Abstract
Introduction
The genus Knema belongs to the family Myristicaceae and consists of small to medium-sized trees found primarily in the tropical regions of Southeast Asia, China, India, Australia, and Africa. These plants are well-known in traditional medicine and pharmacy. 1 In modern research, Knema species possess many important pharmacological properties, including antitumor, antibacterial, anti-inflammatory, neuroprotective, antidiabetic, antioxidant, and hepatoprotective. 2 Phytochemical studies of Knema species demonstrated that this genus contains a diverse range of naturally secondary metabolites substances, such as phenols, flavonoids, 3 aceto-phenones, 4 lignans, polyketides, sterols, terpenoids, quinones, and stilbenoids.5,6
Knema globularia (synonym Knema corticosa), one of the most important species of the Knema genus, is also called “maucho” in the local Vietnamese dialect. Its seeds are famous for curing scabies 6 and other skin diseases, especially in traditional usage. 7 Additionally, the stem bark is used as a blood tonic. 8 Several phytochemicals were isolated from the barks, seeds, and roots of this plant, such as quercetin and kaempferol glycosides, 9 proanthocyanidin, luteolin, taxifolincatechin, 7-megastigmene-36,9 triol, 3′,4′,6′-trihydroxyaurone, β-sitosterol, daucosterol, 10 kneglobularic acid A and B, kneglobularone A, 6-tridecylsalicylic acid, and kneglomeratanone A. 11 To the best of our knowledge, none of the chemical substances derived from K. globularia was known for inhibitory effect despite all scientific efforts. The results regarding the anti-inflammatory effects of Knema genus extracts and chemical components are limited due to several research difficulties, with only a few results of substances obtained from K. furfuracea showing NO inhibitory effect. 12 This research lays the groundwork for the NO inhibition effect of extractions and chemical compositions from Vietnamese K. globularia by identifying study gaps needed to determine the anti-inflammatory aspects for further investigations.
Results and Discussion
Chemical Structure Identifications
The EtOAc fraction was subsequently subjected to column chromatography (CC) using silica gel to isolate six secondary metabolites (

