Abstract
Keywords
Introduction
Stress exposure activates not only the hypothalamic–pituitary–adrenal (HPA) axis but also the sympathetic nervous system.1,2 Sympathetic activation triggered in the brain regions such as the paraventricular hypothalamic nucleus (PVN) stimulates peripheral release and/or secretion of noradrenaline and adrenaline.3,4 These peripheral catecholamines elicit systemic and/or local responses in their target organs, resulting in various sympathetic responses including elevation of blood pressure and heart rate. We previously reported that various stress-related neuropeptides such as corticotropin-releasing factor (CRF) and vasopressin5,6 as well as several kinds of stressors, such as restraint stress (RS) and glucoprivation stress,7–9 increase plasma catecholamine levels, and that prostanoids and their synthases in the brain mediate these responses in rats.
Yokukansan (YKS) is a traditional Japanese Kampo medicine approved by the Ministry of Health, Labour and Welfare of Japan, and has indications for neurosis, insomnia, night crying, and irritability in children. 10 YKS is composed of 7 kinds of medicinal herbs, namely, Atractylodes Lancea (rhizome of Atractylodes japonica Koidz. ex Kitam., Compositae, 4.0 g), Poria (strain of Pinus densiflora Siebold and Zucc., Polyporaceae, 4.0 g), Cnidium Rhizome (rhizome of Cnidium officinale Makino, Umbelliferae, 3.0 g), Uncaria Thorn (hook-bearing stems of Uncaria rhynchophylla (Miq.) Miq., Rubiaceae, 3.0 g), Japanese Angelica (root of Angelica acutiloba (Siebold and Zucc.) Kitag., Umbelliferae, 3.0 g), Bupleurum (root of Bupleurum falcatum L., Umbelliferae, 2.0 g), and Glycyrrhiza (root of Glycyrrhiza uralensis Fisch., Laguminosae, 1.5 g), and was kindly provided by Tsumura & Co. (Tokyo, Japan) (http: www.worldfloraonline.org). Additionally, clinical studies have recently reported that YKS is also effective, without serious side effects, against the behavioral and psychological symptoms of dementia (BPSD), such as aggression and hallucinations, in patients with Alzheimer's disease11–13 and other diseases or symptoms including borderline personality disorder and preoperative anxiety (see review by Mizoguchi and Ikarashi 14 ). Similar to these clinical studies, such ameliorative effects of YKS have been reported in rodents that show BPSD-like behaviors. YKS improved aggression in various animal models such as amyloid precursor protein (APP) transgenic mice, 15 thiamine-deficient rats, 16 and rats with cholinergic degeneration in the nucleus basalis of Meynert, 17 and anxiety-like behaviors in APP transgenic mice. 15 These findings from clinical and experimental animal studies suggest that YKS has therapeutic effects on psychological/psychiatric symptoms.
In addition to the effects on BPSD-like symptoms, YKS seems to be involved in regulation of stress responses. It is well-known that corticosterone levels, which are the main indicator of HPA responses to stress in rodents, are elevated in response to various stressors. The effects of YKS on stress-induced corticosterone levels vary considerably, ranging from no effects18–20 to suppressive effects.21–23 This discrepancy is thought to be due, in part, to differences in the type and strength of stressors and the doses of YKS administered. YKS treatment also seems to affect stress responses other than corticosterone responses. In the brain, YKS administration decreases RS-induced Fos expression in the amygdaloid nuclei and prelimbic cortex, but not in the PVN, although neither single nor repeated administration of YKS had an effect on the basal or stressed levels of corticosterone. 18 In contrast, in the PVN, YKS administration was shown to normalize stress-induced reductions in glucocorticoid receptor protein levels by regulating microRNA-18 expression. 23 Furthermore, YKS is known to ameliorate stress-induced behavioral abnormalities. Many previous studies reported that repeated administration of YKS ameliorated stress-induced behavioral changes such as anxiety-like behaviors, aggressive behaviors, and decreased social behaviors.18,20,24,25 These findings suggested that oral administration of YKS affects central regulation of stress-induced behavioral responses. However, it remained unclear as to whether YKS can affect sympathetic responses to stress exposure or not.
