Abstract
Objectives: The up-regulation of proinflammatory cytokines in the hypothalamic paraventricular nucleus (PVN) is well demonstrated to be involved in the development of neurogenic hypertension, including stress-induced hypertension (SIH). IL-17A has been found to be increased in the PVN of several hypertensive animal models and has been shown to play a key role in the development of hypertension. Although IL-36γ was found to be expressed in spinal neurons, its role in hypertension remains elusive. Here, we investigated the co-expression of IL-17 receptor A (IL-17RA) and IL-36γ in the PVN cells of SIH rats. Methods: The electric foot shock combined with buzzer noise stressors were used to make hypertensive rat model. The immunochemical staining or immunofluorescence staining was used to reveal cells as requested. The Western blot was used to detect the related protein levels. Results and Conclusion: In the PVN of the SIH rats, the number of CD3+CD4+ T cells was significantly increased by the immunochemical staining. Additionally, the protein levels of RORγt and IL-17A were significantly upregulated by Western blot, confirming the infiltration of CD4+ T cells and differentiation into Th17 cells in the PVN of SIH rats. Immunofluorescence staining revealed abundant expression of IL-17RA in PVN neurons, with relatively less expression in astrocytes or microglia. Furthermore, IL-36γ positive cells and protein expression of IL-36R were significantly increased. Notably, this study demonstrates for the first time that most IL-36γ cells were strongly colocalized with IL-17RA positive cells in the PVN of SIH rats, and the colocalized cells were significantly higher in SIH rats. This suggests that IL-17A secreted by infiltrated Th17 cells may stimulate PVN neurons to produce IL-36γ via IL-17RA, indicating that the combination of IL-17 and IL-36γ might produce strong pro-inflammatory effects in the PVN of SIH rats.
Introduction
Neurogenic hypertension is characterized by excessive sympathetic nerve activity (SNA). Although a large body of studies has been carried out to unravel the mystery of increased SNA, the mechanisms that drive excessive SNA in neurogenic hypertension remain to be elucidated. The pre‐sympathetic neurons of the paraventricular nucleus (PVN) project to the rostral ventrolateral medulla (RVLM) and intermediolateral column (IML) of the spinal cord, both of which are the major centers in the control of SNA.
An impairment of neuronal activity in in the cardiovascular centers of the PVN and RVLM is linked to an augmentation of sympathetic activity, which has considered to be a crucial mechanism for the pathogenesis of essential hypertension. Multiple research evidence has demonstrated that neuroinflammation characterized by an increase in pro-inflammatory factors and a decrease in anti-inflammatory factors has a strong correlation with sympathoexcitation. 1 Zhu and his colleagues showed that microinjection of exogenous tumor necrosis factor-α (TNF-α) or interleukin-1β (IL-1β) into the PVN significantly increased sympathetic outflow and blood pressure. 2 Furthermore, PVN microinjection of TNF-α-blocker or IL-1β inhibitor significantly attenuated the capsaicin-induced increases in SNA and blood pressure. 3 These findings suggest a role of inflammatory mechanisms in driving SNA and elevation of blood pressure.
Accumulating evidence indicated that the levels of proinflammatory cytokines such as TNF-α, IL-1β, interleukin-1 (IL-6) and interleukin-17 (IL-17) increased and anti-inflammatory cytokines including transforming growth factor-β (TGF-β) or interleukin-10 (IL-10) decreased in the PVN in various forms of hypertension models.1,2,4–7 In the central nervous system (CNS), cytokines are primarily produced by activated microglia and reactive astrocytes in the brain parenchyma. Various pro-hypertensive stimuli such as Ang II, high salt and psychological stress, etc., have been found to prime microglia and astrocytes into pro-inflammatory phenotype, leading to a dysregulated neuroinflammatory state.4,8,9 In these pathological conditions, the disruption of blood brain barrier (BBB) and the upregulation of chemokines facilitate the infiltration of peripheral immune cells into the brain. Additionally, The Th17 cells levels have been observed to increase in the brain of mice subjected to chronic restraint stress. 10 Previous findings have indicated that Th17 cells infiltrated the parenchyma of the PVN in response to stress and play an important role in the pathophysiology of the stress-induced hypertension (SIH). 11 Th17 cells are major producers of IL-17, with IL-7A being the most extensively studied member of the IL-17 families. Studies have shown that IL-17A is upregulated in animal hypertensive models or hypertensive patients and mediates the regulation of blood pressure (BP). 12 An injection of anti-IL-17 antibody in the PVN evoked a reduction of blood pressure of the SIH rats, 11 indicating that the infiltrated immune cells in the CNS are involved in neuroinflammation and development of the SIH. IL-17A signals through its receptor IL-17RA. However, how IL-17A/IL-17RA signaling contributes to the pathogenesis in the SIH remains unclear.
