferric Reducing Antioxidant Power) assays. Both assays were conducted across a range of concentrations.
In the DPPH assay, the ability of the samples to reduce the DPPH radical, which has a deep violet color, was monitored spectrophotometrically. The reduction leads to a color change, allowing for quantification of antioxidant activity. The IC50 value was calculated and the fraction with the highest antioxidant properties was identified and selected. 20
The FRAP assay supports these findings by measuring the sample's ability to reduce ferric ion (Fe³+) to ferrous ion (Fe²+) in a colored complex, which can also be quantified spectrophotometrically. This method provided complementary data on the samples’ overall reducing power.17,21
Anti-Inflammatory Assays
The anti-inflammatory properties of the extracts were evaluated using both in vitro and in vivo models. The study was started with obtaining the necessary ethical approvals for the utilization of cell cultures and animal models from the Ethics Committee of Kerman University of Medical Science (Approval ID: IR.KMU.REC.1403.398).
Cytotoxicity Assay
The J774A.1 macrophage cell line was cultured in RPMI-1640 medium, supplemented with 10% fetal bovine serum and 5% penicillin, under standard conditions (37 °C, 5% CO2, 95% O2 atmosphere). Cells were seeded in 96-well plates at a density of 1 × 102 cells per well and incubated for 24 h prior to the experiments.
The MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay was employed to assess cellular viability. This method relies on the mitochondrial reduction of MTT to formazan, which is then quantified spectrophotometrically at 570 nm. 22
Induction of Acute Paw Edema with λ-Carrageenan
Forty-eight)48(male BALB/c mice, weighing 20-25 g and aged 9-10 weeks, were housed under controlled conditions (25 ± 3 °C, 50%-60% humidity, 12-h light-dark cycle) with unrestricted access to standard diet and water.
Mice were divided into eight groups of six: negative control, λ-carrageenan (CGN), CGN + diclofenac (250 mg/kg) (positive control), and five treatment groups (CGN + extract, CGN + F6, CGN + F3, CGN + F1, and CGN + SF1). The negative control and CGN groups were given 1 mL of normal saline sub-plantar injection in the left hind paw. Paw edema was induced by injecting 30 μL of 1% λ-carrageenan into the left hind paw. Treatment groups received intraperitoneal injections of their respective compounds 1 h after λ-carrageenan injection. Paw size measurements were taken at 0, 1, 3, 5, and 7 h after λ-carrageenan injection, and the inhibitory effect was calculated. 23
Evans Blue Dye Extravasation
To assess vascular permeability, mice received a tail vein injection of 200 μL of 0.5% Evans Blue dye 7 h after λ-carrageenan injection. The paws were then processed to extract the dye, and its concentration was measured spectrophotometrically at 630 nm. 23
Detailed Analysis of Active Compounds
The active compounds in the extract were characterized using HPLC with a YL9100 HPLC system featuring a RP-C18 reversed phase column (250 × 4.6 mm, 5 µm). The analysis was carried out using Clarity version 6.2.0.208 software. The total run time for the analysis of the standards of RA and TQ was set at 40 min, with isocratic elution comprising 70% solvent A (0.04% TCA in water) and 30% solvent B (methanol) at a flow rate of 1.0 mL/min and 30 °C. For thymol, the analysis was completed in 20 min, using 40% solvent A (water) and 60% solvent B (methanol) with UV detection at 282 nm. Calibration curves were established by preparing standard solutions of each compound at varying concentrations after initial sonication and filtration to ensure accuracy
Further structural elucidation of the identified compounds was performed using NMR spectroscopy and Fourier Transform Infrared (FTIR) spectroscopy, providing comprehensive insights into their chemical structures. Limits of detection (LOD) and quantification (LOQ) were calculated using the signal-to-noise ratios from analyses of eight blanks, applying established formulas: LOD = 3.3 × (SD of blank / Slope) and LOQ = 10 × (SD of blank / Slope). This thorough characterization process affirms the identity and concentration of the active compounds present in the samples.
Statistical Analysis
Statistical analyses were carried out using SPSS Software, version 25. The differences between two groups were assessed using Student's t-test, while differences among multiple groups were evaluated using a one-way analysis of variance (ANOVA) followed by Duncan's Honestly significant difference test (Duncan's HSD). All experiments were conducted independently and the results are presented as mean ± SD. In all experiments, mouse samples were randomly collected, with significance considered at P < .05.