YKS has been shown to affect neurotransmitter systems, including the serotonergic (5-HTergic), glutamatergic, and GABAergic systems.26–28 Moreover, several studies demonstrated that the ameliorative effects of YKS on behavioral abnormalities are blocked by intraperitoneal administration of antagonists for the 5-hydroxy tryptamine (5-HT1A), gamma-aminobutyric acid (GABAA), or N-methyl-D-aspartate (NMDA) receptors in rats and mice.20,24,26,29,30 Although the underlying detailed mechanisms remain unclear, accumulating evidence raises the possibility that YKS acts directly or indirectly in the brain. It has been shown that YKS affects neurotransmitter levels in the brain, specifically, 5-HT in the prefrontal cortex 31 and the hypothalamus, 26 acetylcholine (ACh) in the hippocampus,27,32 and glutamate and GABA in whole brain homogenates. 28 However, it is unknown at present whether neurotransmitter levels are modulated by YKS treatment in the PVN, the major integrative center for sympathetic function and stress responses in the brain. 2
In this study, we focused on the potential for YKS treatment to affect RS-induced elevation of plasma catecholamine (noradrenaline and adrenaline) levels and related prostanoid (prostaglandin [PG] E2 and thromboxane [Tx] B2) production in the rat PVN (Figure 1). In addition, we measured neurotransmitter levels in the PVN during stress exposure under YKS treatment.

Experimental schedule. Arrows indicate daily administration of Yokukansan (YKS). On day 14, YKS was administered 4 h prior to the start of the dialysis experiment. Gray squares above the line on day 14 show the dialysate collection every 20 min.
Results
Effects of a Single Administration of YKS on RS-Induced Elevation of Plasma Catecholamine Levels
Exposure to RS for 60 min significantly increased plasma levels of noradrenaline and adrenaline (Figure 2). One-way ANOVA indicated that a single administration of YKS 4 h prior to RS exposure did not affect the stress-induced elevation of plasma noradrenaline or adrenaline levels (Figure 2).

Effects of a single administration of YKS on the RS-induced elevation of plasma catecholamine levels. YKS was administered 4 h prior to the start of RS exposure. Plasma catecholamine levels after RS exposure for 60 min were measured. *Significantly different (P < .05) from the vehicle (V) + RS (−) group.
Effects of Repeated Administration of YKS on Body Weight
In our main experiment, YKS was administered for 14 days. Body weight was measured daily, just before YKS administration. Repeated administration of YKS for 14 days had no significant effect on body weight (Figure 3). On day 14, mean body weight was 355 ± 6.6 g for the vehicle + RS group (n = 12), and 352 ± 6.1 g for the YKS + RS group (n = 16).

Effects of repeated Yokukansan (YKS) administration on body weight. Body weight was measured just before YKS administration for determination of dosage every day for 14 days.
Effects of Repeated Administration of YKS on RS-Induced Elevation of Plasma Catecholamine Levels
Two-way analysis of variance (ANOVA) indicated a significant main effect for time [F(3, 68) = 25.028, P < .01] on plasma noradrenaline levels, but no significant main effect for treatment or interaction between treatment and time (Figure 4). Plasma noradrenaline levels were increased during stress exposure compared to basal levels in both vehicle-treated and YKS-treated rats, and YKS did not affect the elevation of plasma noradrenaline. In contrast, there were significant main effects for treatment [F(1, 68) = 6.836, P < .05] and time [F(3, 68) = 30.241, P < .01] on plasma adrenaline levels and a significant interaction between treatment and time [F(3, 68) = 2.782, P < .05; Figure 4]. RS increased plasma adrenaline levels in both vehicle-treated and YKS-treated rats at 20 and 40 min. Furthermore, post-hoc analysis revealed that plasma adrenaline levels in YKS-treated rats were significantly suppressed compared with those of vehicle-treated rats after 60 min of RS, and the levels at 60 min showed no significant difference compared with the basal levels in YKS-treated rats.