Interleukin-36 (IL-36) cytokines, including IL-36α, β and γ, are produced primarily by monocytes/macrophages, T cells, keratinocytes and bronchial epithelial cells. 13 Although the expression of IL-36 and its receptor IL-36R in the CNS is less well-known, IL-36 cytokines have been implicated in neuroinflammation.14,15 In mice treated with intraplantar injections of complete Freund’s adjuvant, IL-36γ but not IL-36α or IL-36β was persistently upregulated in spinal neurons, with IL-36 receptor (IL-36R) primarily expressed in spinal astrocytes, 16 indicating the role of IL-36γ/IL-36R signaling in the inflammatory diseases of the CNS.
IL-17A has been shown to stimulate keratinocytes to express IL-36, which plays a crucial role in the pathogenesis of psoriasis.17–19 However, the synergistic effect of IL-17A and IL-36 in neuroinflammation is less studied. In the present study, the expression of IL-36γ/IL-36R and IL-17/IL-17RA in the PVN of the SIH rats was investigated, along with the co-expression of IL-17RA and IL-36γ. We found that IL-17A and IL-36γ were upregulated with co-expression of IL-17RA and IL-36γmainly in neurons of the PVN of the SIH rats. This study provides a new mechanism underlying the SIH and suggests a potential therapy in clinical hypertension management.
Materials and methods
Animal preparation
Male adult Sprague-Dawley (SD) rats (200–250 g) used in this study were purchased from the Laboratory Animal Center, Zhejiang Academy of Medical Sciences (SCXK (zhe) 2019–0002). The rats were housed in a temperature-controlled room (22 ± 1°C) under a 12 h light-dark cycle with food and water ad libitum. All procedures were approved by the Animal Care and Use Committee of Jiangsu Vocational College of Medicine (Ethics review form XMLL-2022-061).
Rats were randomly assigned into two groups: sham, and stress. The stress group rats were individually placed in a cage (22 cm × 22 cm × 28 cm) with a grid floor and exposed to electric foot shock combined with buzzer noise stress for 2 h twice a day, with a 4-h interval between sessions, for consecutive 14 d. 20 Control rats were also individually placed in a similar cage but without receiving stressors (electric shocks and noises). Consistent with our previous findings,4,11,20,21 the stressed rats exhibited a gradual increase in systolic blood pressure in the first 9 days, plateauing on day 10 but without reaching hypertension until day 14. On day 14 when the stress stopped, the rat brains were sampled for the following experiments.
Immunohistochemical staining (IHC)
Immunohistochemical staining was performed as previously described. 20 In brief, the fixed rat brains were mounted on a freezing microtome (CM 1850, Leica, Germany) and coronal sections (18 μm in thickness) of the PVN (before and after bregma −1.80 mm) were sliced according to the atlas of Paxinos and Watson (1996). To inactivate endogenous peroxidase, sections were immersed in a solution of 30% H2O2/methanol (1:50). Then, the sections were respectively incubated with rabbit polyclonal antibody to CD3 (ab5690, abcam, UK; 1:50), mouse monoclonal antibodies to CD4 (MA5-17390, thermo fisher scientific, CN; 1:25) and to IL-36γ (ab239526, abcam, UK; 1:50) overnight at 4°C. After washing, the slices were incubated with biotin-labeled rabbit immunoglobulin G (IgG) secondary antibodies and StreptAvidin-Biotin Complex (SA1028, BOSTER, Wuhan, China) at room temperature for 30 min. The sections were visualized by 3, 3′-diaminobenzidine (DAB), mounted with neutral gum, observed by Olympus B×51 microscope, and photographed with a digital camera. Immunoreactive cells were counted by ImageJ software. Six slices were counted for each rat brain, and the average was taken as the number of positive cells.