Structure of compounds
Anti-Inflammatory Activity
In the initial experiment, a cytotoxicity assay was conducted to establish the safe and non-toxic concentrations of the isolated compounds (
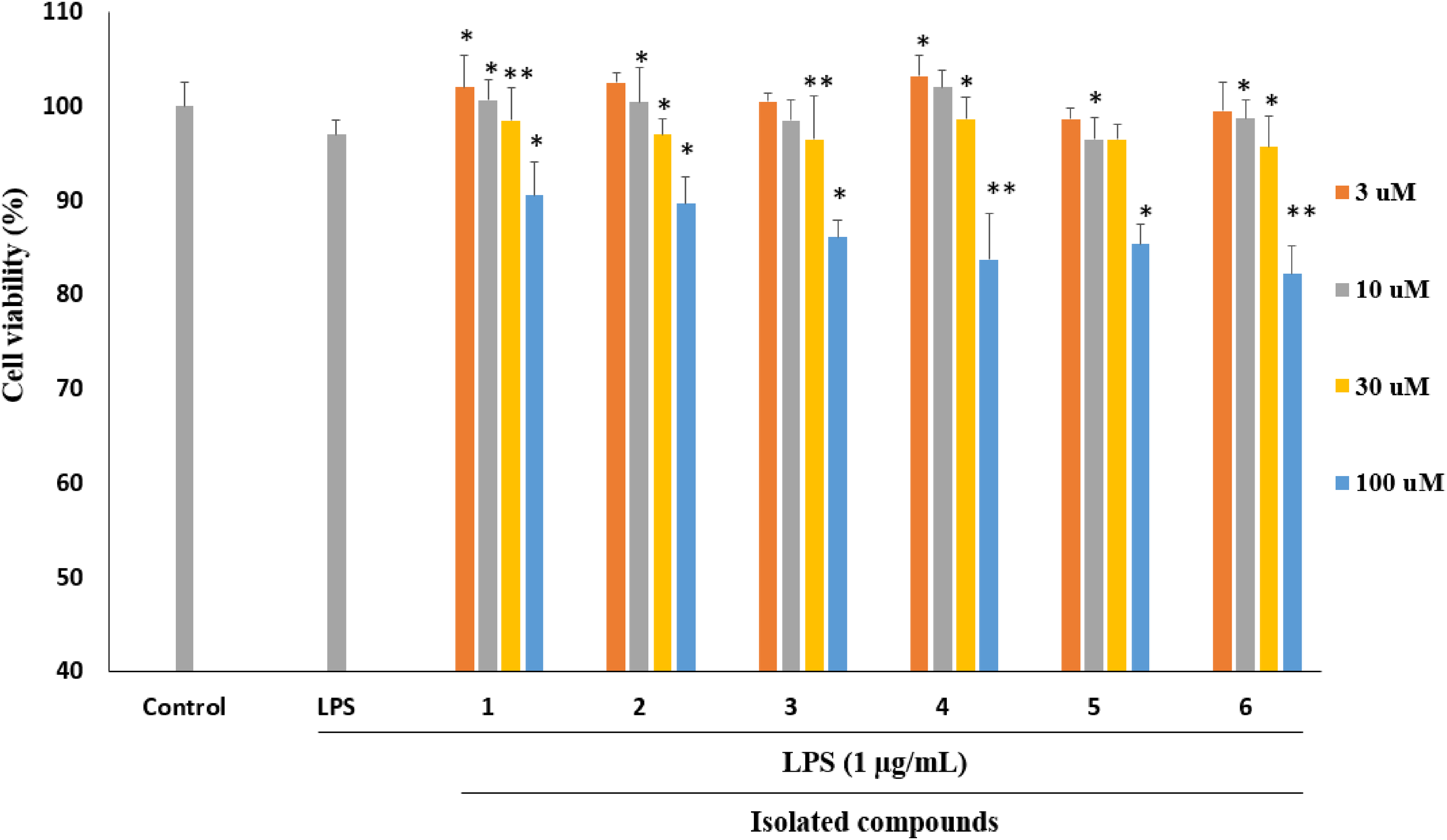
The impact of LPS stimulation on cell viability in the presence of compounds
To evaluate the inhibitory activity on NO production, RAW 264.7 cells were exposed to varying concentrations (1-30 μM) of the isolated compounds, and the level of NO production was measured using the Griess reaction.
19
As shown in Table 1, compounds
NO Production Inhibition in RAW264.7 Cells of Isolated Compounds (

a The inhibitory effects are represented as the molar concentration (μM) giving 50% inhibition (IC50) relative to the vehicle control; Values are mean ± S.D (n = 3); b Positive control; *p < 0.05 compared with the control group.
The control group was not exposed to either LPS or the samples. Therefore, any observed inhibitory effects of these compounds on NO production were not influenced by cytotoxic impacts. Upon stimulation with LPS (1 µg/mL), the control group exhibited an approximately 13-fold increase in NO production after 24 h. In contrast, compounds

Inhibitory effect of compounds
Molecular Docking
Three compounds (

Compounds and iNOS interactions.
Docking Results Toward the iNOS.

The docking simulations of the COX-2 protein with the three studied ligands unveiled the potential of these compounds as COX-2 inhibitors, owing to the robust interactions observed between the ligands and residues in the receptor's active site. Figure 5 showcases the optimal docking poses of the ligands, highlighting their binding interactions with the receptor. Additionally, the free binding energies of the ligands were calculated using AutoDock Vina, providing a basis for evaluating the affinity of the ligands towards the COX-2 target (Table 3).

Compounds and COX-2 interactions.
Docking Results Toward the COX-2.

Interestingly, there was a similarity in the value of free binding energy of compounds
Figure 6 presents the optimal docking poses of the ligands, elucidating their binding interactions with the IL-8 receptor. Furthermore, the free binding energies of the ligands were computed using AutoDock Vina, serving as a foundation for evaluating the affinity of the ligands towards the IL-8 target (Table 4).

Compounds and IL-8 interactions.
Docking Results Toward the IL-8.