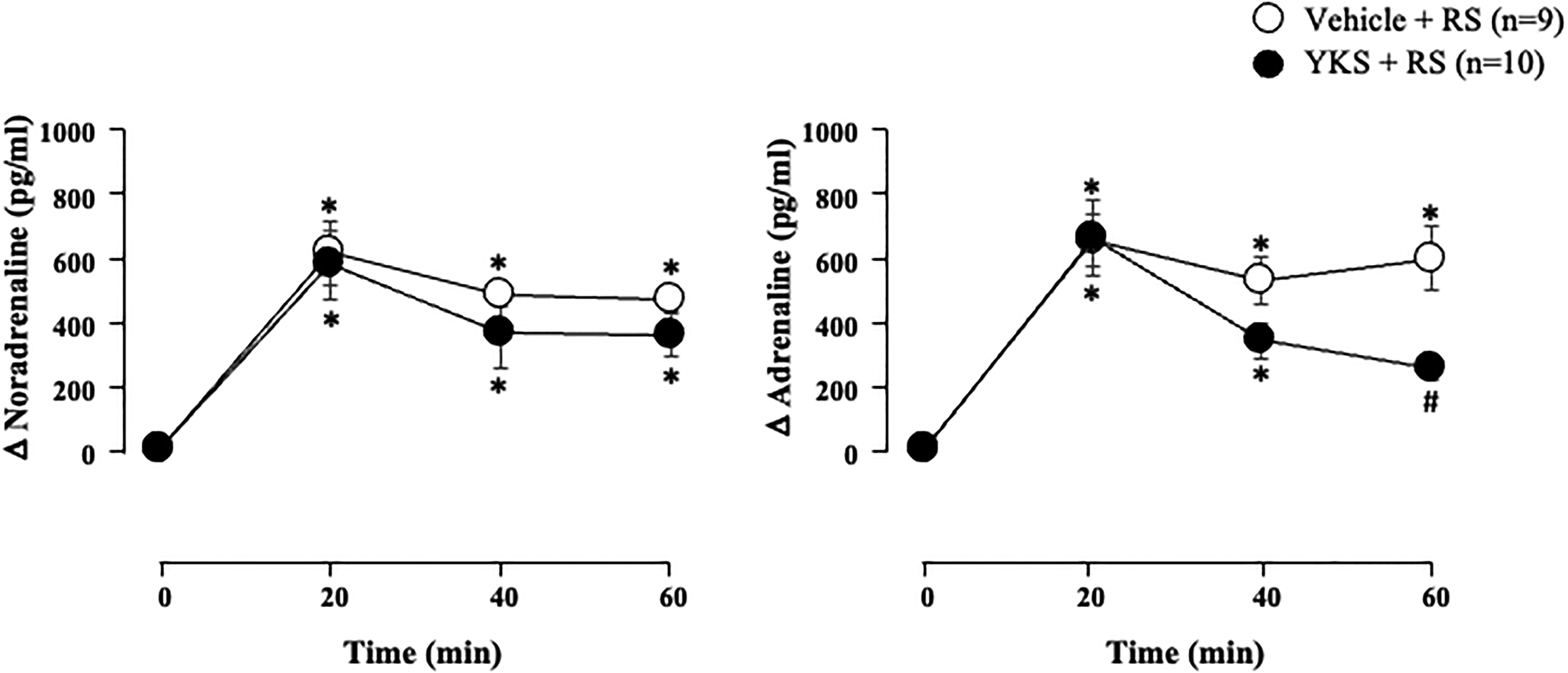
Effects of repeated YKS administration on the RS-induced elevation of plasma catecholamine levels. After administration of YKS for 14 days, plasma samples were collected during a 60-minute RS exposure on day 14. ○, vehicle + RS (n = 9); ●, YKS + RS (n = 10). *Significantly different (P < .05) from 0 min in each group; #significantly different (P < .05) from the vehicle + RS group.
The actual values for noradrenaline and adrenaline at 0 min were 73.9 ± 41.1 and 35.5 ± 14.5 pg/mL for the vehicle + RS group (n = 9), and 99.2 ± 31.6 and 92.7 ± 38.6 pg/mL for the YKS + RS group (n = 10), respectively.
Effects of Repeated YKS Administration on RS-Induced Elevation of PGE2 and TxB2 Levels in the PVN
There were significant main effects for treatment [F(1, 48) = 8.699, P < .01] and time [F(3, 48) = 3.244, P < .05] on the levels of PGE2 in the PVN, but no significant interaction was found between them (Figure 5). 33 In vehicle-treated rats, the PGE2 levels in the PVN were significantly increased at 40 min of RS (Figure 5; 156.8% of the basal level). In contrast, PGE2 levels did not change during stress exposure in YKS-treated rats, and YKS significantly suppressed PGE2 levels at 40 min compared with vehicle-treated rats.