Immunofluorescence staining
We performed Immunofluorescence staining as described previously. 11 The rat brain samples were fixed (4% paraformaldehyde, PFA), gradient sugar deposition (10%, 20%, 30% sucrose), and then coronal sections (35 mm in thickness) of the PVN were sliced using a freezing microtome (CM 1850, Leica, Germany). Sections were incubated in rabbit monoclonal antibody to CD3 (ab135372, abcam, UK; 1:50) and mouse monoclonal antibody to CD4 (MA5-17390, Thermo Fisher Scientific, CN; 1:25); rabbit polyclonal antibody to IL-17RA (ab180904, abcam, UK; 1:50) and mouse monoclonal antibody to GFAP (cst3670, Cell Signaling Technology, USA; 1:50) or goat polyclonal antibody to Iba-1(NB100-1028, Novus Biologcials, USA; 1:50) or mouse monoclonal antibody to NeuN (ab104224, abcam, UK; 1:50); rabbit polyclonal antibody to IL-17RA (ab180904, abcam, UK; 1:50) and mouse monoclonal antibody to IL-36γ(ab239526, abcam, UK; 1:50), at 4°C overnight. After the sections were washed with 0.01 PBS, they were incubated with goat anti-mouse IgG H&L (Alexa Fluor® 488) (ab150113, abcam, UK; 1:200) or donkey anti-goat IgG H&L (Alexa Fluor® 488) (ab150129, Abcam, UK; 1:200) and goat anti-rabbit IgG H&L (Cy3 ®) preadsorbed (ab6939, abcam, UK; 1:200) as secondary antibody at room temperature for 2 h. A laser scanning confocal microscope (LSM 900, Zeiss, Germany) was used to observe the positive cells.
Western blot
The protocol of western blot was the same as that previously reported. 11 Briefly, 200 μL of pre-cooled extraction reagent was added to the rat PVN sample. The extraction reagent was pre-added with 2 μL of protease inhibitor, 10% PMSF, and phosphatase inhibitor. Protein concentration was measured by the BCA method. 10% separation gel and 60 V constant voltage were first used, then 120 V constant voltage was used for electrophoresis until the bromophenol blue color reached the bottom of the separation gel. Subsequently, proteins were transferred onto a PVDF membrane that was washed with TBST solution 3 times, 5 min each time. The membrane was then blocked with 5% milk in PBS-Tween 20 for 1 h, and incubated overnight at 4°C with rabbit polyclonal antibody to RORγt (PA5-86733, Thermo Fisher Scientific, CN; 1:50), rabbit polyclonal antibody to IL-17A (PA5-79470, Thermo Fisher Scientific, CN; 1:50), mouse monoclonal antibody to β-Actin (AF0003, Beyotime, China; 1:1000), respectively. Then, the membrane was washed 3 times (10 min each time), incubated with anti-rabbit or anti-mouse secondary antibodies at room temperature for 60 min, washed, immersed in ECL solution for about 1 min, and photographed with a gel imaging system. The bands were quantified by ImageJ software.
Antibody array assay
At the end of modeling, the rats were killed by 10% chloral hydrate injection and decapitated, then the brains were quickly taken out, snap-frozen in liquid nitrogen, and stored at −80°C. PVN tissue was taken out from the brain as previously described. 4 Rat cytokine antibody array GS67 (GSR-CAA-67, RayBiotech, USA) was used to detect cytokines in the protein extracts of PVN according to the manufacturer’s instructions. Briefly, protein extracts were diluted to 500 µg/ml with blocking buffer, added to the array pools printed with 75 corresponding anti-cytokine antibodies, and incubated overnight. After washing, a biotin-conjugated anti-cytokine mix was incubated with the pools for 2 h. Finally, Cy3-conjugated streptavidin was used for glass series arrays. Afterwards, the glass slides were scanned to detect the fluorescent signals of microarrays using InnoScan 300 microarray scanner (Innopsys, France). The membrane arrays were exposed to HRP-catalyzing chemiluminescent solution using an Image Quant LAS4000 scanner (GE Healthcare, Waukesha, WI, USA). The signal values were read and normalized using an internal positive control by a RayBiotech analysis tool specifically designed to analyze the data of rat cytokine antibody arrays.