Results
Extract Yield, Phytochemical Screening and Total Phenolic Content
The Percentage yields (w/w) of petroleum ether, dichloromethane and ethyl acetate extracts were found to be 6.11%, 7.55% and 4.41% respectively. Phytochemical screening of the plant confirmed the presence of flavonoids, terpenoid and tannins. The total phenolic content of ZM was calculated to be 6.25%(W/W) Gallic acid equivalent, as determined from the calibration curve (y = 0.0016x − 0.0174, R2 = 0.9945).
Spectral Analysis of Active Compounds
The composition of the fractions collected during column chromatography, along with the concentrated fractions, was analyzed for determination of active ingredients by UV spectroscopy.
The FTIR spectrum of fraction F1 (Figure 1A) exhibited a prominent band at 3225 cm−¹, which corresponds to the stretching of phenolic -OH groups, indicative of hydrogen bonding interactions. The aromatic character of F1 is further supported by a C=C stretching vibration of the benzene ring observed at 1622 cm−¹. Additionally, the spectrum reveals two distinct bands at 2958 cm−¹ and 2926 cm−¹, associated with the asymmetric stretching vibrations of -CH groups.

FTIR spectrum of F1 (A) and SF1 (B) separated from ZM EtAc extract.
In contrast, the FTIR spectrum of fraction SF1 (Figure 1B) showed a significant band at 1656 cm−¹, representative of the strong stretching vibration of the carbonyl (C=O) group. A band located at 2965 cm−¹ correlates with the C-H stretching vibrations of aliphatic groups. Furthermore, the band at 2925 cm−¹ is attributed to the C-H stretching of tertiary carbon, while the band at 2870 cm−¹ corresponds to the symmetric stretching modes of methyl groups. Finally, a band at 1458 cm−¹ is assigned to the antisymmetric bending of methyl (−CH3) groups (Figure 1).
Interpretation of the NMR Spectrum
The analysis of the IR and NMR spectra reveals that F1 possesses a molecular formula of C10H14O, correlating with the chemical structure of 2-isopropyl-5-methylphenol (Figure 2A). Thus, F1 is identified as thymol. In contrast, SF1 demonstrates a molecular formula of C10H12O2, consistent with the chemical structure of 2-isopropyl-5-methyl-1,4-benzoquinone(Figure 2B). Therefore, SF1 is confirmed to be TQ. The interpretation of the 1H-NMR and 13C NMR spectra for F1 and SF1 is presented in Figure 3 - Table 1 and Figure 4 - Table 2, respectively.

The chemical structure of the F1 (A) and SF1 (B). carbon and hydrogen in the structure of compounds are represented by numbers and letters, respectively.

1H-NMR spectrum of F1 (A) and SF1 (B) separated from ZM EtAc extract.