Effects of repeated YKS administration on the RS-induced elevation of PGE2 and TxB2 levels in the PVN. (A) After administration of YKS for 14 days, PVN dialysates were collected during a 60-minute RS exposure on day 14. ○, vehicle + RS (n = 6); ●, YKS + RS (n = 8). *Significantly different (P < .05) from 0 min in each group; #significantly different (P < .05) from the vehicle + RS group. (B) Diagrammatic representation of rat brain frontal plates from Paxinos and Watson, 33 showing the placement of the microdialysis probe. (C) Representative ion chromatograms for standard prostanoids and their internal standards from LC-ITMSn analysis.
Two-way ANOVA indicated a significant main effect for treatment [F(1, 48) = 7.930, P < .01], but no significant main effect for time nor an interaction between treatment and time on TxB2 levels in the PVN (Figure 5). Similar to PGE2, TxB2 levels in the PVN were significantly increased at 40 min (173.7% of the basal level) in vehicle-treated rats. In YKS-treated rats, TxB2 levels showed no significant change during exposure to RS.
Effects of Repeated YKS Administration on Neurotransmitter Levels in the PVN
The 4 neurotransmitters measured in the dialysis experiments (5-HT, GABA, ACh, and histamine) showed different responses to 60 min of RS exposure in this study (Figure 6). 33 In the PVN dialysate, two-way ANOVA indicated a significant main effect of treatment for both 5-HT and GABA [5-HT: F(1, 28) = 18.460, P < 0.01; GABA: F(1, 28) = 18.240, P < .01], but no significant main effect for time nor interaction between treatment and time. In vehicle-treated rats, RS significantly increased 5-HT levels at 20 and 40 min and GABA levels at 60 min (Figure 6). In contrast, neither 5-HT nor GABA levels changed during the 60-min stress exposure in YKS-treated rats. Post-hoc analysis revealed that the RS-induced increase in 5-HT (at 20 and 40 min) and GABA (at 60 min) levels were suppressed by YKS treatment. Moreover, there were significant main effects of treatment [F(1, 28) = 16.362, P < .01] and time [F(3, 28) = 4.515, P < .05] on ACh levels and also a significant interaction between treatment and time [F(3, 28) = 4.768, P < .01]. In the PVN, ACh levels significantly increased at 40 min (Figure 6), and this increase was suppressed to basal levels. No significant main effects on histamine levels or interactions were found.

Effects of repeated YKS administration on neurotransmitter levels in the PVN. (A) After administration of YKS for 14 days, PVN dialysates were collected during a 60-minute RS exposure on day 14. ○, vehicle + RS (n = 4); ●, YKS + RS (n = 6). *Significantly different (P < .05) from 0 min in each group; #significantly different (P < .05) from the vehicle + RS group. (B) Diagrammatic representation of rat brain frontal plates from Paxinos and Watson 33 , showing the placement of the microdialysis probe. (C) Representative ion chromatograms for standard neurotransmitters and their internal standards from LC-ITMSn analysis. Abbreviations are the same as in Figure 5.
Discussion
Stress exposure causes sympathetic activation such as hypertension and tachycardia. Stress-induced increases in plasma catecholamine levels trigger these sympathetic responses. Previous studies clearly revealed that acute RS exposure greatly stimulates release and/or secretion of catecholamines.9,34,35 Consistent with these previous studies, we also found that acute RS caused a large increase in plasma levels of both noradrenaline and adrenaline in vehicle-treated rats, as shown in Figure 2. In the present study, to determine if YKS treatment can affect stress-induced sympathetic activation, we examined the effects of YKS treatment on RS-induced catecholamine responses and prostanoid production in the PVN.