Statistical analysis
Data are expressed as mean ± SEM. For data with a normal distribution, the difference between two group means was tested with Student’s t-test, paired or unpaired as appropriate. The null hypothesis was rejected at p < .05. For data that did not follow a normal distribution or had an unknown distribution, the difference between two group means was tested with equivalent non-parametric tests. The difference among multiple means was tested with one-way ANOVA followed by Sidak’s multiple comparisons test. Statistical analysis was performed using GraphPad Prism (version 8.0.1, GraphPad Software, San Diego, CA, USA).
Results
Infiltration of CD3+CD4+ lymphocytes into the PVN of SIH rats
IL‐17 is primarily produced by Th17 cells which are one of the subsets of T helper cells that express CD4. To assess the infiltration of peripheral T cells in the PVN in rats exposed to SIH, we used immunochemical staining to identify the infiltrated CD3+ T cells (pan T-cells) characterized by expression of the cluster of differentiation cell surface marker CD3. As shown in Figure 1(a) and 1(b), the number of CD3+T cells significantly increased in the PVN of the SIH rats compared to that in the sham rats. Similarly, the number of CD4+T cells also increased significantly in the PVN of the SIH rats compared to the sham rats as shown in Figures 1(c) and 1(d). The further immunofluorescence staining revealed the presence of CD3+CD4+ (double positive) T cells in the PVN of the SIH rats (Figure 1(e)). CD3+ -, CD4+ - and CD3+CD4+ T cells were not detected by immunofluorescence staining in the PVN of the sham rats. Infiltration of CD3+CD4+T cells into the PVN of SIH rats. (a) Representative images of CD3+ immunostaining cells (arrows indicated) in the PVN from foot-shock stressed rats and sham rats. (b) Statistical results of CD3+-positive cells from the indicated rats. (c) Representative images of CD4+ immunostaining cells (arrows indicated) in the PVN from foot-shock stressed rats and sham rats. (d) Statistical results of CD4+-positive cells from the indicated rats. (e) Representative images of CD3+CD4+ double positive T cells (arrows indicated) in the PVN of the foot-shock stressed rats by immunofluorescence staining. **p < .01 versus sham group, n = 3.
Upregulation of RORγt and IL-17A in the PVN of SIH rats
RORγt is a member of the retinoic acid-related orphan receptor (ROR) family of transcription factor and is the key transcription factor of Th17 cells, inducing the Th17 cell differentiation from CD4+T cells through encoding IL-17 gene and IL-17 expression. Therefore, the protein levels of RORγt were measured for the investigation of the filtrated Th17 cells in the PVN in the stressed rats. Analysis by western blot revealed that the protein levels of RORγt in the PVN of SIH rats were significantly higher than that in the sham rats (Figure 2(a) and 2(b)), indicating the infiltration of CD3+CD4+T cells (Th17) into the PVN of SIH rats. Th17 cells are characterized by the production of the inflammatory cytokine interleukin (IL)-17
22
and Th17 cells have been implicated in the pathogenesis of SIH.