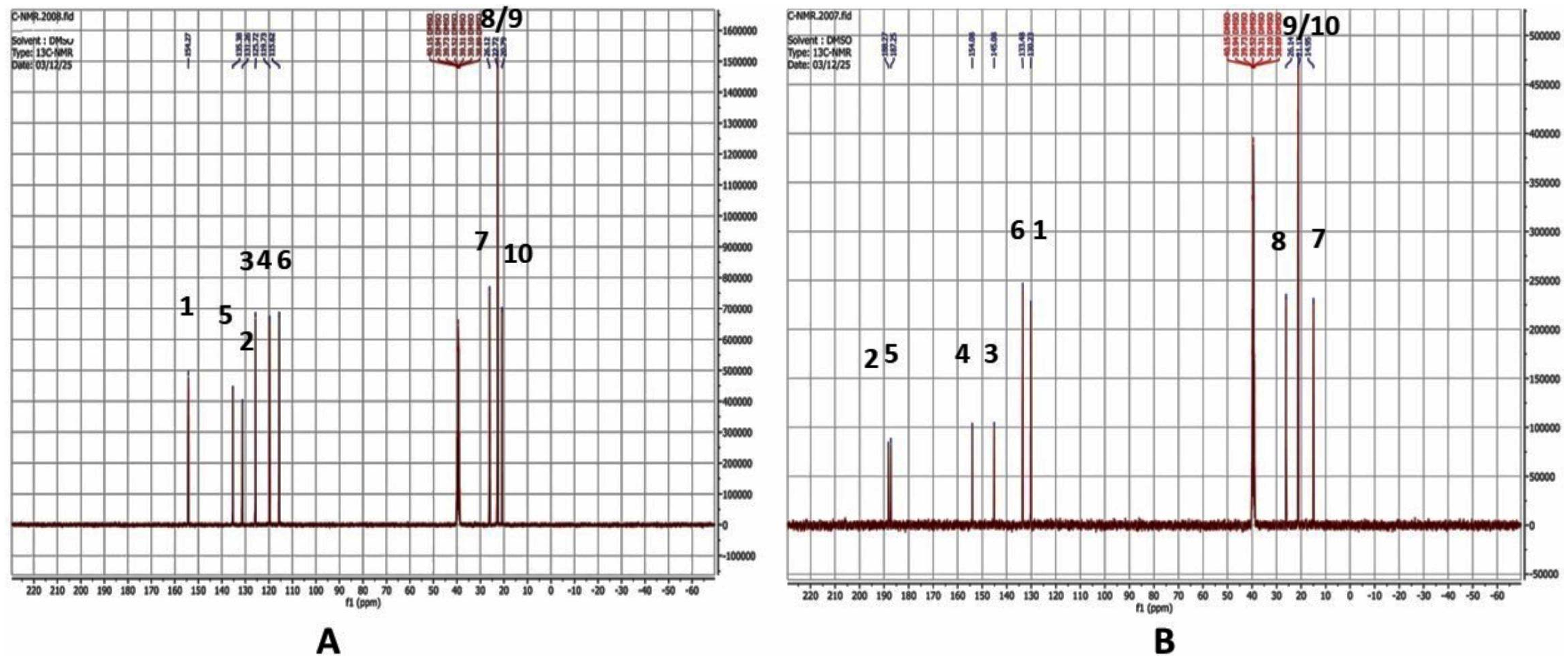
13C-NMR spectrum of F1 (A) and SF1 (B) separated from ZM EtAc extract.
Antioxidant Assays
The DPPH free radical scavenging activity of the samples exhibited a concentration-dependent increase in antioxidant capacity. Notably, F6 displayed the highest DPPH scavenging activity, achieving an impressive inhibition of 93.28%, compared to other fractions. The inhibition levels correlate with strong antiradical activity, as represented in Figure 5.

Percentage of DPPH radical inhibition for various fractions obtained from the EtAc extract of ZM at 200 μg/mL. The experiment was conducted in triplicate, and the results are expressed as Mean±SD. Abbreviations: F, Fraction.
Samples with IC50 values of 50 to 100 µg/mL demonstrate intermediate antioxidant activity, while those with values between 10 to 50 µg/mL indicate strong activity. In this context, F6 showed an IC50 of 62.95 µg/mL, categorizing it as having intermediate antioxidant activity. In contrast TQ and RA displayed strong antioxidant activities, with IC50 values of 7.13 and 7.7, respectively.
In FRAP Assay, The Fe²+ (µmol) content of the ZM extract was found to be 0.1 at a concentration of 500 μg/mL, as per the FeSO4·7H2O calibration curve (y = 0.05714 + 0.2387, R² = 0.9981). Among the fractions, F6 exhibited the highest FRAP value, while F7 also showed superior FRAP compared to thymol (0.15 vs 0.09). TQ and RA demonstrated strong FRAP values of 0.19 and 0.10 at 10 µg/mL, but were weaker relative to green tea, which had a FRAP value of 0.50
Anti-Inflammatory Assays
Cytotoxicity Assay
As depicted in Figure 6, TQ exhibited the highest viability of J774.1 cells across a range of concentrations in comparison to the other samples. After 72 h of treatment, the IC50 values for TQ, F3, F6, thymol, and ZM extract were determined to be 34.797, 0.397, 27.039, 13.336, and 2.628 mg/mL, respectively. Notably, the IC50 value for TQ was higher than those of the other samples, suggesting that TQ demonstrates lower toxicity.