Both compound
ADMET Predictions of Studied Compounds
The “Lipinski's rule of five” has long been utilized to assess the druggability of a compound. In this study, the three investigated compounds including compounds
Moreover, the number of rotatable bonds, total polar surface area (TPSA), and aqueous solubility (log S) were evaluated as part of assessing physicochemical parameters. For optimal oral bioavailability and intestinal absorption, the number of rotatable bonds and the TPSA value should not exceed 10 and 140 Å2, respectively. 21 According to the predicted results by SwissADME, the number of rotatable bonds and TPSA values for these compounds fall within favorable ranges, indicating good physicochemical properties. The log S values of these compounds are −3.70, −2.22, and −2.22, respectively, indicating their solubility. The characteristics of the studied compounds are comprehensively presented in Table 5.
Physicochemical Properties Analyzed with SwissADME.

MW: molecular weight; log P: log of octanol/water partition coefficient; nHBD: number of hydrogen bond donor(s); nHBA: number of hydrogen bond acceptor(s); TPSA: total polar surface area; MR: molar refractivity; log S: log of solubility; nRotB: number of rotatable bond(s).
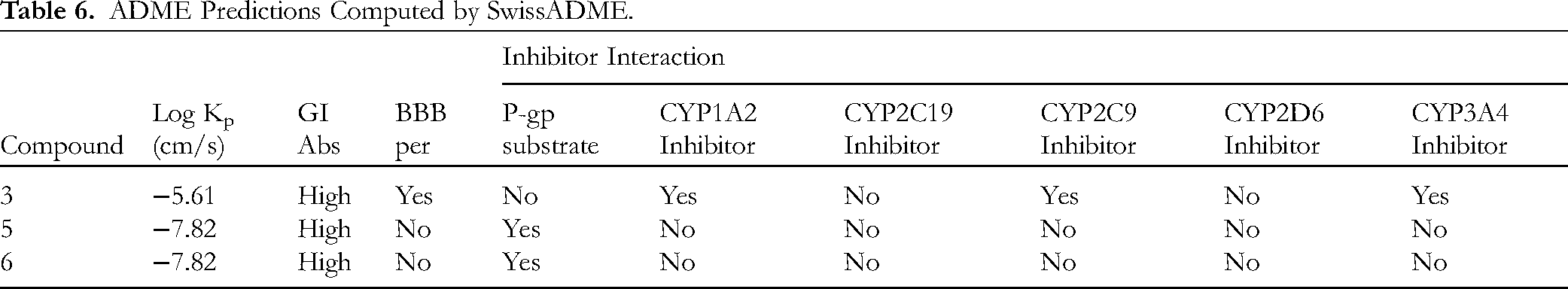
Table 6 presents the in silico predictions of ADME for the three investigated compounds. The range of log skin permeability (log Kp) values spans from −7.82 to −5.61. All compounds are predicted to demonstrate high gastrointestinal absorption. Additionally, both compounds
ADME Predictions Computed by SwissADME.
Furthermore, the DL-AOT prediction server was employed to calculate the LD50 values of compounds
Toxicity Predicted by DL-AOT Prediction Server.