11
Further analysis by western blot revealed that the protein levels of IL-17A (primary subtypes of the IL‐17 family) in the PVN of SIH rats were significantly higher than that in the sham rats (Figure 2(a)–2(d)). Consistent with our previous reports showing the infiltration of IL-17 positive Th17 cells in the PVN by using immunofluorescence, these results suggest that the infiltrated Th17 cells produced IL-17A cytokines. Upregulation of RORγt and IL-17A in the PVN of SIH rats. (a) Representative images of RORγt protein levels in the PVN from foot-shock stressed rats and sham rats. (b) Statistical results of RORγt protein levels from the indicated rats. **p < .01 versus sham group, n = 4. (c) Representative images of IL-17A protein levels in the PVN from foot-shock stressed rats and sham rats. (d) Statistical results of IL-17A protein levels from the indicated rats. *p < .05 versus sham group, n = 4.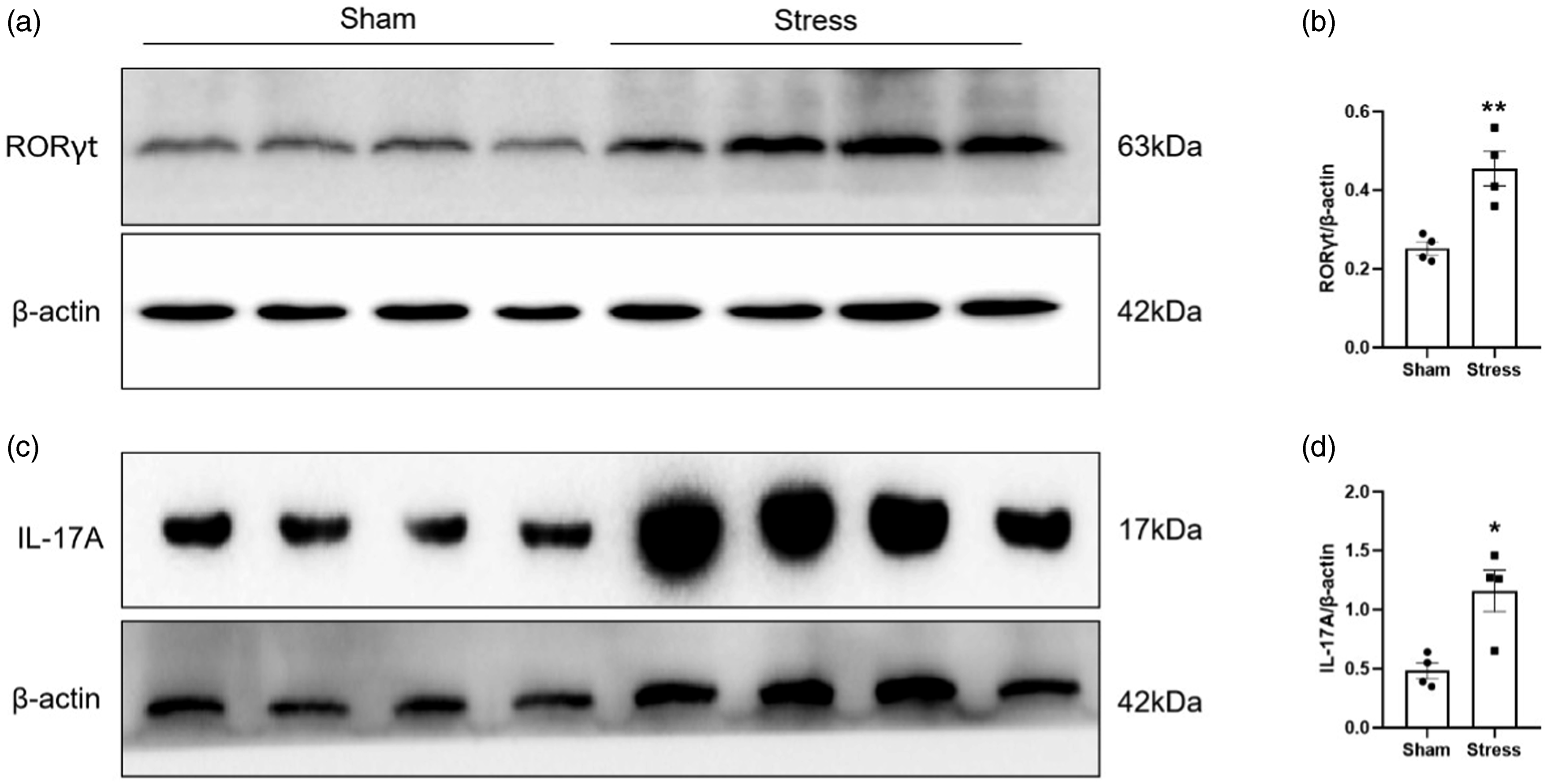
Distribution of IL-17RA in the PVN of SIH rats
IL-17A signals through its receptor IL-17RA. To determine the potential role of IL-17A in the development of SIH, we first examined the expression of IL-17RA in the microglia, astrocytes and neurons in the PVN of the SIH rats. The immunofluorescence staining revealed that IL-17RA expression was observed abundantly in NeuN-positive neurons (>20 cells/per field, Figures 3(a)–3(b)), moderately in GFAP-positive astrocytes (<15 cells/per field, Figure 3(c)–3(d)) and Iba1-positive microglia (<7 cells/per field, Figure 3(e)–3(f)). In comparison with the sham rats, IL-17RA-positive cells increased significantly in neurons, astrocytes and microglia in the PVN of the SIH rats, respectively. Cellular expression of IL-17RA in the PVN. (a) Representative images showing double positive staining of IL-17RA and NeuN by immunofluorescence staining in the PVN from foot-shock stressed rats and sham rats. (b) Statistical results of IL-17RA and NeuN double-positive cells from the indicated rats. (c) Representative images showing double positive staining of IL-17RA and GFAP by immunofluorescence staining in the PVN from foot-shock