Cell viability of TQ (A), F3 (B), F6 (C), Thymol (D), and ZM extract (E): statistical analysis was performed using one-way ANOVA followed by Duncan's multiple range test (Mean±SD). Statistically significant differences are indicated for P< .05.
λ-Carrageenan-Induced Paw Edema
Paw swelling in mice was measured using a digital Vernier caliper. As depicted in Figures 7 and 8, TQ and diclofenac exhibited maximum inhibition rates of 46.15% and 65.38% respectively, in comparison to control group (normal saline treated).

Paw Edema in different treated group. Abbreviations: CGN, Carrageenan group; positive Ctrl, positive control group; Neg.Ctrl, negative control group; TQ, Thymoquinone group; F3, fraction3 group; F6, fraction6 group.

Comparison of paw thickness (Edema) in various animal treatment groups. Thickness was measured using a digital vernier caliper. Data represent Mean±SD of three experiments; statistically significant differences (P < .05) Are indicated between groups. The differences between two groups were assessed using student's t-test and all groups showed a significant difference compared to the control group at a specific hour. Abbreviations: Neg., Negative; Ctrl, Control; CGN, Carrageenan; TQ, Thymoquinone; F6, Fraction 6; F3, Fraction3.
Extravasation of Evans Blue Dye in Paw Tissues of Mice
Acute inflammation caused by λ-carrageenan can increase the vascular permeability, as indicated by the extravasation of Evans blue from the tissues. The λ-carrageenan induced paw edema significantly increased the absorption of Evans blue at 630 nm. As shown in Figure 9, treatment with both TQ and diclofenac exhibited the greatest suppression of Evans blue dye extravasation in comparison to the other samples.

Comparison of Evans blue dye levels in paw Edema tissue among different experimental groups. numerical data are presented as Mean±SEM (n=3). A significance level of P <.05 Was considered significant when compared to the control group. Abbreviations: Neg., Negative; Ctrl, Control; CGN, Carrageenan; TQ, Thymoquinone; F6, Fraction 6; F3, Fraction 3.
Histopathological Examination
The effects of different treatments (T1 = F6, T2 = Thymol, T3 = TQ, T4 = Extract, T5 = F3) on histopathological changes in paw edema were assessed by HE staining. As shown in Figures 10 and 11, in the acute inflammatory response induced by λ-carrageenan, the paws of mice showed severe edema characterized by blisters of epithelial and conjunctival tissue with a significant number of infiltrated inflammatory cells, most of which were neutrophils. At the site of paw edema, areas equal to the red dotted rectangles were selected at ˟100. Quantification of cell numbers was performed by counting cells in circles with an area equal to the densest cells. Figure 8 showed that the acute inflammatory response with neutrophil infiltration was significantly reduced by T3(TQ) and diclofenac compared with mice induced with λ-carrageenan. According to histopathological findings, the percentage of neutrophils, Mean Inflammatory cell counts, Edema, Inflammatory score were significantly decreased in both T3(TQ) and diclofenac groups, and the percentage of lymphocytes was significantly increased in both TQ and diclofenac groups.

Histopathological examination and assessment of cell counting (H& E Stain) (T1=F6, T2=Thymol, T3=TQ, T4=Extract, T5 =F3). Numerical data are presented as Mean±SEM (n=3). *P < .05 were considered as significant difference compared to the CGN group. Abbreviations: T, Treatment; CGN, Carrageenan; Control−, Negative Control; Control+, Positive Control.

Histopathological microphotography under microscope with the magnification × 100 (H& E stain, scale bar=200 micrometer). Yellow arrow show keratin layer, red arrow show epidermis and green arrow show muscular layer. Abbreviations: T, Treatment; CGN, Carrageenan; Control−, Negative Control; Control+, Positive Control.