While our study focused exclusively on inhibiting NO production, it did not assess other inflammatory agents such as prostaglandin E2 (PGE2) or proinflammatory cytokines like ILs, tumor necrosis factor-alpha (TNF-α), and interferon-gamma (IFN-γ). Moreover, we did not investigate the inhibitory mechanisms at a molecular level using techniques such as western blotting to assess iNOS and COX-2. Despite these limitations, our study yielded positive outcomes, including the isolation of compounds 1, 3, and 4 for the first time from Vietnamese K. globularia, evaluation of isolated compounds on NO inhibitory activity, and docking studies. These novel findings will serve as the groundwork for more comprehensive studies in the future.
Conclusions
The investigation of phytochemicals in K. globularia stem bark from Vietnam yielded the isolation and structural elucidation of six compounds: lupeol (1), formononetin (2), isoliquiritigenin (3), 2-[4-(3-hydroxypropyl)-2-methoxyphenoxy]propane-1,3-diol (4), (+)-catechin (5), and (−)-epicatechin (6). Notably, this study reports the first identification of compounds 1, 3, and 4 from Vietnamese K. globularia. These compounds were evaluated for their anti-inflammatory activity by assessing their ability to inhibit NO production induced by LPS in macrophage RAW264.7 cells. Among the compounds tested, compound 5 exhibited the most potent inhibitory activity, with an IC50 value of 5.61 μM. This was followed by compounds 3 and 6, which showed IC50 values of 11.52 and 6.76 μM, respectively. However, compounds 1, 2, and 4 were inactive, with IC50 values exceeding 30 μM. In consistency with the results from the in vitro assay, molecular docking was conducted. The results revealed compound 3 as an inhibitor of iNOS, COX-2, and IL-8 receptors, with binding free energies of −6.9, −8.2, and −5.3 kcal/mol, respectively. For compound 5, the binding free energies were −7.1, −8.2, and −5.8 kcal/mol, while for compound 6, the energies were −6.8, −9.3, and −5.7 kcal/mol, respectively. This research enhances our understanding of the potential pharmacological applications of compounds derived from Vietnamese K. globularia in the context of inflammation-related conditions.
Material and Methods
General Experimental Procedure
The 1H NMR (400 MHz) and 13C NMR (100 MHz) spectra were acquired using a Varian Unity Inova 400 MHz spectrometer (Varian, Inc., California, USA). For column chromatography (CC), all solvents, analytical purposes, and visualization were described in Supplemental Materials.
Plant Material
The stem bark of Vietnamese K. globularia was gathered from Son La province, Vietnam, in May 2021. Botanical identification was expertly conducted by Nguyen Quoc Binh, PhD, of the Vietnam National Museum of Nature, Vietnam Academy of Science and Technology (VAST). To ensure traceability, a voucher specimen (KC-SB-2710) was meticulously archived at the Natural Product Research and Development Lab, Phenikaa University, Vietnam.
Extraction and Isolation
The dried powdered stem bark of Vietnamese K. globularia (2.0 kg) was subjected to reflux extraction using methanol (MeOH, 8.0 L × 3 times) for 1 h. The extraction and isolation scheme was described in detail in Supplemental Materials.
Biology
Cell culture, cell viability assay, and determination of NO production were experimented with according to the methods described in Supplemental Materials.
Molecular Docking Study
Protein and ligand preparation for molecular docking and molecular docking 22 were experimented with according to the methods described in Supplemental Materials.
ADMET Predictions of Studied Compounds
All three studied compounds were screened based on “Lipinski's rule of five”. 20 SwissADME web tool was used to calculate data relating to the pharmacokinetics of these compounds. 23 In the case of acute oral toxicity prediction, a DL-AOT prediction server was applied. 24
Statistical Analysis
Data are presented as the mean ± standard deviation (SD). Graphs were generated, and statistical analyses were conducted using SigmaPlot 6.0 and SigmaStat 3.1 (Systat Software, San Jose, CA, USA). For the analysis, ANOVA followed by Tukey's test on pre-validated data was employed. Alternatively, Prism (GraphPad Software, San Diego, CA, USA) was used for comparisons between two groups, utilizing an unpaired Student's t-test. For comparisons involving more than two groups, the one-way nonparametric ANOVA with Tukey's test followed by Bonferroni post hoc analysis or correlation analysis, as appropriate was used. Statistical significance levels were set at *p < 0.05; **p < 0.01.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241280043 - Supplemental material for Nitric Oxide Production Inhibitors from Vietnamese Knema globularia: An in Vitro and in Silico Study
Supplemental material, sj-docx-1-npx-10.1177_1934578X241280043 for Nitric Oxide Production Inhibitors from Vietnamese Knema globularia: An in Vitro and in Silico Study by Dao Cuong To, Phu Chi Hieu Truong, Phi-Hung Nguyen, Le Minh Hoang, Hoa Thi Nguyen, Truong Thi Viet Hoa, Truong Thi Thuy Nhung, Phuong Dai Nguyen Nguyen, Ngu Truong Nhan and Manh Hung Tran in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Vietnam Academy of Science and Technology under the grant number: UDPTCN 03/21-23.
Ethical Approval
Ethical approval is not applicable to this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