stressed rats and sham rats. (d) Statistical results of IL-17RA and GFAP double-positive cells from the indicated rats. (e) Representative images showing double positive staining of IL-17RA and Iba1 by immunofluorescence staining in the PVN from foot-shock stressed rats and sham rats. (f) Statistical results of IL-17RA and Iba1 double-positive cells from the indicated rats. **p < .01 versus sham group, n = 3.
Upregulation of IL-36γ and IL-36R in the PVN of SIH rats
Immunochemical staining was used to detect the expression of IL-36γ in the PVN of the SIH rats. Compared to the sham rats, IL-36γ positive cells significantly increased in the PVN of SIH rats (p < .01) Figure 4(a)–4(b). The RayBiotech antibody array GSR-CAA-67 was used to detect proteins of IL-36R (IL-1R6) in the PVN of SIH rats. The protein levels of IL-36R were significantly higher in SIH rats than that in sham rats (p < .01) Figure 4(c)–4(d). Expression of IL-36γ and IL-36R in the PVN. (a) Representative images of IL-36γ immunostaining cells in the PVN from foot-shock stressed rats and sham rats. (b) Statistical results of IL-36γ-positive cells from the indicated rats (n = 3). (c) Positions of proteins as indicated in (d) on the antibody array. (d) Statistical analysis of expressions of protein (n = 3). *p < .05; **p < .01.
Co-expression of IL-17RA and IL-36γ in the PVN of SIH rats
To investigate the potential synergistic effects of IL-17A and IL-36γ in the development of SIH, the double immunostaining was performed to examine the coexistence of IL-17RA and IL-36γ. As shown in Figure 5, most of IL-36γ-immunoreactivity (IR) cells were strongly colocalized with IL-17RA-IR in the PVN of SIH rats and the colocalized cells were significantly higher in SIH rats than that in sham rats (p < .01). Co-expression of IL-17RA and IL-36γ in the PVN. (a) Representative images showing double positive staining of IL-17RA and IL-36γ by immunofluorescence staining in the PVN from foot-shock stressed rats and sham rats. (b) Statistical results of IL-17RA and IL-36γ double-positive cells from the indicated rats. **p < .01 versus sham group, n = 3.