HPLC Analysis
The outcomes of the antioxidant and anti-inflammatory activity tests were utilized to choose bioactive sub-fractions for the isolation of their components. Fractions F3, F6, and F11 exhibited the highest concentrations of thymol, TQ and RA, respectively (HPLC chromatograms shown in Figures 12C, 13, and, 14C). HPLC analysis of the EtAc extract of ZM and its chromatographic fractions identified and quantified RA, thymol, and TQ as major active compounds (HPLC chromatograms shown in Figures 12B and 14B). The extract contained 5.38% thymol, 0.005% TQ and 0.0064% (w/w) RA based on the calibration curves (thymol: y = 8.5122x + 3.3308, R2 = 0.9926; TQ: y = 23.495x − 0.329, R2 = 0.9936; RA: y = 11.13x + 0.1853, R2 = 0.99260) (HPLC chromatograms of standard compound shown in Figures 12A and 14A). In this study, three compounds, thymol, TQ, and RA, were identified in the ZM plant, but only two compounds, thymol and TQ, were isolated because the amount of RA in the ZM plant extract and fractions obtained from column chromatography of this plant was low and could not be separated.

HPLC chromatograms of (RA) (Blue Line) and (TQ) (Red Line) at: 5 μg/mL (A), ZM extract (B), and F11 (C). Abbreviations: RA, Rosmarinic Acid; TQ, Thymoquinone.
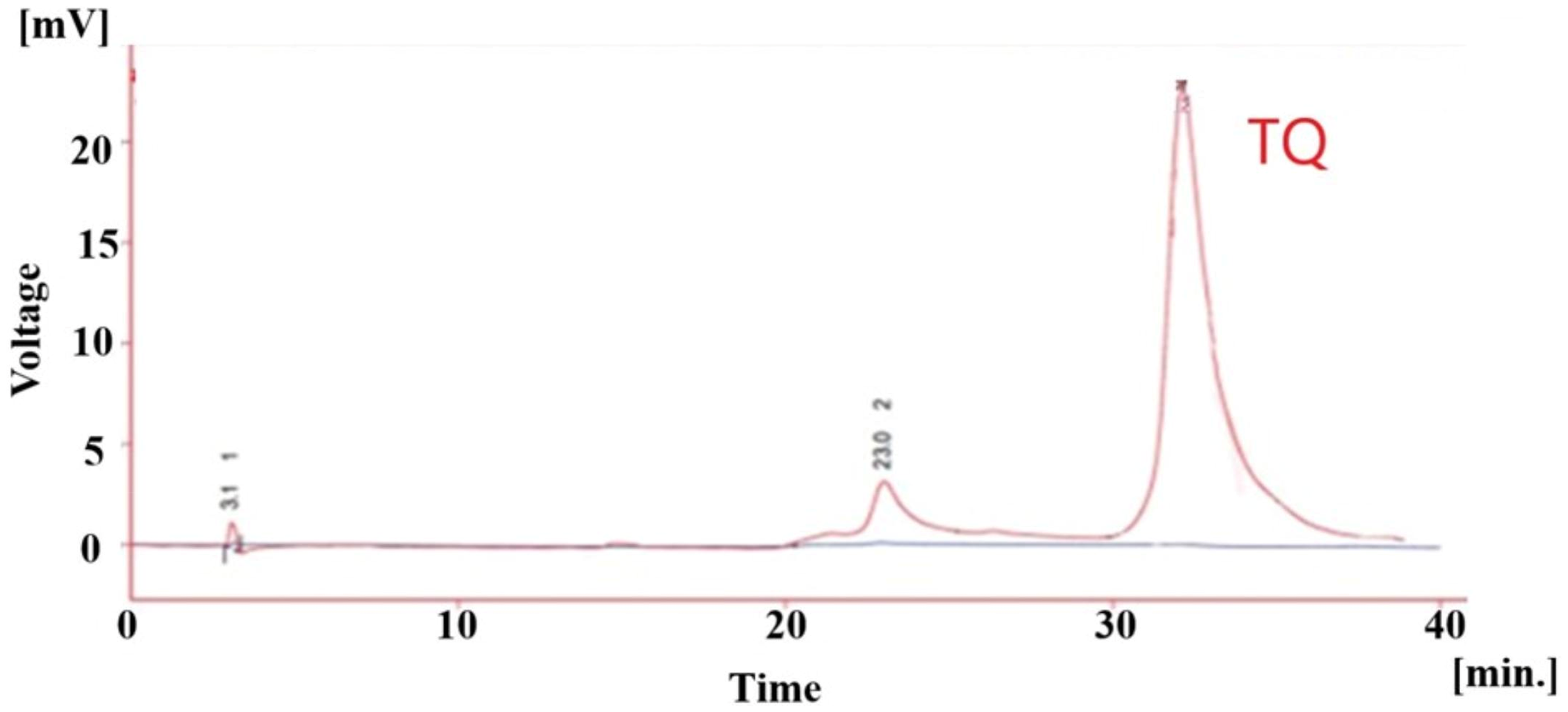
HPLC chromatograms of the F6. Abbreviations: TQ, Thymoquinone.