Several lines of evidence have demonstrated that repeated administration of YKS is more effective than a single administration in ameliorating anxiety-like behavior18,20,25 and aggressive behavior. 24 However, several studies have suggested that single and repeated administration of YKS have different effects. Yamaguchi et al 20 reported that a single administration of YKS suppresses innate fear as measured by elevated plus maze test, whereas repeated administration shows anxiolytic effects for both innate fear and conditioned fear in a contextual fear conditioning test. Other studies indicated that single administration of YKS affects only social abnormalities but repeated administration ameliorates aggression in addition to sociality,24,26 suggesting that YKS might have an acute effect on social behavior and a chronic effect on aggressive behavior. Taken together, most previous findings suggested the benefits of repeated YKS treatment for abnormal behaviors and the possibility that a single treatment may also be effective in some cases. Therefore, in our first experiment, we tried to reveal whether a single administration of YKS exerts effects on stress-induced sympathetic activation. However, our results showed that a single administration of YKS did not affect the RS-induced elevation of either noradrenaline or adrenaline levels. Therefore, we next aimed to examine the effects of chronic YKS treatment on RS-induced sympathetic activation and changes in neurotransmitter levels in the PVN.
It has been reported that YKS added to food at a dose of 300 mg/kg for 10 days has no influence on arterial blood pressure during cerebral ischemia and reperfusion in mice. 36 However, the effects of YKS treatment on sympathetic activation during stress exposure remain unknown. In this study, we examined the effects of repeated YKS administration on acute RS-induced elevation of plasma catecholamine levels. Our results showed that repeated administration of YKS significantly suppressed the RS-induced increase in adrenaline but not noradrenaline plasma levels. In general, plasma noradrenaline levels reflect noradrenaline spillover from sympathetic nerve terminals to plasma (and/or noradrenaline secreted from the adrenal medulla), whereas plasma adrenaline is only secreted from the adrenal medulla.37,38 These differences suggest that plasma noradrenaline acts mainly in a target organ-specific manner, whereas plasma adrenaline acts systemically. Based on previous findings and our current results, the combined data raise the possibility that repeated administration of YKS has a selective effect on sympathetic responses induced by stress exposure and that YKS can suppress systemic sympathetic responses to stress via suppression of adrenaline secretion. Although the detailed mechanisms underlying YKS-mediated suppression of catecholamine levels remain unclear, one possible mechanism may involve neuroinflammatory mediators. We previously reported that central inhibition of inducible nitric oxide synthase (iNOS) suppresses CRF- and RS-induced elevation of plasma catecholamine levels and activation of presympathetic PVN neurons,8,39 and that iNOS is involved in the interleukin (IL)-1β-induced elevation of plasma noradrenaline levels. 40 A previous in vitro study showed that YKS treatment suppressed the expression of iNOS and IL-1β mRNA and protein in lipopolysaccharide-stimulated BV2 cells. 41 These pathways, including iNOS and IL-1β, might also be involved in the suppressive effects of YKS on plasma catecholamine elevation.
It is well-known that the PVN is the major integrative center for sympathetic function and stress responses in the brain. 2 The PVN contains presympathetic neurons which directly or indirectly project to sympathetic preganglionic neurons in the spinal cord.42,43 We have previously reported that prostanoids in the brain, especially in the PVN, are involved in CRF- and RS-induced elevation of plasma catecholamine levels.5,6,9 In addition, it has also been shown that inhibition of cyclooxygenase (COX), which is the rate-limiting enzyme in the synthesis of prostanoids, results in CRF- and RS-induced elevation of both noradrenaline and adrenaline levels.5,8 Therefore, in this study, we examined whether repeated YKS administration can affect prostanoid production in the PVN during RS exposure, as prostanoids are important factors in activating sympathetic function. We found that acute RS significantly increased both PGE2 and TxB2 (a metabolite of TxA2) levels, which peaked in the 40-min fraction, in PVN microdialysates. These results are generally consistent with a previous report using RS-exposed rats. 9 Furthermore, in the present study, our results showed that YKS administration suppressed the elevation of both PGE2 and TxB2 levels in the PVN. It has also been reported that YKS decreased substance P-induced COX-2 expression in U373 MG glioblastoma astrocytoma cells. 44 Consistent with this, we previously found that the inhibition of COX-1 and COX-2 suppresses the RS-induced elevation of plasma catecholamine levels and neuronal activation of presympathetic PVN neurons. 8 Taken together, it is highly possible that YKS induces the downregulation of COX(s) in the PVN and then decreases prostanoid production in the PVN, leading to suppression of the RS-induced elevation of plasma catecholamine levels.