Discussion
Previous data from both animal and human studies have demonstrated the critical role of T cells in hypertension.23,24 The activated T cells infiltrate tissues and produce cytokines leading to hypertension. 25 The infiltration of peripheral T cells into the central nervous system is one of the important mechanisms underlying neuroinflammation involved in the development of hypertension. We previously found the increased Th17 cells in the PVN of the SIH rats. 11 Th17 cells are differentiated from naïve CD4+ T cells. In this study, we found the increased CD3+CD4+ T cells in the PVN of the SIH rats, suggesting the infiltration of the naïve CD4+ T in the PVN. As CD3+CD4+ T cell infiltration into the PVN is observed in the stressed rats but is almost undetectable in the control group by immunostaining, it is largely a stress-specific pathological phenomenon. The presence of both IL-6 or IL-21 and TGF-β is essentially required for the differentiation of CD4+ T cells into Th17 cells. In our previous reports, we have found the upregulation of IL-6 and TGF-β in the PVN of SIH rats. 4 These upregulated cytokines could promote the differentiation of CD4+ T cells into Th17 cells. Taking the previous results together with the present findings, the infiltrated CD4+ T cells are differentiated into Th17 cells in the PVN of the SIH rats. The increased protein levels of transcription factor RORγt further confirm the presence of Th17 cells in the PVN of the SIH rats. Th17 cells could also directly infiltrate into PVN as shown by the previous study. 11
IL-17A cytokines are potent proinflammatory cytokines secreted primarily by Th17 cells. In this study, the expression of IL-17A was increased in the PVN of SIH rats, suggesting that the infiltrated CD4+T cells were differentiated into Th17 cells and produced IL-17A. The injected recombinant IL-17A in the PVN of normal rats resulted in the elevation of blood pressure (unpublished data) and the blockage of IL-17A by an injection of anti-IL-17 antibody in PVN reduced the elevated blood pressure of rats subjected to chronic stress, suggesting the involvement of IL-17A in the development of stress-induced hypertension. 11 IL-17A has been shown to play a critical role in hypertension and its associated complications. 26 IL-17A was elevated in hypertensive mouse models and in humans with hypertension. 27 Peripherally, IL-17A elevated blood pressure through promoting multiple inflammatory mechanisms leading to vascular constriction and renal sodium retention. There is also emerging data demonstrating the involvement of the central IL-17A in the pathogenesis of hypertension. Recently, Cao et al. has demonstrated that PVN microinjection of IL-17A directly induced dramatic and prolonged increases in blood pressure. 28 Taken together, the increased IL-17A from the infiltrated Th17 cells in PVN of SIH rats would contribute to the development of stress-induced hypertension.
IL-17A signals through a heterodimeric receptor consisting of IL-17 receptor A (IL-17RA) and IL-17 receptor C (IL-17RC). 29 IL-17RA has been shown to be constitutively expressed in CNS tissue and upregulated in the pathological conditions. 30 IL‐17RA is found on various cell types in the CNS such as microglia, neurons, and astrocytes. 31 In the present study, we identified the cellular types expressing IL-17RA in the PVN of the SIH rats and sham rats. We found that IL-17RA was substantially expressed in neurons but less in astrocytes and microglia. The different expression of IL-17RA among different cells may implicate their different roles in the development of SIH. The increased number of IL-17RA positive neurons or astrocytes/microglia in the PVN of SIH rats suggests the IL-17RA might be involved in the stress-induced elevation of blood pressure. This was supported by the Cao’s findings showing that PVN microinjection of IL-17RA siRNA significantly attenuated IL-17A-induced sympathetic over-activation and elevation of blood pressure. 28 The high abundance of IL-17RA in the neurons highlights the importance of the neurons of PVN in mediating inflammation in the pathogenesis of SIH. There are three functionally different neurons in the PVN including the hypophysiotrophic PVN neurons that directly control the hypothalamic-pituitary-adrenocortical (HPA) axis, magnocellular neurons which secrete neurohypophysial peptides and brainstem projecting neurons that regulate autonomic function. These neuroendocrine neurons and pre-sympathic neurons both participate in the blood pressure regulation. Evidence suggests that all three neurons contribute to stress-induced hypertension.32,33 The wide-spread distribution of Th17-RA in the whole section of PVN in the present study would imply that more than one type of neurons has upregulated Th17-RA.
The present study revealed that IL-17RA is relatively less expressed in astrocytes and microglia in the PVN of SIH rats. The peripheral or central injection of IL-17A resulted in substantial activation of astrocytes and microglia via IL-17RA, accompanied by the upregulation of various inflammatory cytokines and chemokines in the PVN, 28 implying that IL-17RA expressed in these glia cells would plays a critical role in inducing chronic inflammation by IL-17 secreted by the infiltrated Th17 cells in the PVN of SIH rats. A functional IL-17RA enables astrocyte and microglia to be responsive to IL-17 by producing several pro-inflammatory cytokines which amplify neuroinflammation and by producing chemokines which facilitate the recruitment of Th17 cells within PVN.34,35 Thus, IL-17RA plays a key role in the cross-talk between Th17 cells and astrocytes and microglia, which form a positive-feedback loop to further aggravate the neuroinflammation, promoting hypertension. In the PVN of the SIH rats, we have found the activated microglia, the increased proinflammatory cytokines including IL-17 and inhibition of microglia by amphotericin diminished the elevated blood pressure. 4 These results suggest that IL-17A/IL-17RA signaling exerts effects on blood pressure via neuroinflammation mechanisms.