HPLC chromatograms of Thymol at 2.5 μg/mL concentration (A), ZM extract (B) and F3 (C).
The comparison and determination of the content of active antioxidant and anti-inflammatory compounds reveal that the highest concentration of RA was found in fractions F11, F10 and F9, with respective values of (0.092, 0.024 and 0.021% w/w). In contrast, the greatest amount of thymol was observed in fractions F3, F2 and F4, with values of (69.39, 25.43 and 10.5%w/w). Finally, the analysis indicated that the highest concentration of TQ was identified in F6, followed by a significant decrease in F5, with respective values of (0.56 and 0.077).
Chromatographic characteristics in ZM extract chromatogram: Retention time (RT) for RA, TQ and thymol was calculated to be 12.69, 35.6, and 7.1 min., and capacity factor (K) was 3.06, 10.42 and 10.4., respectively.
The LOD for RA, thymol, and TQ was calculated to be 0.21, 0.057, and 0.026 (μg/mL), and the LOQ was 0.72, 0.191 and 0.085(μg/mL), respectively.
Discussion
The findings of this study underscore the significance of ZM as a potent source of bioactive compounds capable of modulating inflammatory and oxidative pathways. Our investigation reveals that thymol and TQ are the primary active constituents within the EtAc fraction of ZM, each exhibiting distinct but complementary biological activities. While thymol is present in higher concentrations in the extract, it is TQ that predominantly drives the antioxidant and anti-inflammatory effects observed. This highlights the importance of ZM in traditional medicine and provides underlying scientific mechanisms for its potential therapeutic applications. The current study emphasizes TQ's superior efficacy as both an antioxidant and an anti-inflammatory agent, supported by its lower toxicity profile. TQ's mechanism of action includes scavenging superoxide anions, modulating cytokine release, and influencing key signaling pathways such as COX-2, nuclear factor erythroid 2–related factor 2 (Nrf2), phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT), and nuclear factor kappa-light-chain-enhancer of activated B (NF-κβ). 24 These pathways are crucial for alleviating inflammation and providing protection against oxidative stress and this processes central to the pathophysiology of chronic inflammatory diseases. The ability of TQ to reduce the expression of the NLRP3 inflammasome and inflammatory cytokines, such as IL-6 and IL-8, further supports its potential as a therapeutic agent.25,26 TQ activates polymorphonuclear neutrophils (PMN) both in vitro and in vivo. PMNs play a protective role against pathogens secondary to the production of superoxide anion by NADPH oxidase. In addition, oral treatment of mice with TQ reduced several inflammatory regulators such as interleukin 1 beta (IL-1β), IL-6, TNFα, interferon γ (IFNγ), and prostaglandin E2 PGE(2). 27 Also, intraperitoneal injection of TQ into mice suppressed the increase in blood leukocyte counts as well as plasma IL-6 concentrations. In mice with allergic asthma, TQ reduced the levels of T helper type 2 (Th2) cytokines and suppressed the expression of iNOS and transforming growth factor-β1 (TGF-β1) mRNAs, preventing inflammatory changes associated with asthma. 25 TQ also showed excellent antioxidant and anti-arthritic activity in Wistar rats and reduced the expression of inflammatory factors such as IL-1β, TNF-α, IFN-γ, and IL-6. These findings indicate an important role of TQ in the anti-inflammatory process. 28
Thymol, despite being more abundant in the extract, also significantly contributes to the overall biological effects observed. Its role in inhibiting cytokine release and enhancing antioxidant defenses highlights its potential utility in managing inflammatory conditions. The ability of thymol to stimulate GSH production indicates its important role in maintaining cellular redox homeostasis. 29 Furthermore, thymol's antimicrobial properties may enhance the overall therapeutic effects of ZM in inflammatory diseases where microbial infection often plays a significant role. Thymol has shown anti-inflammatory properties in in vitro and in vivo studies. In mice and macrophages, thymol inhibits TLR4 expression and subsequently inhibits the activation of NF-κB signaling, which reduces the production of inflammatory cytokines such as TNF-α and IL-1β. In mice with ulcerative colitis, thymol reduces COX-2 expression and NO levels in the colon of the mice. These studies demonstrate the anti-inflammatory function of thymol through inhibition of the NF-κB signaling pathway. 30 Thymol, as a natural monoterpene phenol, acts against oxidative stress and inflammatory processes. In a study, the anti-inflammatory effects of thymol were investigated in formalin-induced mice and egg albumin-induced chickens. Thymol significantly reduced the paw edema profile in mice and chickens compared to control groups in a dose- and time-dependent manner, with these anti-inflammatory effects probably being mediated through the COX-2 interaction pathway. 31 Numerous studies have been conducted on the properties of thymol, and it has been found that thymol has beneficial properties, including antioxidant, anti-inflammatory, disinfectant, antibacterial, antifungal, and analgesic. 32