Finally, we examined the effects of YKS on stress-dependent changes in neurotransmitter release in the PVN. In the present study, we analyzed 4 neurotransmitters (5-HT, GABA, ACh, and histamine) in PVN dialysates from rats that were repeatedly administered YKS for 14 days. Our results showed that RS exposure significantly increased 5-HT, GABA, and ACh, but not histamine, in the PVN, and that repeated administration of YKS suppressed the RS-induced increase in these neurotransmitters. It has been reported that the activities of 5-HT, 45 GABA (via GABAB, but not GABAA, receptors),46,47 and ACh 48 in the PVN are involved in sympathetic activation. Therefore, it is possible that the YKS-induced decrease in these transmitter levels in the PVN shown in our study might be related to YKS-dependent suppression of sympathetic activation during stress exposure. Several previous studies have revealed that repeated YKS administration affects neurotransmitter levels in various brain regions. For example, dietary zinc deficiency increases the concentration of GABA, glutamate, and dopamine in brain homogenates of socially isolated mice, and YKS treatment restores the transmitters to normal levels. 28 In contrast, other studies have revealed that decreases in neurotransmitters in the brain, that is, ischemia-induced reduction of ACh release in the dorsal hippocampus 27 and age-dependent reductions in 5-HT concentration in the prefrontal cortex, 31 are improved by YKS treatment in rodents. Although the cause of this discrepancy remains unknown, there is a possibility that YKS can normalize neurotransmitter levels—whether decreasing or increasing. The relationship between the YKS-induced normalization of neurotransmitter levels and the YKS-induced suppression of catecholamine levels during RS exposure shown in our study remains unclear at this time. These details need to be further investigated in future studies.
Conclusions
In this study, we investigated the effects of YKS on stress-induced sympathetic activation in rats. Our results showed that repeated administration of YKS suppressed the RS-induced elevation of plasma adrenaline, but not noradrenaline, levels. Moreover, the YKS administration also suppressed the elevation of both PGE2 and TxB2 levels in the PVN, which is the major integrative center for sympathetic regulation. We also found that YKS administration suppressed the RS-induced increase in 5-HT, GABA, and ACh, but not histamine, in the PVN. Our results suggest that YKS can ameliorate stress-induced sympathetic activation via inhibition of stress responses in the brain.
Materials and Methods
Animals
Male Wistar rats weighing ∼ 380 g were maintained in an air-conditioned room at 22 °C to 24 °C under a constant day-night rhythm for more than 3 weeks with food and water provided ad libitum. All experiments were conducted in compliance with the guiding principles for the care and use of laboratory animals approved by Aichi Medical University (Nos. 2019-1 and 2020-39).
Experimental Design
We first examined whether a single administration of YKS can affect the stress-induced elevation of plasma catecholamine (noradrenaline and adrenaline) levels (n = 18). In the next experiments, to determine the effects of repeated administration of YKS on stress-induced activation of the sympathetic nervous system, we examined plasma catecholamine levels and PVN prostanoid levels (PGE2 and TxB2) during stress exposure in YKS-administered rats (n = 19). In addition to the effects of YKS on stress-induced sympathetic activation, the effects of YKS on neurotransmitter levels in the PVN during stress exposure (n = 10) were also analyzed. In the 2 later experiments, all rats were administered YKS or vehicle daily for 14 days, then were exposed to RS for 60 min on day 14 (Figure 1). In parallel, PVN dialysate was collected before and during exposure to RS.
Drug Administration
A dried extract powder of YKS (Lot No. 2170054020) was kindly provided by Tsumura & Co. (Tokyo, Japan). YKS was prepared by Tsumura & Co.: all 7 herbs were extracted with purified water (95 °C, 1 h), and the extraction liquid was separated from the insoluble waste and concentrated by drying under diminished pressure. Spray drying was used to produce an extract powder, and the yield of the extract was about 15.9%. 49
YKS was dissolved in distilled water (1000 mg/10 mL). The dose of YKS (1000 mg/10 mL/kg, p.o.) was determined based on previous studies showing ameliorative effects on stress-induced behavioral abnormalities and corticosterone elevation.17,20,23,24 It has been reported that repeated administration, but not single administration, of YKS for 14 days had ameliorative effects on anxiety and emotional abnormalities.18,25 In our first experiment, a single administration of YKS 4 h prior to stress exposure did not affect the stress-induced elevation of plasma catecholamine levels (Figure 2). We thus administered YKS daily for 14 days in the subsequent experiments. Four hours after the last administration on day 14, all rats were subjected to dialysis experiments.