In the present study, we also found a significant increase both in IL-36γ positive cells and protein expression of IL-36R in the PVN of SIH rats, suggesting the involvement of IL-36γ/IL-36R signaling in the stress-induced neuroinflammation and hypertension. In CNS, IL-36γ was found to be expressed in spinal neurons of mice with intraplantar injections of complete Freund’s adjuvant. 16 IL-36γ could activate spinal astrocytes via IL-36R-mediated JNK pathway. 16 A study by Bozoyan et al. showed that IL-36γ activated microglia in the spinal cord of experimental autoimmune encephalomyelitis (EAE) mice. 14 These results indicated the implication of IL-36γ in neuroinflammation. We further conducted a double staining of IL-36γ with IL-17RA and found a large degree of colocalization of IL-36γ with IL-17RA.The co-existence of IL-36γ with IL-17RA indicates that IL-17A could stimulate PVN neurons, astrocytes and microglia to produce IL-36γ by binding to IL-17RA under stress. IL-17A alone is not a powerful inflammatory cytokine and its potent inflammatory action is mostly related to its synergistic actions with other proinflammatory cytokines. 36 The increased IL-36γ might exert its synergistic actions with IL-17A. IL-17A has been demonstrated to induce the upregulation of IL-36γ in keratinocytes of the skin and to be involved in the pathogenesis of inflammatory skin psoriasis through synergistic actions with IL-36γ.37,38
Limitations of this study. At present study, we demonstrated that IL17RA was mainly expressed in the PVN neurons. Furthermore, IL17RA was co-expressed with IL-36γ. However, there are some limitations for this study. Firstly, an incubation of the dissociated PVN neurons with IL-17A is needed to investigate whether IL17A IL17RA signaling would induce the release of IL-36γ from the PVN neurons. Secondly, a co-injection of IL-17A with IL-36γ in the PVN of normal rats is required to observe the synergistic effects of IL-17A and IL-36γ in raising blood pressure.
Conclusion
In summary, the present study provided the first data revealing that IL-17RA was substantially localized in PVN neurons of the SIH rats and IL-17RA was mostly co-expressed with IL-36γ in PVN neurons. The up-regulated IL-17A from the infiltrated Th17 cells in the PVN of the SIH rats might directly excite PVN neurons leading to sympathetic overactivation and an increase in blood pressure or induce the release of IL-36γ from PVN neurons via IL-17RA. The released IL-36γ from PVN neurons stimulated by the Th17 cells-derived IL17 A might further interact with IL17A, promoting the development of SIH. These findings will open a new direction of future exploration into potential therapies targeting IL-17RA for the treatment of hypertension.
Footnotes
Acknowledgements
CW acknowledges the support from Biotechnology and Biological Sciences Research Council [BB/P004695/1] and National Institute of Aging [1R01AG049321-01A1]. The funders have no roles in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Authors’ note
Qin Wu conceived and designed the study. Hong Zhang, Tingyu Ding, and Wenying Zhang acquired the data. Qin Wu, Yuping Chen and Jinjin Jiang analyzed and interpreted the data. Jihu Sun and Qin Wu wrote and prepared the manuscript. Changhao Wu critically revised the manuscript. Jihu Sun coordinated the study. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by the Six Major Talent Peak Expert of Jiangsu Province (No.WSN-120), the research Start-up Fund for high-level talents of Jiangsu Vocational College of Medicine (20191001), the College Breeding Program for the National Natural Scientific Foundation (20204301) and the Scientific Research Foundation of Jiangsu Vocational College of Medicine for Zhang Hong (20204109) and Wu Qin (20190402). CW acknowledges the support from Biotechnology and Biological Sciences Research Council [BB/P004695/1] and National Institute of Aging [1R01AG049321-01A1].
Ethical statement
Informed consent
Written informed consent was obtained from all subjects before the study.
Animal welfare
The present study followed institutional guidelines for humane animal treatment and complied with relevant legislation.