Additionally, the use of advanced analytical techniques, such as FTIR and proton nuclear magnetic resonance (1H NMR) spectroscopy, in our study plays a crucial role in characterizing the bioactive constituents of ZM. The spectral data not only confirms the identity of compounds such as thymol and TQ but also provides insights into their structural features, which correlate with their biological activities. The integration of chromatographic methods ensured the isolation of pure compounds, facilitating further biological evaluations and reinforcing the reliability of the findings. Such detailed analytical characterization provides valuable information regarding specific functional groups and molecular bonds responsible for their activity, enhancing our understanding of their mechanisms of action.
The clinical implications of our findings are evident, as ZM extract has shown promise in improving symptoms associated with asthma and COPD by modulating inflammatory cytokines and enhancing pulmonary function. These observations align with clinical trials demonstrating significant reductions in inflammatory biomarkers and improvements in antioxidant status among patients. In particular, the reductions in levels of malondialdehyde (MDA), nitrite, and C-reactive protein, alongside increased total thiol content and activities of superoxide dismutase (SOD) and catalase (CAT) further highlight the efficacy of ZM extract in alleviating symptoms associated with COPD. 33
Furthermore, the cytotoxicity assays conducted in this study further establish the safety profile of TQ, showing high cell viability at varying concentrations, which supports its candidacy for further development as a therapeutic agent. The relatively high therapeutic index of TQ signifies its potential for safe use in clinical settings. However, further research is necessary to determine the optimal dosing and administration routes for maximizing efficacy and safety in clinical studies.
There were notable limitations to this study, such as the in vivo assessments being short-term and acute. Long-term safety, efficacy, and pharmacokinetics of the isolated compounds remain unexplored.
Conclusion
This study highlights the prominent roles of TQ and thymol as key bioactive constituents within the ethyl acetate fraction of ZM, with TQ demonstrating superior antioxidant and anti-inflammatory efficacy. The integration of phytochemical analysis with biological evaluation establishes a comprehensive framework for understanding the therapeutic potential of ZM. Future investigations should focus on delineating the synergistic interactions among TQ, thymol and RA, as well as their pharmacokinetics and long-term safety in clinically relevant models. Expanding the scope to include other inflammatory pathologies and unraveling the molecular mechanisms underlying their effects could pave the way for the development of ZM-based interventions in chronic inflammatory and oxidative stress-related disorders. The findings are based on animal models, and extrapolation to human physiology requires caution and further validation through clinical studies.
Footnotes
Abbreviations
Ethics Approval
The study obtained the necessary ethical approvals for the utilization of animal models from the Research Ethics Committee of Kerman University of Medical Science and the project was found to be in accordance to the ethical principles and the national norms and standards for conducting Medical Research in Iran. (Approval ID: IR.KMU.REC.1403.398).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Research Ethics Committee of Kerman University of Medical Science and approved by this Committee.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Vice Chancellor of Research and Technology, Kerman University of Medical Sciences (Grant No: 402000449).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All data used to support the findings of this study are included in the article.