Restraint Stress
Exposure to RS was performed according to our previous reports.8,9 All rats were restrained by taping their body trunk and 4 limbs to a metal mesh for 60 min.
Microdialysis
Microdialysis experiments were performed according to our previously published methods.6,9 On day 9 (5 days before the dialysis experiments), rats were anesthetized intravenously with a mixture of medetomidine, midazolam, and butorphanol (0.75, 4.0, and 5.0 mg/kg, respectively), then a guide cannula (Eicom, Kyoto, Japan) was implanted for a microdialysis probe.6,9 After the last administration of YKS on day 14, a microdialysis probe (Eicom, Kyoto, Japan) was inserted into the right side of the PVN.6,9 After stabilization for 4 h, dialysis samples for the pre-RS time point were collected for 60 min prior to the stress exposure; rats were then subjected to RS for 60 min (Figure 1).
Measurement of Plasma Catecholamines
Catecholamine levels in plasma samples were assayed according to our previously published methods3,5,9 by high-performance liquid chromatography (HPLC) with electrochemical detection (HTEC-510, Eicom, Kyoto, Japan).
Measurement of PGE2 and TxB2 in PVN Microdialysates
PGE2 and TxB2 (a metabolite of TxA2) in PVN microdialysates were assayed according to our previous reports9,50 by liquid chromatography-ion trap tandem mass spectrometry (LC-ITMSn) analysis. To measure prostanoid concentrations in the dialysates, the peak area ratio relative to an internal standard was calculated and determined from the corresponding calibration curve.
Measurement of Neurotransmitters in PVN Microdialysates
Neurotransmitters (5-HT, GABA, Ach, and histamine) in PVN microdialysates were analyzed according to our previously published methods47,51 by LC-ITMSn analysis. To measure neurotransmitter concentrations in the dialysates, the peak area ratio relative to an internal standard was calculated and determined from the corresponding calibration curve.
Data Analysis and Statistics
Data are expressed as the mean ± SEM Plasma catecholamine levels are displayed as the net change above the respective basal values, and data from the microdialysates (prostanoids and neurotransmitters) are shown as a percentage of the respective basal values. The data were analyzed by a one-way ANOVA followed by a post hoc analysis with the Bonferroni method (Figures 2 and 3) and by a two-way ANOVA for the main effects of treatment (vehicle or YKS) and time (0, 20, 40, or 60 min from the start of RS) and their interaction with SPSS v25.0 (IBM, New York, NY, USA) followed by a post hoc analysis with the Bonferroni method (Figures 4 to 6). P values < .05 are defined as being statistically significant.
Footnotes
Abbreviations
Acknowledgments
We thank Natsumi Kodama and Makoto Naruse (Division of Advanced Research Promotion, Institute of Comprehensive Medical Research, Aichi Medical University) for expert assistance in the LC-ITMSn experiment, and thank also Atsushi Iida, Shotaro Takemoto, Koshiro Tanamoto, Momoko Higuchi, Marimo Yonesu, and Mayuko Watanabe (School of Medicine and Aichi Medical School) for assistance in the preliminary experiments. We thank Tsumura & Co. (Tokyo, Japan) for generously providing Yokukansan.
Authors Contributions
Naoko Yamaguchi: conceptualization, methodology, formal analysis, investigation, writing–original draft, writing–review and editing, funding acquisition. Kenta Maruyama: formal analysis, writing–review and editing. Shoshiro Okada: writing–review and editing, funding acquisition.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by Aichi Medical University (Nos. 2019-1 and 2020-39).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI Grant Number 20K07982 (to N.Y.) and the Research Foundation for Oriental Medicine (to S.O.).
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the guiding principles for the care and use of laboratory animals approved by Aichi Medical University (Nos. 2019-1 and 2020-39).
Supporting Information
The data presented in this study are included in this article. Further inquiries can be directed to the corresponding author.
